Abstract
Enhancement of the subarachnoid space after intravenous administration of gadolinium contrast agent is not common. Enhancement usually occurs in pathological conditions that increase the permeability of the blood–cerebrospinal fluid barrier, most notably in meningitis. We herein describe possible subarachnoid enhancement in patients with no apparent effect on the meninges. These patients had clinical signs of Meniere’s disease and underwent specific magnetic resonance imaging of the inner ear to possibly visualize endolymphatic hydrops. The endolymphatic space can be noninvasively imaged by intravenous administration of contrast agent, usually at a double dose, 4 hours before the scanning process. During this time, the contrast agent penetrates not only the perilymph but also the subarachnoid space, where the highest concentration occurs after 4 hours according to some studies.
Keywords
Introduction
In recent years, magnetic resonance imaging (MRI) of the inner ear has been introduced as a useful diagnostic tool for Meniere’s disease (vertigo, tinnitus, and hearing impairment). The pathophysiological causes of Meniere’s disease remain unclear, but the underlying anatomical condition is endolymphatic hydrops (EH)—dilation of the inner ear structures (sacculus, utriculus, and ductus cochlearis) secondary to volume changes of the endolymph. 1 Recent studies have shown that the grade of EH is closely correlated with hearing impairment. 2 In the MRI protocol for EH, gadolinium contrast agent (GCA) is administered intravenously usually at a double dose of 0.2 mmol/kg; the scan then takes place after 4 hours to allow the GCA to reach its highest concentration in the perilymphatic space. 3
At present, macrocyclic nonionic GCA is mainly used because of its low risk of allergic reactions and suitable pharmacokinetics in the body after intravenous administration. The vast majority of MRI examinations of the central nervous system involve the use of GCA, which is distributed to the extracellular space (intravascular and interstitial). More than 95% of the GCA is excreted unchanged by the kidneys within 24 hours of administration in patients without impaired renal function. Elimination is generally divided into a rapid phase (first 2 hours after administration) and a slow phase (up to 6 days after administration).4,5 An unimpaired blood–brain barrier prevents penetration of the GCA into the brain parenchyma because of the macrocyclic and hydrophilic nature of GCA. 6 The importance of the blood–cerebrospinal fluid barrier (BCSFB) has been gradually elevated to the level of the blood–brain barrier because it may play an important role in some pathological processes. 7
Case report
The reporting of this study conforms to the CARE guidelines. 8 All patients have been de-identified.
MRI of the inner ear for diagnosis of EH began in our hospital in 2016, and 67 patients had been examined at the time of this writing. In this study, we used 3T heavily weighted T2 three-dimensional fluid-attenuated inversion recovery (3D-FLAIR) scans of the pyramids of the temporal bones to evaluate the endolymphatic space, and gadobutrol was used in every patient. We added a whole-brain T2 FLAIR axial scan to our protocol as a screening method for possible incidental findings. The parameters of this sequence were as follows: field of view, 220 mm; slice thickness, 3 mm; inversion time, 2500 ms; repetition time, 9000 ms; echo time, 87 ms; flip angle, 150 degrees; matrix, 217 × 230; bandwidth, 256 Hz/Px; turbo factor, 16; voxel size, 0.7 × 0.7 × 3 mm; and acquisition time, 4 minutes 32 s. In one patient, we detected a relatively extensive homogeneous increase in the signal of the left frontal subarachnoid (SA) space (Figure 1(a)) on the T2 FLAIR sequence.
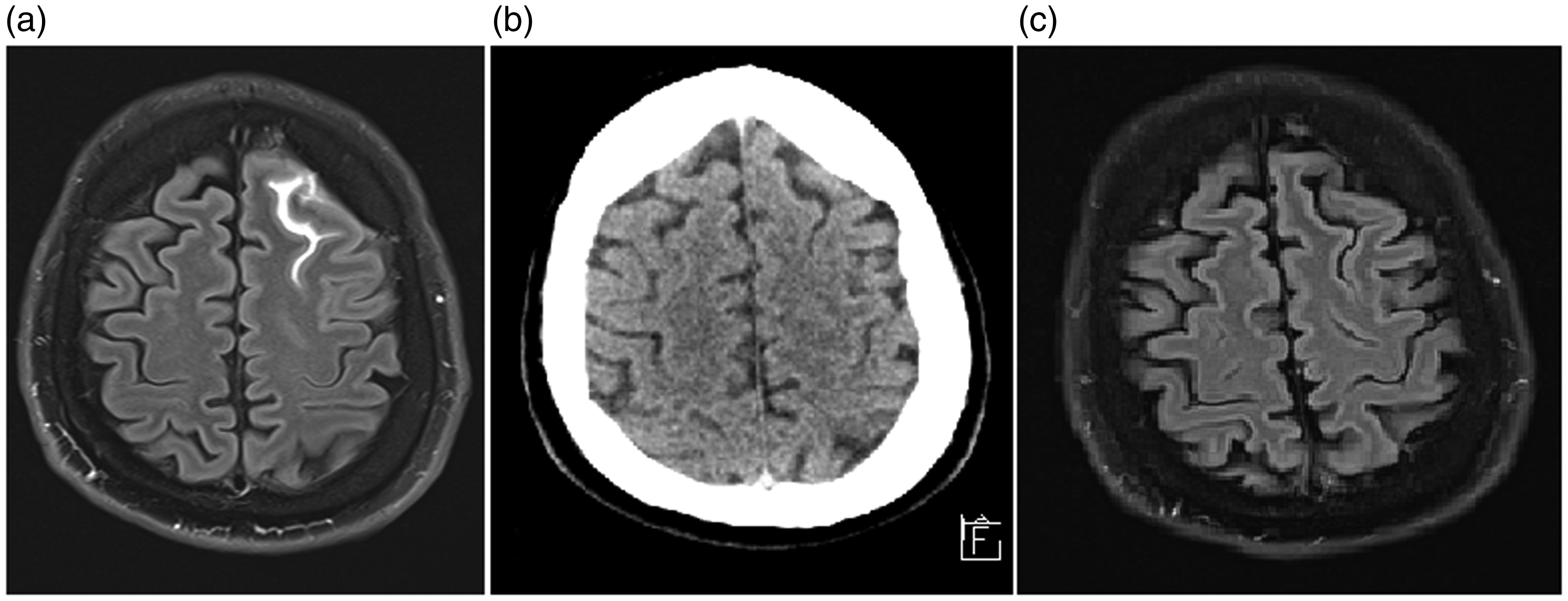
Images of a 58-year-old man examined for Meniere’s disease. (a) 3T T2 fluid-attenuated inversion recovery (FLAIR) sequence (axial plane, 3-mm-thick slices) obtained 4 hours after intravenous administration of 0.2 mmol/kg of gadobutrol shows enhancement of the subarachnoid space of the left frontal sulcus. (b) Unenhanced axial computed tomography scan of the brain rules out fresh subarachnoid bleeding. (c) Unenhanced 1.5T T2 FLAIR sequence (axial plane, 5-mm-thick slice) at the 2-week follow-up examination shows no signs of enhancement in the aforementioned area.
The patient, a 58-year-old man, had no symptoms other than the underlying chronic problems (vertigo, tinnitus) for which he was being examined (the patient did not have EH). Nevertheless, we ruled out fresh SA bleeding with an unenhanced computed tomography scan of the patient’s head (Figure 1(b)). An unenhanced MRI follow-up examination after 2 weeks showed normal findings (Figure 1(c)). By retrospective analysis of the MRI examination findings of other patients, we found another eight patients in whom SA enhancement was present to a lesser degree (short parts of the sulci, parasellar space, and Meckel’s cave) (Figure 2). Unfortunately, we did not acquire unenhanced T2 FLAIR scans to exclude possible pulsation or flow artifacts. The mean age of these patients was 69.67 years (range, 50–89 years), and they comprised five women and four men. All patients had normal renal function. Seven of the patients had positive findings for EH (Figure 3). There was no evident connection between the patients in terms of age, sex, body mass index, occupation, family history, or other diseases. The remaining 58 examined patients showed no clear signs of possible SA enhancement.
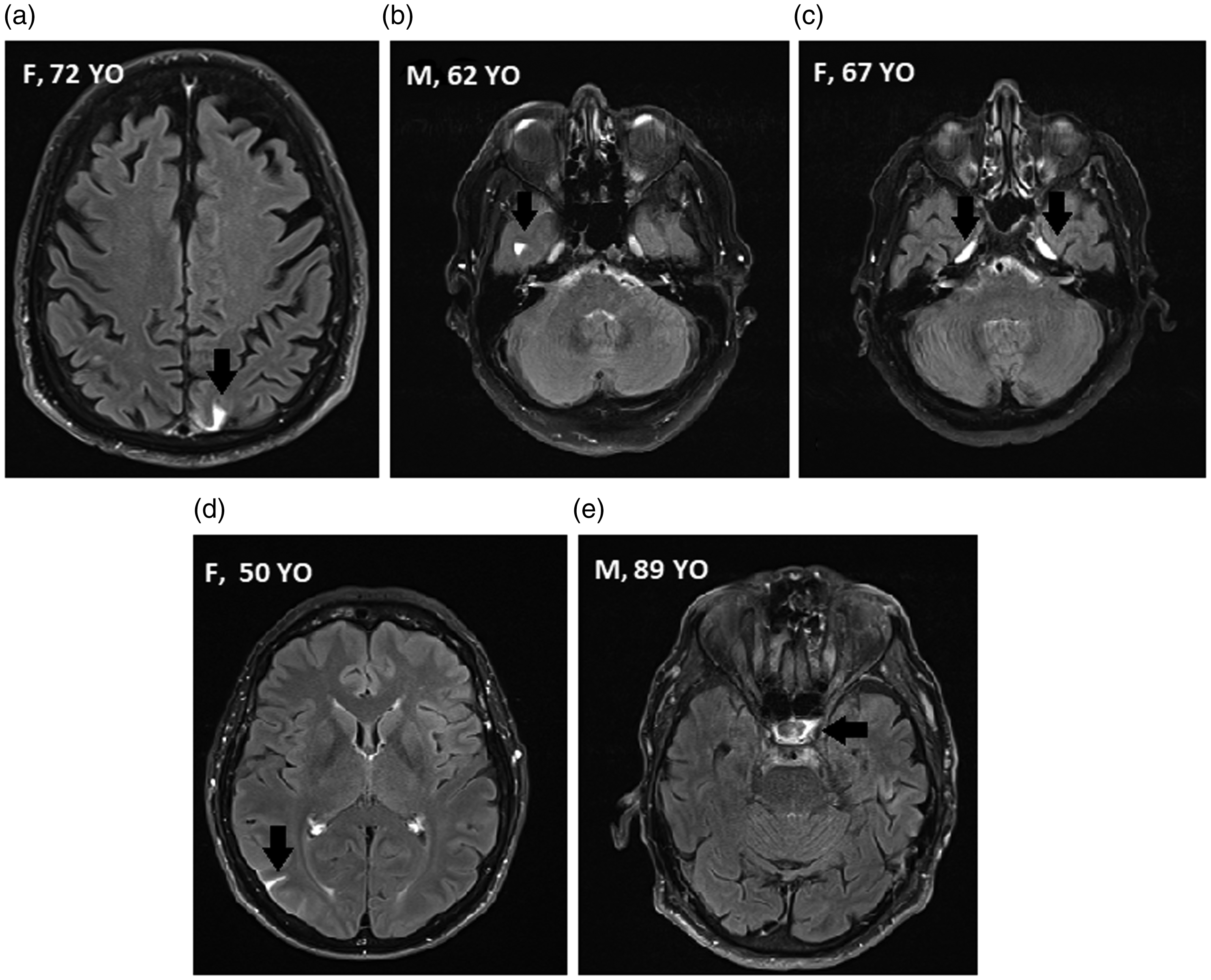
3T T2 fluid-attenuated inversion recovery (FLAIR) sequences (axial plane) obtained 4 hours after intravenous administration of 0.2 mmol/kg of gadobutrol. Retrospective examination indicates possible subarachnoid enhancement in these patients examined for Meniere’s disease, although the enhancement was not as extensive (arrows). All of these patients were diagnosed with endolymphatic hydrops. M, male; F, female; YO, years old. (a, b, d) Short parts of sulci. (c) Meckel’s cave (c). (e) Parasellar region.

3T T2 three-dimensional fluid-attenuated inversion recovery (3D-FLAIR) sequences of left inner ears (axial plane, submillimeter slices) obtained 4 hours after intravenous administration of 0.2 mmol/kg of gadobutrol. (a) Normal finding in the inner ear without endolymphatic hydrops. There is no dilation of the sacculus or utriculus, only small physiological defects of perilymphatic contrast filling are present (arrow). (b) Moderate endolymphatic hydrops. There is a large defect of perilymphatic contrast filling, with a thin rim of enhancement preserved peripherally (arrow).
Discussion
A study from 2016 confirmed that GCA penetrates the BCSFB and is detectable in the CSF up to 60 days after intravenous administration (maximum concentrations were detected in the first 8 hours), especially in patients with some degree of renal insufficiency. 9 In contrast, patients with normal renal function showed almost complete disappearance of the GCA after 48 hours. 9 After application of GCA at a dose of 0.1 mmol/kg and subsequent examination without delay, enhancement of the SA space is almost never visible when using conventional MRI sequences (T1-weighted, T2-weighted, FLAIR, and fat-suppressed sequences) despite the proven penetration of the CSF by GCA. The literature describes cases in which CSF enhancement occurred, but this was in pathological conditions such as a suprasellar oligodendroglioma in a patient with congenital hydrocephalus and in a case of posterior reversible encephalopathy syndrome with stage 4 chronic renal disease. It should be noted that a double dose of GCA (0.2 mmol/kg) was administered in these cases. 10
MRI for EH is grounded in the fact that GCA enters the perilymph through the permeable barrier between the blood and the labyrinthine space and does not cross the Reissner and basilar membranes that define the endolymphatic space. The endolymphatic system is then indirectly imaged by the enhanced surrounding perilymph. A 1992 study showed that in guinea pigs, the permeability of the blood–perilymph barrier was 3-fold lower than the permeability of the BCSFB. 11 The authors of that study used the cation of trimethylphenylammonium (the molecular weight of which is roughly 4- to 8-fold lower than the molecular weight of conventional GCA) to compare the permeability of the barriers. 11 The most sensitive agent for imaging the perilymphatic system is the heavily weighted T2 3D-FLAIR sequence, which allows selective nulling of the endolymphatic space by selecting an inversion time such that there is no net transverse magnetization of endolymph at equilibrium. This technique produces an image in which the hypointense endolymphatic space can be differentiated from the hyperintense perilymph fluid, but not from the surrounding bone, which shows a signal intensity similar to that of endolymphatic fluid on this sequence.3,12,13
GCA penetrates the BCSFB, but enhancement can be seen in pathological conditions that increase BCSFB permeability such as meningitis. The aforementioned 2016 study, in which the concentration of GCA in CSF was evaluated, showed that the maximum concentration is attained in the first 8 hours, and the average time of CSF sampling was 4.6 hours after intravenous administration. 9 This finding may explain why CSF enhancement occurs after 4 hours.
Some studies have shown that abnormalities of CSF pressure (usually in patients with hydrocephalus) may cause pathologies involving the inner ear fluids, such as the perilymph and endolymph. These pathologies are attributed to direct transmission of the pressure. 14 Additionally, glaucoma is sometimes associated with hydrocephalus. 15 Changes in the pressure within the SA space, inner ear fluid compartments, and anterior eye segment sometimes occur together, and enhancement of these cavities is codependent.3,13 Furthermore, the clinical symptoms of high intracranial pressure and EH overlap. 16 This suggests that there are strong connections among these fluid compartments that should be more thoroughly studied. None of our patients had signs of intracranial hypertension or hydrocephalus. The patient with vast SA enhancement did not even have MRI signs of EH, but the other seven patients with a lesser degree of SA enhancement were diagnosed with EH.
There is a theory suggesting that the CSF flows through so-called glymphatic pathways. According to this theory, the CSF courses perivascularly and centripetally around arteries and through aquaporin-4 channels into the interstitial space, from which it then flows to the perivascular space around veins and into the bloodstream. Studies suggest that clearance of glymphatic pathways becomes delayed with advancing age.17,18 This phenomenon might have played a role in our series.
In conclusion, vast enhancement of the SA space during an MRI examination is not common; such enhancement is usually seen when the BCSFB is affected, such as in patients with meningitis. Our case report indicates that SA enhancement might be an incidental finding in patients with no evident effect on the meninges during the use of a relatively new MRI technique for EH in which the inner ear is examined 4 hours after intravenous administration of GCA, usually at a double dose.
Footnotes
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards. Ethical approval was not needed for this type of article (case report); according to the institutional review board, such articles do not meet the criteria for research. Formal consent was not required because of the nature of this study (retrospective case report).
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Funding
This work was supported by MH CZ-DRO (UHHK, 00179906).
