Abstract
Importance
Vestibular migraine (VM) is diagnosed using limited consensus-based criteria and presents with symptoms that overlap those of other vestibular disorders. Accurate diagnosis of VM is important to facilitate adequate and timely symptom management as well as to avoid mismanagement.
Objective
To characterize ocular findings during vestibular testing in adults with VM.
Design
Systematic review.
Setting
This systematic review was completed following the Preferred Reporting Items for Systematic Reviews and Meta-Analyses guidelines. Database searches of Embase, MEDLINE, and Cochrane Library were conducted.
Participants
Studies evaluating participants with a diagnosis of definite or probable VM and no concomitant vestibular disorders were included.
Intervention or Exposures
Positioning tests, positional tests, caloric testing, and video head impulse testing (vHIT).
Main Outcome Measures
Positioning and positional testing of nystagmus, caloric testing for unilateral weakness, as well as vHIT gain and saccades.
Results
This review included 31 studies. Positioning testing nystagmus was present in 2% to 100% of patients during Dix-Hallpike (DH) and in 7.9% to 37.5% of patients during the supine head roll testing (SHRT). Nystagmus direction, when present during DH and SHRT, included horizontal, vertical, and torsional nystagmus. Positional testing nystagmus was present in 0% to 100% of patients during upright positional testing and in 42.1% to 95.5% of patients during supine positional testing. Horizontal, vertical, and torsional nystagmus in upright positional testing and direction-changing nystagmus in supine positional testing were observed. Unilateral weakness was present in 0% to 100% of patients; however, most studies reported a rate of <50% of unilateral weakness in tested patients. During vHIT, both abnormal gain and corrective saccades were observed, with the latter being more common.
Conclusion and Relevance
VM can present with ocular movement patterns seen in both central nervous system disorders and peripheral vestibular system disorders on vestibular testing. Future studies should focus on further describing the characteristics of nystagmus and associated symptoms experienced during vestibular testing in VM.

Keywords
Key Message
- During vestibular testing, patients with vestibular migraine can present with ocular movements similar to those seen in central nervous system and peripheral vestibular system disorders
- In suspected vestibular system disorders, ocular movements seen in central nervous system disorders should not be dismissed, as they may point to a vestibular migraine diagnosis
Introduction
Vestibular Migraine (VM) is the most common cause of dizziness seen in tertiary dizziness clinics, with a prevalence of 1% to 2.7% and a female preponderance.1,2 It is a condition characterized by migraine headaches and vestibular symptoms.1,2
VM symptoms are not specific to a diagnosis of VM. Commonly associated vestibular symptoms are spontaneous vertigo or vertigo triggered by either positional changes or head movements, which mimic Benign Paroxysmal Positional Vertigo (BPPV).2 -4 In addition, VM can also present with otologic symptoms, such as aural fullness, tinnitus, and sensorineural hearing loss that overlap with those of Meniere’s disease (MD). 5 Misdiagnosing VM as MD can lead to unnecessary and irreversible ablative audio-vestibular treatments.
VM is a clinical diagnosis with criteria, for its episodic variant, created by the International Headache Society and Bárány Society. 6 However, these criteria fail to capture VM symptoms occurring outside of distinct “episodes” or migraine-related dizziness without distinct vestibular events. In addition, the temporal relationship between vestibular symptoms and migraine headaches in VM is highly variable, creating diagnostic uncertainty regarding whether symptoms represent separate, distinct diseases or are a manifestation of a single disease. 7
Disease symptom variability and clinical features that overlap with other vestibular disorders underscore the importance of identifying vestibular testing findings more specific to a diagnosis of VM. Due to the neurological link between the vestibular system and the movements of the eyes, ocular movements have served an essential role in diagnosing vestibular conditions. 8
Therefore, the primary objective of this study was to review and summarize the current literature on the ocular findings during positioning and positional testing in patients with VM. The secondary objective was to summarize the results of caloric testing and video head impulse testing (vHIT) in patients with VM.
Methods
The review follows the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) reporting guidelines. 9 The review was registered on the Prospective Register of Systematic Reviews (PROSPERO, CRD42024568274) on July 28, 2024. This study did not involve human participants; therefore, Ethics Committee approval was not required.
Database Search
A search model was developed in collaboration with an academic medical research librarian. Electronic searches were conducted on Embase, MEDLINE, and Cochrane Library from database inception to May 2024. Gray literature and citations searches were also performed. Searches were centered around the terms: VM, positioning testing, positional testing, caloric testing, vHIT, nystagmus, and saccades.
Study Selection and Data Extraction
Primary literature with adult patients (≥18 years) diagnosed with definite or probable VM according to criteria by Lempert et al, 6 or with complaints of migrainous vertigo, and reporting results from positioning testing, positional testing, vHIT, or caloric testing, were included. Studies were excluded if patients had reported concomitant vestibular disorders (BPPV, MD, acoustic neuromas, superior semicircular canal (SCC) dehiscence, perilymph fistula, inflammation of the middle or inner ear).
Retrieved studies were uploaded to Covidence, 10 and duplicates were removed. Two reviewers (R.M. and J.S.) individually performed title and abstract screening, followed by full-text screening. Discrepancies were discussed, and consensus was reached after each round of screening. Both reviewers individually performed data extraction, and discrepancies were resolved by team consensus. The following data points were extracted: author; year; study design; number of and age of sample; criteria for diagnosing VM; period of testing (ictal vs inter-ictal); definitions of abnormal vestibular testing results; and vestibular testing results.
Risk of Bias Assessment
Risk of bias assessment was performed using the Newcastle-Ottawa Scale (NOS) for cohort and case-control studies. 11 Cross-sectional studies were assessed using an adapted version of the NOS presented by Herzog et al. 12 Assessments were graded based on scales from Kim et al 13 and Langan et al 14 for cross-sectional and cohort/case-control studies, respectively. Case reports were assessed using the Joanna Briggs Institute critical appraisal tool. 15 Assessments were graded based on which response was most reported (Yes: Low RoB; Unclear: Moderate RoB; No: High RoB). Both reviewers independently performed risk of bias assessment, and discrepancies were resolved through team consensus.
Statistical Analysis
Descriptive statistics, such as the measures of central tendency (mean), were collected from the outcomes of interest in the included studies. Narrative synthesis was performed. Ranges of abnormal vestibular testing results are reported in this review. Subgroup analyses comparing ictal and inter-ictal testing results were performed.
Results
Baseline Characteristics of Included Studies
A total of 751 studies were identified from database searches, and 31 were included in the review.16 -46 The selection process and reasons for exclusion are detailed in Figure 1. Study characteristics are summarized in Table 1. The included studies reported on 1510 patients diagnosed with VM, probable VM, or migranous vertigo with a range of 1 to 226 patients per study and duration of symptoms ranging from less than a year up to 17.6 years. Control groups, when present, were small and ranged from 14 to 78 patients. After risk of bias assessment, 21 studies were found to be at a high risk of bias, 4 studies at a moderate risk of bias, and 6 at a low risk of bias. Cross-sectional studies accounted for most of the high-risk-of-bias studies (20/21) because sex and other demographic factors were not controlled for in these studies.
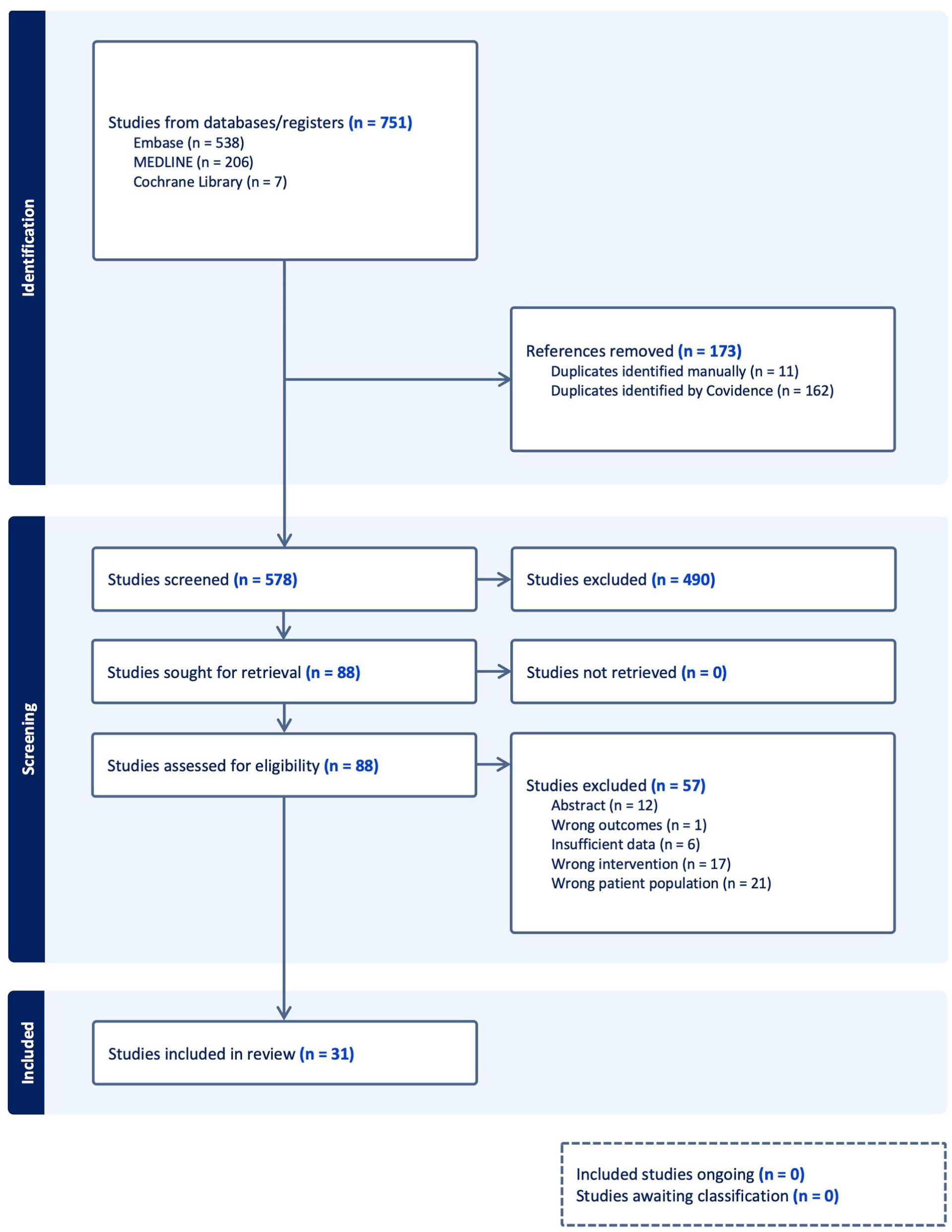
PRISMA flow diagram.
Summary of Studies Included in the Systematic Review.

Derived from age of migraine of vertigo onset and mean age.
Data presented as aggregates that include other groups of patients.
Positioning Testing
Of the included studies, 8 reported ocular findings during positioning testing, with two24,38 reporting ictal results, four19,42,43,46 reporting inter-ictal results, one 33 reporting both ictal and inter-ictal results, and one 17 not specifying when testing was performed. Positioning testing was performed on 519 patients. Supplemental Tables 2 and 3 contain detailed positioning testing data.
Positioning nystagmus was present in 15% to 100% of patients (total n = 91; 3 studies) tested during active vestibular symptoms (ictal period). When reported, the direction of the nystagmus during ictal positioning testing was variable, with nystagmus during Dix-Hallpike (DH) testing including up-beating and torsional nystagmus as well as down-beating nystagmus and horizontal nystagmus. Supine Head Roll Testing (SHRT) nystagmus directions included horizontal nystagmus (both geotropic and apogeotropic) as well as vertical and torsional nystagmus. Duration, latency, and symptoms experienced during testing were not reported.
In the periods between episodes of active vestibular symptoms (inter-ictal), abnormal ocular movements were reported in 2% to 80% of tested patients (total n = 391; 5 studies). Nystagmus direction was only reported by Li et al 33 where both geotropic and apogeotropic nystagmus were reported during SHRT and horizontal nystagmus during DH (n = 50). Duration, latency, and presence or absence of symptoms during testing were not reported. Li et al 33 tested patients both during the ictal (n = 50) and inter-ictal (n = 40) periods, demonstrating slightly higher frequency of abnormal positioning testing in the ictal period as compared to the inter-ictal period (30% and 37.5% positive findings on SHRT and DH, respectively, during ictal periods as compared to 22% and 2% positive findings on SHRT and DH, respectively, during inter-ictal periods).
Manual sub-group analysis (where possible) demonstrated that abnormal nystagmus findings during positioning testing were more common during the ictal period (77%) as compared to the inter-ictal period (13%). Furthermore, DH testing was more likely to have abnormal findings than SHRT (60% vs 38% in ictal periods and 27% vs 16% in inter-ictal periods).
Positional Testing
Ocular findings during positional testing were reported in fifteen of the included studies, with four20,25,34,38 reporting ictal results, six19,26,41 -43,46 reporting inter-ictal results, two33,45 reporting both ictal and inter-ictal results, and three27,28,32 not specifying when testing was performed. Positional testing was performed on 999 patients. Supplemental Tables 2 and 3 contain detailed positional testing data.
All 6 studies reporting positional testing findings during the ictal period tested patients in the upright position. Nystagmus was present in 5% to 100% of patients in the upright position during an ictal period. Direction of observed nystagmus, reported in 5 of these 6 studies, included both horizontal and vertical nystagmus.
Interestingly, Young et al 45 reported findings over multiple ictal periods for some patients. Although the majority were found to have nystagmus that did not change direction between recordings, 5 had spontaneous nystagmus in the upright position that changed direction between ictal periods (n = 4) or even within the same ictal period (n = 1).
Young et al 45 reported on ictal supine positional testing. In this study, 95.5% (85/89) of patients demonstrated nystagmus in at least one of the tested positions and, in all cases, nystagmus persisted throughout maintenance of the position. Most patients (62/85) who had nystagmus in one of the supine positions also had nystagmus in the upright position, whereas the remaining 23/85 demonstrated nystagmus in at least one of the supine positions in the absence of spontaneous nystagmus in the upright position. The supine positional nystagmus, when present, was direction-changing when lying on either side in 15.7% (14/89; geotropic in 10 and apogeotropic in 4). The remaining 71 patients who demonstrated positional nystagmus had non-direction-changing nystagmus, though the direction was not specified in the report.
Among the 8 studies reporting on inter-ictal positional testing, all reported on the frequency of nystagmus in the upright position, whereas only 2 reported on supine positional nystagmus. In the inter-ictal period, 0% to 32.3% of patients had nystagmus in the upright position. Supine position nystagmus during the inter-ictal period was present in 3.6% to 67.5% of tested patients. Reported directions of nystagmus in these positions included horizontal, vertical, torsional, and direction-changing nystagmus in lateral supine positions.
Manual sub-group analysis (where possible) demonstrated that abnormal nystagmus findings during positional testing were more common in the supine position compared to upright, and ictal periods compared to interictal (supine vs upright: 96% vs 41% during ictal periods, 46% vs 12% inter-ictal periods). Vivek et al 42 were excluded from this subgroup analysis due to the inability to extrapolate the total numbers of positive findings.
Caloric Testing
Of the included studies, eighteen reported ocular findings during caloric testing, with one 34 reporting ictal results, eight19,30,39,41,43 -46 reporting inter-ictal results, one 33 reporting both ictal and inter-ictal results, and eight17,18,27,28,32,35,36,40 not specifying when caloric testing was done. Caloric testing was performed in 776 patients. Supplemental Table 4 contains further caloric testing details.
The reported definition of unilateral weakness ranged from 20% to 35% unilateral weakness among studies, although 5 studies did not specify their definition of unilateral caloric weakness, and 1 study defined slow phase velocity (SPV) of <10 deg/s as abnormal, without further specification. Abnormal caloric tests were seen in 0% to 100% of patients. If we exclude the 1 case report that reported 100% presence of caloric weakness (n = 1), the remaining studies all reported less than 50% rate of caloric weakness in patients.
Interestingly, Li et al 33 reported bilateral caloric (lateral canal) weakness in 3 of 6 patients who were tested during the inter-ictal period. Woźniak et al 43 found that abnormal caloric testing results were nearly twice as common in patients with recurrent symptoms for more than 10 years (48% [n = 42]) compared to those whose symptoms had been present for less than 1 year (24% [n = 42]).
Manual sub-group analysis (where possible) demonstrated that unilateral caloric weakness was more likely to be present if patients were tested during the ictal period rather than the inter-ictal period (32% vs 15%). Overall, 26% of patients with VM in the included studies showed the presence of unilateral caloric weakness.
Video Head Impulse Testing
Ocular findings during vHIT were reported in 19 studies, with four20,25,31,34 reporting ictal results, eight16,21 -23,26,29,37,40,42,46 reporting inter-ictal results, one 45 reported both ictal and inter-ictal results, and six18,21,23,24,35,36 not specifying when testing was done. vHIT was performed on 834 patients. Supplemental Table 5 contains further vHIT details from the included studies.
Gain (ratio of eye movement velocity to head movement velocity), as well as the presence of corrective saccades (involuntary refixation eye movements occurring after rapid head movement), were reported. Reported criteria for abnormal gain ranged from <.64 to <.86, and some studies reported abnormal gain as ≤2 standard deviations from that of age-matched controls. Seven studies did not indicate their criteria for abnormal gain.16,22,23,26,29,34,42
The presence of abnormal vHIT gain varied widely among the 19 studies, from 0% to 100%, with 5 of the studies having reported no cases of abnormal gain among tested patients. Fifteen studies reported the presence or absence of corrective saccades with vHIT testing and displayed varying results, once again, ranging from 0% to 100% of patients demonstrating corrective saccades. Two studies reported no corrective saccades at all among their patients.
Manual sub-group analysis (where possible) demonstrated that patients were more likely to demonstrate low gain and corrective saccades when tested in the ictal period as opposed to the inter-ictal period (48% and 44% during the ictal period as compared to 5% and 16% during the inter-ictal period). Overall, among tested patients, 18% had abnormally low vHIT gains and 30% had corrective saccades.
Thirteen studies reported on both abnormal gain and corrective saccades in their patients. Among these, 5 studies (n = 81) showed that abnormal gain occurred at the same rate as corrective saccades. Six studies (n = 403) showed that corrective saccades occurred more frequently than did abnormal gain (n = 102 patients; 25% vs n = 41; 10%, respectively). Finally, 2 studies (n = 126) showed that abnormal gain occurred more frequently than did corrective saccades (n = 68 patients; 54% vs n = 34; 27%).
Discussion
Patients with VM can display ocular movements consistent with both central nervous system disorders and peripheral vestibular system disorders during positioning testing, positional testing, caloric testing, and vHIT.
Positioning and Positional Testing
Positioning tests such as DH and SHRT are typically used to identify BPPV. During positioning testing, rapid head movement is utilized to evoke nystagmus and dizziness. BPPV is confirmed by identifying nystagmus in a direction appropriate for the tested SCC, such as torsional up-beating nystagmus in posterior SCC BPPV. In this review, nystagmus was observed in VM patients during both ictal and inter-ictal positioning testing, with a higher prevalence of up to 77% during the ictal period.
Within the limitations of this review, some patients displayed nystagmus that could be consistent with BPPV during positioning testing; however, this was not consistently the case. There were notable reports of horizontal nystagmus during DH testing as well as vertical and torsional nystagmus during SHRT, which are not consistent with BPPV for the SCCs tested during these maneuvers. Although BPPV is recognized to be more prevalent in VM patients than the general population, 47 these results suggest that positioning testing nystagmus in VM patients may not always be due to BPPV. Importantly, some of the nystagmus directions observed resemble those associated with central nervous system disorders. Macdonald et al 48 identified atypical directions of nystagmus during DH in 97.5% of patients with brainstem or cerebellar lesions, and horizontal nystagmus was most observed in 36.8% (n = 82) of patients. Therefore, when positioning testing nystagmus is not consistent with the tested SCC, such as when nystagmus direction aligns with that seen in central nervous system disorders, a diagnosis of VM should be considered.
During positional testing, rapid head movement is not the evoking stimulus; instead, it is the final position assumed and maintained that elicits symptoms and nystagmus. The positions tested most often include the upright position (assessing for spontaneous nystagmus) and supine positions. In this review, VM patients were found to have the highest rate of abnormal positional nystagmus findings in the supine position when tested during the ictal period, where (>95% of tested patients demonstrated abnormal positional nystagmus). Positional nystagmus was also identified in the supine position during the inter-ictal period, albeit less frequently than during active symptoms. Finally, spontaneous nystagmus in the upright position can also be detected in VM patients. Just as in supine testing, spontaneous nystagmus is more likely to be detected in VM patients during the ictal period (41%) rather than the inter-ictal period (12%). Within the limitations of data reported by authors of included studies, we note that VM patients displayed horizontal as well as vertical nystagmus during upright positional testing. Torsional nystagmus was also reported; however, this was only in 1 case report. 34 Spontaneous nystagmus, when vertical or torsional, is not consistent with peripheral vestibular pathology. 49 With these findings during positional testing, a central cause of nystagmus, including VM, should be considered.
In addition, nystagmus that changes direction in a single head position is not consistent with peripheral vestibular pathology. Janiak-Kiszka et al 28 and Young et al, 45 both reported direction-changing nystagmus in 1 head position during supine positional testing during 1 episode and between multiple episodes of VM. It should be noted that supine positional testing can provoke lateral SCC BPPV, and that differentiating this from central positional nystagmus in these cases was not possible with the available data. However, direction-changing in a single head position or non-direction-changing between lateral lying sides is not consistent with BPPV. Therefore, based on the available information, we can interpret that during positional testing, VM patients can present with spontaneous nystagmus patterns seen in central nervous system disorders or peripheral vestibular system disorders, such as BPPV. Detailed observation of the nystagmus direction in addition to latency, duration, presence of symptoms, and pattern of intensity over time must be noted in cases that appear peripheral to differentiate BPPV from non-BPPV positional and positioning nystagmus that can be seen in VM.
Caloric Testing
The caloric test has been considered the gold-standard for evaluating unilateral vestibulopathies such as MD. 50 However, this review demonstrates that VM may also be associated with unilateral weakness during caloric testing, as was reported in up to 48.1% of patients by Zhang et al 46 These findings are similar to those of a recent retrospective cohort study involving 2101 patients (released after the screening and data collection periods of this systematic review), which identified caloric testing unilateral weakness in 20.9% of VM patients. 51 In addition, in cases of MD, caloric testing results show considerable fluctuation based on when testing is done. 52 Therefore, despite the unknown pathophysiology of vestibular weakness in VM, patients with VM may present with unilateral weakness, classically thought to only be seen in peripheral vestibular system disorders. Caution should be taken in using caloric weakness to distinguish VM from MD.
Video Head Impulse Testing
The vHIT assesses the vestibulo-ocular reflex, where reduced gain indicates diminished transmission of head movement amplitude to the extraocular eye muscles, resulting in inadequate eye movement velocity and a need for corrective saccades to refix gaze. 53 In this review, both reduced gain and corrective saccades were observed in patients with VM. In some cases, these occurred together, as can be seen in peripheral vestibular dysfunction, while in others, there was discordance between low gain and corrective saccades. Preserved gain with corrective saccades has been observed in central nervous system disorders. Nam et al 54 found that patients with stroke-related cerebellar disorder were more likely to exhibit corrective saccades with normal gain than patients with isolated vestibular neuritis. The vHIT findings in this review demonstrate that VM can present with vHIT findings consistent with both peripheral vestibular and central nervous system disorders.
Limitations
Limitations of this review include limited reporting of nystagmus characteristics in the literature (duration, latency, response to head movements), methodology heterogeneity (difference in positioning and positional tests performed), inclusion of case reports, small sample sizes, and poor delineation of ictal and inter-ictal period testing. Case reports were included due to limited literature on this topic; however, present a high risk of selection bias and have poor generalizability. Consistent definitions of ictal and inter-ictal periods of testing are clinically important because certain findings may be specific to when vestibular testing is performed. Overall, these limitations underscore the need for standardized testing and reporting protocols to ensure reliable and comparable data in future VM research.
Conclusion
VM can present with ocular findings characteristic of both central nervous system and peripheral vestibular system disorders during positional testing, positioning testing, caloric testing, and vHIT. In patients presenting with a clinical history suggestive of VM, with either central or peripheral patterns of ocular movements during vestibular testing, a diagnosis of VM should be considered. Future studies reporting consistent and comprehensive vestibular testing data on VM patients are needed to validate the results observed in this study.
Supplemental Material
sj-docx-1-ohn-10.1177_19160216261429361 – Supplemental material for Ocular Findings During Vestibular Testing in Vestibular Migraine: Systematic Review
Supplemental material, sj-docx-1-ohn-10.1177_19160216261429361 for Ocular Findings During Vestibular Testing in Vestibular Migraine: Systematic Review by Margaret Aron, Richard Mageto, Jayant Seth, Kevan Lu and Brian D. Westerberg in Journal of Otolaryngology - Head & Neck Surgery
Footnotes
Acknowledgements
The authors would like to acknowledge Jane Jun of the University of British Columbia, Southern Medical Program Library Services, for assistance with database searches.
Author Contributions
RM and JS were involved in study preparation, data collection, data analysis, and manuscript preparation. BDW and KL were involved in the study and manuscript preparation. MA was involved in study preparation, data analysis, and manuscript preparation.
Data Availability Statement
The dataset used and analyzed during this study is available from the corresponding author upon request.*
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethical Approval and Informed Consent
This study was exempt from ethics approval, and informed consent was not required based on the methodology and publicly available data.
Supplemental Material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
