Abstract
Importance
Proton pump inhibitors (PPIs) do not demonstrate superiority over placebo in laryngopharyngeal reflux disease (LPRD). While poorly used, many alternative medical treatments exist for controlling the gastroduodenal and gastro-pharyngeal reflux processes.
Objective
To investigate the clinical findings of controlled studies comparing therapeutic regimens for treating LPRD.
Design
PubMED, Scopus, and Cochrane Library systematic review without meta-analysis using the PRISMA statements.
Setting
Review of prospective or retrospective cohort studies comparing 2 medical regimens in patients with suspected or confirmed LPRD.
Participants
Patients with suspected or confirmed LPRD.
Intervention
Comparison of PPI therapy to other therapeutic regimens for LPRD.
Outcomes
Symptom, sign changes, and therapeutic responses were considered from pre- to posttreatment. Bias analysis was conducted with Methodological Index for Non-Randomized Studies (MINORS). Implications for practice were summarized with a focus on the pharmacological and biological findings of all drug classes in the LPRD pathophysiology.
Results
Fourteen studies met the inclusion criteria, including 1269 patients. There were 675 females (59.6%) and 458 males (40.4%). The mean age of patients was 49.1 years. Substantial variability was observed among studies regarding both pharmacological interventions and participant selection criteria. Two of the four comparative studies demonstrated superior therapeutic efficacy with prokinetic-PPI combination therapy compared with PPI monotherapy. The alginate-PPI combination yielded enhanced symptom amelioration compared with PPI alone.
The mean MINORS score of 9.1 ± 1.2 indicated an important heterogeneity between randomized and non-randomized controlled studies for inclusion and exclusion criteria, LPRD diagnosis, therapeutic regimens, and outcomes. Recommendations are provided for future randomized controlled studies.
Conclusion
To date, only a few studies investigated the effectiveness of alternative medications to PPIs in the treatment for LPRD, which represents an important gap with the gastroesophageal reflux disease literature. Although the limited number of studies and their heterogeneity preclude definitive conclusions, the preliminary findings from this review support the need for future randomized controlled trials examining the therapeutic potential of alginates and prokinetics in patients with primary or refractory LPRD.

Key Messages
The number of studies comparing proton pump inhibitors (PPIs) with other medication is still limited.
Prokinetic-PPI combination therapy should be superior to PPI monotherapy, while the alginate-PPI combination yielded enhanced symptom amelioration compared with PPI alone.
Future randomized controlled trials examining the therapeutic potential of alginates and prokinetics in patients with primary or refractory laryngopharyngeal reflux disease are needed.
Introduction
Laryngopharyngeal reflux disease (LPRD) is defined as a disease of the upper aerodigestive tract resulting from the direct and/or indirect effects of gastroduodenal content reflux, inducing morphological and/or neurological changes in the upper aerodigestive tract. 1 The diagnosis of LPRD consists of the identification of more than one pharyngeal acidic, weakly-acidic, or alkaline reflux events at the 24 hour hypopharyngeal-esophageal multichannel intraluminal impedance-pH testing (HEMII-pH), while gastroesophageal reflux disease (GERD) diagnosis is based on Lyon criteria and acidic exposure in low esophagitis or related complications. 1 Nowadays, the primary treatment for LPRD includes diet and lifestyle recommendations, and the prescription of once-daily– or twice-daily–proton pump inhibitors (PPIs).2,3 However, the treatment for LPRD remains challenging with moderate effectiveness of PPIs, 4 ensuring symptom relief in approximately 50% of cases. 4 Moreover, the last meta-analyses reported that PPIs do not still demonstrate superiority over placebo. 3 The limited efficacy of PPIs in LPRD can be attributed to fundamental pathophysiological distinctions between GERD and LPRD. While GERD is characterized primarily by acidic liquid reflux, typically defined by greater than 6% acid exposure time in the distal esophagus—leading to complications such as esophagitis, stricture formation, and potential Barrett’s metaplasia—the pathophysiology of LPRD is more complex. In GERD, PPI therapy makes sense as it effectively raises gastric pH, thereby reducing both acid-mediated injury and pepsin activity.1,2 However, this therapeutic approach may be insufficient for LPRD, where even mildly-acidic or nonacidic refluxate containing activated pepsin can cause significant laryngopharyngeal injury. The pathophysiology of LPRD involves the gaseous weakly-acidic and alkaline back-flowed droplets of gastroduodenal content into the upper aerodigestive tract mucosa through the transient relaxations of both lower and upper esophageal sphincters.5,6 As for GERD, many alternative medical treatments exist for controlling the gastroduodenal reflux processes and the related esophageal dysmotility, including sodium alginates, antacids, prokinetics, H2 blockers, transient lower esophageal sphincter relaxation inhibitors, or potassium- competitive acid blockers. 7
This systematic review aimed to review the controlled studies investigating the effectiveness of alternative medical treatments to PPIs in the management of LPRD.
Materials and Methods
The criteria for study inclusion were based on the population, intervention, comparison, outcome, timing, and setting (PICOTS) framework. 8 The study data were reviewed and collected regarding the PRISMA checklist for systematic reviews. 9
Types of Studies
A systematic review of the content of controlled prospective or retrospective studies published between 1990 and 2024 was conducted if the studies investigated the effectiveness of alternative treatments to PPI therapy for LPRD. The studies were published in English, Spanish, or French peer-reviewed journals. The authors only considered studies reporting data of 10 or more adult patients.
Populations, Inclusion, and Exclusion Criteria
To be included, the authors had to report the inclusion and exclusion criteria, diagnosis criteria of LPRD, treatment regimens, and therapeutic outcomes. The studies including the pediatric population were excluded. In study’s populations, the diagnosis of LPRD was confirmed if patients had more than one pharyngeal reflux event at the 24 hour HEMII-pH, which is consistent with the Dubai consensus. 1 The use of oropharyngeal pH monitoring, dual-probe or triple-probe pH monitoring with pharyngeal pH sensor can support but not confirm the diagnosis. 1 The LPRD diagnosis was suspected but not confirmed for patients included through the use of patient-reported outcomes questionnaires [eg, reflux symptom index (RSI), 10 reflux symptom score (RSS) 11 ] and validated sign instruments [eg, reflux finding score (RFS), 12 reflux sign assessment (RSA) 13 ]. Patients with LPRD symptoms completing GERD diagnosis criteria (eg, Montreal, Lyon consensus) 1 were suspected of LPRD diagnosis. In this review, patients with confirmed or suspected LPRD were included, as well as those with GERD and LPRD symptoms.
Outcomes
The following outcomes were reviewed: study design; number of patients; gender ratio; age; diagnosis criteria; therapeutic regimens; diet and lifestyle recommendations, therapeutic outcomes, and duration of treatments. The following medications were considered as an alternative therapy to PPIs: alginates, antacids, prokinetics, H2 blockers, potassium-competitive acid blockers, transient lower esophageal sphincter relaxation inhibitors (ie, GABA receptor agonists, metabotropic glutamate receptors, CB1 receptor agonists/antagonists, neuromodulators).
Intervention and Comparison
The following therapeutic comparisons were considered: PPIs versus alginate, antacids, prokinetics, or transient lower esophageal sphincter relaxation inhibitors; PPIs + an alternative medication versus transient lower esophageal sphincter relaxation inhibitors. The nonmedical therapeutic approaches were not included in the present review. Heterogeneity among included articles in LPRD diagnosis, inclusion/exclusion criteria, therapeutic regimens, diet and lifestyle recommendations, therapeutic duration, and outcomes precluded statistically pooling the data into a formal meta-analysis, thereby limiting the analysis to a qualitative rather than quantitative summary of the available information.
Timing and Setting
There were no criteria for specific timing in the “disease process” of the study population.
Search Strategy
Two investigators (J.R.L. and a librarian) conducted the PubMed, Scopus, and Cochrane Library database research for relevant peer-reviewed controlled studies related to medical therapies of LPRD. The following keywords were used “Laryngopharyngeal,” “Reflux,” “Laryngitis,” “Extra-esophageal,” “Alginate,” “Antacids,” “Proton pump inhibitors,” “Prokinetics,” “H2-blockers,” “potassium- competitive acid blockers,” “transient lower esophageal sphincter relaxation inhibitors” to identify clinical controlled prospective, and retrospective studies, reviews, and meta-analyses. The 2 investigators considered only studies with database abstracts, available full-texts, or titles containing the search terms. The findings of the research strategy were reviewed for relevance and the reference lists of these articles were examined for additional pertinent studies. Implications for practice were summarized with a focus on the pharmacological and biological effects of alternative medication in the LPRD pathophysiology. Ethics committee approval was not required.
Bias Analysis
The bias analysis was carried out with the Methodological Index for Non-Randomized Studies (MINORS) tool, 10 and the Tool to Assess Risk of Bias in Cohort Studies 11 developed by the Clarity Group and Evidence Partners. The MINORS tool includes items related to the analysis of methodological points of comparative studies. Each item was rated as 0 if absent; 1 when reported but inadequate; and 2 when reported and adequate. The aim of the study was rated as clearly stated (2), unclear (1), or absent (0). The patients were included consecutively (2), or not (1), with prospective data collection as perfectly prospective (2), retrospective analysis of prospective recruited patients (1), or absent (0). The quality of endpoints (therapeutic outcomes) was judged as high (2) when authors carefully evaluated the pretreatment to posttreatment outcomes with validated, standardized, and structured outcomes (including patient-reported outcome questionnaires or validated clinical instruments). The use of unvalidated structured outcomes was judged as incomplete (1), while the use of subjective unvalidated and nonstructured outcomes was judged as low (0). A 2 to 3 month treatment was considered as an adequate therapeutic duration (2) according to the Dubai consensus. 1 A duration of less than 2 months was less reliable (1), while a shorter duration was considered inadequate. The consideration of a <5% rate of patients who were lost to follow-up was rated as adequate (2). The last MINORS outcome evaluates the report of a study size prospective calculation (2). The ideal MINORS score was 24 for comparative studies. 10
Some items that are not evaluated in MINORS were assessed with the Tool to Assess Risk of Bias in Cohort Studies, 11 which provides an analysis of selection; detection; performance; attrition; and reporting biases. 11 The following items were evaluated: diagnosis method of LPRD (optimal with 24 hour HEMII-pH, moderate with 24 hour dual-probe or triple-probe pH monitoring, low with validated thresholds or patient-reported outcome questionnaire and clinical finding instruments, and very low with evaluation of symptoms and findings without validated and standardized instruments), and exclusion criteria of confounding factors. The evaluations of symptoms and findings met the same criteria as the evaluation of the diagnosis method. Note that the fiberscope findings need to be evaluated blindly regarding symptoms and patient features (groups). The quality of the randomized process of randomized controlled trials was evaluated in the bias analysis. 11
Results
Of the 230 identified studies, 14 studies met our inclusion criteria and data were extracted (Figure 1 and Table 1).12-25 There were 5 monocentric controlled prospective studies [evidence level (EL): IIB],12,14,15,17,18 and 9 randomized-controlled studies (IB),13,16,19,20-25 accounting for 1269 patients with suspected LPRD. There were 675 females (59.6%) and 458 males (40.4%). The mean age of patients was 49.1 years (n = 1123). The diagnosis approach and the therapeutic regimen features are reported in Table 2.

PRISMA flow chart.
Controlled Studies Evaluating Alternative Therapies to Proton Pump Inhibitors.
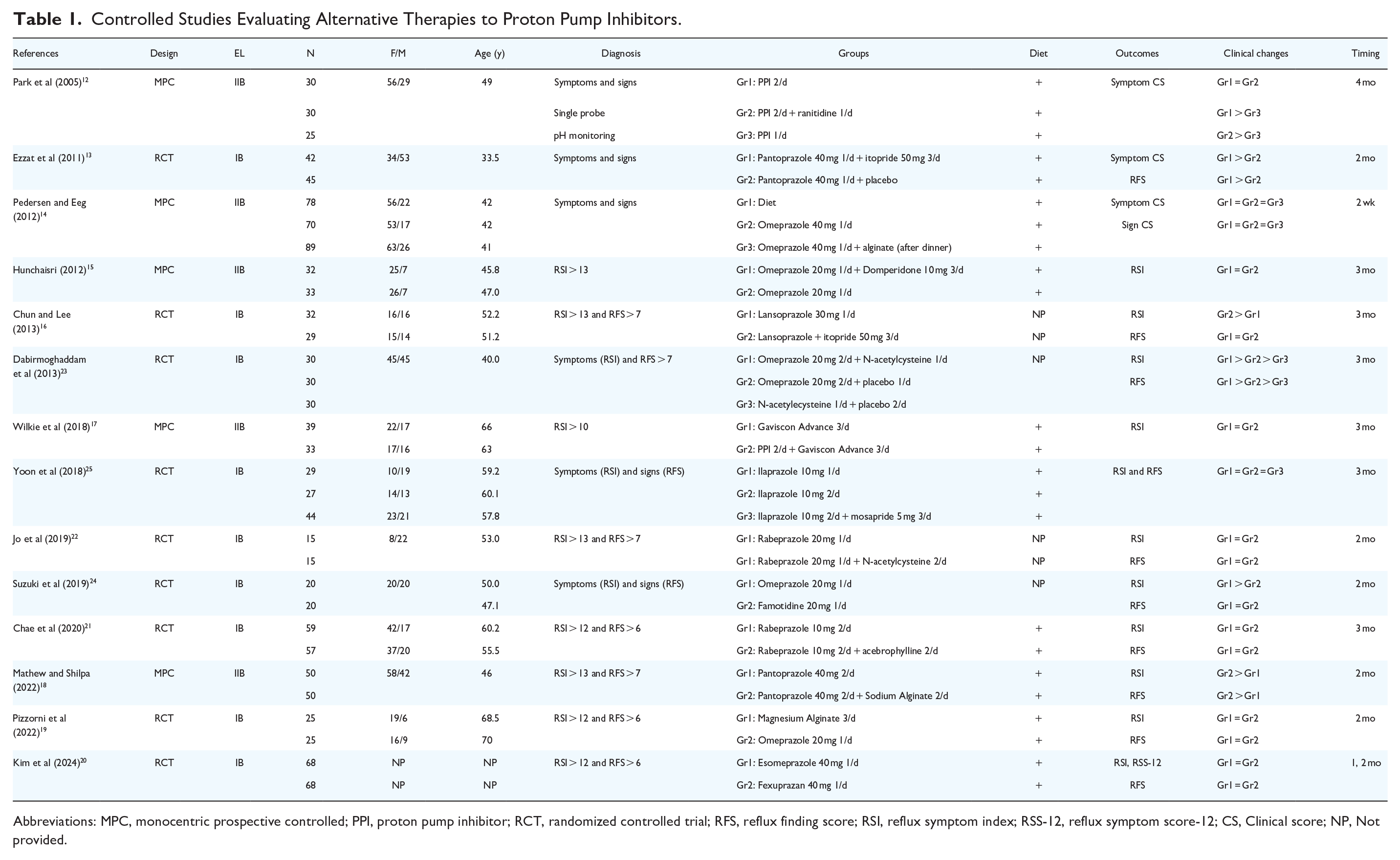
Abbreviations: MPC, monocentric prospective controlled; PPI, proton pump inhibitor; RCT, randomized controlled trial; RFS, reflux finding score; RSI, reflux symptom index; RSS-12, reflux symptom score-12; CS, Clinical score; NP, Not provided.
Diagnosis and Therapeutic Outcomes of Controlled Studies.
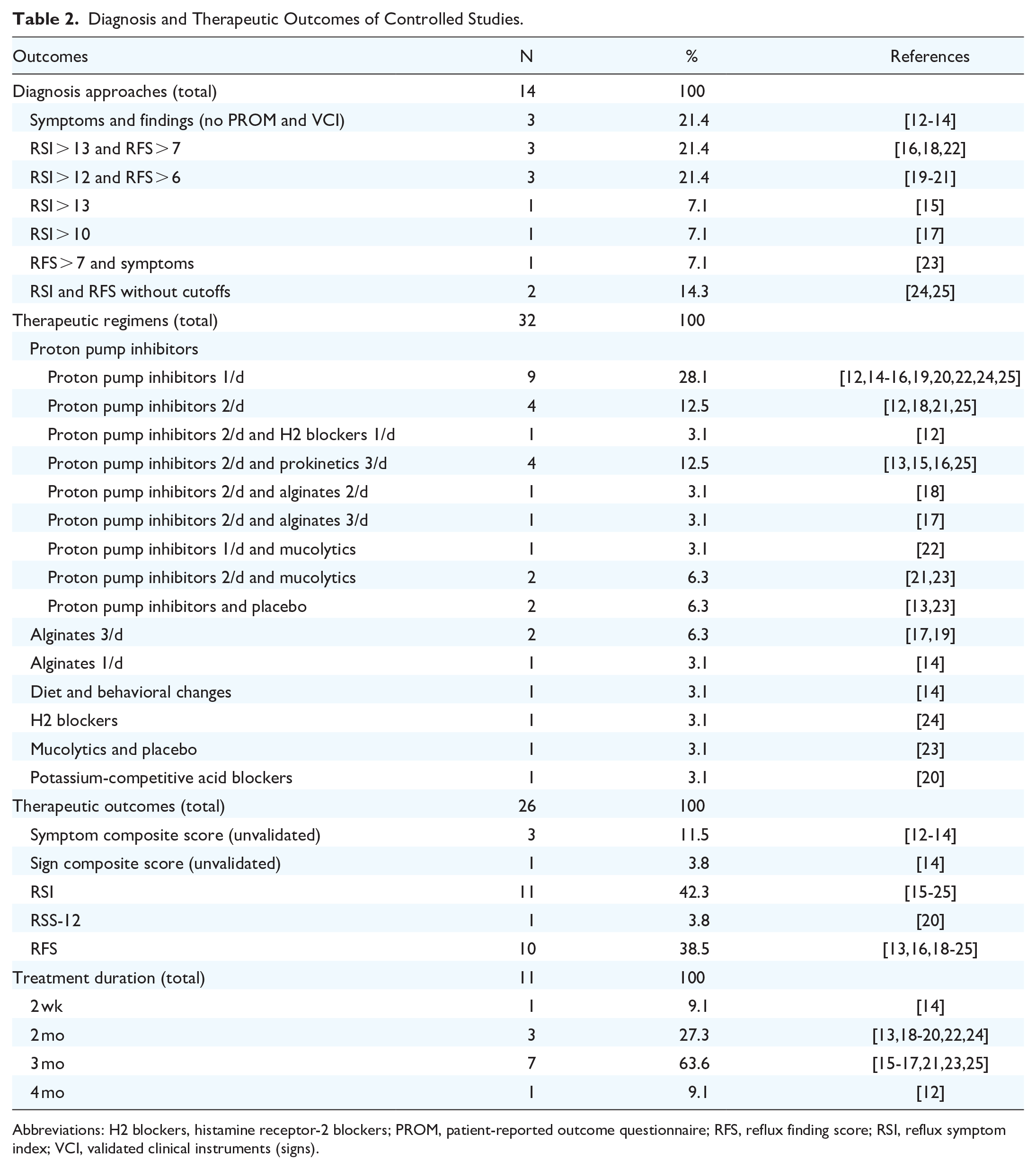
Abbreviations: H2 blockers, histamine receptor-2 blockers; PROM, patient-reported outcome questionnaire; RFS, reflux finding score; RSI, reflux symptom index; VCI, validated clinical instruments (signs).
Thirteen diagnosis methods have been used in the studies, all using symptom and/or sign evaluation. Among them, validated patient-reported outcome questionnaires (RSI and RFS) were used in 76.8% of cases. However, the initial thresholds defined by Belafsky et al for suggesting the LPRD diagnosis (RSI > 13 and RFS > 7)26,27 were used in only 2 studies.16,18,22 In twelve studies, the authors conducted a controlled study with once-daily or twice-daily PPI as the treatment for the control group (Table 2).12-16,18-22,24,25 The pretreatment to posttreatment outcomes were primarily the composite or validated patient-reported outcome questionnaires or clinical instruments used for the diagnosis (Table 2). The duration of treatment ranged from 2 weeks to 4 months (Table 2). Four studies did not detail the diet and lifestyle recommendations for patients.16,22-24
Benefits of Prokinetics
Prokinetics enhance gastric motility and accelerate gastric emptying through multiple mechanisms, including antagonism of dopamine D2 receptors (metoclopramide, domperidone) or activation of 5-HT4 receptors (prucalopride), while simultaneously increasing lower esophageal sphincter pressure and improving esophageal clearance. 13 Then, prokinetics reduce reflux episodes by minimizing the retrograde flow of gastroduodenal contents and decreasing the volume of material available for reflux. The therapeutic benefits of prokinetics were investigated in 4 studies.13,15,16,25 Ezzat et al observed that the combination of PPI with prokinetics (3/day) was associated with a significantly-higher reduction in a composite laryngeal symptom score (eg, hoarseness, irritative cough, throat pain, and throat clearing) and a composite laryngeal sign score (eg, laryngeal mucosa erythema, edema, granulation, and ulceration) after 2 months of treatment compared to the intake of PPI with a placebo. 13 These findings were corroborated in the study of Chun and Lee, who observed a superiority of the association between lansoprazole and itopride over the lansoprazole treatment in terms of RSI reduction. 16 In the study of Hunchaisri and Yoon et al, there were no significant differences between PPI and PPI + prokinetic groups.15,25 However, both teams used lower doses of PPIs than those by Ezzat et al 13 and Chun and Lee 16 (Table 1).
Benefits of Alginates
Alginates form a rapid-acting, floating mechanical barrier on the top of the gastric contents within minutes of contact with gastric acid, physically blocking reflux events. 14 The raft formation occurs through a chemical reaction where sodium alginate reacts with gastric acid to form alginic acid, while sodium bicarbonate releases CO2 that gets trapped within the gel matrix, creating a low-density foam barrier that can persist for up to 4 hours. Moreover, the potassium bicarbonate or calcium carbonate components provide local buffering effects. Alginate is the second drug class that was investigated in controlled studies.14,17-19 Wilkie et al reported that the prescription of twice-daily PPIs and thrice-daily alginates reported similar symptom relief to the use of thrice-daily alginate. 17 In the study of Matheuw and Shilpa, the patients treated with a combination of twice-daily PPIs and sodium alginates had higher symptom and sign reliefs than those treated with twice-daily PPIs only. 18 The daily doses of PPIs in these 2 studies were significantly higher than the doses used by Pizzorni et al who did not report a superiority of once-daily omeprazole (20 mg) over thrice-daily magnesium alginate. 19 The non-inferiority of alginates over the PPIs was however not supported by Pedersen and Eeg, who did not observe significant differences between PPI and PPI + alginate groups. 14 It is however important to note that alginate was prescribed after dinner (once-daily) in the study of Pedersen. 14
Benefits of Mucolytics
Three studies evaluated the effectiveness of oral21,23 or inhaled 22 mucolytics in addition to PPIs. Jo et al and Chae et al did not find the superiority of PPI + mucolytics over PPI at the 2nd and 3rd month posttreatment,21,22 while Dabirmoghaddam et al reported that the association between twice-daily omeprazole (20 mg) and mucolytics led to a higher reduction in RSI and RFS scores at the 3rd month posttreatment than by twice-daily omeprazole (20 mg) and placebo, and mucolytics and placebo. 23
Benefits of H2 Blockers and Potassium-Competitive Acid Blockers
H2-receptor antagonists competitively block histamine type 2 receptors on gastric parietal cells, thereby inhibiting one of the major pathways of acid secretion and reducing both basal and food-stimulated acid production by approximately 70%. 24 The 3 last studies compared PPI with potassium-competitive acid blockers, 20 H2 blockers, 24 or PPI + H2 blockers. 12 No study demonstrated the superiority of H2 blockers or PPI and H2 blockers over the twice-daily PPI therapy.20,24 In these 2 last studies, only the twice-daily PPI regimen was suggested as superior to the use of once-daily PPI. 12 Note that Park et al used several PPIs with high doses (lansoprazole 30 mg 2/day, omeprazole 20 mg 2/day, or esomeprazole 40 mg 4/day). 12
Epidemiological Analysis
The mean MINORS of studies was 9.1 ± 1.2, which indicates a low score for controlled studies (Table 3). There was no study reaching the cutoff of 24. A few teams included consecutive patients,12,16,17,19 and none reported an adequate rate of patients who were lost to follow-up (Table 3). The study size population was calculated in 2 studies,14,20 while the randomized process was detailed and adequate in most studies (Table 3). Their diagnosis was not based on 24 hour HEMII-pH, MII-pH, or oropharyngeal pH monitoring in studies. Considering a clinical LPRD diagnosis, the exclusion criteria of confounding conditions were judged as adequate in 2 studies (Appendix 1).17,25 Others included patients with systemic or ear, nose, and throat conditions with nonspecific symptoms and findings that can mimic LPRD. For example, Chae et al and Wilkie et al included smokers, drinkers (chronic laryngopharyngitis), and patients with psychiatric illnesses.17,21 Respiratory diseases (chronic obstructive pulmonary disease, asthma) and their related treatments (inhaled corticoids), which can be both associated with respiratory symptoms or drug-induced laryngitis 1 were excluded in only 3 studies.18,19,22 Similar observations can be made for chronic rhinosinusitis or allergy (Appendix 1). There was an important heterogeneity between studies for the therapeutic regimens of the control group (PPIs) in terms of molecules, doses, and intake. Pedersen and Eeg recommended Omeprazole 40 mg at bedtime, 14 which does not match with the pharmacological recommendations for taking PPIs (30 minutes to 1 hour before meals). 1 Some teams recommended twice-daily PPIs,12,18,21,25 while others prescribed once-daily low doses.15,19,21 At the exception of one study, 14 the therapeutic duration was adequate according to consensus paper. 1 The outcomes used for analyzing the therapeutic response were judged as incomplete. The validated RSI and RFS were used in most studies.15-25 However, RSI is an incomplete patient-reported outcome questionnaire that does not document all LPRD symptoms. Only Kim et al used both RSI and reflux symptom score-12 (RSS-12), 20 which report higher sensitivity and specificity than RSI. 28 RFS was not evaluated blindly regarding the patient history and symptoms, which can bias the analysis. 29
Bias Analysis.
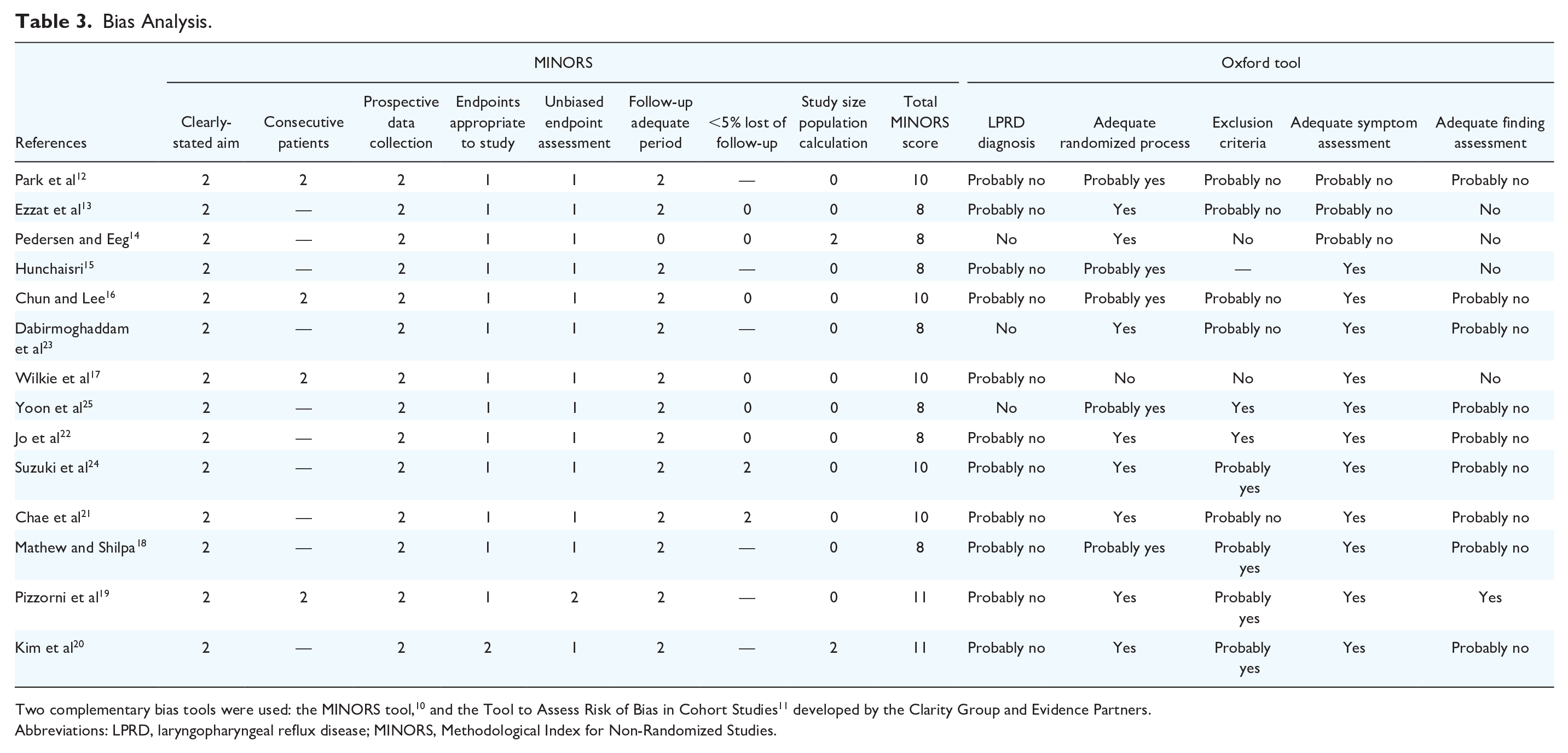
Two complementary bias tools were used: the MINORS tool, 10 and the Tool to Assess Risk of Bias in Cohort Studies 11 developed by the Clarity Group and Evidence Partners.
Abbreviations: LPRD, laryngopharyngeal reflux disease; MINORS, Methodological Index for Non-Randomized Studies.
Discussion
The treatment for LPRD has long time been based on the GERD recommendations. Indeed, a recent review of the literature suggests that of the 53 prospective clinical studies of LPRD treatment conducted in the past 25 years, PPIs were used as primary medication in 94.0% of therapeutic regimens (33% in association with other drugs). 30 This observation corroborates the data of a recent international survey, highlighting that more than 50% of otolaryngologists exclusively treat LPRD with PPIs. 31 However, LPRD differs from GERD for many pathophysiological and biological mechanisms,1,32 with the most blatant being the weakly-acidic and alkaline nature of most gastroesophagopharyngeal reflux events,5,6 which supports the lower effectiveness of PPI therapy in LPRD than in GERD. 33
The low number of clinical controlled studies comparing PPIs to alternative medical therapies is the primary finding of the present review. However, some interesting preliminary reports suggested a potential effectiveness in associating prokinetics with PPIs for treating LPRD symptoms.13,16 The prokinetics’ effectiveness can be attributed to the increase in the lower and upper sphincter tonicities, which reduces the number and duration of transient relaxation episodes of both sphincters and, consequently, the gaseous droplets with gastroduodenal enzymes reaching the upper aerodigestive tract. This hypothesis is supported by recent studies showing that patients with LPRD have a longer duration of upper esophageal sphincter (UES) transient relaxation episodes than controls, 34 or a high prevalence of proximal esophageal dysmotility disorders. 35 The weakly-acidic or alkaline nature of most pharyngeal reflux events at the 24th hour HEMII-pH supports the use of alginates or antacids in the treatment for LPRD.5,6 Of the 4 studies investigating the benefits of alginates, 3 supported the non-inferiority of alginates over PPIs in the treatment for LPRD.17-19 The epidemiological quality of the study reporting no benefits of alginate was low, with a bias analysis supporting the occurrence of many inclusion and therapeutic biases that can limit the conclusion of the study 14 (Table 3). The potential interest in using alginates or antacids in the treatment for LPRD was strengthened by recent uncontrolled large-cohort studies of patients with a confirmed LPRD diagnosis. Thus, it has been found that patients with LPRD treated with alginate and antacids had higher therapeutic response rates 36 than the mean therapeutic response rates found in the literature. 4 Magaldrate is the primary antacids used in Western countries. Magaldrate is a complex aluminum-magnesium hydroxide compound, acting as a rapid antacid by neutralizing gastric acid through chemical buffering, resulting in the formation of aluminum and magnesium chlorides plus water, thereby increasing gastric pH within minutes of administration. In addition, magaldrate exhibits cytoprotective effects by forming a protective barrier on the mucosa and binding to pepsin and bile salts, while its crystalline structure provides a longer duration of action compared to traditional aluminum/magnesium hydroxide combinations. The compound adsorbs lysolecithin and bile acids, potentially reducing their damaging effects on the esophageal mucosa. 1
In this review, only one study investigated the benefits of H2 blockers, while another compared PPIs to potassium-competitive acid blockers, which is insufficient to conclude for these 2 drugs.
Regardless of the alternative medication to PPIs, the findings of this review support the need to conduct future randomized controlled studies investigating the effectiveness of alternative medications in the treatment for patients with LPRD. The epidemiological analysis supports the need to consider some further methodological points. First, because LPRD is associated with nonspecific symptoms and findings,37,38 the use of 24-h HEMII-pH is required for including patients with a confirmed diagnosis. Indeed, the inclusion of patients with some prevalent confounding conditions, such as allergy, chronic rhinitis, or tobacco-induced laryngopharyngitis, can undoubtedly bias the pretreatment to posttreatment assessments.39,40 Second, despite a confirmed diagnosis, the control of the additional ear, nose, and throat disorders has to be optimal to differentiate the symptoms related to LPRD from those due to other disorders. Third, critical attention must be paid to the diet and lifestyle recommendations as an influencing factor on the therapeutic effectiveness. In this way, some controlled and uncontrolled studies have demonstrated that an alkaline, low-fat, low-high-released sugars, high-protein diet can lead to LPRD symptom relief in a few months.41-43 The adherence to an antireflux diet and the reduction in triggers of autonomic nerve dysfunction (stress, anxiety, depression) can be considered as a treatment and, consequently, can influence the posttreatment outcomes. They should be controlled and matched between groups in future studies. Fourth, adherence to the medication is an additional confounding factor in the assessment of the effectiveness of medications. Pisegna et al reported that 62.7% of patients treated with PPIs described an incorrect routine in taking their medication, taking it with other pills, with food/drink, and uncertainty about which pill is for reflux. 44 The adherence to diet, lifestyle changes, and good practice in taking the medications have been not investigated in the controlled studies. Moreover, from a pharmacological standpoint, the several drug regimens used in LPRD treatments were based on GERD regimens, and they require future investigations. Thus, some authors used once-daily PPI, 20 while others recommended twice daily. 12 There was an important heterogeneity in the doses of PPIs, ranging from 20 mg/day to 80 mg/day. According to some pharmacological investigations, the steady-state inhibition on once-a-day dosing is about 66% of maximal acid output for a PPI taken at least 30 minutes beforehand, while the intake of a morning dose, and an evening dose before meals (twice-daily-PPI regimen), results in about 80% inhibition of maximal acid output. 45 Physiologically, studies suggested that an increase in the PPI dose (80 mg/day) has virtually no effect once the optimal dosage has been reached. 45 For alginates and antacids, the GERD recommendations consist of an intake 1 hour post-meals (3/day) or at bedtime to neutralize the acidity of the gastric pocket during daytime and/or nighttime.1,46 Given the occurrence of most pharyngeal reflux events during daytime, upright, and after meals, as well as the weakly-acidic or alkaline nature of events, this regimen can be put into question. The bedtime intake of alginates or antacids does not make sense regarding the lack of pharyngeal reflux events at nighttime. Moreover, practitioners could favor the immediate post-meal intake of alginate and antacids to reduce the number of gaseous pharyngeal reflux events that occur in the immediate post-meal time. 47
The heterogeneity between studies in inclusion criteria, diagnosis approaches, therapeutic regimens, and outcomes is the primary limitation of this review, making difficult the comparison across studies. The low number of patients included in the study groups, the lack of full information on the diet and lifestyle recommendations, the use of incomplete patient-reported outcomes measures, and the adherence to the medication regimens are additional limitations. Concerning patient-reported outcomes measures, RSI 26 does not consider some prevalent LPRD symptoms, such as throat pain, halitosis, odynophagia, or ear pain, which has been found to reduce its accuracy for the clinical diagnosis and the follow-up of symptoms through treatment.28,32 Table 4 reports some potential recommendations for further studies comparing several drug classes in the treatment for LPRD.
Recommendations for Future Controlled Studies.

The recommendations were elaborated with the last consensus statements of laryngopharyngeal reflux disease management.1,4
Abbreviations: GERD, gastroesophageal reflux disease; GI, gastrointestinal; HEMII-pH, hypopharyngeal-esophageal multichannel intraluminal impedance-pH monitoring; LPRD, laryngopharyngeal reflux disease; PPIs, proton pump inhibitors; QoL, quality of life.
Conclusion
To date, only a few studies investigated the effectiveness of alternative medications to PPIs in the treatment for LPRD, which represents an important gap with the GERD literature. Some preliminary results suggest a non-inferiority of some drugs to PPIs (alginates, potassium-competitive acid blockers), but the low number and the quality of studies limit the drawing of reliable conclusions. These data can encourage the conduction of future randomized controlled studies investigating the benefits of alternative drugs in the treatment for patients with a primary or recalcitrant LPRD.
Footnotes
Appendix 1
Exclusion Criteria of Studies.

| References | Exclusion criteria | ||
|---|---|---|---|
| Respiratory conditions | GI conditions | Drugs and others | |
| Park et al (2005) 12 | — | GERD surgery | GERD medical treatment, cancer |
| Ezzat et al (2011) 13 | Endotracheal intubation (3 mo), vocal fold lesions | GERD surgery | Pregnancy, obesity, continuous medication for systemic disease, NSAIDs, antidiabetics, anti-HT Psychiatric illness, chronic disease |
| Pedersen and Eeg (2012) 14 | Cancer | Pregnancy | |
| Hunchaisri (2012) 15 | NP | ||
| Chun and Lee (2013) 16 | Vocal cord paralysis, laryngeal cancer | GERD diagnosis or complications, GI surgery | PPI (1 mo) |
| Dabirmoghaddam et al (2013) 23 | Laryngeal cancer |
— | Medication reaction |
| Wilkie et al (2018) 17 | NP | ||
| Yoon et al (2018) 25 | Airway infection, allergies |
Gastroenterological disease |
Pregnant or lactating NSAIDs, glucocorticoids, H2 blockers, PPIs |
| Jo et al (2019) 22 | Acute/chronic rhinosinusitis infection, asthma, COPD, pneumonia, lung cancer, pharynx, larynx, oral lesions | Pregnant |
|
| Suzuki et al (2019) 24 | Smokers, cancer history | Surgery history | Pregnant |
| Surgery history, infection | Infection | ||
| Radiation history | |||
| Chae et al (2020) 21 | Laryngeal/neck surgery | GERD, gastritis (3 mo) |
PPI intake, polypharmacy |
| Mathew and Shilpa (2022) 18 | Previous surgery, premalignant or malignancy, COPD, rhinosinusitis, laryngitis | Upper GI cancer | Allergy to PPIs and alginates |
| Psychiatric disorders | |||
| Pregnancy, breastfeeding | |||
| Pizzorni et al (2022) 19 | Laryngopharyngeal surgery, cancer, dysphonia not due to LPRD, respiratory diseases | GERD surgery, gastroenterological diseases | Immunosuppression/deficiency, diabetes, cystic fibrosis, cardiac disease, alcohol/drug abuse, pregnancy, breastfeeding |
| Kim et al (2024) 20 | Cancer, radiotherapy history | Cancer, GERD, GERD complications | Antireflux medication (2 wk) |
Abbreviations: COPD, chronic obstructive pulmonary disease; GERD, gastroesophageal reflux disease; GI, gastrointestinal; HT, hypertension; NP, not provided; NSAID, nonsteroidal anti-inflammatory drugs; PPI, proton pump inhibitor(s).
Acknowledgements
The author would like to thank the librarian for the review.
Author Contributions
Jérôme R. Lechien: design, acquisition of data, data analysis and interpretation, drafting, final approval, and accountability for the work; final approval of the version to be published; agreement to be accountable for all aspects of the work in ensuring that questions related to the accuracy or integrity of any part of the work are appropriately investigated and resolved.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
This article does not contain any studies with human participants or animals performed by any of the authors.
