Abstract
Importance
The retrograde cricopharyngeus dysfunction (R-CPD) is an emerging topic in otolaryngology, gastroenterology, and primary care.
Objective
To review the current literature about the etiology, clinical presentation, and management of retrograde cricopharyngeus dysfunction (R-CPD).
Design
Systematic review.
Setting
Three investigators conducted the PubMED, Scopus, and Cochrane Library review of the literature related to the etiologies and management of patients with R-CPD through the PRISMA statements.
Participants
Studies that incorporated R-CPD patients.
Interventions
In-office, or operating room botulinum toxin injection, or no treatment.
Main outcome measures
Etiology, clinical presentation, and therapeutic outcomes.
Results
Seventeen studies met the inclusion criteria (826 patients). A family history was reported in 28.0% of cases with most patients developing R-CDP symptoms in childhood (55.5%). In addition to the cardinal symptom of the inability to belch, associated complaints of bloating and chest pain, gurgling noises, and excessive flatulence were found in 95.7%, 86%, and 80.2% of cases, respectively.The diagnosis was recognized by patients themselves in 78.9% of cases. The immediate success rate of botulinum toxin injection into the cricopharyngeal sphincter in facilitating burping was 92.5%. Recurrence occurred in the first month of follow-up in 9.5% of cases, whereas 12.6% and 27.9% of patients had recurrence during the 1–5 months and more than 6 months posttreatment, respectively. Transient dysphagia is the most prevalent complication after botulinum toxin injection (31.1%). The heterogeneity was high across studies, limiting the establishment of further combined analyses.
Conclusion and relevance
R-CPD is a recently-recognized and likely-underdiagnosed condition associated with severe symptoms affecting the quality of life of patients. Future studies are needed to elucidate the etiology of this condition, develop adequate patient-reported outcome questionnaires for the baseline and follow-up evaluations of symptoms, and propose therapeutic consensus.

Keywords
Introduction
Retrograde cricopharyngeal dysfunction (R-CPD), known as the inability to burp, can be defined as a condition with absent or incomplete upper esophageal sphincter (UES) relaxation in response to abrupt esophageal distention by gastroesophageal gas reflux, which leads to bothersome symptoms.1,2 The first cases of R-CPD were initially reported by Kahrilas (1987), 2 Waterman and Castell (1989), 3 and Tomizawa et al. (2001). 4 Without clear diagnostic criteria and more widespread knowledge of therapeutic approaches, the condition remained underappreciated despite these isolated reports. In 2019, Bastian and Smithson published the first large case series of patients with the inability to burp, codifying this constellation of symptoms as R-CPD. 5 R-CPD is thought to be related to the loss of the belch reflex, that is, otherwise thought to be a normal reaction to the gaseous pressurization of the esophagus that occurs when transient lower esophageal sphincter relaxation allows gas from the stomach to reach the esophagus; if the upper esophageal sphincter does not relax in the setting of this pressurization, gas is trapped in the esophagus and then carried back into the stomach by peristalsis—resulting in the gurgling noises, bloating, chest pain, and excessive flatulence, which often accompany the inability to burp.1,6 Some additional symptoms have been associated with R-CPD, including difficulty vomiting, hiccups, nausea, globus sensation, dysphagia, and anorexia, but all of them remain nonspecific. 1 To date, most series on R-CPD document treatment of this condition through the injection of botulinum toxin into the cricopharyngeus muscle in the office or in the operating room, with or without esophageal dilation.1,5,6 Since the publication of the case series by Bastian and Smithson, 5 the number of clinical research dedicated to R-CPD is increasing in the United States and Europe. These recent studies improved the knowledge about the etiology, clinical presentation, and management of R-CPD. However, R-CPD is still an undiagnosed condition with most patients making the diagnosis themselves after having seen many practitioners and having passed a myriad of additional examinations. 7 The unclear origin of R-CPD, and the nonspecific clinical presentation support the lack of attention toward this troublesome disorder by primary care, otolaryngology, or gastroenterology providers.
In this systematic review, the authors aimed to review the current literature on the epidemiology, etiologies, clinical presentation, diagnosis, and treatment of R-CPD.
Materials and Methods
The criteria for study inclusion and exclusion were based on the population, intervention, comparison, outcome, timing, and setting (PICOTS) framework. 8 The data review and collection were performed by 3 independent authors (J.R.L., S.H., and M.M.) according to the PRISMA checklist for systematic reviews. 9
Types of studies: The 3 authors carried out a critical review of the content of retrospective case series, and uncontrolled or controlled prospective studies published between January 1987 and November 10, 2024, if the studies investigated the etiology, clinical presentation, and/or the therapeutic management of R-CPD. The studies were published in English, Spanish, or French peer-reviewed journals. The authors considered preprint papers. Case reports were excluded, and only studies reporting data for ≥5 patients have been considered.
Populations, inclusion, and exclusion criteria: Studies were included in this review if the authors reported the diagnosis criteria of R-CPD (inclusion), which consists of the inability to belch. According to the Bastian and Smithson criteria, the diagnosis can be based on the association of the 4 primary symptoms: inability to belch, chest pain/bloating, gurgling noises, and excessive flatulence. Because some patients can have an incomplete clinical picture, 1 the inability to belch was considered necessary for the diagnosis, while the other symptoms supported the diagnosis. The overlap between clinical studies published by the same teams was evaluated, and the smallest cohorts were excluded for the overlapped outcomes (eg, symptom prevalence, etiologies). However, if the outcomes presented in the overlapped studies substantially differed, the authors included all studies in the analysis.
Outcomes: The following outcomes were reviewed: study design; number of patients; gender ratio; age (mean/median); etiology; diagnosis method; examinations before the diagnosis; initial and posttreatment symptom prevalence; additional examination findings; medical or surgical procedure(s); duration of follow-up; number of procedure(s) per patient; complications; re-intervention(s); and surgical outcomes (eg, effectiveness of treatment on symptoms, swallowing, and duration of treatment).
Intervention and comparison: The following procedures were considered: botulinum toxin injection into the cricopharyngeus muscle, sphincter balloon dilatation, or surgical myotomy. The authors paid careful attention to the doses of botulinum toxin and the surgical approach: in the office (with or without electromyography [EMG] control) or in the operating room (suspension esophagoscopy). Note that the present review differs from the review of Miller et al., 1 which was a narrative review, and the review of Jonsson and Plaschke, 6 who only focused on the clinical presentation and the postoperative outcomes of 13 studies/case reports. In the present review, the authors reported findings related to the etiology, diagnosis method, and the pre- to posttreatment evolution of symptoms, which are all points never systematically reported in previous reviews.
Timing and setting: There were no criteria for specific stages or timing in the “disease process” of the study population.
Search Strategy
The literature research was conducted by 3 investigators (J.R.L., S.H., and M.M.) through PubMed, Scopus, and Cochrane Library databases for relevant peer-reviewed publications related to epidemiology, etiology, clinical presentation, diagnostic features, therapeutic management, and outcomes of R-CPD. The following keywords were used “cricopharyngeus,” “cricopharyngeal,” “sphincter,” “dysfunction,” “disorder,” “belch,” “belching,” “inability,” “treatment,” “management,” and “procedure,” to identify case reports, clinical prospective/retrospective studies, reviews, and meta-analyses. The authors considered studies with and without (case reports) database abstracts. The papers had available full-texts or titles containing the search terms. The findings were reviewed for relevance, and the reference lists of these articles were examined for additional pertinent studies. Implications for practice were summarized. Ethics committee approval was not required.
Bias Analysis
The authors carried out a bias analysis with the Methodological Index for Non-Randomized Studies (MINORS) tool, which is a validated instrument designed for assessing the quality of non-randomized surgical studies. 10 The MINORS tool includes 12 items related to the analysis of methodological points of studies. The items were scored 0 if absent; 1 when reported but inadequate; and 2 when reported and adequate. Based on a previous systematic review, 11 the inclusion of patients was evaluated in terms of consecutive inclusion (0 or 2), while the prospective data collection was rated as perfectly prospective (2), retrospective analysis of prospectively-recruited patients (1), or absent (0). The quality of endpoints was judged as high (2) when authors evaluated the pre- to post-intervention outcomes with objective outcomes. The use of a subjective but validated patient-reported outcome questionnaire was judged as incomplete (1). The use of subjective unvalidated assessment and the lack of evaluation of treatment effectiveness were considered as low (0). Given the duration of botulinum toxin, 1 a follow-up period of more than 6 months was considered as adequate (2). A shorter follow-up was considered less reliable in the accurate evaluation of botulinum toxin procedures (1). The lack of posttreatment evaluation was considered as low (0). The 5% rate of lost to follow-up patients was considered as the threshold in the MINORS, while the study size prospective calculation needed to be carried out (2), mentioned as unnecessary (1), or absent (0). The ideal MINORS score was 16 for non-comparative studies and 24 for comparative studies. 10
Results
Of the 168 identified studies, 17 publications met our inclusion criteria and data were extracted (Figure 1).2-5,7,12-30 There were 8 prospective controlled27,28 or uncontrolled5,14,18,25,29,30 studies, 7 retrospective chart reviews,7,12,13,17,21,22,26 and 2 cross-sectional studies.23,24 Eight case reports were excluded.2-4,15,16,19,20,31 One cross-sectional study consisted of an oriented interview, 23 while the second was a survey. 24 Two references published in 2019 5 and 2020 7 appeared to be connected with a potential overlap of patients, but outcomes were different. Two additional papers were published by the same team in 2023 22 and 2024, 28 but the authors reported different outcomes from their initial study to the largest. Excluding the overlapped smallest cohort studies,5,22 and case reports, this systematic review included the data of 826 patients (Table 1). There were 573 females and 253 males. The mean age of diagnosis was 27.0 years, ranging from 14.0 years to 32.5 years. The demographics, clinical, and therapeutic findings of studies are reported in Table 1.

PRISMA flow chart.
Clinical Studies Conducted on Retrograde Cricopharyngeus Dysfunction.

Some studies reported percentage of symptom relief at an intermediate time of follow-up that is specified into brackets in the result’s column.
Abbreviations: BT, botulinum toxin; CT, control; EMG, electromyography; ESO, esophagoscopy; F/M, female/male; GAD7, general anxiety disorder-7; GRE, gaseous reflux event; HRM, high-resolution manometry; IO, in office; Man, manometry; mo, month; N, number; NF, nasofibroscopy; NP, not provided; PRE, proximal reflux event; RSI, reflux symptom index; TNO, transnasal esophagoscopy; U, units; UES, upper esophageal sphincter; VESS, videoendoscopic swallowing study; we, week.
Etiology, Ethnicity, and Comorbidities
The etiology of R-CPD was investigated in 8 studies (Table 2).7,14,23-26,29,30 There were no overlapped data across the studies. Out of 518 patients with data for the presence or absence of family history, 145 patients (28.0%) were reported as having a family history of R-CPD. In all studies, the family history was based on the parent’s information who described in the patient some combination of gassiness, colic, incessant crying from pain, projectile vomiting, and difficulty burping. 7 The family history was, however, difficult to obtain in studies with 82.3% of responders who were unaware of any family history in the study of Chen et al., 24 and 25.4% of patients without information about their infancy in the study of Hoesli et al. 7 R-CPD developed during childhood was lifelong in 55.5% (325/586 cases), while 2.9% (17/586) of patients reported having first symptoms during adolescence (Table 2). In 6 studies,14,17,18,24,26,29 authors comment on the direct or indirect relationship between R-CPD and gastroesophageal reflux disease (GERD), some using pH impedance. Oude Nijhuis et al. reported a median number of gaseous reflux events of 33, and their impedance-pH monitoring data indicated air entrapment in the esophagus. 14 Prolonged air trapping in the esophagus was similarly reported in 60% of children including in the impedance-pH monitoring study of Dorfman et al. 17 Siddiqui et al. noted a history of GERD in 42.4% of patients, with 28.2% being treated with proton pump inhibitor or H2 blocker therapy. 18 Moreover, approximately one-third of patients had a diagnosed mood disorder, the majority with anxiety, which can be associated with autonomic nerve dysfunction and reflux disease. 18 In the study of Mailly et al., 51.9% of patients complained of acid brash. 29 In the survey of Chen et al., 28.7% of R-CPD patients reported being diagnosed with GERD. 24 Finally, Arnaert et al. found 8% of R-CPD patients with intestinal atonia and 50% with GERD. 26 Importantly, Arnaert et al. reported that 92.3% of patients had significant improvement in heartburn complaints 1 month after Botulinum toxin injection 26 —leading to the question as to how much “reflux” symptomatology may be secondary to substernal pressure/pain from R-CPD itself rather than actual reflux. The relationship between R-CPD and ethnicity was indirectly investigated in 3 studies where authors reported the ethnicity data of patients who underwent botulinum toxin treatment.24,27,28 The white, black, and Asian proportion of patients ranged from 85.0% to 91.3%, 0.0% to 1.7%, and 0.0% to 7.0%, respectively.
Etiological Findings.
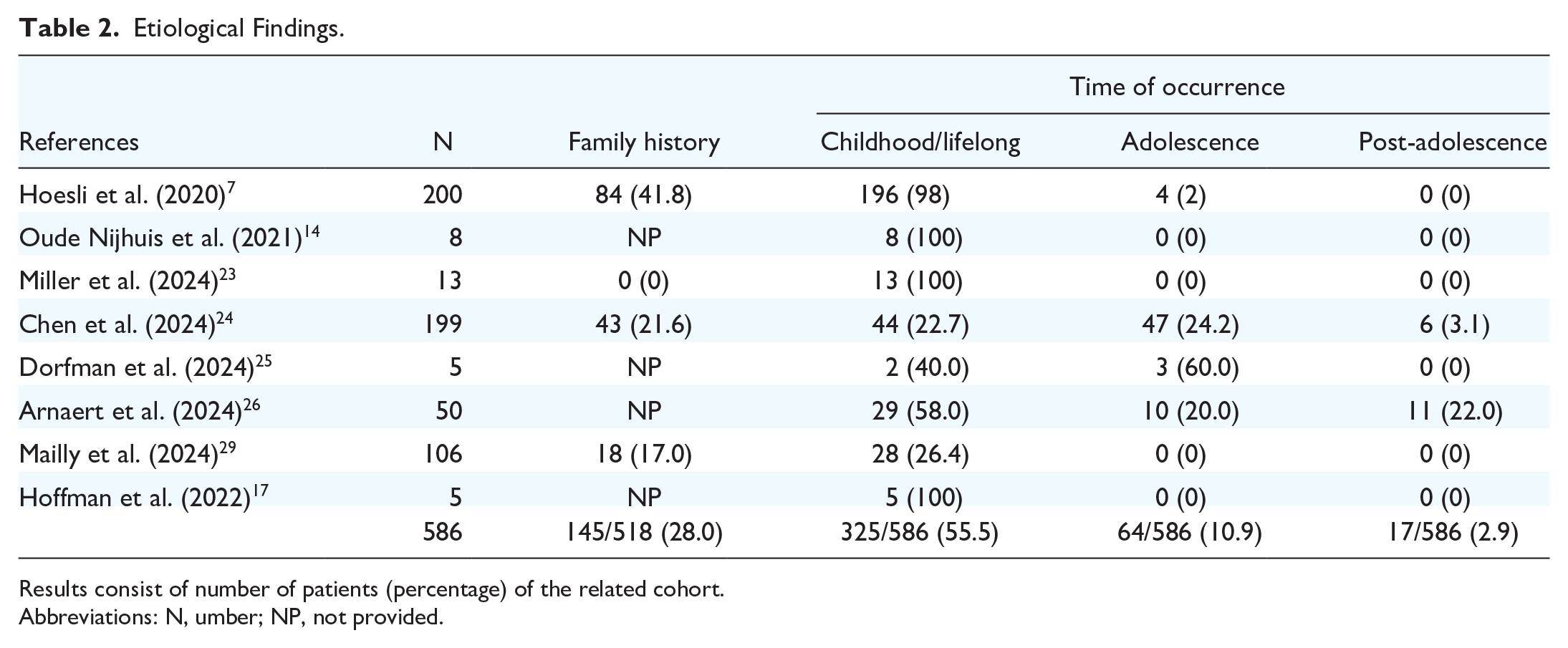
Results consist of number of patients (percentage) of the related cohort.
Abbreviations: N, umber; NP, not provided.
Clinical Presentation
The prevalence of the primary symptoms at presentation is reported in Figure 2. Three patients (0.3%) had a mild and partial inability to belch, but they were considered as R-CPD by authors according to clinical findings18,24; otherwise, all included patients had a pathognomonic inability to belch. Bloating and chest pain, gurgling noises, and excessive flatulence were found in 95.7%, 86%, and 80.2% of cases, respectively (Figure 2). Some additional symptoms potentially associated with R-CPD were reported in a few studies, including difficulty vomiting,23,24,27 hiccups,14,20 nausea,14,20,23,26,27,29 dysphagia,14,18,27 globus pharyngeus,14,18,27 and anorexia. 23 Importantly, some of these symptoms, such as hiccups or nausea, significantly resolved after R-CPD treatment (Appendix 1).20,26 Note that in some studies, it was unclear whether some of these additional R-CPD symptoms would overlap some GERD symptoms.25,26The authors described symptoms as exacerbated after eating or drinking that would increase abdominal gas, especially carbonated drinks and beer.14,23 Some patients reported that they forced themselves to burp (“air vomit”) by sticking their finger into the back of their throat, while others get temporary relief by lying supine on their left side to allow the gas to pass down the alimentary tract to later get expelled as excessive flatulence. Xie et al. suggested that symptoms can be aggravated when the patients cry. 16 Only Chen et al. reported the symptom frequency, which consisted of daily (87.6%), weekly (10.3%), or monthly (2%). 24

Pre- to Posttreatment Prevalence of Symptoms.
Diagnosis
Diagnostic criteria varied by study. Most included studies made the diagnosis based on symptoms alone,17-19,21-26,28 symptoms and transnasal esophagoscopy, 13 symptoms and nasofibroscopic examination,14,27,29 symptoms, and (high-resolution) manometry (HRE) findings.15,16,30 Most authors considered the inability to belch as the primary symptom, but some teams based the diagnosis on the presence of all 4 cardinal symptoms (Table 1).13,28,29
The use of objective examinations led some authors to support the diagnosis. Oude Nijhuis et al. observed a significant increase in esophageal pressure and air entrapment in the proximal esophagus (periods of continuous high impedance levels) in 8 patients who underwent HRE and multichannel intraluminal impedance-pH testing (MII-pH). The authors demonstrated the botulinum toxin effectiveness with a decrease in UES basal pressure after the injection of 50 units of toxin botulinum. 14 Similar objective findings have been reported in a pediatric population. 25 Dorfman et al. observed normal UES pressure during swallowing but abnormal UES relaxation with concurrent high esophageal impedance reflecting air entrapment in the proximal esophagus. 25 The HRE features of R-CPD patients were compared with asymptomatic individuals in a recent study. 27 Yousef et al. reported that R-CDP patients have higher UES length, basal pressure, and higher rates of ineffective swallows than controls. 27 Moreover, an incomplete bolus clearance was more frequently reported in R-CDP patients than in asymptomatic individuals. 27 Anderson et al. reported that 53/85 (63%) R-CPD patients had abnormal HRM study, with 40 (47%) patients with ineffective esophageal motility or absence of motility. 30 The mean eating assessment tool-10 value of the R-CPD population of Anderson et al. was abnormal, whereas reflux symptom index and voice handicap index-10 means were normal. 30
Most patients had sought initial medical attention from those unfamiliar with the syndrome, which led to delayed or eventual self-diagnosis. Of the 5 studies evaluating the diagnosis approach, the diagnosis was carried out by patients themselves in 61.5% to 99.1% of cases, based on information found by patients on social media or web forums (Appendix 2).
Prior testing or treatments before the diagnosis were documented in 8 papers.5,7,17,18,23,25,27,29 Patients underwent esogastroduodenoscopy (32.7%), barium swallow study (15.9%), and high-resolution manometry or manometry (7.3%) without diagnosis. Among medication, proton pump inhibitors, antispasmodics, and antibiotics were empirically prescribed in 32.3%, 5.6%, and 1.1% of cases, respectively (Table 3). In the study of Hoesli et al., 7 15.8% of patients had already undergone 4 or more procedures, while in the study of Mailly et al., 29 19 patients (17.9%) had more than one procedure before the diagnosis. Among the specialists seen by patients, Siddiqui et al. reported that all patients (N = 85) consulted a gastroenterologist, with one patient also having seen an allergist and another also having seen an otolaryngologist. 18 Miller et al. showed that the diagnosis was carried out by gastrointestinal specialists and otolaryngologists in 7.7% and 30.8%, respectively. 23
Prior Testing or Treatment Before Diagnosis and Methods of Diagnosis.

Results consist of number (percentage) of patients of the related cohort.
Abbreviations: AB, antibiotics; Aspas, antispasmodic; BBS, barium swallow study; CT, CT-scan; EGD, esogastroduodenoscopy; GI/OTO, gastroenterologist/otolaryngologist; HRM, high-resolution manometry; MD cons, prior consultation with medical doctor; MII-pH, impedance-pH testing; PPI, proton pump inhibitors; Provo, carbonated water provocation test (HRM) with paradoxical sphincter contraction in response to gas.
Treatment Features and Outcomes
The primary treatment consisted of injection of botulinum toxin into the cricopharyngeal sphincter in all studies reporting therapeutic data (Table 4). Among them, only one team did not report details about the type and dose of botulinum toxin use. 23 The injections were performed in the operating room in 10 studies,5,7,13,14,17,18,21,23,26,27 and in the office in 2 studies.12,22,29 Among them, Mailly et al. carried out some operating room injections in patients with recalcitrant R-CPD, 29 and Doruk et al. 22 compared both approaches. The doses of botulinum toxin significantly varied from one study to another, or, within a study. The following doses were primarily used: 25U,17,18,29 30U,22,28 50U,5,7,12-14,17,21,27 75U,7,12,13,26,29 80U,22,28 and 100U.13,18,21,27 Authors used several doses in the same patient population in 9 studies, making it difficult to estimate the success rate according to the dose (Table 4).7,12,13,17,18,21,22,27,28 A trend of increase in dose since the first study can be observed with more recent studies using higher doses. The pre- to posttreatment information was available for 695 patients (Table 4). Note that the data of Bastian and Smithson 5 were not considered for the effectiveness analysis given the overlap of patients with the study of Hoesli et al. 7 ; these patients in the latter study were included. Of the 640 patients analyzed, 48 (7.5%) remained unable to belch after initial treatment, leading to the group success rate of botulinum injection of 92.5%. Follow-up data regarding recurrence were available for 552 patients. Among them, 87 (15.8%) patients reported recurrence of inability to belch after 1 month to 29 months of follow-up. The treatments of recurrences and their related success rates are described in Table 4. Note that four teams reported data for patients with several recurrences. Bastian and Smithson performed 3 injections in one patient, 5 while Mailly et al. 29 performed 2, 3, and 4 injections in 21, 4, and 1 patient(s), respectively. Karagama similarly carried out 2 injections for second recurrence in two patients and used balloon dilatation for the treatment of primary and second recurrences. 13 In the study of Hoesli et al., 2 patients were treated with cricopharyngeal myotomy for recurrence. 7
Treatment and Recurrence Outcomes.

Abbreviations: EMG, electromyography; IO, in office; N, number; NP, not provided; OR, operating room; U, Units.
Data including injections for both recurrences and failed primary injections.
The pre- to posttreatment evolution of symptoms is described in Figure 2. At the time of presentation, bloating/chest pain was reported in 95.7% of patients, gurgling noises in 86.0%, and excessive flatulence in 80.2%, respectively. According to studies, the inability to belch recurred in the first month of follow-up in 9.5% of cases, whereas 12.6% and 27.9% of patients had recurrence during the 1–5 months and more than 6 months posttreatment (Figure 2). The patterns of recurrence of gurgling noises and bloating/chest pain throughout the follow-up were similar with 20.8% to 28.0% recurrence after 6 months of follow-up. The data related to the recurrence of excessive flatulence differ from the other symptoms at 1 to 5 months of follow-up, but they were based on a low number of patients. Hoesli et al. indicated that the age of patients was a predictive factor of recurrence because the mean and median age of patients who lost their primary response were 39.2 and 38 years as compared to the mean and median age of those who kept their response of 28.0 and 26 years. 7 There is no specific analysis of a potential dose effect on the response in any study. However, Karagama observed that patients who underwent 100 units of botulinum toxin at the initial treatment did not require a second injection over the follow-up. 13
Complications
The complications were reported in 13 studies (Table 5).5,7,12-14,16-18,22,23,25-27,29 Transient dysphagia is the most prevalent complication after botulinum toxin injection, seen in 31.1% of patients (94 out of 302 with available data). Dysphagia resolved as early as after a few days of onset or after a few weeks. Nine teams reported an increase in acid brash, regurgitation, or laryngopharyngeal reflux symptoms after treatment.5,13,14,17,18,22,23,27,29 Aspiration and stridor were not reported as complications in studies assessing swallowing postinjection.13,17,22,23,29 The other complications included tooth injury or pharyngeal injury from esophagoscopy, transient change in breathing, dysphonia, hiccups, and globus (Table 5). Vocal cord paresis was reported in one patient and lasted for 2 months. 22 Arnaert et al. reported that 11 (22.0%) of patients had throat pain after the procedure, which was attributed to the esophagoscopy rather than the botulinum toxin injection. 26
Complications.

Results consist of number of patients with the related complication/total number of assessed patients. Abbreviations: +, unspecified number; GERD, gastroesophageal reflux disease; N, number; UnDe, undetermined.
one was vocal cord paresis.
Bias Analysis
Heterogeneity among included articles in R-CPD definition, inclusion/exclusion criteria, surgical procedure features (in office vs in operating room; botulinum toxin doses), postoperative functional, and surgical outcomes precluded statistically pooling the data into a formal meta-analysis, thereby limiting the analysis to a qualitative rather than quantitative summary of the available information. The MINORS analysis was only carried out on case series and prospective studies (Appendix 3). The mean MINORS score was 10.0 ± 2.2. There was no study reaching the minimal MINORS score (16) to be considered an unbiased study. 10 The primary biases consisted of the lack of prospective data collection, the lack of study size prospective calculation, and the lack of unbiased outcomes in most studies. Indeed, there were 7 prospective studies,5,14,18,25,27-29 and, among them, only one was controlled. 27 The outcomes used in studies were mostly subjective and consisted of the evaluation of symptom prevalence or severity without an R-CPD-validated patient-reported outcome questionnaire. Objective findings were used in 4 studies, consisting of high-resolution manometry,14,25,27,30 or impedance-pH monitoring.14,25
Discussion
R-CPD is a new disorder with an unknown prevalence. Since 2019, the number of studies reporting etiological, clinical presentation, and therapeutic outcomes has increased, but the pathophysiology of R-CDP remains unelucidated. In this systematic review, the authors summarized key findings about etiology, diagnosis approach, clinical presentation, treatment, and posttreatment outcomes. Most patients report childbirth and a family history in 17% to 48% of cases. In this way, the family environment and genetic patterns could be investigated in future studies to resolve the etiology of R-CPD. Several authors reported a high prevalence of GERD in R-CDP patients.14,17,18,24,26,29 The investigation of laryngopharyngeal reflux disease and enzyme-related esophageal mucosa injuries could be investigated in this population of patients using hypopharyngeal-esophageal multichannel intraluminal impedance-pH monitoring, and enzyme measurements. The diagnosis is a challenging point for future studies. Indeed, this review reports that most patients made the diagnosis themselves, which highlights an unawareness of a substantial number of otolaryngologists, gastroenterologists, or primary care practitioners. Despite evidence about the effectiveness of botulinum toxin injection, there is no standardization of doses and procedures used. The important heterogeneity between studies about botulinum toxin doses, procedure steps, and additional therapeutic advice (eg, careful chewing, washing solids down with liquids, body position changes during nighttime) makes difficult the interpretations of the current data of the literature, especially for a potential dose effect or for the complications or the recurrences. Finally, the development of an R-CPD patient-reported outcome questionnaire could be an additional helpful project to better evaluate the prevalence of symptoms and potential symptom overlapping with other GI conditions, and the effectiveness of treatment regimens. The prevalence of symptoms is not enough to appreciate the clinical picture of R-CPD given the several degrees of symptom relief after treatment, 26 and the impact of symptoms on quality of life.
Conclusion
R-CPD involves a constellation of symptoms, which are still poorly diagnosed by practitioners. Its etiology remains unelucidated and should be associated with genetic patterns and contributing conditions, such as reflux. The administration of botulinum toxin into the cricopharyngeal sphincter is safe and associated with a high percentage of transient or definitive relief of the inability to belch. Future studies are needed to better understand the etiology of R-CPD, and to develop adequate patient-reported outcome questionnaires, and consensus for botulinum toxin injection procedures.
Footnotes
Appendix
Bias Analysis.

| Clearly | Inclusion of | Prospective | Endpoints | Unbiased | Follow-up | <5% of lost | Study size | Total | |
|---|---|---|---|---|---|---|---|---|---|
| Stated | consecutive | data | appropriate | endpoint | adequate | of | prospective | MINORS | |
| References | Aim | patients | collection | to study | assessment | period | follow-up | calculation | score |
| Bastian and Smithson (2019) 5 | 2 | 2 | 2 | 2 | 1 | 2 | 1 | 0 | 12 |
| Hoesli et al. (2020) 7 | 2 | 2 | 2 | 2 | 1 | 2 | 2 | 0 | 13 |
| Wajsberg et al. (2021) 12 | 2 | 1 | 1 | 1 | 1 | 2 | 1 | 0 | 9 |
| Karagama et al. (2021) 13 | 2 | 1 | 1 | 1 | 1 | 2 | 2 | 0 | 10 |
| Oude Nijhuis et al. (2021) 14 | 2 | 2 | 2 | 2 | 2 | 1 | 0 | 0 | 11 |
| Hoffman et al. (2022) 17 | 2 | 1 | 1 | 1 | 1 | 2 | 0 | 0 | 8 |
| Siddiqui et al. (2023) 18 | 2 | 1 | 2 | 2 | 1 | 1 | 1 | 0 | 10 |
| Silver et al. (2023) 21 | 2 | 1 | 1 | 1 | 1 | 0 | 2 | 0 | 8 |
| Doruk et al. (2024) 22 | 2 | 1 | 1 | 1 | 1 | 2 | 1 | 0 | 9 |
| Miller et al. (2024) 23 | 2 | 1 | 2 | 1 | 1 | 0 | 1 | 0 | 8 |
| Chen et al. (2024) 24 | 2 | 0 | 2 | 0 | 0 | 0 | 0 | 0 | 4 |
| Dorfman et al. (2024) 25 | 2 | 1 | 2 | 2 | 2 | 2 | 0 | 0 | 11 |
| Arnaert et al. (2024) 26 | 2 | 2 | 1 | 2 | 1 | 2 | 1 | 0 | 11 |
| Yousef et al. (2024) 27 | 2 | 2 | 2 | 2 | 2 | 1 | 2 | 0 | 13 |
| Doruk et al. (2024) 28 | 2 | 1 | 1 | 2 | 2 | 1 | 1 | 0 | 10 |
| Mailly et al. (2024) 29 | 2 | 2 | 2 | 1 | 1 | 2 | 2 | 0 | 12 |
| Anderson et al. (2024) 30 | 2 | 1 | 2 | 2 | 2 | 2 | 0 | 0 | 11 |
Acknowledgements
The authors thank the librarian (G.B.) for the review.
Author Contributions
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethical approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Informed consent
Not applicable.
