Abstract
Importance
The objective response (ORR) rate in patients with locally advanced hypopharyngeal squamous cell-carcinoma (LA-HPSCC) following neoadjuvant chemotherapy (NACT) of albumin-bound paclitaxel plus carboplatin is low. At present, it is unclear whether the addition of pembrolizumab could increase the ORR or not.
Objective
To investigate whether the addition of pembrolizumab could increase the ORR, and to develop a nomogram to predict the response of pembrolizumab addition.
Design
Retrospective cohort study.
Setting
This study was conducted at a single institution.
Participants
This study included 129 patients who conformed to the inclusion criteria.
Intervention or Exposures
NACT with or without pembrolizumab for patients with LA-HPSCC.
Main Outcome Measures
The ORR was analyzed according to the RECIST 1.1 criteria and a nomogram was developed based on least absolute shrinkage and selection operator and multivariable Cox regression analysis. Predictive accuracy and discriminative ability of the nomogram were evaluated by receiver operating characteristics, precision recall, calibration curves, and decision curve analysis.
Results
Eighty-two patients received NACT and 47 also received pembrolizumab. ORR was higher in patients receiving additional pembrolizumab (66.0% vs 47.6%, χ2 = 4.074, P = .044). The nomogram identified pretreatment levels of lymphocytes and red blood cells as independent predictors of a high ORR, while basophil levels were an independent predictor of a low ORR. Calibration curve showed that the nomogram-based predictions corresponded well with actual observations. C-index of the nomogram was 0.925 (0.848-1.002) and the area under curve was 0.925. Decision curve analysis affirmed that the nomogram had important clinical value.
Conclusions and Relevance
Pembrolizumab could improve the ORR in LA-HPSCC patients treated with NACT. Furthermore, a risk-prediction nomogram incorporating readily assessable routine pretreatment blood parameters can accurately estimate the response to NACT with pembrolizumab, leading to precise treatment and minimizing the waste of medical resources.

Keywords
Introduction
Hypopharyngeal squamous cell carcinoma (HPSCC) accounts for most cases of hypopharyngeal cancer, including cancers of the posterior pharyngeal wall, post-cricoid area, and pyriform sinus. 1 In addition, HPSCC accounts for approximately 3% to 5% of head and neck cancers and <0.5% of all cancers.2,3 HPSCC is a highly aggressive malignancy and patients usually present with loco-regionally advanced disease, leading to the highest mortality rate and worst prognosis among all head and neck cancers.4-6 Despite recent diagnostic and therapeutic advances, the estimated 5-year overall survival rate has not improved significantly and remains at around 40% for all hypopharyngeal cancers and approximately 15% to 25% for locally advanced tumors.1,7,8
The traditional surgical treatment for locally advanced HPSCC is total laryngectomy 9 ; however, the effects of this procedure on the patient’s voice and the risk of intractable postoperative complications, such as pharyngocutaneous fistula,10,11 mean that nonsurgical organ-preserving treatments have attracted increasing interest in clinical practice.2,12,13 Neoadjuvant chemotherapy (NACT), also called induction chemotherapy, has been proposed as an adequate first-line option for hypopharyngeal cancer in recent decades,14,15 followed by concurrent chemoradiotherapy in patients with a good response or by total laryngectomy in patients without an adequate response.16-19 Nonetheless, only about half of all HPSCC patients benefit from NACT.20,21
The development of programmed cell death protein 1 (PD-1)/PD-1 ligand blockers, such as pembrolizumab, has revolutionized the treatment of head and neck squamous cell carcinoma (HNSCC).22-24 Pembrolizumab was approved by the United States Food and Drug Administration in 2016 as second-line monotherapy for recurrent or metastatic HNSCC, and as front-line treatment in 2019.25-27 Few studies, however, have focused on the efficacy of NACT with pembrolizumab for the treatment of locally advanced HPSCC. We therefore conducted a retrospective study to analyze the efficacy of NACT with pembrolizumab in patients with locally advanced HPSCC.
The nomogram is a statistics-based tool, which provides prognostic information by integrating various clinical variables. Moreover, it can generate a numerical probability of a clinical event with a simple visual representation of a statistical predictive model. Herein, we aim to evaluate the objective response rate (ORR) and develop a nomogram to predict the response of pembrolizumab addition, which may help to make more rational clinical treatment decisions in the future.
Methods
Participants
This was a retrospective observational study based on the review of medical records of patients with locally advanced HPSCC. The inclusion criteria were: (i) Histologically diagnosed with HPSCC, (ii) clinical stage III to IV according to the American Joint Committee on Cancer (eighth edition), (iii) received at least 3 courses of NACT with or without pembrolizumab as the initial treatment in the Department of Otolaryngology, The First Affiliated Hospital, Sun Yat-Sen University, Guangzhou, China, between March 2019 and April 2023. Exclusion criteria included: (i) coinstantaneous second primary tumor or recurrent disease, (ii) previous treatment including chemotherapy, radiotherapy, immunotherapy, and surgery other than a biopsy, (iii) unsuitable candidate for the full course of NACT treatment or poor tolerance to it. The First Affiliated Hospital of Sun Yat-Sen University Institutional Review Board approval was obtained ([2023]283). The need for informed consent was waived because only retrospective, de-identified data were used.
NACT and Pembrolizumab
Patients were divided into a NACT group and a NACT plus pembrolizumab group. Patients in the NACT group received albumin-bound paclitaxel (body surface area × 260 mg/m2) and carboplatin [area under the curve (AUC) = 5] once every 3 weeks (referred to as TP treatment). Patients in the NACT plus pembrolizumab group received TP plus 200 mg pembrolizumab once every 3 weeks (TPK treatment).
Evaluation of ORR
Enhanced computerized tomography (CT) scan combined with electronic laryngoscopy was performed at baseline and 2 weeks after the third course of treatment. The ORR was assessed independently by 3 clinicians blinded to the group information. All assessments were independently completed by 2 experienced radiologists initially, and disagreement was solved through discussion or by the decision of the third person who was an experienced surgeon. The ORR was categorized as progressive disease (PD), stable disease (SD), partial response (PR), and complete response (CR) according to the RECIST 1.1 criteria. Patients with PD or SD were considered as the nonresponse group, and patients with PR or CR were considered as the response group.
Clinical Data Collection
Clinicopathologic baseline characteristics and blood test results were extracted retrospectively from the Hospital Information System. Blood tests in all patients were conducted within 3 days before the first course of treatment, and all the covariates were measured at the same time. We collected information on the following potential predictive variables: age, sex, differentiation grade, T-stage, N-stage, metastasis, clinical stage, white blood cell, neutrophils, lymphocyte (LY), monocyte, eosinophils, basophil (BASO), red blood cell (RBC), hemoglobin (Hb), platelet, alanine aminotransferase, aspartate aminotransferase, serum albumin, serum globulin, calcium, phosphorus, sodium (natrium), potassium (kalium), chlorine, and creatinine. The missing covariate data were little and considered “Missing Completely at Random” because drop-out occurrences were unrelated to the missing data items, hence, all of them were left out and not imputed. The data were used for the following analysis and are available as detailed in the data availability statement. Moreover, we also calculated the neutrophil-to-lymphocyte ratio (NLR), platelet-to-lymphocyte ratio (PLR), lymphocyte-to-monocyte ratio (LMR), and lymphocyte to white blood cell ratio (LWR).
Variable Selection and Nomogram
We applied the least absolute shrinkage and selection operator (LASSO) regression to minimize the potential collinearity of variables using the R package GLMNET (version 4.1.7). The 10-fold cross-validation method was adopted to confirm the appropriate tuning parameters (lambda, λ) for LASSO analysis and the most significant features were screened using LASSO. The screening indicator for 10-fold cross-validation method was the deviance, and the random seed was set to 1221. The variables selected in the LASSO regression were then further analyzed by univariable and multivariable logistic regression. 95% confidence intervals are calculated with the Wald method. We then constructed a nomogram (R package rms version 6.4.0) based on the multivariable logistic regression results. Finally, the discriminative performance of the nomogram was evaluated by receiver operating characteristic (ROC) curve (R package pROC version 1.18.0 and ggplot2 version 3.3.6), precision-recall (PR) curve (another approach aside from ROC for assessing the performance of the prediction model, R package pROC version 1.18.0 and ggplot2 version 3.3.6), calibration curve (to compare the congruence between the prediction model and real-world evidence, R package rms version 6.4.0), and decision curve analyses (DCA) (a method for evaluating the benefits of the prediction model, R package rmda version 1.6 and ggplot2 version 3.3.6).
Statistical Analysis
The study followed the TRIPOD-AI guidelines, 28 and the checklist was shown in Supplemental Table 1. Statistical analysis was carried out using GraphPad Prism 7 (GraphPad Software, San Diego, CA, USA) and R version 4.2.1 (R Foundation for Statistical Computing, Vienna, Austria; https://www.R-project.org/). Differences between groups were analyzed by χ2 tests. Two-tailed t-tests assuming equal variances were used to compare 2 groups, and Welch’s correction was used if the P-value of the F test to compare variances was <.05. Non-normally distributed data were analyzed by Mann-Whitney U-tests. The model assumptions were tested using the likelihood ratio test. Hosmer-Lemeshow goodness of fit test was used to assess the goodness of fit of the model. A P-value <.05 was considered statistically significant.
Results
Baseline Patient Characteristics
A total of 129 patients with advanced HPSCC were selected for the present study. Their baseline characteristics, including age, sex, tumor grade, T-stage, N-stage, metastasis, and clinical stage are presented in Table 1. There was no significant difference in age (t = 0.669, P = .505) or sex (χ2 = 1.196, P = .274) between the nonresponse and response groups. Forty-seven patients received TPK (Table 2), with significant differences among the various N-stages (χ2 = 8.625, P = .035) in the TPK group. The baseline characteristics of the patients in the TP group are shown in Supplemental Table 2.
Baseline Characteristics of All Patients.

Baseline Characteristics of Patients in the TPK Group.

The Higher ORR in the TPK Group
We compared the blood test results between responders and nonresponders in the TPK and TP groups separately. In the TPK group, lymphocytes (1.623 ± 0.830 and 1.073 ± 0.415, P = .012), RBCs (4.465 ± 0.706 and 3.855 ± 0.649, P = .006), Hb (131.355 ± 14.373 and 116.688 ± 24.234, P = .037), and LMR (3.036 ± 1.541 and 2.231 ± 1.233, P = .035) levels were all significantly higher while basophils (0.028 ± 0.018 and 0.044 ± 0.020, P = .003) and PLR (194.790 ± 123.195 and 299.067 ± 169.853, P = .007) levels were significantly lower among the responders compared with the nonresponders (Figure 1A; Supplemental Figure 1A). In the TP group, neutrophils (4.577 ± 2.219 and 5.225 ± 2.030, P = .036) and calcium levels (2.252 ± 0.090 and 2.332 ± 0.134, P = .002) were significantly lower in the responders compared with the nonresponders (Figure 1B; Supplemental Figure 1B). Notably, the ORR was significantly higher in the TPK group (66.0%) compared with the TP group (47.6%) (χ2 = 4.074, P = .044) (Figure 1C).

Higher ORR in the TPK group. (A) Lymphocytes (U = 136, P = .012), RBCs (t = 2.880, P = .006), and Hb levels (t = 2.227, P = .037) were higher in responders compared with nonresponders in the TPK group, while basophil levels were lower (U = 379.5, P = .003). (B) In the TP group, neutrophils (U = 1065.5, P = .036) and calcium (U = 1152.5, P = .002) were lower in responders compared with nonresponders. (C) The ORR was higher in the TPK group (16 nonresponders and 31 responders, 66.0%) compared with the TP group (43 nonresponders and 39 responders, 47.6%) (χ2 = 4.074, P = .044). ORR, objective response; RBC, red blood cell; Hb, hemoglobin.
Correlations of Lymphocytes, Basophils, and RBC Levels With ORR in the TPK Group
We conducted LASSO, univariable, and multivariable logistic regression analyses to screen the potential independent predictors. LASSO identified 7 characteristics (lymphocytes, eosinophils, basophils, RBCs, Hb, globulin, creatinine) that were strongly associated with response when the optimal λ value was 0.046 (Figure 2A,B). Univariable logistic regression identified lymphocytes [odds ratio (OR) 4.594, 95% confidence interval (CI) 1.313-16.081, P = .017], basophils (OR 0.000, 95% CI 0.000-0.000, P = .013), RBCs (OR 4.297, 95% CI 1.380-13.374, P = .012), and Hb (OR 1.044, 95% CI 1.007-1.083, P = .019) as potential predictive factors (Figure 2C), and multivariable logistic regression revealed that lymphocytes (OR 17.009, 95% CI 1.515-190.927, P = .022), basophils (OR 0.000, 95% CI 0.000-0.000, P = .006), and RBCs (OR 14.065, 95% CI 1.129-175.238, P = .040) were independent predictors of response to TPK treatment (Figure 3A).

Variable selection using LASSO and univariable logistic regression. (A) Tenfold cross-validation for tuning parameter selection in the LASSO model. (B) Seven characteristics were strongly associated with response when the optimal λ value was 0.046. (C) Univariable logistic regression identified lymphocytes (OR 4.594, 95% CI 1.313-16.081, P = .017), basophils (OR 0.000, 95% CI 0.000-0.000, P = .013), RBCs (OR 4.297, 95% CI 1.380-13.374, P = .012), and Hb (OR 1.044, 95% CI 1.007-1.083, P = .019) as potential predictive factors. LASSO, least absolute shrinkage, and selection operator; RBC, red blood cell; Hb, hemoglobin; OR, odds ratio; CI, confidence interval.
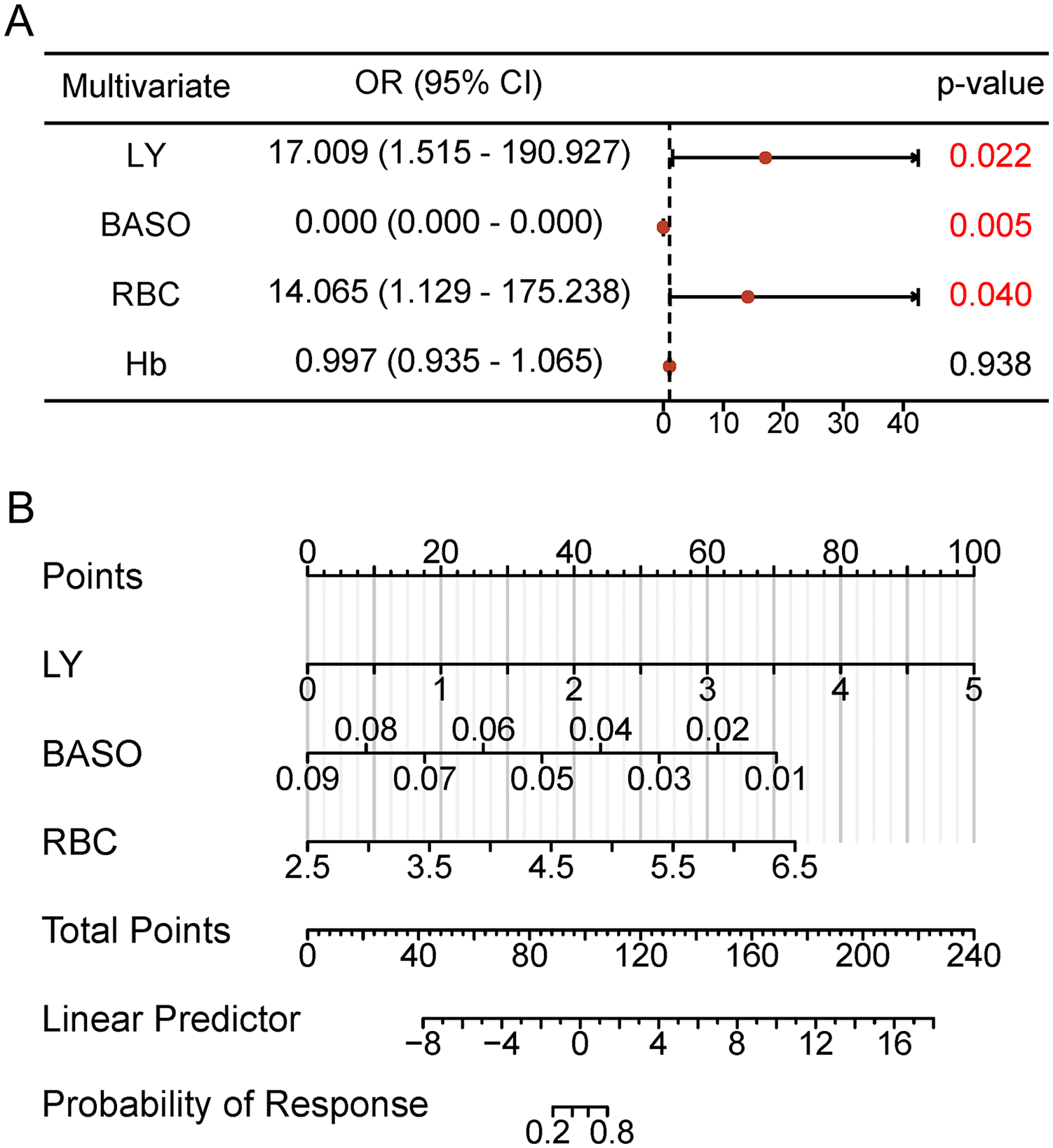
Multivariable logistic regression and nomogram. (A) Multivariable logistic regression identified lymphocytes (OR 17.009, 95% CI 1.515-190.927, P = .022), basophils (OR 0.000, 95% CI 0.000-0.000, P = .006), and RBCs (OR 14.065, 95% CI 1.129-175.238, P = .040) as independent predictors. (B) A nomogram was constructed based on the levels of lymphocytes, basophils, and RBCs for predicting the response in the TPK group. To estimate the probability of response, calculate points by drawing a straight line from the variable value to the axis labeled “Points” for each variable. Sum all points and draw a straight line from the axis labeled “Total Points” to “Probability of Response.” RBC, red blood cell; OR, odds ratio; CI, confidence interval.
Development and Validation of Nomogram
Finally, we constructed a nomogram to predict the response to TPK treatment, based on the results of multivariable logistic regression analysis (likelihood ratio test, χ2 = 29.793, P = 1.53e-06). The Hosmer-Lemeshow goodness of fit test indicated that the model fitted the data well (χ2 = 4.271, P = .832). The C-index of the nomogram for response prediction was 0.925 (0.848-1.002) (Figure 3B; Supplemental Table 3). The detailed logistic regression model was shown in the Supplemental Figure 2A. To avoid the low OR of BASO and misunderstandings, we incorporated a new variable called 100*BASO (BASO value multiplied by 100) and built another nomogram through LY, 100*BASO and RBC (Supplemental Figure 2B,C), the 100*BASO generated an OR of 0.288 (0.120-0.693).
The ROC and PR curves both showed that the resulting risk score had good discrimination, with an AUC of 0.925 (95% CI 0.848-1.000, sensitivity 90.323%, specificity 87.5%, Youden index 0.778) (Figure 4A,B). The calibration plot of response probabilities showed good agreement between the nomogram prediction and actual observations (Figure 4C). We also assessed the clinical usefulness of the nomogram by DCA, which indicated that using this nomogram with most threshold probabilities to predict the response would be better than the “All” or “None” schemes (Figure 4D).

Validation of the nomogram and evaluation of predictive performance. (A, B) The ROC and PR curves showed that the resulting risk score had good discrimination, with an AUC of 0.925. (C) The calibration plot of response probabilities showed good agreement between the nomogram prediction and actual observations. (D) DCA affirmed that the nomogram had important clinical value. ROC, receiver operating characteristic; PR, precision recall; AUC, area under the curve; DCA, decision curve analyses.
Discussion
Hypopharyngeal cancer, especially environmentally-related HPSCC, is characterized by submucosal tumor spread and vascular invasion, making it the most lethal type of head and neck cancer.1,29 The anatomy and location of the hypopharynx mean that most patients with HPSCC are asymptomatic in the early phase and thus show late presentation, 30 with about 70% to 90% of newly diagnosed patients presenting with stage III or IV disease.31,32 The incidence of HSPCC is relatively low and shows geographical variation, being <0.5 per 100,000 population in Eastern Asia, Africa, and Northern Europe, and approximately 2.5 per 100,000 in India, Brazil, and Central and Western Europe. 2 About 84,000 new cases and 39,000 new deaths were reported in 185 countries in 2020. 3 HPSCC is thus often studied together with laryngeal squamous cell carcinoma, even though they differ greatly in terms of their biology and prognosis.33-36 We therefore consider that it is not appropriate to simply extrapolate the results of studies of laryngeal squamous cell carcinoma and subgroup analyses of multi-site HNSCC research to the hypopharynx. The current study thus focused specifically on patients with locally advanced HPSCC, with the aim of analyzing the efficacy of NACT plus pembrolizumab.
The present results showed that the ORR of HPSCC to NACT was approximately 48%. NACT is considered to be an attractive laryngeal-preservation strategy and has been increasingly applied as the initial treatment for locally advanced HPSCC in clinical practice in recent years. 37 The current standard NACT regimens for hypopharyngeal carcinoma are based on TPF, including taxanes (docetaxel, paclitaxel, albumin paclitaxel), platinum (cisplatin, carboplatin), and 5-fluorouracil38-40; however, the toxicity of standard TPF is greater in Asian patients compared with white patients, thus limiting its clinical application, 41 while acute toxicity also impedes the completion of subsequent therapies. 42 Increasing studies have thus focused on developing new NACT treatments with reduced toxicity, including TP, which has been widely used to treat HPSCC in clinical practice.17,20,43 Li et al compared the ORRs of TP and TPF in patients with HPSCC and revealed no differences in ORR or overall survival, with an ORR for TP of about 50.98% (26 responders and 25 nonresponders). 20 These data are consistent with the current results (χ2 = 0.147, P = .701).
To the best of our knowledge, the present study is the first to show that pembrolizumab could significantly improve the ORR compared with TP alone in patients with HPSCC. Furthermore, our clinical observations suggested that TPK treatment was also tolerable in terms of adverse events. Pembrolizumab is a PD-1/PD-L1 blocker and can improve the antitumor ability of T cells.26,27 Previous studies established pembrolizumab as a first-line treatment for recurrent or metastatic HNSCC,23,26,27,44 and pembrolizumab plus NACT has been applied to various types of tumors, such as breast cancer,45-47 esophageal squamous cell carcinoma, 48 bladder cancer, 49 and lung carcinoma.50,51 Nanda et al conducted a randomized phase 3 clinical trial including 300 patients with breast cancer and revealed that pembrolizumab more than doubled the estimated pathologic CR and improved event-free survival rates. 45 Wakelee et al analyzed 797 patients with non-small-cell lung cancer and showed that pembrolizumab with chemotherapy followed by resection and adjuvant pembrolizumab could improve the pathological CR, major pathological response, and event-free survival compared with chemotherapy alone followed by surgery. 50
Few studies, however, have focused on the efficacy of NACT with pembrolizumab in HNSCC or HPSCC. Wang et al conducted a prospective, single-arm, single-center clinical trial including 22 patients with HNSCC, and reported that preoperative treatment with pembrolizumab and NACT had a high pathological CR rate, with no significant impact on surgical safety. 52 NACT combined with pembrolizumab has been used in our department as an alternative treatment for locally advanced HPSCC since 2019. The current study retrospectively analyzed 129 patients with HPSCC and revealed that pembrolizumab significantly improved the ORR of TP. Furthermore, to predict the response of patients to TPK, we also constructed a nomogram based on the blood test results. The nomogram showed that pretreatment levels of lymphocytes and RBCs were independent predictors of a high ORR, while basophils were an independent predictor of a low ORR.
Lymphocytes, mainly including T cells, B cells, and natural killer cells, are critical cells for the immune surveillance of malignant tumors. 53 Several studies have focused on improving the antitumor ability of lymphocytes in various tumors.54,55 In addition, NLR, PLR, LMR, and LWR have been shown to predict the response to NACT in patients with various tumors, including gastric cancer, esophageal squamous cell carcinoma, and breast carcinoma.56-59 In the present study, there were no differences in these ratios between the nonresponders and responders in the TP group; however, PLR was significantly higher and LMR was significantly lower in nonresponders in the TPK group. The main reason for this could be the higher lymphocyte levels among responders. Pan et al analyzed 25 patients with HNSCC and 25 with salivary gland cancer treated with pembrolizumab and vorinostat and revealed that higher pretreatment NLR and lower pretreatment lymphocytes correlated with worse overall and progression-free survival, but there was no correlation between NLR and ORR. 60 This is consistent with the current results.
The present study also showed that pretreatment RBCs and basophil levels were independent predictors of response in the TPK group. RBCs and basophils are widely used in clinical practice and are routinely measured in blood tests. RBCs, including Hb, are an important index of anemia, with few functions other than transporting oxygen.61,62 Herein, we found that a higher pretreatment RBC level was related to the response in the TPK group. This may be related to the ability of RBCs to enhance drug delivery.63,64 To the best of our knowledge, this study provides the first evidence to suggest that lower basophil levels could predict the response to TPK treatment.
Basophils account for a small proportion of the circulating leukocytes in the bloodstream and play an important role in the induction of inflammatory and type I allergic responses. 65 The role of basophils in tumors has recently attracted increasing attention, especially in terms of the relationship between allergy and cancer. 66 Moreover, clinical studies found that basophil levels were associated with a worse prognosis in various tumors, such as prostate cancer, gastric carcinoma, and bladder cancer.67-69 He et al enrolled 448 patients with gastric cancer and found that tumor-infiltrating basophils determined a significantly inferior therapeutic response to fluorouracil-based adjuvant chemotherapy. 68 Herein, we revealed that basophil level was an independent adverse predictive factor for the response to TPK treatment in patients with locally advanced HPSCC.
Despite our efforts to ensure data accuracy and statistical robustness, this study has some limitations. First, its retrospective nature introduces the possibility of selection bias and confounding factors. Second, confinement to a single center limits the generalizability of our results. Third, owing to the small number of patients who received TPK treatment, it was not feasible to split the data set into separate training and validation sets. Hence, an independent validation of the findings in a larger sample size will need to be conducted in future studies.
Conclusions
In summary, the current study showed that pembrolizumab could increase the ORR from 48% to 66% in patients with locally advanced HPSCC treated with TP neoadjuvant chemotherapy. Furthermore, we constructed a nomogram based on pretreatment levels of lymphocytes, RBCs, and basophils to predict the response to TPK treatment, which might help to guide treatment decisions and reduce the waste of medical resources.
Supplemental Material
sj-docx-3-ohn-10.1177_19160216251318255 – Supplemental material for Nomogram Prediction of Response to Neoadjuvant Chemotherapy Plus Pembrolizumab in Locally Advanced Hypopharyngeal Squamous Cell Carcinoma
Supplemental material, sj-docx-3-ohn-10.1177_19160216251318255 for Nomogram Prediction of Response to Neoadjuvant Chemotherapy Plus Pembrolizumab in Locally Advanced Hypopharyngeal Squamous Cell Carcinoma by Zhangwei Hu, Yi Chen, Renqiang Ma, Wei Sun, Lin Chen, Zhimou Cai, Weiping Wen and Wenbin Lei in Journal of Otolaryngology - Head & Neck Surgery
Supplemental Material
sj-docx-4-ohn-10.1177_19160216251318255 – Supplemental material for Nomogram Prediction of Response to Neoadjuvant Chemotherapy Plus Pembrolizumab in Locally Advanced Hypopharyngeal Squamous Cell Carcinoma
Supplemental material, sj-docx-4-ohn-10.1177_19160216251318255 for Nomogram Prediction of Response to Neoadjuvant Chemotherapy Plus Pembrolizumab in Locally Advanced Hypopharyngeal Squamous Cell Carcinoma by Zhangwei Hu, Yi Chen, Renqiang Ma, Wei Sun, Lin Chen, Zhimou Cai, Weiping Wen and Wenbin Lei in Journal of Otolaryngology - Head & Neck Surgery
Supplemental Material
sj-tif-1-ohn-10.1177_19160216251318255 – Supplemental material for Nomogram Prediction of Response to Neoadjuvant Chemotherapy Plus Pembrolizumab in Locally Advanced Hypopharyngeal Squamous Cell Carcinoma
Supplemental material, sj-tif-1-ohn-10.1177_19160216251318255 for Nomogram Prediction of Response to Neoadjuvant Chemotherapy Plus Pembrolizumab in Locally Advanced Hypopharyngeal Squamous Cell Carcinoma by Zhangwei Hu, Yi Chen, Renqiang Ma, Wei Sun, Lin Chen, Zhimou Cai, Weiping Wen and Wenbin Lei in Journal of Otolaryngology - Head & Neck Surgery
Supplemental Material
sj-tif-2-ohn-10.1177_19160216251318255 – Supplemental material for Nomogram Prediction of Response to Neoadjuvant Chemotherapy Plus Pembrolizumab in Locally Advanced Hypopharyngeal Squamous Cell Carcinoma
Supplemental material, sj-tif-2-ohn-10.1177_19160216251318255 for Nomogram Prediction of Response to Neoadjuvant Chemotherapy Plus Pembrolizumab in Locally Advanced Hypopharyngeal Squamous Cell Carcinoma by Zhangwei Hu, Yi Chen, Renqiang Ma, Wei Sun, Lin Chen, Zhimou Cai, Weiping Wen and Wenbin Lei in Journal of Otolaryngology - Head & Neck Surgery
Supplemental Material
sj-xlsx-5-ohn-10.1177_19160216251318255 – Supplemental material for Nomogram Prediction of Response to Neoadjuvant Chemotherapy Plus Pembrolizumab in Locally Advanced Hypopharyngeal Squamous Cell Carcinoma
Supplemental material, sj-xlsx-5-ohn-10.1177_19160216251318255 for Nomogram Prediction of Response to Neoadjuvant Chemotherapy Plus Pembrolizumab in Locally Advanced Hypopharyngeal Squamous Cell Carcinoma by Zhangwei Hu, Yi Chen, Renqiang Ma, Wei Sun, Lin Chen, Zhimou Cai, Weiping Wen and Wenbin Lei in Journal of Otolaryngology - Head & Neck Surgery
Footnotes
Acknowledgements
None.
Author Contributions
Z.H.: Acquisition, analysis, and interpretation of data, drafting of the manuscript. Y.C.: Acquisition, analysis, and interpretation of data. R.M.: Acquisition, analysis, and interpretation of data. W.S.: Acquisition, analysis, and interpretation of data. L.C.: Acquisition, analysis, or interpretation of data. Z.C.: Acquisition, analysis, or interpretation of data. W.W.: Design of the research, critical revision of the manuscript for important intellectual content. W.L.: Design of the research, critical revision of the manuscript for important intellectual content.
Data Availability Statement
The data analyzed in this study are available on reasonable request from the corresponding author. A protocol was not prepared. This study was not registered. The data are not publicly available due to privacy or ethical restrictions.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the National Natural Science Foundation of China (82403372, 81972528); Guangdong Basic and Applied Basic Research Foundation (2022A1515110147); Guangzhou Basic and Applied Basic Special Research Project Young PhD “Qi Hang” Foundation (2025A04J4018); China Postdoctoral Science Foundation (2021M703712).
Ethics Approval and Informed Consent
The First Affiliated Hospital of Sun Yat-Sen University Institutional Review Board approval was obtained ([2023]283). The need for informed consent was waived because only retrospective, de-identified data were used.
Supplemental Material
Additional supporting information is available in the online version of the article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
