Abstract
Importance
Idiopathic facial nerve palsy (FNP) has devastating sequelae and is potentially linked to coronavirus disease-19 (COVID-19).
Objective
The rate of FNP was compared in the pandemic versus pre-pandemic periods. Furthermore, the risk of FNP was estimated among the COVID-19 vaccinated group.
Design
Systematic review and meta-analysis.
Setting
An electronic search was conducted in 7 databases: Scopus, Web of Science core collection, PubMed, Cochrane Central Register of Controlled Trials, MEDLINE, Embase, and CINAHL.
Participants
English observational studies investigating an association between idiopathic FNP and COVID-19 or its vaccination were included, irrespective of patients’ demographics.
Exposures
COVID-19 or COVID-19 vaccine.
Main Outcome Measures
Change in FNP incidence between the pre-pandemic and pandemic periods; risk of developing FNP in individuals vaccinated against COVID-19 compared to those who were unvaccinated against COVID-19.
Results
After excluding duplicates, the search yielded 906 related articles, of which 118 articles were included. The risk of FNP was statistically significantly higher during the COVID-19 pandemic than the pre-pandemic period (RR: 1.68, [95% CI: 1.16-2.43], P = .01). A nonsignificant increase in FNP risk was identified among COVID-19 vaccinated individuals compared to unvaccinated individuals (overall OR: 1.07, [95% CI: 0.85-1.35], P = .55).
Conclusions and Relevance
A remarkable increase in FNP rates was identified during the pandemic compared to pre-pandemic, which seemed unlikely to be attributed to COVID-19 vaccination.

Key Message
There has been a significant increase in facial nerve palsy during the pandemic.
There is a lack of supportive evidence linking COVID-19 vaccination with facial nerve palsy.
Introduction
The coronavirus infection disease-19 (COVID-19) pandemic was caused by the emergence of a fast-spreading virus with high morbidity and mortality rates. According to the World Health Organization (WHO), this pandemic has led to approximately 405 million cases and more than 5 million deaths worldwide as of February 11, 2022. 1 Respiratory manifestations are a common presentation of COVID-19; however, a range of signs and symptoms have been reported as first manifestations,2-4 including facial nerve palsy (FNP).5-7
The pathogenesis of FNP is not completely understood; however, the viral inflammation of the nerve sheath is one possible etiology.8,9 It has been hypothesized that the negative neurotropic effect of severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), in which it invades the central nervous system via the olfactory nerve, plays a significant role.10,11 FNP incidence has increased during the COVID-19 pandemic,12,13 and a connection between COVID-19 vaccines and FNP has been suspected. 14 Furthermore, there is evidence of an FNP incidence rate 1.5 to 3 times higher among individuals inoculated with mRNA vaccines than the general population. 15 However, such evidence was insufficient for the U.S. Food and Drug Administration to conclude causality. 16
There is limited evidence addressing comprehensively the association of SARS-CoV-2 infection and vaccination with FNP. 17 To our knowledge, there is a paucity of literature estimating the incidence of FNP during the COVID-19 pandemic using a comparative approach with pre-pandemic data. Therefore, we carried out this meta-analysis to compare pre-pandemic FNP rates with those during the pandemic and to determine the FNP risk among individuals who were vaccinated against COVID-19. Second, we pooled original data of individually reported patients to assess demographics, clinical characteristics, treatments, and outcomes of post-COVID-19 FNP (PC-FNP).
Methods
Protocol and Registration
The protocol of this systematic review has been registered in the International Prospective Register of Systematic Reviews (PROSPERO; ID: CRD42021274868). The Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) recommendations were followed in reporting this systematic review. 18 No ethical approval was required for the current study.
Search Strategy and Study Selection
With the use of relevant search words and controlled vocabularies, a systematic electronic search was conducted in 7 databases: Scopus, Web of Science core collection, PubMed, Cochrane Central Register of Controlled Trials (CENTRAL), MEDLINE (Ovid), Embase, and CINAHL. The eligibility period was from inception to January 6, 2023. Further details regarding the search strategy are found in Appendix A. Epidemiological observational studies in English estimating FNP incidence during the COVID-19 pandemic, regardless of patient demographics, were included. The comparison between pre-pandemic and pandemic periods was drawn from the original studies included in our meta-analysis. These studies independently defined and compared the pre-pandemic and pandemic periods according to their specific designs. Therefore, we did not establish our own timeframe but adhered to the timeframes used by the original studies to align with their methodologies. Similarly, epidemiological observational studies investigating an association between FNP and COVID-19 vaccination, regardless of vaccine type, were included. Further, all relevant original cases reported in the literature—such as cases reports, cases series, or other formats—where idiopathic FNP developed within 45 days of the onset of COVID-19 infection, confirmed by positive polymerase chain reaction (PCR) test, were eligible for inclusion in our PC-FNP original cohort. The 45-day timeframe was chosen to capture cases of FNP that are temporally close to the COVID-19 infection, thereby reflecting a plausible acute post-infection complication. Patients with a concomitant neurological disorder (eg, Guillain–Barré syndrome or stroke) or another secondary cause of peripheral facial palsy, such as otitis media, trauma, or Ramsay Hunt syndrome, were not included in this review. Other exclusion criteria were duplicate records, data that could not be extracted reliably, abstract-only articles, theses, books, preprints, reviews, and conference papers. Only the editorials and commentaries that reported original data were included. All identified articles were independently screened by 2 authors (A.A. and F.N.). Any disagreements were resolved through discussion with a third author (A.M.).
Data Extraction
Data extraction was carried out by 2 independent reviewers (A.A. and S.N.) using the same form. A third independent reviewer (A.M.) audited the data to ensure accuracy. All disagreements and discrepancies were resolved by discussion.
Quality Assessment
Three independent reviewers (A.A., F.N., and S.N.) assessed the risk of bias in the observational cohort, cross-sectional, case-control studies, case reports, and case series included in the review. The NIH Quality Assessment tools were used to assess the quality of observational cohort and cross-section studies, case-control studies, and case series, while The Joanna Briggs Institute Critical Appraisal tool was used to assess the quality of case reports.19,20 Any discrepancy between the reviewers was resolved by discussion.
Quantitative Synthesis (Meta-Analysis) of Epidemiological Studies
A meta-analysis was carried out concerning the change in FNP incidence between the pre-pandemic and pandemic periods (Meta-I). A subsequent meta-analysis (Meta-II) was executed to assess the risk of FNP development among individuals who had mRNA-based COVID-19 vaccine compared to those who were unvaccinated. If a relevant epidemiological study reported separate results for both the first and second doses of a COVID-19 vaccine, we included only the data corresponding to the first dose in our analysis. In Meta-II analysis, we initially pooled data from all relevant epidemiological studies, regardless of study design. Since the event rates of the outcome (FNP) were low across the cohort studies, the risk ratios (RRs) from the respective cohort studies were considered equivalent to odds ratios (ORs) in the initial analysis. Subsequently, a subgroup analysis was performed to address potential sources of heterogeneity, including separating the pooling of RRs and ORs according to study design (cohort and case-control study, respectively). To achieve a more symmetrical distribution and approximate normality, we calculated logarithmic odds ratios (log ORs) and logarithmic risk ratios (log RRs), along with their respective 95% CIs, where applicable, and then back-transformed to obtain exponential effect sizes (with corresponding 95% CIs). A forest plot was generated to demonstrate the summary effect size. To assess the variation across studies (heterogeneity), Cochran’s Q-statistic and I2 statistics were used. If significant heterogeneity was observed (I2 > 50%), random-effects model was employed to pool the effect sizes, and a leave-one-out sensitivity analysis was conducted to evaluate the robustness and stability of the overall results. Both the Funnel plot test and Egger’s test were used to assess for publication bias. The meta-analysis was performed by using Stata Statistical Software (StataCorp. 2023. Stata Statistical Software: Release 18. College Station, TX: StataCorp LLC). All tests were two-sided, and P < .05 indicated statistical significance.
Pooled Analysis of PC-FNP Cohort
Pooled descriptive analyses were performed for demographics such as age and sex, clinical characteristics including latency period, laterality, and severity at presentation, treatments, and recovery outcome, among individually reported cases. The latency period was defined as the interval from the onset of SARS-CoV-2 infection—either the first appearance of symptoms or the date of a positive COVID-19 test, whichever occurred first—until the development of FNP. FNP severity was measured by the House-Brackmann (HB) grading system, which ranges from I (normal) to VI (complete paralysis). Inferential analyses of individually reported data were carried out based on the availability of data for each outcome from the included reports. Univariate logistic regression analysis was performed to identify factors, including age and latency period, potentially associated with complete recovery. Kaplan–Meier curves were plotted for complete recovery estimates with respect to multiple variables: age, sex, latency period, laterality, severity at presentation, and treatment. The survival function analyzed the number of events (complete recovery) with respect to time-to-recovery or last follow-up. Log-rank analysis was employed to compare the survival distributions between groups defined by categorical variables such as age and treatment. All statistical tests were two-sided, and P < .05 indicated statistical significance. Statistical analysis was performed using Stata Statistical Software, as previously described.
Results
Identification and Selection of Studies
After excluding duplicates, 906 articles were identified. Title and abstract screening yielded 241 records for full-text screening. A total of 118 studies were included (Figure 1).

Flow chart depicting the method of study selection.
Study Characteristics and Quality of the Included Studies
Most included articles were individual-level studies (37%), followed by epidemiological cohort studies (27%). Epidemiological studies about FNP association with COVID-19 infection (n = 24) and vaccination (n = 52) are summarized in Appendix B. A total of 44 studies provided individual patient data, which we compiled into the PC-FNP original cohort (Appendix C). This cohort consists of reported cases of FNP associated with COVID-19 infection (n = 64), confirmed through positive PCR testing. Most of the individually reported cases were from Brazil (n = 13), followed by Turkey (n = 10). The quality of the majority of studies varied from good to fair (Appendix D).
FNP Incidence in the Pre-Pandemic Versus Pandemic Period (Meta-I)
Of 24 epidemiological studies on the association of FNP and COVID-19, 5 were comparable and estimated the rate of FNP in the pandemic period versus the pre-pandemic period. Our analysis showed that FNP risk was significantly higher during the COVID-19 pandemic compared with the pre-pandemic period, with a pooled risk ratio of 1.68 (95% CI: 1.16-2.43; P = .01; Figure 2A); this was associated with statistically significant heterogeneity (I2 = 80.4%, P < .001). Upon visual inspection of the forest plot, the study by Hogg et al. 21 was identified as a possible outlier. A subsequent subanalysis excluding the study by Hogg et al. 21 revealed that heterogeneity was slightly reduced but remained statistically significant (I2 = 65.5%, P = .03). Further investigation through a leave-one-out sensitivity analysis failed to determine the source of statistical heterogeneity. However, it revealed that the overall results remained stable and consistent, with no single study being significantly influential (Figure 2B). Funnel plot and Egger’s test demonstrated a significant publication bias for the comparison of FNP rate between pre-pandemic and pandemic (P < .001; Appendix E).

(A) Forest plot showing pooled risk ratio of FNP in the COVID-19 pandemic versus pre-pandemic period, (B) The respective sensitivity analysis using the leave-one-out method.
Association of FNP With COVID-19 Vaccine (Meta-II)
A total of 52 epidemiological studies described the association of FNP with COVID-19 vaccination, 9 of which compared unvaccinated groups with those who received COVID-19 vaccines and were amenable to meta-analysis. The overall findings revealed a nonsignificant increase in FNP risk among COVID-19 vaccinated compared to unvaccinated individuals (overall OR: 1.07, [95% CI: 0.85-1.35], P = .55; Figure 3A). This was associated with statistically significant heterogeneity (I2 = 71.1%). The sensitivity analysis indicated that excluding the study of McMurry et al. 22 showed a statistically significant increase in FNP among the vaccinated group compared to the unvaccinated group (OR: 1.22, [95% CI: 1.04-1.43], P = .02; Figure 3B). A subgroup analysis was executed based on the study design (cohort and case-control study). Pooled results from cohort studies22-27 showed no significant risk of FNP among COVID-19-vaccinated compared with unvaccinated cohorts (RR: 1.01, [95% CI: 0.76-1.33], P = .95; I2 = 78.6%; Figure 3A). Likewise, the case-control studies28-30 yielded nonsignificant evidence of increased odds of FNP in the vaccinated group compared with the unvaccinated group (OR: 1.36, [95% CI: 0.98-1.89], P = .07; I2 = 6.7%; Figure 3A). There was no evidence of publication bias for the relationship of the COVID-19 vaccine with FNP, based on Egger’s test (P = .11; Appendix E).
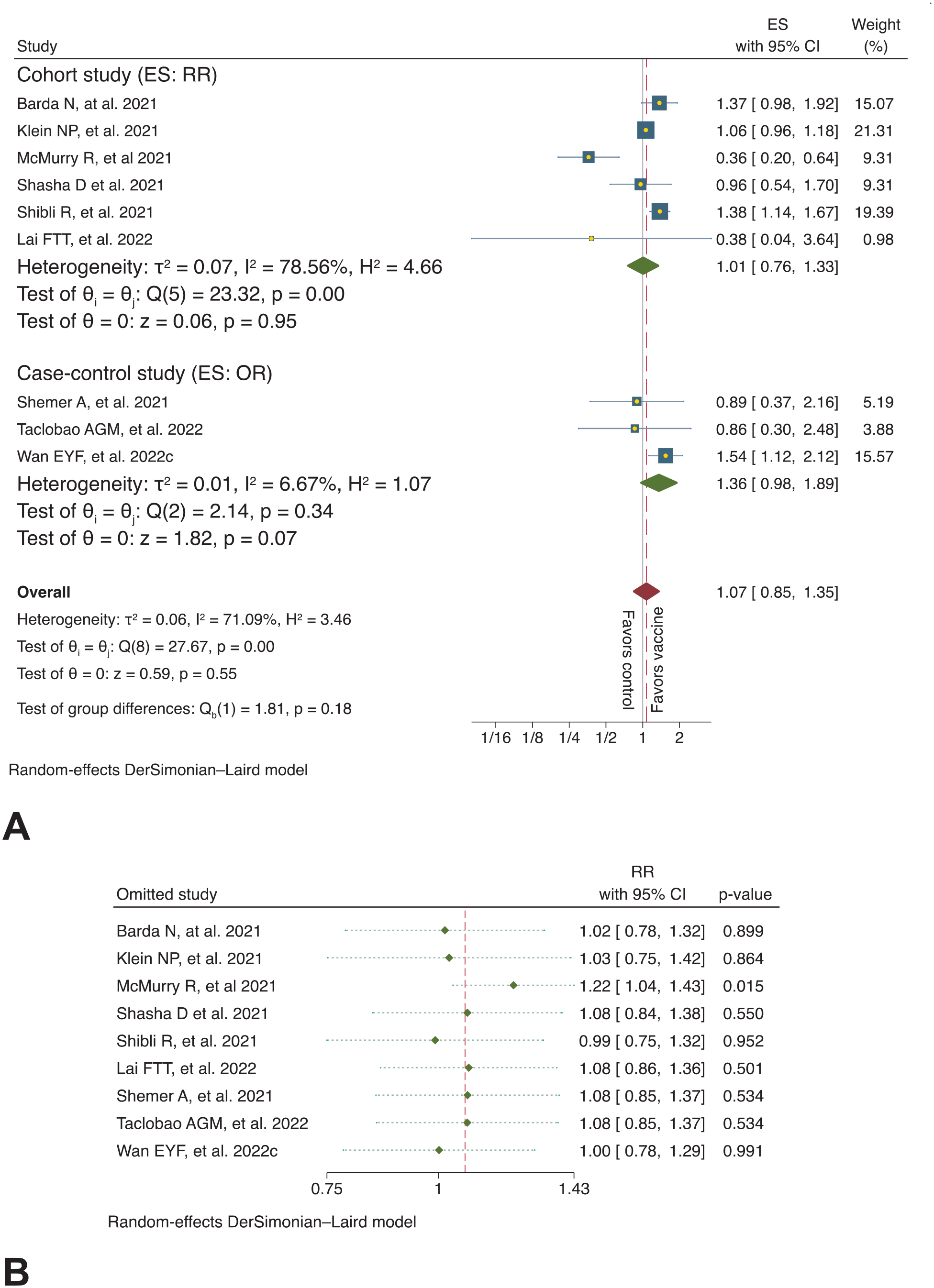
(A) Forest plot showing summary effect size of occurrence of FNP in the COVID-19 vaccinated versus unvaccinated groups, with subgroup analysis (by study design), (B) The respective sensitivity analysis using the leave-one-out method.
Characteristics of PC-FNP Cohort
A total of 64 cases were reported individually; 37 of them were men, with a male-to-female ratio of 1.4:1. Of the reported PC-FNP cases, 22 (46%) developed the condition within the first week following COVID-19 infection (Figure 4A). The majority were unilateral FNP cases (91%), and most of these (62%) involved the right side. In cases including HB grading of FNP (n = 32), grade IV was predominant at the presentation (44%; IQR: III-IV grade). Most patients (84%) were treated with systemic steroids, whereas antiviral agents were used less frequently (39%). Among all cases treated with antiviral regimens, Acyclovir and Valacyclovir were the most frequently used, each accounting for 33%. Table 1 provides a further descriptive summary, while individual-level details are included in Appendix C.

(A) Time points of FNP development following SARS-CoV-2 infection, (B) House-Brackmann grade at presentation, (C) Rate of FNP recovery, (D) Recovery from FNP according to age, (E) Recovery from FNP according to latency period.
Summary of Demographics, Clinical Characteristics, Treatments, and Outcome of Facial Nerve Palsy Following SARS-CoV-2 Infection, From Individually Reported Cases.

Abbreviations: FNP, facial nerve palsy; IQR, inter-quartile range.
Variable has missing values due to unavailability of data.
Data are presented as N (%) unless otherwise specified.
Total number of patients who received an antiviral agent(s).
A few patients received more than one antiviral agent.
Recovery from PC-FNP
Figure 4C displays the recovery rate of the PC-FNP patients. With the median follow-up time of 21 days, complete recovery occurred in 57% of patients. When compared to the expected recovery rate (83.3%) for FNP disease (unrelated to COVID-19), 31 the observed complete recovery rate was significantly lower in our COVID-19 cohort (one-sample proportion z-test; z = −5.46; P < .001). Following the FNP presentation, younger patients were more likely to experience complete recovery compared to older ones (P = .003; Figure 4D). However, both young (<45) and old individuals (≥45) had almost similar median time-to-complete recovery (28 and 30, respectively, P = .62; Figure 5A). There was no statistically significant difference in the complete recovery rate with respect to the latency period (P = .931; Figure 4E). Nevertheless, patients who developed FNP within the first 2 weeks of COVID-19 infection exhibited shorter median time-to-complete recovery compared to those of more than 14 days (30 and 60, respectively; Figure 5C). In addition, individuals with worse features of FNP at presentation (HB grade V and VI; median time: 90) had longer median time-to-complete recovery in reference to their counterparts (HB grade II-IV; median time: 30; Figure 5E). There was not a large difference in the median time-to-complete recovery among those with steroid-only treatment versus combined steroid-antiviral treatment versus the group of other treatments or no treatment (P = .30, Figure 5F).

Kaplan–Meier time-to-event curve depicting time-to-complete recovery for post-COVID-19 FNP cohort with respect to (A) age, (B) sex, (C) latency period, (D) laterality, (E) House-Brackmann grade at presentation, and (F) treatment.
Discussion
Summary of Findings
Our analysis showed that the incidence of FNP was significantly higher during the COVID-19 pandemic compared with the pre-pandemic period. However, there was no evidence for increased FNP risk among COVID-19-vaccinated subjects compared with unvaccinated subjects. Findings provided further insights into the characteristics and recovery of patients with COVID-19-associated FNP. Although complete recovery was the most common patient outcome, its rate was lower than expected based on previous data.
Plausible Mechanisms of COVID-19-Associated FNP
Our analysis found an increased incidence of FNP during the COVID-19 pandemic compared to pre-pandemic times. Although the exact cause is unclear, this rise may involve various mechanisms. Neurological disorders have been linked to SARS-CoV-2 infection in both acute and post-acute phases.32-35 FNP pathophysiology may involve microthrombi, 36 autoimmune responses, 37 hypercoagulability, 38 or direct neural invasion by SARS-CoV-2,39-42 all with potential negative neurotrophic effects. 41 Direct viral invasion also likely explains COVID-19-related anosmia. 42 Conversely, the familiarity with Bell’s palsy, along with the urgency to explore COVID-19’s neurological effects, may have prompted more clinicians to document and publish on FNP during the pandemic. This may have inflated the perceived association between COVID-19 and FNP due to heightened anxiety and increased diagnostic attention from clinicians during the pandemic. In addition, COVID-19 led to widespread lockdowns and increased healthcare-seeking behavior. These factors likely contributed to more frequent reporting and an apparent rise in case frequency, independent of direct viral pathology.
Comparative Epidemiology of COVID-19-Associated FNP
Our systematic review included data from 118 studies, 24 of which focused on FNP incidence during the COVID-19 pandemic. While prior studies have analyzed case reports and series, a comprehensive systematic review of original epidemiological data on FNP incidence amid COVID-19 appears to be lacking. Previous reviews by Turki et al. 43 and Namavarian et al. 44 linked COVID-19 to FNP, with the latter noting differences between COVID-19-related FNP and traditional Bell’s palsy.43,44
Although we found increased FNP incidence during the pandemic, this conclusion could be limited by variability across studies, such as differences in sample size, study design, study duration, study location, and demographics. For example, Mutlu et al. 45 and Gursoy et al. 46 conducted large retrospective studies in Turkey over 12 months but in different years (2019 and 2020), comparing cases to pre-pandemic data. In contrast, Hogg et al. 21 performed a prospective 5-month study in the UK on a smaller, exclusively pediatric population. Omitting Hogg et al.’s 21 data does not alter our overall results, despite their overall finding being perceived as an outlier. Zammit et al. 13 and Codeluppi et al. 12 conducted shorter retrospective studies with smaller populations. Our further analyses indicated that the overall findings of incidence estimates favoring the pandemic are robust and reliable despite the observed statistical heterogeneity.
Association of FNP With COVID-19 Vaccine
Our analysis found that the mRNA-based COVID-19 vaccines do not increase the risk of FNP development, supporting recent meta-analyses.47,48 Earlier in the pandemic, Tamaki et al. 49 reported that vaccinated individuals had a lower incidence of FNP compared to COVID-19-positive patients. Similarly, Shemer et al. 28 found no overrepresentation of vaccinated individuals among FNP cases and no significant change in incidence rates compared to previous pre-pandemic years. Gordon et al., 48 in a previous meta-analysis evaluating various COVID-19 vaccine manufacturers up to May 2022, concluded that the risk of FNP after COVID-19 vaccination is comparable to the unvaccinated population. However, their findings should be interpreted cautiously due to significant statistical heterogeneity that was not fully addressed, even after subgroup analyses. Our updated review revealed that performing a sensitive analysis, excluding McMurry et al.’s 22 data—which significantly influenced our overall conclusion–resulted in an emerging risk of FNP among the vaccinated population. McMurry et al. 22 uniquely compared both BNT162b2 or mRNA-1273 vaccines with unvaccinated participants and had a smaller sample than other studies. Nonetheless, Gordon et al. 48 found no significant difference in FNP risk between these 2 mRNA vaccines. Our study provides more definitive evidence regarding the association between mRNA-based COVID-19 vaccines and FNP risk, highlighting the need for large-scale, well-designed studies to assess rare adverse events, such as FNP, accurately.
During the initial rollout of COVID-19 vaccines, concerns emerged about FNP based on early post-authorization data from the FDA, necessitating further investigation. At that time, a cautious approach was necessary, given the limited data and the critical importance of vaccine safety surveillance. 50 Subsequent large-scale studies and meta-analyses, including ours, have provided a comprehensive overview showing no increased incidence of FNP in vaccinated populations compared to unvaccinated groups.
Clinical Characteristics and Management
Our analysis shows that younger patients have better outcomes after COVID-19-related FNP, consistent with historical data of FNP—unrelated to COVID-19—indicating that younger individuals generally have a better regenerative capacity. 51 Namavarian et al. 44 studied 72 COVID-19 FNP cases, 30 of which involved Guillain-Barré Syndrome (GBS), suggesting COVID-19 may cause a more severe yet paradoxically better-prognosed FNP. Although our study excluded GBS and other neurological conditions, it adds to the understanding of COVID-19-related facial paralysis by focusing on isolated cases.
In 2013, the American Academy of Otolaryngology-Head and Neck Surgery recommended early steroid use, with or without antivirals, for Bell’s palsy, but evidence for steroid use in the context of COVID-19 is limited. 52 Some studies discourage steroid use, generally, in COVID-19 infection due to severe side effects such as increased mortality 53 and delayed viral clearance,53,54 while others support low-to-moderate doses for critically ill patients.55,56 The WHO recommended corticosteroids for severe COVID-19 cases in 2020. In our review, systemic steroids were the main treatment for COVID-19-related FNP. Unexpectedly, the recovery times were similar regardless of treatment. Nevertheless, the timing of steroid administration was not analyzed due to data limitations.
The COVID-19 pandemic led to social isolation, quarantine, economic stress, and delayed medical care, potentially worsening FNP cases, increasing the risk of incomplete recovery, and delaying the recovery process.57-60 This may also explain the similar recovery times between patients who received steroids and those untreated, as steroids may have been initiated later in the disease course.
Limitations
Our study has several limitations. First, the systematic search was conducted without the prior consultation of an institutional librarian. In addition, only studies published in English were included in the analysis, introducing a potential language-related selection bias that may have excluded relevant studies from non-English-speaking countries. This limitation is particularly significant given that COVID-19 first emerged in Asian countries, such as China, where some research may not be readily accessible in English. Consequently, our findings may underestimate the true incidence of FNP related to COVID-19 in these regions. At the time we started our search, we faced certain resource constraints, including limited accessibility to translational resources. Furthermore, the included studies were carried out in different countries with various healthcare systems. This results in heterogeneity of data, impacting the validity of the findings. There were possible confounders that could not be considered and analyzed, including comorbidities, medication use, and specific conditions that might impact the risk of FNP. Finally, differences in follow-up periods across the included reports could result in many censored cases in the survival analysis. Thus, these results should be interpreted with caution, as censored data may reduce the robustness of the estimate.
Conclusion
This analysis suggests a higher incidence of FNP during the COVID-19 pandemic compared to pre-pandemic times. COVID-19-related FNP appears to exhibit a relatively lower rate of full recovery compared to traditional Bell’s palsy, following a variable course with steroid treatment, with or without antivirals, but without a clear indication of accelerated recovery. We found no significant link between COVID-19 vaccination and increased FNP risk. However, due to data limitations and variability, these results should be interpreted cautiously. As our understanding of FNP evolves, more robust research is needed to explore the causes and long-term residual effects of post-COVID-19 FNP, which will ultimately guide improved treatment strategies.
Supplemental Material
sj-docx-1-ohn-10.1177_19160216251315057 – Supplemental material for Facial Nerve Palsy Amid the SARS-CoV-2 Pandemic: A Pooled Analysis
Supplemental material, sj-docx-1-ohn-10.1177_19160216251315057 for Facial Nerve Palsy Amid the SARS-CoV-2 Pandemic: A Pooled Analysis by Ahmad A. Mirza, Abdulaziz H. Almalki, Faisal A. Noori, Sultan A. Neazy, Valerie Dahm, M̈nir Demir Bajin and Vincent Y. Lin in Journal of Otolaryngology - Head & Neck Surgery
Supplemental Material
sj-docx-2-ohn-10.1177_19160216251315057 – Supplemental material for Facial Nerve Palsy Amid the SARS-CoV-2 Pandemic: A Pooled Analysis
Supplemental material, sj-docx-2-ohn-10.1177_19160216251315057 for Facial Nerve Palsy Amid the SARS-CoV-2 Pandemic: A Pooled Analysis by Ahmad A. Mirza, Abdulaziz H. Almalki, Faisal A. Noori, Sultan A. Neazy, Valerie Dahm, M̈nir Demir Bajin and Vincent Y. Lin in Journal of Otolaryngology - Head & Neck Surgery
Supplemental Material
sj-docx-3-ohn-10.1177_19160216251315057 – Supplemental material for Facial Nerve Palsy Amid the SARS-CoV-2 Pandemic: A Pooled Analysis
Supplemental material, sj-docx-3-ohn-10.1177_19160216251315057 for Facial Nerve Palsy Amid the SARS-CoV-2 Pandemic: A Pooled Analysis by Ahmad A. Mirza, Abdulaziz H. Almalki, Faisal A. Noori, Sultan A. Neazy, Valerie Dahm, M̈nir Demir Bajin and Vincent Y. Lin in Journal of Otolaryngology - Head & Neck Surgery
Supplemental Material
sj-docx-4-ohn-10.1177_19160216251315057 – Supplemental material for Facial Nerve Palsy Amid the SARS-CoV-2 Pandemic: A Pooled Analysis
Supplemental material, sj-docx-4-ohn-10.1177_19160216251315057 for Facial Nerve Palsy Amid the SARS-CoV-2 Pandemic: A Pooled Analysis by Ahmad A. Mirza, Abdulaziz H. Almalki, Faisal A. Noori, Sultan A. Neazy, Valerie Dahm, M̈nir Demir Bajin and Vincent Y. Lin in Journal of Otolaryngology - Head & Neck Surgery
Supplemental Material
sj-docx-5-ohn-10.1177_19160216251315057 – Supplemental material for Facial Nerve Palsy Amid the SARS-CoV-2 Pandemic: A Pooled Analysis
Supplemental material, sj-docx-5-ohn-10.1177_19160216251315057 for Facial Nerve Palsy Amid the SARS-CoV-2 Pandemic: A Pooled Analysis by Ahmad A. Mirza, Abdulaziz H. Almalki, Faisal A. Noori, Sultan A. Neazy, Valerie Dahm, M̈nir Demir Bajin and Vincent Y. Lin in Journal of Otolaryngology - Head & Neck Surgery
Footnotes
Acknowledgements
The authors sincerely acknowledge Dr. Brian Healy from the Biostatistics Center at Massachusetts General Hospital, Harvard Medical School, Boston, USA, for the insightful input on the analyses. In addition, we are appreciative of Dr. Mohamed A. Haireche for his assistance with language editing. Finally, we acknowledge the use of AI-based tools to enhance the clarity of complex synthesized ideas and for the organization of thoughts.
Author Contributions
Ahmad A. Mirza: Conception of the work, design of the work, literature search, study selection, formal analysis, and drafting the work; Abdulaziz H. Almalki: Literature search, study selection, data extraction, quality assessment, and drafting the work; Faisal A. Noori: Study selection, quality assessment, and drafting the work; Sultan A. Neazy: Data extraction, quality assessment, and drafting the work; Valerie Dahm: Interpretation of the data, and critically revising the work; Münir Demir Bajin: Interpretation of the data, and critically revising the work; Vincent Y. Lin: Conception of the work, supervision, and critically revising the work. All authors approved the final version for publication, and all agree to be accountable for all aspects of the work.
Availability of Data and Materials
Some datasets supporting the conclusions of this article are included within the article and its additional files. Other datasets used and/or analyzed during the current study are available from the corresponding author on reasonable request.
Consent for Publication
Not applicable.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics Approval and Consent to Participate
Not applicable.
Supplemental Material
Additional supporting information is available in the online version of the article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
