Abstract
Background
Neck dissections (ND) are a routine procedure in head and neck oncology. Given the postoperative functional impact that some patients experience, it is imperative to identify and track quality of life (QoL) symptomatology to tailor each patient’s therapeutic needs. To date, there is no validated French-Canadian questionnaire for this patient-population. We therefore sought to translate and validate the Neck Dissection Impairment Index (NDII) in Canadian French.
Methods
A 3-phased approach was used. Phase 1: The NDII was translated from English to Canadian French using a “forward and backward” translational technique following international guidelines. Phase 2: A cognitive debriefing session was held with 10 Canadian French-speaking otolaryngology patients to evaluate understandability and acceptability. Phase 3: The final version was administered prospectively to 30 patients with prior history of ND and 30 control patients. These patients were asked to complete the questionnaire 2 weeks after their first response. Test-retest reliability was calculated with Spearman’s correlation. Internal consistency was elicited using Cronbach’s alpha.
Results
NDII was successfully translated and validated to Canadian French. Cronbach’s alpha revealed high internal consistency (0.92, lower 95% confidence limit 0.89). The correlation for test-retest validity were strong or very strong (0.61-0.91).
Conclusion
NDII is an internationally recognized QoL tool for the identification of ND-related impairments. This validated Canadian French version will allow clinicians to adequately assess the surgery-related QoL effect of neck surgery in the French-speaking population, while allowing French institutions to conduct and/or participate in multisite clinical trials requiring the NDII as an outcome measure.

Background
The neck dissection (ND) is a hallmark of modern oncological surgery. Following its first description in 1906, it became broadly accepted and integrated in the surgical management of head and neck cancers. 1 At the time, Crile’s 1906 article depicted an en bloc approach for clinically positive nodal disease, sacrificing several adjacent structures including the spinal accessory nerve, ensuing a poor functional and cosmetic outcome. 1
Over time, the ND technique evolved to decrease its functional impact while maintaining oncologic efficacy. In 2002, the American Head and Neck Society and the American Academy of Otolaryngology—Head and Neck Surgery refined the definition of the different types of ND. The radical approach consists of the removal of nodal levels I to V, the internal jugular vein, sternocleidomastoid muscle, and spinal accessory nerve, while the modified technique implies the removal of the same nodes but preserves at least one of the nonlymphatic structures. 2 The selective ND refers to the discriminatory lymphadenectomy based on known lymphatic metastasis patterns. 2 Currently, selective procedures for the reduction of morbidity are widely implemented. 3 Despite these advances, pain and motor impairment, among other, are still frequent complications of ND. 4
Given the postoperative functional impact, ranging from mild to profound,5-8 it is imperative to identify and track symptomology with patient-reported outcomes (PRO) to better tailor each patient’s therapeutic needs. A literature review 9 evaluating various PRO questionnaires for shoulder disability found the Neck Dissection Impairment Index (NDII) 10 to be the only tool adapted specifically for patients having undergone ND, while also being used as a major outcome in multisite surgical oncologic trials. 11 Given the pivotal role language-concordance plays in improved outcomes, 12 we sought to translate and validate the NDII in Canadian French.
Methods
The present study was approved by the Ethics Review Board of the Centre-intégré-universitaire-de-santé-et-de-services-sociaux du de l’Est-de-l’Île-de-Montréal (MEO-12-2022-9405). Written informed consent was obtained from all participants. Permission to use the English NDII for this study was granted by contacting the NDII developers at dchepeha@umich.edu. Future use of the Canadian French version must be granted by contacting dchepeha@umich.edu, who will obtain joint consent from the corresponding author of this study (A.M.).
Neck Dissection Impairment Index
The NDII is a 10-item questionnaire that uses a 5-point Likert scale to evaluate impacts on quality of life (QoL) after ND. Scoring was achieved by patients rating each response item from 1 to 5, with the highest score (5) representing the worse QOL related to the ND. Raw scores were calculated by summating the 10 questions, with total scores ranging from 10 (best QoL) to 50 (worse QoL). For publication purposes, the score was transformed in 2 ways: to a 0 to 100-point scale and the scale was reversed to yield 100 for better QoL related to ND and 0 for the worst QoL related to ND,

French Neck Dissection Impairment Index.
Suite au traitement de votre cou pour votre cancer, au cours des 4 dernières semaines, à quel point avez-vous été incommodé(e) par les éléments suivants?
Êtes-vous incommodé(e) par une douleur ou une sensation d’inconfort au niveau du cou ou de l’épaule?
Êtes-vous incommodé(e) par une raideur au niveau du cou ou de l’épaule ?
Êtes-vous incommodé(e) par la difficulté à réaliser des gestes au quotidien (par exemple, se coiffer, s’habiller, se laver, etc.) à cause de votre cou ou de votre épaule ?
Votre capacité à soulever des objets légers a-t-elle été diminuée à cause de votre épaule ou de votre cou ?
Votre capacité à soulever des objets lourds a-t-elle été diminuée à cause de votre épaule ou de votre cou ?
Votre capacité lever le(s) bras pour saisir un objet en hauteur (par exemple, objet situé sur une étagère, une table ou un comptoir) a-t-elle été diminuée à cause de votre épaule ou de votre cou ?
Êtes-vous incommodé(e) par votre niveau d’activité générale à cause de votre cou ou de votre épaule ?
Est-ce que le traitement de votre cou a affecté votre participation à des activités sociales ?
Votre capacité à participer à des activités de loisir ou récréatives a-t-elle diminuée à cause de votre épaule ou de votre cou ?
Votre capacité à travailler (y compris travailler à domicile) a-t-elle diminuée à cause de votre épaule ou de votre cou ?
1- Pas du tout
2- Très peu
3- De façon modérée
4- Assez5- Beaucoup
Translation
Translation and cultural adaptation of the questionnaire items and Likert scale were accomplished in accordance with the Translation and Cultural Adaptation group from the International Society for Pharmacoeconomics and Outcomes Research. 13 A 3-phased translational approach was employed. In phase 1, forward translation was accomplished by 2 licensed translators and a final harmonized questionnaire was generated. A third translator was solicited to back-translate the questionnaire, ensuring incongruities were addressed. In phase 2, a cognitive debriefing session was held with 10 native Canadian French speakers to identify and address ambiguities, understandability, and acceptability. The questionnaire was refined accordingly, and the final version was devised. In phase 3, the questionnaire was administered prospectively to 30 patients with prior history of ND and 30 control patients. Patients who presented to the otolaryngology clinic for routine follow-up (control group) and patients who had a history of a ND (experimental group) were screened and approached to participate. Patients were approached in a sequential manner. Only patients identifying Canadian French as their first language were enrolled. These patients were asked to complete the questionnaire once more, 2 weeks later.
Statistical Analyses
The estimates were reported as means, standard deviations (SDs), medians, interquartile ranges (IQRs), and percentages, when appropriate. All analyses were carried out using Stata/IC Statistical Software: Release 16. (StataCorp LP).
Internal Consistency
To measure the internal consistency of the NDII, the Cronbach’s alpha was calculated along with a one-sided (lower) 95% confidence limit (95% CL). The alpha ≥.9 was considered excellent, ≥.8 good, ≥.7 acceptable, ≥.6 questionable, ≥.5 poor, and <.5 unacceptable. Several additional alpha-related estimates were reported: item-test correlations, item-rest correlations, average interitem covariances, and alphas with 1 item removed at a time.
Test-Retest Reliability
To investigate the correlation between repeated measures, the Spearman correlation coefficient was calculated along with 95% confidence interval (95% CI). The correlation coefficient of <.19 was considered very weak, .20 to .39 weak, .40 to .59 moderate, .60 to .79 strong, and .80 to 1.0 very strong correlation.
In addition to the Spearman’s correlation, the intraclass correlations (ICC) were calculated for both individual (agreement within the same respondent) and average (average agreement among all the respondents) correlations. The ICC < .5 was considered of poor reliability, .5 to .75 moderate, .75 to .9 indicate good, and >.90 excellent reliability.
Exploratory Factor Analysis
The exploratory factor analysis (EFA) was conducted on the estimates obtained from all 60 respondents and included both quantitative (unrotated principal factors and parallel analysis) and graphical analyses. The cutoff for retaining was set at eigenvalues ≥1.0 (Kaiser rule).
Results
There were 16 (53%) and 17 (57%) females among the cases and controls, respectively. The average age was 64 (range 25-80) years among the cases and 64 (range 25-84) years among the controls. The mean NDII score was 15 (SD 8.4) and 18 (SD 8.6) points among controls and cases, respectively (Table 1).
Total NDII Scores for Case and Control Groups.
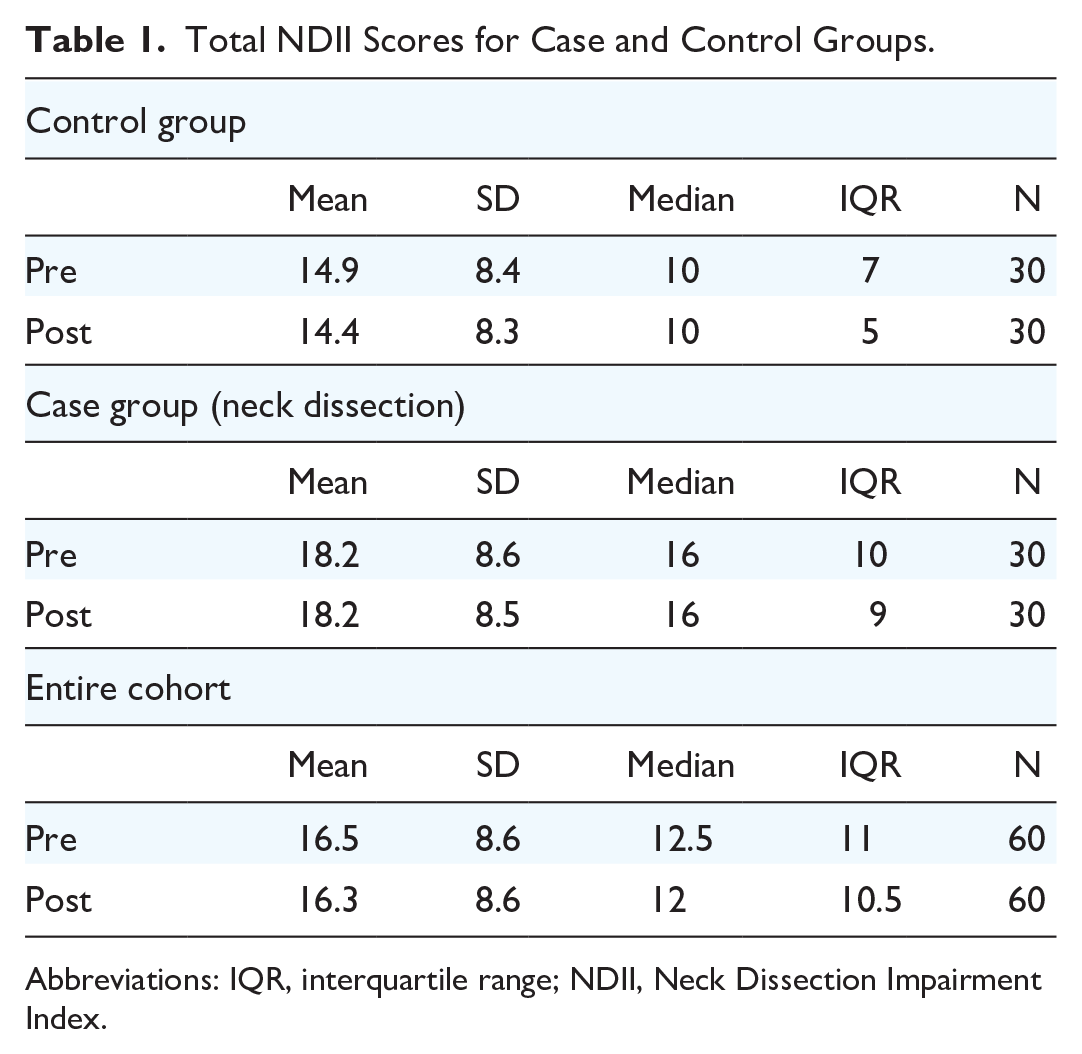
Abbreviations: IQR, interquartile range; NDII, Neck Dissection Impairment Index.
Reliability
Cronbach’s alpha for the entire sample was excellent, at 0.92 (lower 95% CL 0.89; Table 2). All the items demonstrated good item-test an item-rest correlations. Also, excluding one item at a time did not substantially improve alpha for the entire 10-item set. The correlations for test-retest validity were strong or very strong varying from .61 to .91 (Table 3). The individual ICC estimates varied from moderate to excellent: .74 to .93 (Table 4). In turn, the average ICC estimates were higher varying from good to excellent: .75 to .94.
Internal Consistency (Cronbach’s Alpha) With One Item Deleted From the Model at a Time.

Abbreviation: CL, confidence limit.
Test-Retest Validity for All 60 Patients.

Abbreviation: CI, confidence interval.
Test-retest Intraclass Correlations for all 60 Respondents.

Abbreviation: CI, confidence interval.
Validity
The EFA initially resulted in a 2-factor structure, which could be seen both in numerical and graphical models. However, the second factor, with an eigenvalue of 1.20, was close to the Kaiser’s cutoff of 1.0. This is contrasted with the first factor which had an eigenvalue of 5.75. Except for item #8, all the other items had higher loadings on the first factor (even in the case of item #8, the loadings on both factors were very close—0.55 vs 0.58). Thus, the results of factor analysis were considered to be close to one-dimensionality (Tables 5 and 6, Figure 2).
Exploratory Factor Analysis—Item Loadings.

Parallel Analysis. The Cutoff for Retaining Was Set at Eigenvalues ≥1.0 (Kaiser Rule).


Scree plot of exploratory factor analysis along with parallel analysis.
Discussion
The importance of translating previously validated QoL assessment tools is essential for international collaboration, optimization, and personalization of patient rehabilitation treatments. Our aim was to translate the NDII, thereby obtaining a tool for shoulder function-related QoL in head and neck patients in the Canadian French population. A meticulous translational process, guided by international standards, 13 was used to successfully translate the original English NDII to Canadian French.
The Canadian French version of the NDII was shown to be conceptually comparable to the original. The reliability of our questionnaire was shown to be excellent, with a Cronbach’s alpha of 0.92, very similar to the original value of 0.95. 10 Furthermore, the total score for test-retest validity was 0.91, showing minimal variability on the pre- and posttest scores. The original NDII study showed an identical test-retest value. 10 Last, the EFA showed the translated version to be valid. However, the factor structure of a new scale should be investigated in a similar population assuming unidimensionality.
Surgeons must systematically quantify disability rendered after surgery to adapt and follow outcomes related to rehabilitative efforts. A study investigating construct validity and overall appropriateness of PROs using Rasch analysis concluded that the NDII is the most appropriate questionnaire to evaluate outcomes after ND. 14 Currently, the NDII has been translated into Danish, 15 Korean, 16 and efforts are underway to have it translated into Turkish. 17 This study was the first of its kind to generate a Canadian French version of the NDII questionnaire. Given its widespread use in the literature, translational efforts are primordial for interinstitutional and international research collaboration.
As a minor limitation, the translation and cognitive debriefing sessions were conducted with experts and patients who speak Canadian French, more specifically the more prominent variety of Quebec French. While extremely similar to other varieties of French, there may exist some subtilties for which we do not believe impact the internal construct of our study. Validation studies can be performed in other French-speaking regions in the world to further confirm the reliability of our work.
Conclusion
NDII is an internationally recognized QoL tool for the identification of ND-related impairments. This validated Canadian French version will allow clinicians to adequately assess the surgery-related QoL effect of neck surgery in the Canadian French-speaking population. Furthermore, French institutions will now be able to conduct and/or participate in multisite/international clinical trials requiring the NDII as an outcome measure.
Footnotes
Acknowledgements
We would like to thank Dr Chepeha for permission of use of the NDII questionnaire for this study. We would also like to thank Said Dababneh, medical student at the University of Montreal, for his contribution in data collection.
Author Contributions
MK: Study design, data collection, manuscript redaction, manuscript revision. WG: Study design, data collection. CH: Data collection. TA: Manuscript revision. EB: Manuscript revision. AC: Manuscript redaction, manuscript revision. SM: Manuscript redaction, manuscript revision. MJO: Manuscript redaction, manuscript revision. DC: Manuscript redaction, manuscript revision. SYL: Manuscript redaction, manuscript revision. AM: Study design, data collection, manuscript redaction, manuscript revision.
Availability of data and materials:
Available on request.
Consent for Publication
The present study was approved by the Ethics Review Board of the Centre-intégré-universitaire-de-santé-et-de-services-sociaux du de l’Est-de-l’Île-de-Montréal (MEO-12-2022-9405).
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Ethics Approval and Consent to Participate
The present study was approved by the Ethics Review Board of the Centre-intégré-universitaire-de-santé-et-de-services-sociaux du de l’Est-de-l’Île-de-Montréal (MEO-12-2022-9405).
