Abstract
Importance
Mesenchymal stem cells (MSCs) have the capability of providing ongoing paracrine support to degenerating tissues. Since MSCs can be extracted from a broad range of tissues, their specific surface marker profiles and growth factor secretions can be different. We hypothesized that MSCs derived from different sources might also have different neuroprotective potential.
Objective
In this study, we extracted MSCs from rodent olfactory mucosa and compared their neuroprotective effects on auditory hair cell survival with MSCs extracted from rodent adipose tissue.
Methods
Organ of Corti explants were dissected from 41 cochlea and incubated with olfactory mesenchymal stem cells (OMSCs) and adipose mesenchymal stem cells (AMSCs). After 72 hours, Corti explants were fixed, stained, and hair cells counted. Growth factor concentrations were determined in the supernatant and cell lysate using Enzyme-Linked Immunosorbent Assay (ELISA).
Results
Co-culturing of organ of Corti explants with OMSCs resulted in a significant increase in inner and outer hair cell stereocilia survival, compared to control. Comparisons between both stem cell lines, showed that co-culturing with OMSCs resulted in superior inner and outer hair cell stereocilia survival rates over co-culturing with AMSCs. Assessment of growth factor secretions revealed that the OMSCs secrete significant amounts of insulin-like growth factor 1 (IGF-1). Co-culturing OMSCs with organ of Corti explants resulted in a 10-fold increase in IGF-1 level compared to control, and their secretion was 2 to 3 times higher compared to the AMSCs.
Conclusions
This study has shown that OMSCs may mitigate auditory hair cell stereocilia degeneration. Their neuroprotective effects may, at least partially, be ascribed to their enhanced IGF-1 secretory abilities compared to AMSCs.

Introduction
Degeneration of auditory hair cells and spiral ganglion neurons (SGNs) is one of the main causes of sensorineural hearing loss. In particular, aging, noise exposure, and ototoxic medication lead to irreversible loss of both inner hair cell (IHC) and outer hair cell (OHC). Means of preserving and/or regenerating hair cells and spiral ganglion cells, if identified, would have significant clinical implications.
Various growth factors, like insulin-like growth factor 1 (IGF-1), hepatocyte growth factor (HGF), and acidic fibroblast growth factor, have been implicated to mitigate auditory hair cell and synaptic ribbon degeneration,1-6 whereas brain-derived neurotrophic factor (BDNF), neurotrophin 3, and glial-derived neurotrophic factor (GDNF) have been shown to have protective effects on SGNs.7-11 However, these neuroprotective effects are short-lived as local growth factor concentrations decline rapidly after administration.
The paracrine effects of mesenchymal stem cells (MSCs) might offer a potential alternative to provide local and prolonged neurotrophic support to damaged and degenerating hair cell and SGN. Bone marrow (and adipose)-derived MSCs have been shown to produce neurotrophic factors (BDNF) and support repair of the central and peripheral nervous system in animal models.12-14Systemic and local administration of bone marrow and adipose-derived MSCs to the cochlea have shown that these stem cells can survive in the cochlea and support SGN survival as well as the structures of the lateral wall including the stria vascularis.14-16 In addition, perilymph samples of animals treated with adipose-derived MSCs showed elevated levels of BDNF, GDNF, and HGF, implying a local neurotrophic support mechanism instigated by the stem cells.17,18
MSCs have also been identified in olfactory mucosa, and share similar differentiation properties and cell surface markers as bone marrow and adipose-derived MSCs.19,20 Since these olfactory mesenchymal stem cells (OMSCs) are involved in the ongoing neurogenesis of the olfactory neuroepithelium, we propose that these may be a more potent stem cell line for neural tissue repair than bone marrow or adipose-derived MSCs. 14 A comparative study has demonstrated that these OMSCs promote myelination and secrete higher levels of specific neurotrophic factors than MSCs derived from bone marrow.20,21 Olfactory-derived MSCs have also been shown to mitigate SGN degeneration in organ of Corti cultures lesioned with gentamicin. 22 The effects of OMSCs on auditory hair cell degeneration and survival is still unknown, but given their promising paracrine capabilities to secrete neurotrophic factors, OMSCs might have a potential rescuing effect on degenerating hair cells and SGNs.
The first aim of this study was to characterize and assess the acute neuroprotective potential of rat OMSCs on auditory IHC and OHC survival using a rat cochlear explant culture model. Our second goal was to compare the paracrine, protective effects of these olfactory stem cells with adipose MSCs.
Methods
This study was conducted in accordance with protocols compliant with the Canadian Council on Animal Care and with the approval of the local Animal Care Committee at the University of British Columbia (A18-0368).
Isolating and Culturing of Rat Olfactory- and Adipose-Derived MSCs
The olfactory mucosa consists of a lamina propria and neuroepithelium. 14 Although stem cell-like cells can be harvested from both layers, only the cells derived from the lamina propria express specific MSC markers and have the ability to differentiate into the different specific mesenchymal cell lineages, that is, chondrogenic, adipogenic, and osteogenic. 23
Rat OMSCs were obtained from adult rat olfactory mucosa located within the posterior nasal cavity immediately anterior to the cribriform plate. After harvesting, mucosal tissue was placed in a dish, and incubated for 1 hour at 37°C. 24 Under a dissection microscope, the lamina propria was separated from the epithelial layer, and subsequently macerated and centrifuged. The cell pellet was resuspended and plated on a 6-well dish, pre-coated with Matrigel (ThermoFisher), and incubated at 37°C in a humidified 5% CO2 incubator for 48 hours.
Rat adipose mesenchymal stem cells (AMSC) were obtained from adult rat inguinal adipose tissue, as described previously.17,25
Functional differentiation of OMSCs into osteogenic, adipogenic, and chondrogenic lineages was assessed using a specific rat mesenchymal functional identification kit (R&D systems, SC020). After 21 days of culturing, cells were fixed, cryosectioned, and stained with antibodies to detect aggrecan. Negative control cells were labeled with secondary antibodies only.
Cochlear Explant Culture
Cochlear explant cultures were established from wild-type Long-Evans rats. Forty-one cochleae were dissected from 21 postnatal Long-Evans pups (postnatal day 3-7). After decapitation, temporal bones were surgically separated from the skull. After removal of the bony otic capsule, the cochlea was transferred to a culture dish. The spiral ligament, stria vascularis, and Reissner’s membrane were carefully stripped away to expose the organ of Corti. After that, the specimens were dissected into a basal and a medial full turn section. The apical turn and hook region of the basal turn were discarded as they were often damaged during the dissection. If sections were affected by surgical trauma, sections were discarded.
Olfactory and adipose MSCs were seeded on a 6-well plate. When cells reached 90% confluency, basal and medial cochlear turn sections were placed in 2 of the 6 wells. An even number of cochlear turn sections were placed in 2 other wells which only contained culture medium, without any stem cells. This group was labeled “media only.” Two other wells only contained MSCs with identical culture media and served as control. After 72 hours of incubation, the organ of Corti explants were removed for histologic processing. The incubation duration of 72 hours was determined based on observations in pilot experiments and protocols used by other study groups.5,26-28 The supernatant and cell lysate were removed and vials frozen for ELISA experiments. This experiment was performed in duplicate. In the first experiment, experiment 1.1, 8 cochleae were processed for each of the 3 groups, that is, rat OMSCs (8 cochleae), rat AMSCs (8 cochleae), and media only (8 cochleae). An equal number of sections were placed in two 6-well dishes, that is, each well in experiment 1.1 contained sections obtained from 4 cochleae. In the replicate experiment, experiment 1.2, a total of 5 cochleae were processed for each group, that is, rat OMSCs (5 cochleae), rat AMSCs (5 cochleae), and media only (5 cochleae). In all experiments, all groups contained an equal number of sections from the basal and middle turn to control for potential differences. All sections were placed in one 6-well dish; one 6-well dish served as control.
Cochlear sections were incubated with Phalloidin-iFluor 488 Reagent (Abcam) for 45 minutes and mounted on slides with DAPI (4′,6-diamidino-2-phenylindole). Images were acquired using a Zeiss LSM 900 confocal laser scanning microscope. The number of IHCs and OHCs were manually counted per 100 µm length along the cochlea. A cell was considered dead, when there was complete absence of stereocilia Stack images of a random sample of sixteen 100 µm sections were composed. Hair cells in these sections were counted by 2 blinded authors (E.Y. and L.V.S.) to assess inter-rater reliability.
Protein Secretions
Growth factor concentrations were determined in the supernatant and cell lysate using ELISA kits for GDNF, IGF-1, and HGF All samples were analyzed in duplicate. Absorbance at 450 nm was determined with a U-Quant Plate Reader (Bio-Tek Instruments) and averaged over a total of 5 repeats.
Statistical Analyses
Statistical analyses were completed using SPSS version 22.0 software (IBM). Group differences in the number of hair cells, for the groups included in the first experiment, were determined through an analysis of variance (ANOVA). Significant main effects (P < .05) were followed with Bonferroni post hoc tests. Hair cell count differences in the second experiment were calculated with unpaired, 2-tailed t tests. Differences in protein concentrations in the control wells and corresponding experimental wells containing cochlear explants were determined using independent samples t tests.
Results
Characterization of Rat OMSCs
Cells isolated from the rat olfactory mucosa had a fibroblastic morphology, adhered to plastic and were self-renewing. Culturing the cells in adipogenic, chondrogenic, and osteogenic medium resulted in transdifferentiation of the cells into adipocytes, chondrocytes, and osteocytes, respectively (Figure 1). These results indicate that these olfactory-derived fibroblastic cells are multipotent, similar to MSCs derived from bone marrow or adipose tissue.

Functional differentiation of rat OMSCs (top row) and negative controls (bottom row). FABP4, osteocalcin, and aggrecan are stained green, nuclei are stained with DAPI (red). OMSCs, olfactory mesenchymal stem cells.
Co-Culturing Rat OMSCs and AMSCs With Cochlear Explants
Hair cell counts obtained from 17 sections (2 cochleae) which were mounted directly after harvesting, served as controls at t = 0 hours (Figure 2). In this group, the average number of IHC per 100 µm was 9, the average number of OHCs was 37. This is comparable to hair cell counts reported by other study groups.29,30 Culturing of cochlear explants for 72 hours in basal stem cell medium resulted in a significant loss of both IHC and OHC [IHC in “media only” group mean difference (MD) = 2.5, t(59) = −3.912, P < .001; OHC MD = 15.1, t(52) = −7.872, P < .001] (Figures 3 and 4). Co-culturing cochlear explants with rat OMSCs resulted in a significantly better IHC and OHC survival (Figures 3 and 4). Compared to the control “media only” group, co-culturing with OMSCs resulted in a 60% increased IHC survival [F(2), 22.471, P < .001; Bonferroni post hoc test MD = 3.7, P < .001], and a 68% increased OHC survival [F(2), 22.632, P < .001; MD = 15.2, P < .001]. Total hair cell survival improved by 66% [F(2), 30.181, P < .001; MD = 18.9, P < .0001]. When co-cultured with OMSCs, IHC and OHC counts were comparable to counts obtained directly after harvesting [IHC: 10; MD = 1, t(79) = 1.674, P = .10; OHC: MD = 0, t(79) = 0.041, P = .967]. However, the morphology of the stereocilia was less well arranged and signs of degeneration of stereocilia bundles are readily visible (Figure 3).

Temporal bone after removal of the otic capsule and lateral wall. Insert shows a confocal stack image of a medial turn cochlear section. Hair cells are stained with phalloidin (green) and nuclei with DAPI (blue).
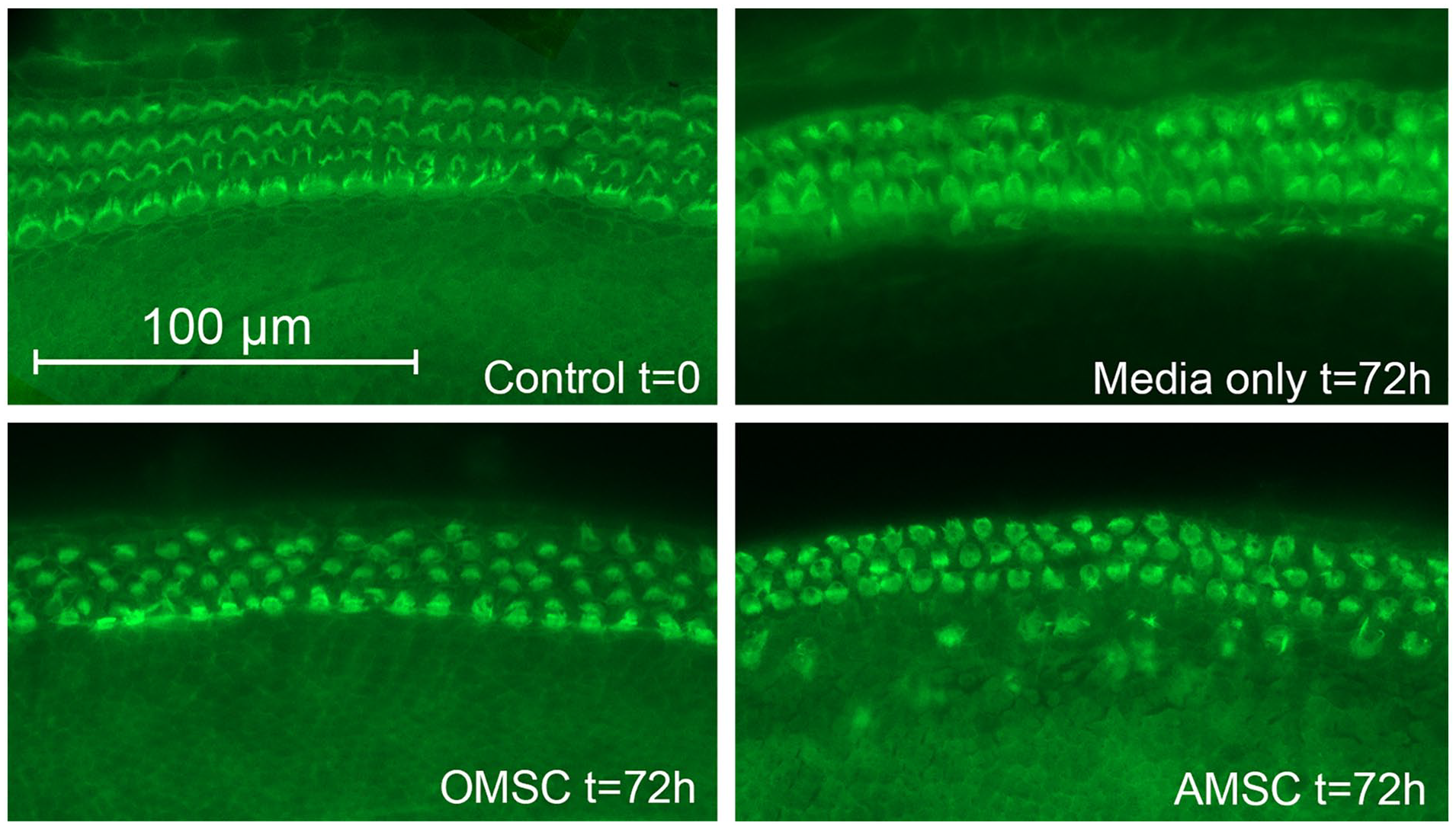
Representative confocal image stacks of cochlear sections of phalloidin labeled hair cells (green) obtained from the control group at t = 0 and after culturing for 72 hours without stem cells (media only t = 72 hours), with rat OMSCs and AMSCs. Degeneration of the stereocilia of both the inner and the 3 outer hair cell rows can be seen. OMSCs, olfactory mesenchymal stem cells; AMSCs, adipose mesenchymal stem cells.
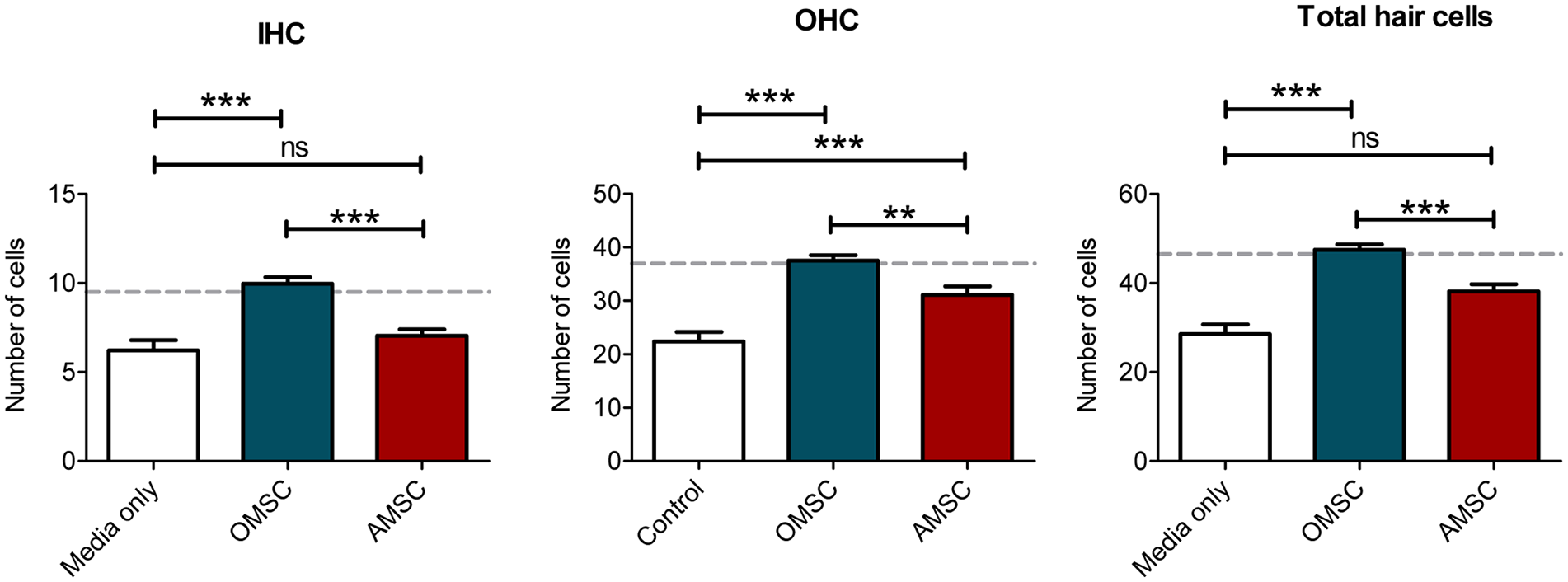
Average hair cell counts measured in 100 µm sections after culturing for 72 hours; control culture media only t = 72 hour group (n = 45 sections), the OMSC (n = 64 sections), and the AMSC (n = 67 sections). For reference: hair cell counts per 100 µm directly after harvesting at t = 0 are 9 inner hair cells, 37 outer hair cells, and 46 total hair cells (dotted gray line). OMSC, olfactory mesenchymal stem cell; AMSC, adipose mesenchymal stem cell; IHC, inner hair cells; OHC, outer hair cells. **P < .01, ***P < .001.
Co-culturing of cochlear explants with AMSCs did not result in an increased survival of IHCs [F(2), 22.471, P < .001; Bonferroni post hoc test MD = 0.8, P = .537], but a protective effect was observed for the OHCs [Figure 3; F(2) 22.632, P < .001], when compared to the control “media only group.” Clear signs of degenerating stereocilia bundles were seen on both IHC and OHC (Figure 3). Comparing the hair cell protective effects of OMSCs and AMSCs revealed a significantly better hair cell protective effect of OMSCs on both IHC and OHC (Figure 4). Compared to co-culturing with AMSCs, co-culturing with OMSCs resulted in a 42% higher IHC survival rate [F(2) 22.471, P < .001, MD = 2.9, P < .001], a 21% improved OHC survival [F(2) 22.632, P < .001, MD = 6.4, P = .006], and a 24% higher total hair cell survival [F(2) 30.181, P < .001, MD = 2.2, P < .001].
Intraclass correlation coefficients (ICCs) revealed a very high agreement for both IHC (ICC = 0.993) and OHC counts (ICC = 0.972) between all 3 independent raters.
Neurotrophic and Growth Factor Secretion by Rat OMSCs and AMSCs
Culturing OMSCs resulted in a basal IGF-1 secretion of 438 pg/ml in experiment 1 and 675 pg/ml in experiment 2. Co-culturing OMSCs with cochlear explants resulted in 4.4- to 4.5-fold increase in IGF-1 concentration, compared to the basal IGF-1 secretion by the OMSCs (Figure 5). In the first experiment, IGF-1 concentration increased from a baseline level of 438 to 1912 pg/ml when cochlear explants were co-cultured with OMSCs [t(3.4) = −24.174, P < .0001]. In the replicate experiment 2, IGF-1 concentrations reached levels above the upper limit of the ELISA test indicating at least a 4.5-fold increase in IGF-1 secretion when cochlear explants were exposed to OMSCs [t(1.9) = −479.400, P < .0001]. Culturing cochlear explants in culture media, without any stem cells resulted in an IGF-1 concentration of 178 pg/ml (Figure 5, “media only”). This basal level can be contributed by release from the cochlear explants. If we compare this basal release from the cochlear explants, with the IGF-1 concentrations when an equivalent amount of cochlear tissue is co-cultured with OMSCs, we found a 10.7-fold increase in IGF-1 levels [t(3.2) = −28.911, P < .0001], indicating active secretion of IGF-1 by the OMSCs. The concentration of intracellular IGF-1, as measured within the cell lysate of the OMSCs in the co-culture increased 3-fold compared to the baseline production of the OMSCs [experiment 1 t(3.1) = −9.131, P = .002; experiment 2 t(1.9) = −9.694, P = .013].
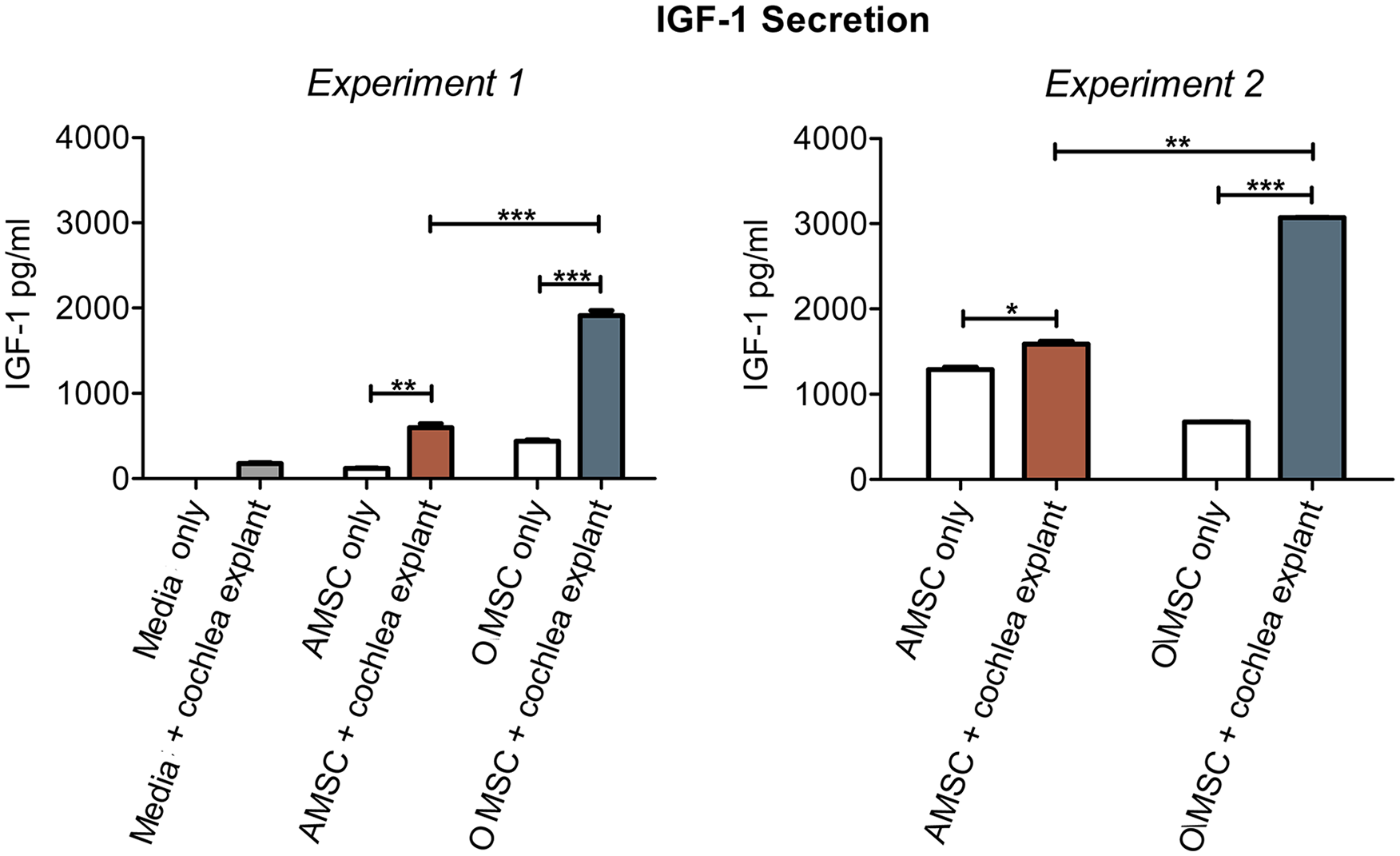
IGF-1 secretion by the rat OMSCs and AMSCs. IGF-1 secretion was determined by measuring the IGF-1 concentration in the supernatant. Filled bars denote the growth factor concentration in the wells with organ of Corti explants; open bars denote the concentrations in the control wells without organ of Corti explants. IGF-1, insulin-like growth factor 1; OMSCs, olfactory mesenchymal stem cells; AMSCs, adipose mesenchymal stem cells. *P < .05, **P < .01, ***P < .001.
Culturing AMSCs resulted in baseline IGF-1 secretion of 121 pg/ml in experiment 1, and 1292 pg/ml in experiment 2. Co-culturing AMSCs with cochlear explants resulted in a 1.2- to 5.0-fold increase in IGF-1 secretion, compared to the basal IGF-1 secretion by the AMSCs (Figure 5). In the first experiment, IGF-1 concentration increased from a baseline level of 121 to 598 pg/ml when cochlear explants were co-cultured with AMSCs [t(3.1) = −9,718, P = .002]. In the replicate experiment 2, IGF-1 levels increased from 1292 to 1588 pg/ml [t(1.8) = −6.882, P = .026]. Compared to the basal IGF-1 release of 178 pg/ml by the cochlear explants, co-culturing with AMSCs resulted in 3.4-fold higher IGF-1 concentrations [t(3.3) = −8.351, P = .002]. In both experiments, IGF-1 secretion by OMSCs was 2 to 3 times higher than the secretion by AMSCs [experiment 1.1 t(6) = 17.191, P < .0001; experiment 1.2 t(2) = 42.126, P = .001].
The intracellular IGF-1 concentration in the AMSCs in the first experiment was twice as high when co-cultured with cochlear explants [t(3,2) = −7.901, P = .003] and 2.6-fold higher in the second experiment (Figure 6), but this was not significantly different, due to large variance in the intracellular IGF-1 concentration in the second experiment [t(1.0) = −4.081, P = .153].

Intracellular IGF-1 concentration in rat OMSCs and AMSCs. The intracellular IGF-1 concentration was measured in the cell lysate of the stem cells. Filled bars denote the growth factor concentration in the wells with organ of Corti explants; open bars denote the concentrations in the control wells without organ of Corti explants. IGF-1, insulin-like growth factor 1; OMSCs, olfactory mesenchymal stem cells; AMSCs, adipose mesenchymal stem cells. *P < .05, **P < .01.
Analysis of both supernatant and cell lysate for HGF secretion demonstrated that OMSCs did not produce or secrete detectable amounts of HGF. Neither did the cochlear explants alone (data not shown). In turn, AMSCs were found to have a high basal HGF secretion of 7.7 ng/ml in the first experiment and 3.1 ng/ml in the second experiment. HGF secretion by AMSCs increased 1.2- to 1.5-fold in the presence of cochlear explants [experiment 1 t(6) = −2.993, P = .024; experiment 2 t(2) = −9.588, P = .011]. Intracellular HGF concentrations were lower compared to the supernatant (experiment 1: 5.7 ng/ml; experiment 2: 2.7 ng/ml) and only changed significantly in the first experiment when co-cultured with cochlear explants [experiment 1 t(6) = −6,967, P < .001; experiment 2 t(2) = 2.660, P = .117].
Only AMSCs were found to produce GDNF levels above the lowest detection threshold of 60 pg/ml (concentration range in supernatant: 80-101 pg/ml, and cell lysate: 69-107 pg/ml). Both secreted and intracellular GDNF concentrations were not influenced by co-culturing with cochlear explants. OMSCs did not produce significant amounts of GDNF, nor did the cochlear explants release detectable levels of GDNF (data not shown).
Discussion
Co-culturing of rat OMSCs with a rat cochlear explant mitigates auditory stereocilia degeneration. This effect may be mediated by the trophic effects of IGF-1, secreted by the OMSCs. The neuroprotective effects on auditory stereocilia survival of olfactory-derived MSCs are superior to adipose-derived MSCs. This may, at least partially, be ascribed to the enhanced IGF-1 secretory abilities of OMSCs.
In this rat cochlear explant culture model, co-culturing with OMSCs resulted in significantly improved survival of stereocilia on both IHC and OHC. This protective effect was more evident with olfactory-derived MSCs than with adipose-derived MSCs. In addition, IGF-1 secretion by OMSCs was 2 to 3 times higher compared to the secretion by AMSCs.
MSCs are known to promote the repair of lesions in different regions of the central nervous system.12,14,25 Studies investigating the effects of MSCs in the inner ear have also shown that MSCs mitigate degeneration of auditory hair cells and SGNs,14,17,18 and promote secretion of neurotrophic factors such as BDNF and GDNF. 17 Olfactory-derived MSCs are mainly found in the lamina propria and are involved in the ongoing neurogenesis of the olfactory neuroepithelium.14,31 Cells obtained from this lamina propria stained positive for mesenchymal markers CD90, CD105, and Stro-1,19,22,23 but also express nestin, a marker for neural stem cells.22,23 Compared to bone marrow-derived MSCs, OMSCs derived from the lamina propria express more nestin and secrete the cytokine Cxcl12 which promotes myelination.20,21,32,33 Our results are in line with these reports, indicating differences in functional properties between different MSC lines.
IGF-1 is known to play an essential role in neurogenesis, but also in survival and neuroprotection. 34 Within the cochlea, the IGF-1 receptor is found in the hair cells and supporting cells of the organ of Corti. 35 Hayashi et al found that IGF-1 treatment results in inhibition of hair cell apoptosis, and initiates proliferation of the Hensen’s and Claudius’ cells, which also contribute to the maintenance of OHCs. 35
The protective effects of IGF-1 on auditory hair cells have also been demonstrated in an animal model of noise exposure. Round window application of a gelatin hydrogel-containing IGF-1 before or 5 hours after noise exposure was found to improve hair cell survival.3,4 Local round window application of IGF-1 after ischemia-induced cochlear damage also resulted in attenuation of hair cell damage. 36 Malgrange et al assessed the effects of various growth factors, including IGF-1, HGF, BDNF, and GDNF, on cochlear explant cultures. Treatment with IGF-1 resulted in OHC survival, whereas BDNF did not protect hair cells from degeneration. 5 Growth factor concentrations in our study showed that the cochlear explant cultures with the highest IGF-1 levels showed better stereocilia survival. In addition, there appears to be a quantitative effect of IGF-1 concentrations on stereocilia survival, that is, culture wells with OMSCs had the highest IGF-1 concentrations and better survival compared to the wells with AMSCs which had lower IGF-1 concentrations and poorer survival. It seems likely that the stem cells, rather than the organ of Corti explants, are responsible for the production of the neurotrophic factors, given the elevated IGF-1 concentrations found in the stem cell lysates. Interestingly, the relatively low intracellular IGF-1 concentration in the OMSCs may indicate a rapid secretion of IGF-1 into the supernatant. This rapid and active growth factor secretion supports the OMSCs’ potential to provide paracrine trophic support. These paracrine capacities of the OMSCs have also been revealed by other groups, which demonstrated the ability of OMSCs to secrete IGF-1.37,38
Limitations
Although, it seems likely that IGF-1 is predominantly produced in the stem cells, it was beyond the scope of this study to exactly determine the mechanisms and pathways involved in the IGF-1 secretion. From our ELISA analyses, it seems likely that the stem cells actively produce and secrete IGF-1. First, IGF-1 levels are more than 10-fold higher in the wells with OMSCs compared to the control wells, which only contain the exact same amount of cochlear tissue. Second, the secretion by the AMSCs is also significantly higher in the presence of cochlear explants, but concentrations found in the co-cultures with OMSCs are still 2 to 3 times higher compared to the concentrations found in the co-cultures with AMSCs. This difference can only be explained by a superior IGF-1 secretion by the OMSCs. Third, intracellular stem cell concentrations also revealed an active production of IGF-1 in the cells.
In our cochlear explant model, we did not make use of aminoglycosides, pro-inflammatory cytokines, or mechanical implant insertion trauma to induce hair cell and stereocilia loss,39,40 since these ototoxic additives can also have undesired and unknown toxic effects on the stem cells. Instead, organ of Corti explants were cultured for 72 hours in basal media without the addition of essential supplements.5,27 The stress reaction caused by dissection, explanting, and consecutive 72 hour incubation is known to release pro-inflammatory cytokines, induce apoptosis, and cause hair cell degeneration, typically seen in the basal and middle turns after 48 hours of culturing.5,26-28 Hair cell counts, in our study, may therefore be higher as sometimes expected in experiments using ototoxic medication. However, our control group clearly indicated that substantial hair cell degeneration occurred after 72 hours of incubation, and the induced degeneration was adequate to study the differences between the stem cell lines on hair cell survival. Of particular importance is the fact that our experimental model was designed to only reveal differences in neuroprotection between the 2 stem cell lines, rather than providing accurate quantifications of neuroprotection on stereocilia and hair cell survival. Future experiments should be focused at providing detailed quantifications on the extent of neuroprotection of the OMSCs in both in vitro and in vivo models, while controlling for the potential confounding effects of aminoglycosides or pro-inflammatory cytokines on both the organ of Corti and stem cells.
Conclusions
Olfactory-derived mesenchymal stem cells may have the potential to mitigate auditory stereocilia degeneration. The OMSCs not only have specific neurogenic characteristics, but they also seem to secrete high levels of the pivotal growth factor IGF-1, while preserving their MSC characteristics. These abilities, as well as the fact that these stem cells are relatively easy to harvest in humans, make the OMSCs a promising stem cell line for therapeutic use in neurodegeneration, including auditory hair cell loss.
Footnotes
Author Contributions
M.J.W.L., E.Y., A.Y., B.D.W., and K.G.E.: research design and conceptualization. M.J.W.L., E.Y., A.Y., I.A.V., T.N.L., and L.V.S. performed research. M.J.W.L., E.Y., A.Y., T.N.L., and L.V.S. analyzed data. B.D.W., A.Y., K.G.E., and M.J.W.L.: resources, project administration, and funding. M.J.W.L., E.Y., A.Y., K.G.E., and B.D.W. wrote the manuscript. All authors contributed to the article and approved the submitted version.
Availability of Data
The raw data supporting the conclusions of this article will be made available by the authors, without undue reservation.
Consent for Publication
Not applicable.
Declaration of Conflicting Interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was funded by an unrestrictive research grant by the Niels Stensen Fellowship, the Netherlands.
Ethics Approval
This study was conducted in accordance with protocols compliant with the Canadian Council on Animal Care and with the approval of the local Animal Care Committee at the University of British Columbia (A18-0368).
