Abstract
Background
The Huntington's Disease (HD) Everyday Functioning (Hi-DEF) is a novel patient-reported outcome (PRO) scale developed to assess the impact of cognitive impairment on daily functioning in early HD patients.
Objective
To examine the psychometric properties, including reliability and validity, of the Hi-DEF. Findings from psychometric analyses using classical test theory (CTT) approach are presented here.
Methods
A non-interventional validation study was conducted across nine HD Centers of Excellence across the US.
Results
Patients with HD (n = 151) were recruited: 59% were female, mean (SD) age was 47 (12) years, and mean (range) Total Functional Capacity (TFC) score was 11.4 (8–13). Excellent internal consistency reliability was observed for the Hi-DEF scale total score (Cronbach's alpha: 0.98) and across subscales (alpha range 0.87–0.96). Most targeting and scaling assumptions were met, although there were ceiling effects for three subscales. Construct validity was demonstrated by moderate to high Spearman's rank-order correlations with TFC scores (range r = −0.38 to −0.62) and known and validated measures such as the HD-PRO-TRIADTM (range r = 0.75–0.90), and ability to discriminate between levels of functional impairment (TFC 13, 12–11, and 10–8; p < 0.001). Correlations with Cambridge Neuropsychological Test Automated Battery (CANTAB) cognitive performance measures were low to moderate and ranged from 0.06 to 0.38.
Conclusions
The Hi-DEF is a reliable and valid PRO scale measuring the impact of cognitive impairment on daily functioning, assessing facets of cognitive functioning in context of activities of daily living impacted in early stages of HD.
Keywords
Introduction
Huntington's disease (HD) is an inherited, autosomal dominant, and progressive neurodegenerative disorder which presents through a triad of symptom classes: motor, neuropsychiatric, and cognitive.1,2 Patients are diagnosed with HD at an average age of 49 years, when motor symptoms first appear (e.g., chorea, extrapyramidal movements, and impaired voluntary movements). 3 Although diagnosis is based on motor manifestations of HD, cognitive symptoms present early in the course of disease and precede the development of motor symptoms4,5; cognitive decline typically manifests around 40 years of age (range 30–59 years) and develops over the next 10 to 20 years.3,6
Cognitive symptoms in HD—most notably difficulties in executive functioning, organization, reasoning, planning, judgment, impulse control, and decision making—have substantial adverse impacts on patients’ quality of life and independence.7–10 Attributable to cognitive impairment during the early stages of the disease, patients may need job modifications or lose their ability to work. 11 Patients indicate that cognitive changes may be the most important and impactful symptoms of HD, both in the early stages of HD and after motor symptoms develop, as they create substantial burden for patients and their families.12,13
Neuropsychological testing is used in research and in clinical trials to establish normative states of functioning and subsequently to evaluate change in functioning over time. 14 For example, the Symbol Digit Modalities Test (SDMT) is used to assess cognitive functioning in clinical trials for the early stages of HD, as well as in the new Huntington's Disease Integrated Staging System (HD-ISS).15–18 Similarly, the Huntington's Disease Cognitive Assessment Battery (HD-CAB) is a battery of tests designed to assess cognitive functioning in clinical trials of early stages of HD. While SDMT and HD-CAB can provide sensitive objective assessments of cognitive performance, they do not assess the impact of cognitive deficits on patients’ daily living and general functioning. Such cognitive testing scores could be complemented by the Huntington’s Disease Everyday Functioning (Hi-DEF), which measures the impact of cognitive impairment on functioning. Traditional neuropsychological tests are critical for identifying affected brain regions and cognitive domains, but they may lack sensitivity to how these impairments manifest in daily life. The Hi-DEF can help bridge this gap by offering a patient-centered perspective on functional difficulties, adding essential context. As such, the Hi-DEF is not a substitute for neuropsychological assessments, but instead provides a complementary, ecologically valid tool that captures the real-world impact of cognitive impairment. Though the Hi-DEF cannot replace cognitive testing, it may also provide a useful tool in instances where cognitive testing may not be feasible in clinical practice. In such cases, as the Hi-DEF can be easily administered, it could be used to provide insights into whether patients have difficulties in their daily activities that are related to cognitive impairment.
Qualitative studies indicate that cognitive decline adversely affects patients’ ability to carry out day-to-day tasks, such as driving, social interactions, household chores, and work-related activities.19–22 Patient-reported outcome (PRO) measures are great tools often used to capture patient experiences with the disease and to quantify such impact on functioning. For example, the HD-PRO-TRIAD, which is a HD-specific PRO instrument for the symptom triad, 23 Huntington Disease Health-Related Quality of Life (HDQLIFETM), 24 and Functional Rating Scale 2.0 (FuRST 2.0) 25 have been developed to provide valid and reliable measures of HD-specific health-related functioning and quality of life. While the existing PROs can broadly assess the impact of HD symptoms, they lack specific questions for detecting subtle cognitive changes that affect executive functioning and independence early in the disease course.
Given the negative effects of cognitive impairment on patients’ ability to maintain their work, home, social functioning, and independence,22,26 it is important to better understand the patient's perspective on these complex day-to-day tasks. To address these gaps, the Hi-DEF was developed. The Hi-DEF is a new PRO scale designed to evaluate the impact of cognitive impairment on daily functioning, particularly in higher-order activities of daily living, among individuals living with HD. Unlike currently available PROs, the Hi-DEF aims to be a more specific measure of complex cognitive changes occurring in the early stages of HD.
Two prior studies have reported on the development of the Hi-DEF scale. The first discusses how the scale was developed based on an in-depth qualitative exploration, including interviews with patients in the early stages of HD and their care partners.22,27 In these concept elicitation interviews, participants were asked open-ended questions to identify how cognitive symptoms and cognitive impairment impacted functioning. Patients reported changes in complex cognitive tasks involving adaptability, planning, multitasking, and processing information, which in turn impacted their work, driving, financial management, social interactions, and other daily functioning abilities. 22 Based on these interviews, a conceptual framework was used as the foundation in the development of the Hi-DEF scale, and the draft Hi-DEF item-set underwent cognitive debriefing with additional patients with HD. During the cognitive debriefing process, patients verbalized their thoughts while completing the draft Hi-DEF and were then interviewed to assess the content validity, relevance, clarity, interpretation, and acceptability of the item-set. 27 Following these interviews, the final Hi-DEF item-set was developed.
The final scale comprises 40 items to assess the impact of cognitive impairment on daily functioning, distributed over four subscales: ‘Home,’ ‘At work,’ ‘Driving,’ and ‘Communicating,’ and is intended for use in both clinical trials and clinical practice. The subscales were developed based on the settings in which patients identified challenges in functioning, in line with the concept elicitation interviews. 27 The functional impact of cognitive impairment is assessed in each subscale via items assessing challenges with daily tasks, such as cooking a meal or completing financial tasks (‘Home’); developing project schedules, interpreting written materials, and completing typical work tasks (‘At work’); paying attention while driving or taking new or unfamiliar routes (‘Driving’); and processing and remembering information or expressing their thoughts (‘Social/Communication’). 27 A sample of items included in the Hi-DEF is included in Supplemental Table 1.
In the second published study, psychometric assessment using Rasch Measurement Theory (RMT) indicated that the Hi-DEF is a cohesive, valid scale that demonstrated good targeting and excellent reliability, with minimal items showing misfit or dependency. 28 Here, we build upon the two prior studies to present additional psychometric analyses to examine the scale targeting, scaling assumptions, reliability, and convergent and known-groups validity, in line with the traditional and widely-used Classical Test Theory (CTT) psychometric approach.29,30
Methods
Study population and sampling
A non-interventional validation study was conducted across nine HD Centers of Excellence in the United States. The entire study visit was completed on a computer, either at the clinical site or at the patient's home, lasting up to 3 h in total. It included a demographic questionnaire, two PRO scales (Hi-DEF scale and HD-PRO-TRIAD) 23 with an estimated duration of approximately 30 min total (for both PROs), and a 30-min practice session on a battery of online cognitive performance assessments from the Cambridge Neuropsychological Test Automated Battery (CANTAB) 31 to familiarize themselves with the test procedures and computer interface. Following a mandatory break of at least 1 h, patients then completed a 30-min scored cognitive assessment (CANTAB) session. Although in other studies CANTAB assessments are conventionally administered on touch screens in clinics, browser-based versions of the CANTAB assessments were used in this study, to accommodate remote testing at home. Patient medical history, including date of genetic testing, CAG repeat length, and Unified Huntington's Disease Rating Scale Total Functional Capacity (TFC) score, were entered into the platform by study coordinators. All data were collected on the CANTAB Connect platform, a cognitive assessment research software (http://www.cambridgecognition.com/products/cognitive-research/web-based-testing).
Included patients were HTT gene mutation positive carriers with either a TFC score of 8 or higher, or investigator-confirmed cognitive symptoms, between the ages of 25 and 65 inclusive, and were able to read and respond to questionnaire items in English. Motor and psychiatric symptoms were not assessed as part of the inclusion criteria. Exclusion criteria included any ongoing neurological conditions other than HD that may impact one's ability to think, concentrate, or make decisions, or any history of neurosurgical intervention or significant head injury. Patients received compensation for participation. This study received ethical approval from Advarra IRB (ref# Pro00048743). Additional details on the study methods can be found elsewhere. 28
Instruments
Hi-DEF scale
The Hi-DEF includes 40 items which ask patients about difficulties in daily functioning due to cognitive impairment during the past week. Three subscales, Home (15 items), At work (11 items), and Driving (8 items), are scored on a 5-level difficulty scale (0 = No difficulty, 1 = A little difficulty, 2 = Some difficulty, 3 = A lot of difficulty, 4 = Cannot do this anymore, N/A = Didn’t have the opportunity to do this in the past week). The fourth subscale, Communicating (6 items), is scored on a 4-level frequency scale (0 = Never, 1 = Sometimes, 2 = Often, 3 = Almost always, N/A = Didn’t have the opportunity to do in the past week). The scale requires a response for each item, resulting in no true missing data. However, the response option ‘N/A’ in both the difficulty and frequency scales was recorded as missing data for the purposes of analyses.
To generate subscale and full scale scores, the item-level scores are summed within subscales and for the Hi-DEF overall. Higher scores indicate greater effects of cognitive impairment on daily functioning. As described previously, 28 scores were transformed to a 0–100 scale for ease of interpretation, in which 0 represents no difficulty and 100 represents maximum difficulty.
HD-PRO-TRIAD
The 47-item HD-PRO-TRIADTM, an HD-specific PRO scale developed for clinical and research settings, 23 assesses the triad of HD symptoms: motor function (19 items), cognition (14 items), and emotional and behavioral dyscontrol (14 items). Each item is answered for the past week and scored on either 5-point frequency or difficulty response scale, where higher scores indicate worse functioning or health-related quality of life. The HD-PRO-TRIAD total score was used in convergent validity analyses.
CANTAB tests
CANTAB is a battery of neuropsychological tests shown to be sensitive to early cognitive changes in neurodegenerative disorders, including HD, 31 and the assessments are among the most widely used cognitive tests for clinical trials of neurological conditions.32,33 The CANTAB tests administered in this study included: Emotional Recognition Task (ERT; social cognitive test of the ability to identify emotions in facial expressions), One Touch Stockings of Cambridge (OTS; executive function test of problem solving, spatial planning, and working memory), Paired Associated Learning (PAL; test of visual learning and memory), Spatial Working Memory (SWM; executive function test of strategy formation and working memory), and Spatial Span (SSP; visuospatial test of working memory).
CANTAB assessments were chosen to closely align with domains assessed by HD-CAB subtests (HD-CAB subtests include Symbol Digit Modalities Test [SDMT], Paced Tapping, One Touch Stockings of Cambridge [OTS], Emotion Recognition Task [ERT], Trail Making B [TMB], and the Hopkins Verbal Learning Test [HVLT]). The CANTAB OTS and ERT in this study are close analogues of the versions included in the HD-CAB, with slight variations. Descriptions of the differences between the CANTAB OTS and ERT and the analogues included in the HD-CAB are provided in the Supplemental Methods.
The PAL was selected as part of the CANTAB assessments as a measure of learning and memory to substitute for the HVLT. Visual search and working memory aspects of the TMB and SDMT are captured by the SWM and SSP in the CANTAB assessments; however, the CANTAB measures eliminate the psychomotor demands of the HD-CAB tests. CANTAB tests selected for this study did not include an equivalent of the paced tapping test of the HD-CAB. Although psychomotor speed and coordination are recognized as important factors mediating performance of some clinically administered paper-pencil tasks in HD, it was not possible to capture these aspects of performance using a self-administered browser-based test platform. Accordingly, while performance of the CANTAB tests can confidently be interpreted as measures of their associated cognitive domains when administered to patients with HD, performance may appear less impaired in comparison to traditional tests (e.g., TMB, SDMT) that assess similar cognitive requirements using a timed psychomotor response format.
While each cognitive test in CANTAB assessments generates a variety of outputs, the following scores were selected as the key outcome measures to track with the outcomes from the HD-CAB, given the browser-based platform that was utilized (https://cambridgecognition.com): SWM = total errors (TE), PAL = first attempt memory score (FAMS), ERT = total hits (TH), OTS = mean choices to correct (MCC), SSP = Forward span length (FSL). Secondary outcome measures were also chosen, specifically SWM = between errors (BE) and strategy (S), PAL = total errors adjusted (TEA), ERT = overall median reaction time (OMDRT) and unbiased hit rate (UHR) per emotion (happiness, H; anger, A; sadness, S; disgust, D; fear, F; surprise, SU), and OTS = median latency to first choice (MDLFC) and problems solved on first choice (PSFC).
Z scores (estimated on basis of the raw score minus the mean score of healthy controls, divided by the standard deviation of healthy controls) were provided by Cambridge Cognition for key outcomes PAL-FAMS, SSP-FSL, and ERT-TH, and secondary outcomes PAL-TEA, OTS-PSFC, SWM-BE, and SWM-S. This enabled the comparison of HD patients and healthy controls on these CANTAB assessments.
Unified Huntington's Disease Rating Scale (UHDRS) - Total Functional Capacity (TFC)
The Unified Huntington's Disease Rating Scale (UHDRS) – Total Functional Capacity score (TFC) is a 5-item clinician-reported instrument that globally assesses occupation, finances, domestic chores, activities of daily living, and level of care. Scores on each item range from 0 to either 2 or 3 (e.g., Occupation: 0 = unable, 1 = marginal work only, 2 = reduced capacity for usual job, 3 = normal). The items are summed into a TFC score ranging from 0 to 13, where greater scores indicate higher functioning.
Psychometric analysis methods: classical test theory
All analyses were conducted using SPSS (version 26, IBM, Armonk, NY, USA). Analyses included examinations of the targeting, scaling assumptions, reliability, and validity of the Hi-DEF as described in Table 1. Results were interpreted with reference to published guidelines wherever possible.
Psychometric measurement properties and assessment criteria.

Targeting and validity analyses were performed on the Hi-DEF 0–100 interval-level scales; all other analyses were run using the raw, item-level data.
Results
Demographics
A total of 151 patients with early stages of HD met the inclusion criteria and chose to participate in this study; 59% were female, the mean age was 47.3 years (SD 11.6; range 25 to 65), and the mean TFC score was 11.4 (range 13 to 8; score 13, n = 55; score 12, n = 30; score 11, n = 21; score 10, n = 13; score 9, n = 15; score 8, n = 15; missing data on TFC score, n = 2).
In terms of employment at the time of the study, 41% (n = 62) were working full-time, 24% (n = 36) on disability (related to HD), 11% (n = 16) working part-time, 10% (n = 15) retired, 7% (n = 10) homemaker, 6% (n = 9) not employed, and less than 1% (n = 1) on disability (not related to HD; n = 2 missing data on employment). In terms of education, 34% (n = 51) had a bachelor's degree, 28% (n = 42) had a postgraduate degree, 14% (n = 21) had some college education, 13% (n = 19) were high school graduates or had a General Educational Development (GED) equivalent, 7% (n = 11) had an associate's degree, 3% (n = 5) had a trade or technical certification, and 1% (n = 2) had some high school education. Additional demographics and other baseline characteristics are described elsewhere. 28 Of the 55 patients with TFC scores of 13, 30 (54.5%) were high functioning—they were working full or part-time and had an associate's, bachelor's, or post-graduate degree. This subgroup of the recruited sample could give insights to the performance of the Hi-DEF at the top end, where cognitive performance begins to change, and daily functioning may be only subtly impacted. On the other end, 94 patients with impaired functioning (TFC <13) were analyzed.
CANTAB results indicated patients’ cognitive performance was generally in the low average range (Supplemental Table 3). The CANTAB scores were not normally distributed and varied between assessments, with some instances of skewness towards the ceiling. See Supplemental materials for CANTAB assessment scores and HD-PRO-TRIAD scores.
CTT psychometric analysis of the Hi-DEF scale
Targeting
No patients were at the floor (score of 100 = maximum difficulties) on any of the Hi-DEF subscales. Ceiling effects (score of 0 = no difficulties) exceeded the >15% criterion in the Home (17%), Driving (29%), and Communicating (16%) subscales. Most patients at the ceiling had a TFC score of 13 for all subscales (Home, 77%; At work, 76%; Driving, 74%; Communicating, 63%) and for the total score (89%). Skewness values were within −1 and +1 for all subscales (−0.49 to 0.54) and for the total score (−0.49; see Table 2 for full data across scales).
Targeting and scaling assumptions of the Hi-DEF.
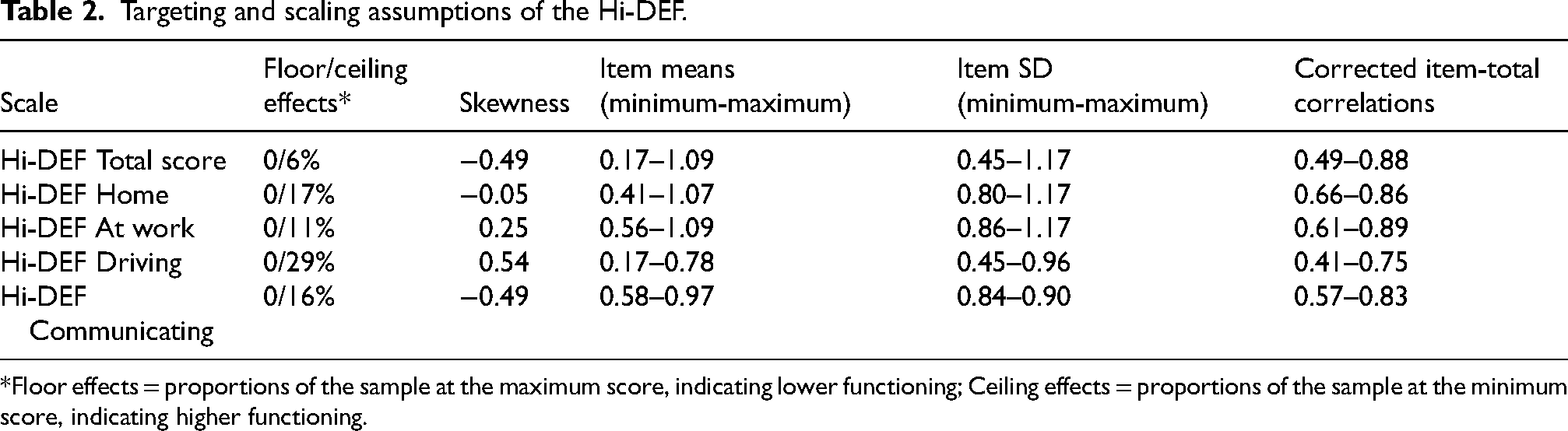
Floor effects = proportions of the sample at the maximum score, indicating lower functioning; Ceiling effects = proportions of the sample at the minimum score, indicating higher functioning.
This indicates that the scale was well targeted for this sample, with the range of difficulties measured by the scale matching the difficulties present in the sample. In line with expectations given the characteristics of the sample, ceiling effects for the Home, Driving, and Communicating subscales indicated patients had fewer difficulties in those environments.
Scaling assumptions
The Hi-DEF scale individual item means and SDs were similar, and corrected item-total correlations were ≥0.30 (Table 2), supporting the presence of a common underlying construct for all Hi-DEF items and the legitimacy of summing items to provide a single total score.
Reliability
Internal consistency was high across the total score and all subscales, with Cronbach's alpha ranging from 0.98 for the total score to 0.87 for the Driving subscale, and mean inter-item correlations and ranges of item-total correlations were ≥0.30 (Table 3), providing supportive evidence for the interrelationships and homogeneity among scale items.
Reliability of the Hi-DEF: Test of internal consistency of items.

Validity
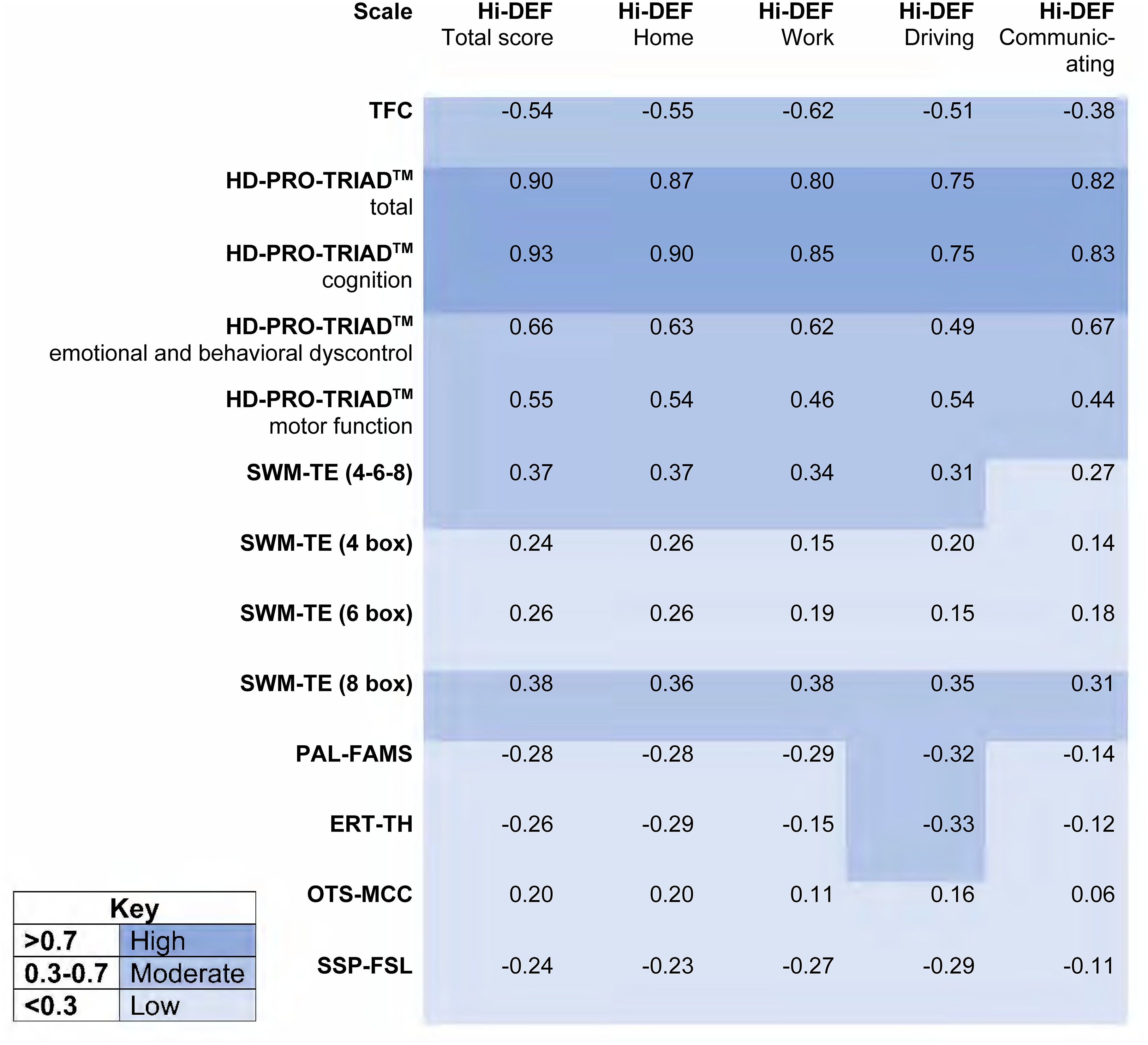
Convergent validity results: Correlations between Hi-DEF and HD PRO TRIAD and CANTAB tests.
The moderate to high correlations of the Hi-DEF with HD-PRO-TRIAD and TFC scores are in line with expected relationships between these measures of related concepts, indicating the Hi-DEF does measure what it was designed to measure. Given that the Hi-DEF is intended to measure the functional impact of cognitive impairment, the higher level of correlation between the Hi-DEF total score and the cognition subscale of the HD-PRO-TRIAD (vs. the other HD-PRO-TRIAD subscales) aligns with expectations. Similarly, the low to moderate correlations between the Hi-DEF and CANTAB tests are in line with expectation due to inherent measurement differences between a subjective, self-reported PRO and objective, performance-based cognitive tests. The CANTAB tests measure cognitive functioning and thus differ from the Hi-DEF, which measures the patient-reported impact of cognitive impairment on daily life.45,46
Convergent validity analyses were also run for the HD-PRO-TRIAD with TFC and CANTAB tests to provide further insight into these correlations. Though these tests do not, in themselves, validate the Hi-DEF, they can contextualize the level of correlation observed between the Hi-DEF and other assessments. Correlations between HD-PRO-TRIAD and TFC scores were low to moderate, slightly lower than with the Hi-DEF, with a magnitude ranging from 0.24 to 0.56. Correlations between HD-PRO-TRIAD scores and CANTAB key measures were similar to that of the Hi-DEF correlations: low to moderate for SWM (range 0.15 to 0.40), PAL (range 0.07 to 0.36), ERT (range 0.06 to 0.44), and SSP (range 0.01 to 0.32), and low for OTS (range 0.14 to 0.27; see Supplemental Material for full results). This low-to-moderate correlation between the HD-PRO-TRIAD and the CANTAB suggests that the similar level of correlation observed for the Hi-DEF may indicate an inherent measurement difference between PRO and performance-based measures.
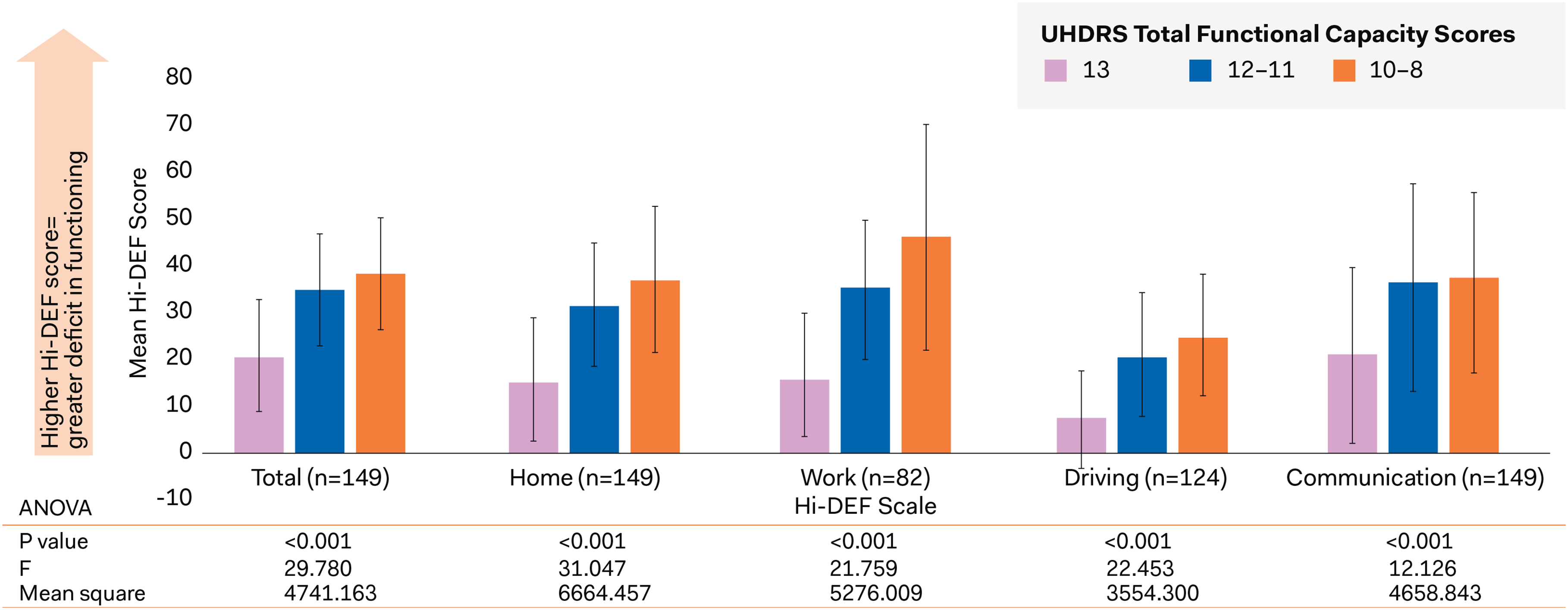
Known-groups validity results: Hi-DEF total score and subscales by TFC score groups. Leftmost bars for each scale/subscale represent the group of patients with a TFC score of 13. Middle bars represent patients with a TFC score of 12–11; rightmost bars represent patients with a TFC score of 10–8.
Known-groups validity of the Hi-DEF: Discrimination between TFC groups.
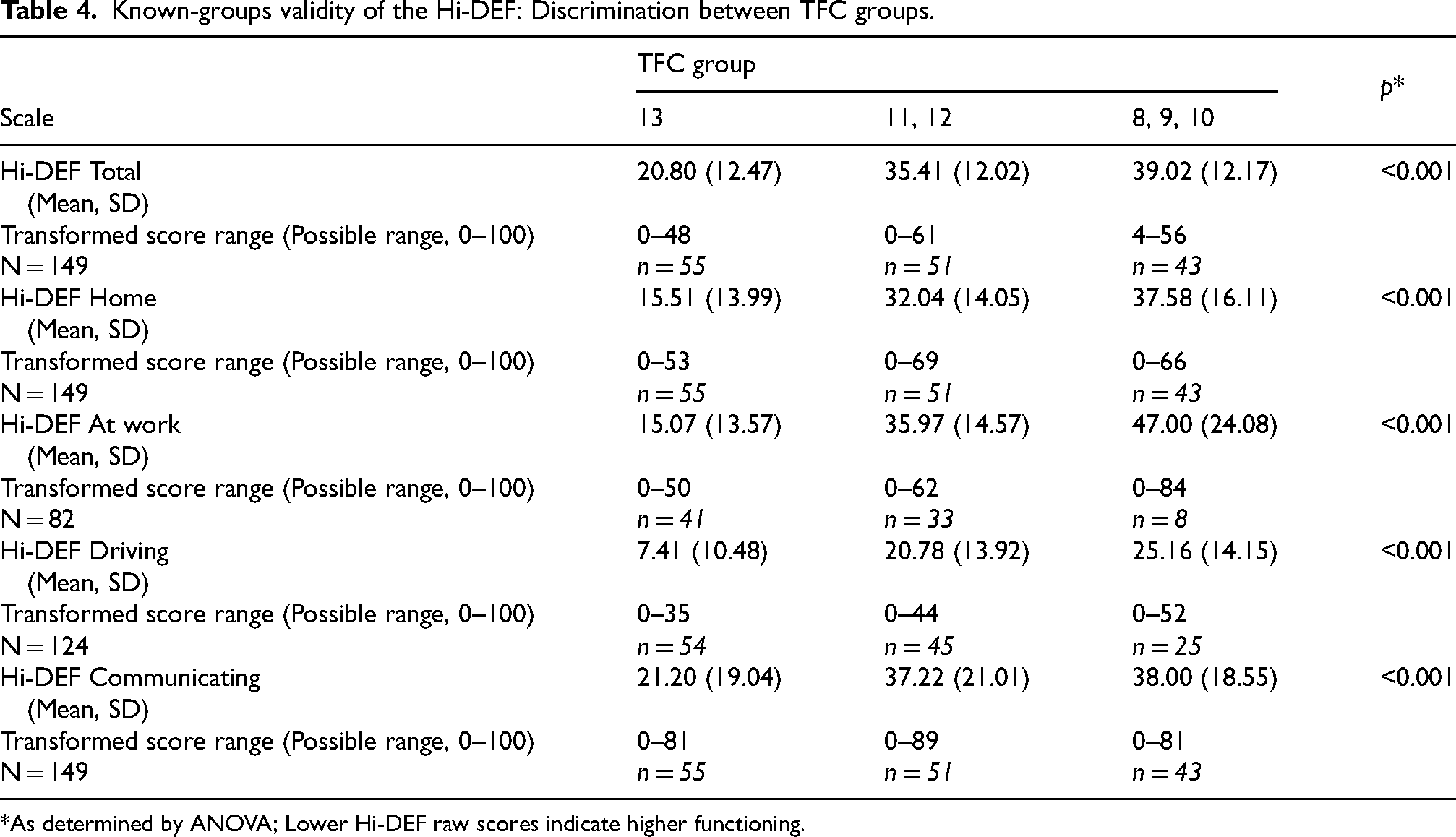
As determined by ANOVA; Lower Hi-DEF raw scores indicate higher functioning.
For the majority of CANTAB variables (SWM-BE, SWM-S, OTS-PSFC, SSP-FSL, & PAL-TEA) the Hi-DEF scale did not discriminate between patients who were impaired or non-impaired. However, the Hi-DEF Total, Home, and Driving scores discriminated between groups who were impaired or non-impaired on the PAL-FAMS assessment (Total: p = 0.030; Home: p = 0.025; Driving: p = 0.026; Supplemental Table 5), and the Hi-DEF Home and Driving scores discriminated between groups who were impaired or non-impaired on the ERT-TH assessment (Home: p = 0.042; Driving: p = 0.027; Supplemental Table 6).
Discussion
The Hi-DEF scale was previously analyzed using RMT, 28 which was supportive of the item performance, overall scale structure, selected item set with minimal dependency, and scoring of the Hi-DEF. In this study, CTT analyses were supportive of the reliability and construct validity of the Hi-DEF, including the ability to discriminate between different levels of functional impairment (based on TFC), and of convergent validity with TFC and the HD-PRO-TRIAD. This first observational study is therefore supportive of the 40-item Hi-DEF as a fit-for-purpose PRO scale for assessing the impact of cognitive impairment on daily functioning in patients with HD. This second psychometric analysis of the Hi-DEF, using CTT, provides further evidence on the reliability and validity properties for its use in clinical research.
Of note, the TFC and the Hi-DEF measure different constructs: the TFC uses a clinician's assessment of overall functional capacity, 47 and what the patient is capable of doing (not only what they might be actually doing), while the Hi-DEF presents a patient's self-reported assessment of the impact of HD on their actual day-to-day functions. As such, it is not expected that the two scales show a strong correlation but rather are complementary. The moderate correlations between the two scales, and the Hi-DEF's ability to distinguish between TFC score groups, both suggest that impaired functioning, as measured by the Hi-DEF, may be reflective of actual differences in functional capacity, as indicated by the TFC. At the same time, for the constructs that are similar across the two scales, the Hi-DEF may offer additional information on the difficulty that patients experience in performing daily activities, as compared to what their clinicians might assess as their ability to perform those activities based on TFC.
Patients with impaired performance on PAL-FAMs, a measure of visual memory and learning, or ERT-TH, a measure of emotion recognition, had significantly higher scores on the Hi-DEF (total score, Home subscale, or Driving subscale). Patients who show more cognition dysfunction on CANTAB assessments may also report greater impairment due to cognitive dysfunction on the Hi-DEF, as is in line with expectations. The Hi-DEF was also moderately correlated with the most difficult version of the SWM cognitive test of executive functioning, though it had low correlations with the other test of executive functioning (OTS). These differences in correlation results may reflect the multifaceted nature of executive functioning as a cognitive domain, as the two tests involve different aspects of executive functioning, or may indicate that, in early-stage HD, distinction between levels of functioning is better characterized by more difficult assessment conditions. Additionally, the SWM's key outcome scores were more widely distributed than the OTS scores in this sample, suggesting that SWM may perform better in correlational analyses. The correlations of CANTAB outcomes and HD-PRO-TRIAD were similar in magnitude.
These validity results align with work in other conditions which has shown small to moderate correlations between PROs and performance-based measures,48–50 suggesting inherent measurement differences between a subjective self-report scale and an objective performance-based measure. 46 CANTAB was designed to assess discrete facets of cognition in isolation and was developed based on animal models. 51 Impairments in specific, individual aspects of cognition may not translate directly to broader aspects of everyday activities such as work or driving, as measured by the Hi-DEF, which utilizes multiple, interconnecting cognitive functions simultaneously. In addition to these intrinsic measurement differences, another contributing factor for the lack of a strong correlation between CANTAB and Hi-DEF scores could be that the study population was patients with early stages of HD with generally higher functioning (with lower cognitive functioning difficulties), and the magnitude of the relationship between subjective self-report and objective performance may not be noticeable before further cognitive impairments develop. Test difficulty may also play a role in highlighting this relationship: the highest difficulty level of the SWM (8-box) was more strongly correlated with the Hi-DEF than the lower-difficulty versions of the SWM. In patients with relatively low levels of cognitive impairment, more difficult cognitive assessment conditions may better capture the level of impairment that patients self-report on the Hi-DEF, as a PRO measure.
Low correlations between performance-based and PRO scales may also indicate that they capture different aspects of cognitive dysfunction.48,50 This divergence may be important for acquiring a comprehensive assessment of the broad construct of cognitive ability. When used alongside CANTAB or other performance-based outcome assessments (e.g., HD-CAB), the Hi-DEF could provide distinct and complementary information for measuring the impact of cognitive impairment in patients in early stages of HD. The Hi-DEF could also be considered a more ecological approach to cognitive testing since it measures functional impairment due to cognitive performance across time and contexts within a patient's routine environment. 52 As such, the Hi-DEF provides an important complement to performance-based measures by capturing essential context on the real-world impact of cognitive impairment.
While other PROs such as the HD-PRO-TRIAD have been developed to measure the totality of symptoms in HD including motor and psychiatric symptoms, the Hi-DEF is specifically designed to measure the impact of cognitive impairment and complex cognitive tasks on daily functioning in HD. This specific focus on the effects of cognitive impairment allows the Hi-DEF to provide a more granular assessment of functioning, including in complex activities, such as driving. The Hi-DEF, unlike the HD-PRO-TRIAD, was also specifically developed to be sensitive to impairment occurring in the early stages of HD. As such, the Hi-DEF provides a key PRO measure with the potential to detect cognitive impairment early in the disease course, to assess changes over time, and to identify where patients with HD may need additional support as indicated by its performance in the high-functioning population recruited in this study. It has previously been suggested that, due to the variety of impairments in cognitive function in HD, a comprehensive assessment should include at least memory, language, executive functions, and visuo-perceptive functions.32,53 The Hi-DEF scale includes items addressing these as well as additional facets of cognition (e.g., multi-tasking, organizing thoughts, planning, and decision-making) and therefore offers a broad assessment of the functional impact of cognitive impairment. In terms of targeting, three subscales of the Hi-DEF demonstrated ceiling effects (patients with no difficulties), which is not surprising given the high-functioning sample included in this study. However, only 6% of the sample (n = 9) had ceiling scores when the total Hi-DEF score was used; therefore, the majority of patients reported some difficulty in a least one functional domain.
Despite the rigorous methods employed in this study, a few limitations must be noted. With browser-based CANTAB assessments used in this study, we were not able to assess deficits in psychomotor function, which could contribute to impairment in activities such as driving. Additionally, the sample (predominantly White and non-Hispanic, with relatively high education levels) lacked diversity. Future research should examine the psychometric properties of the Hi-DEF in more diverse populations and across all stages of HD. Since the sample in this study was relatively high-functioning, the Hi-DEF should also be psychometrically assessed in a sample with greater cognitive impairment. This is especially important considering respondents may have an increasing lack of insight into their issues (anosognosia) as cognitive deficits worsen. 46
Furthermore, in this study, early HD was defined by TFC score range ≥ 8 or investigator-confirmed cognitive changes. Since the motor diagnostic status of participants was not collected, the portion of patients who were manifest/pre-manifest, or pre-/post-clinical motor dysfunction diagnosis, is unknown. The stages of the HD-ISS also cannot be stated with confidence, as Independence Scale scores were not obtained. Future work should investigate the potential utility of the Hi-DEF in identifying the functional effects of cognitive impairment in patients with lower HD-ISS stages.
The timeframe when TFC data were collected in relation to the Hi-DEF and CANTAB data was also not collected in this study, and we do not have information on whether care partners were allowed to attend or participate in the interview during the TFC. The known-groups validity analyses were also limited in terms of the use of TFC scores. TFC scores are produced from a hybrid rating scale which has reduced content and nuance as compared to the Hi-DEF. In particular, there is a lack of overlap in content between TFC and the Hi-DEF Communication subscale. These differences constrain the comparability between scores. Nonetheless, TFC scores were chosen for these analyses since their familiarity enables interpretation by a wider audience.
The lack of caregiver input may also be a limitation in populations with more severe cognitive impairment, in which there may be lack of insight. Moreover, the ceiling effects found in this study should be further investigated during future research. Since the Hi-DEF aims to measure early-stage cognitive impairment, some ceiling effects can be anticipated; however, future research should examine whether scale modifications to address ceiling effects would be beneficial. Additionally, since the Hi-DEF was developed recently, there is currently a lack of longitudinal data for this scale, which prevented the assessment of test-retest reliability and changes over time in clinical trial settings. This should be explored in future studies. Future work should also involve longitudinal assessment to ascertain the ability to detect clinical change and to establish a meaningful change threshold, which may guide the use of the Hi-DEF in clinical practice and decision-making. While the Hi-DEF scale may be too long for clinical practice, a short form (7-items) has also been developed for instances where the full scale may not be feasible or when more frequent assessments may be needed. 54
The Hi-DEF meets the increasing demand for PRO scales to use in research and clinical practice. The Hi-DEF assesses aspects of cognitive impairment which are not measured by performance-based cognitive tests; specifically, the Hi-DEF measures the functional impact of cognitive impairment from the patient's perspective. Therefore, using the Hi-DEF scale in clinical practice may complement existing measures and provide better insight into the patient experience of cognitive impairment associated with HD.
Supplemental Material
sj-docx-1-hun-10.1177_18796397251358375 - Supplemental material for Reliability and validity of the Huntington's Disease Everyday Functioning (Hi-DEF): A patient-reported measure of cognitive capacity on daily functioning in Huntington's disease
Supplemental material, sj-docx-1-hun-10.1177_18796397251358375 for Reliability and validity of the Huntington's Disease Everyday Functioning (Hi-DEF): A patient-reported measure of cognitive capacity on daily functioning in Huntington's disease by Jennifer Petrillo, Ruta Sawant, Rebecca Rogers, Sophie Cleanthous, Stefan Cano, Rajeev Kumar, Jennifer Klapper, Luis A Sierra, Jee Bang, Karen Elta Anderson, Deborah A Hall, Susan Perlman, Henry Moore, Danny Bega, Jason Johannesen and Emma Elliott in Journal of Huntington's Disease
Footnotes
Acknowledgements
We want to thank the HD community and patients with HD who offered their time and effort to participate in this validation study, without them none of this would be possible.
We would also like to thank Aaron Koenig and Sarah Baradaran (formerly Sage Therapeutics, Inc.) for their contributions to the study and Kathryn Wall (Sage Therapeutics, Inc.) for her support during manuscript development. We would also like to thank Aviva Gillman (Modus Outcomes) for help coordinating the study, Flora Mazerolle (Modus Outcomes) for help with data preparation and Trais Pearson (Modus Outcomes) for scientific writing support.
ORCID iDs
Ethical considerations
Patients received compensation for participation. This study received ethical approval from Advarra IRB (ref# Pro00048743).
Consent to participate
Each participant provided written informed consent prior to enrollment.
Consent for publication
Not applicable – results are presented as aggregated data only.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The study was funded by Sage Therapeutics, Inc.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: RS is an employee of Sage Therapeutics and may have stock and/or stock options. JP and JJ were employees of Sage Therapeutics at the time the study was conducted and may have stock.
RR, SC, and SC are employees of Modus Outcomes, a division of THREAD, which received payment from Sage Therapeutics to conduct this research. EE was an employee of Modus Outcomes at the time this study was conducted.
Medical writing support was provided by Modus Outcomes, a division of THREAD (funded by Sage Therapeutics, Inc.). Medical writing and logistical support were provided by Boston Strategic Partners, Inc. (funded by Sage Therapeutics, Inc.).
KEA has served as a consultant for Teva, AskBio, Roche, Biogen, Neurocrine, Sage Pharmaceuticals and Novartis. She has served as a scientific advisor for Neurocrine, Medscape, atheneum, GLG and CHDI. She is also an Editorial Board member of this journal but was not involved in the peer-review process nor had access to any information regarding its peer review. DB is on the speaker's bureau for Teva Pharmaceuticals (Teva), Neurocrine Biosciences, Supernus Pharmaceuticals, Acorda Therapeutics, Abbvie and Kyowa Kirin, and has served as a consultant for Teva, Deerfield Institute, Alexion Pharmaceuticals, Orphalan, Medscape and GLG. He has received grants from the Huntington Disease Society of America and the Parkinson Foundation. HM has served on the advisory board for Ipsen Pharma, served as a consultant for Abbvie Neuroscience, and received grants from MODUS Outcomes, Teva, Sage, Bukwang Pharmaceutical, Neurocrine Biosciences, CHDI Foundation, American Parkinson's Disease Association and Huntington's Disease Society of America. DH has served as an editor for AAN, Elsevier - Parkinsonism and Related Disorders, and Annals of Neurology. She has received research grants from CHDI, Neurocrine, Uniqure and Sage. LS and JK have nothing to disclose. JB serves on the scientific advisory board for Wave Life Sciences. RK has served as a consultant for Supernus, Abbvie, Teva, Annexon, Roche, Acorda Therapeutics, and Cerevel Therapeutics. He has served as a scientific advisor for Impel Pharma. He is on the speaker's bureau for Teva, Supernus, Acorda Therapeutics and Kyowa Kirin. He has served as an officer or member of the board of directors Research Catalyst, LLC (and holds stock) and CenExel RMCR. He has received research grants from Supernus, Sage, Prilenia Therapeutics, Roche, Triplet Therapeutics, CHDI Foundation, Neurocrine Biosciences, Biovie, Neuroderm, Sanofi, Addex Pharma, Integrative Research Laboratories, Takeda, Neuraly, Abbvie, Cerevel Therapeutics,Transposon Therapeutics, Lundbeck, Biohaven, Revance Therapeutics, Impax Laboratories, Pharma Two B, Enterin, CND Life Sciences, Neuron23, Annexon Biosciences, Annovis, PTC Therapeutics, Uniqure, Alexza, SparkNeuro, Praxis, Scion Neurostim, Cognition Therapeutics and Eli Lilly.
Data availability
The data supporting the findings of this study are available on request from the corresponding author. The data are not publicly available due to privacy, ethical restrictions, or other concerns. The Hi-DEF is available for licensing and use by the HD and research communities by accessing the ePROVIDE link (![]() ). Those who wish to see a full copy of the Hi-DEF and all prior research that supports its use can find them by accessing the ePROVIDE link.
). Those who wish to see a full copy of the Hi-DEF and all prior research that supports its use can find them by accessing the ePROVIDE link.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
