Abstract
Given the growing interest in functional foods, blueberry fermentation with probiotics is being explored as a method to enhance the health benefits associated with blueberries. This systematic review investigates the modulatory effects of blueberry fermentation on probiotic growth dynamics and metabolism. In accordance with the Preferred Reporting Items for Systematic reviews and Meta-analyses (PRISMA) guidelines, a comprehensive literature search was conducted across multiple databases, identifying forty-three relevant studies published up to August 2025. The studies were primarily in vitro experiments, and the findings indicate that blueberry fermentation significantly enhances the growth and viability of various probiotic strains. The fermentation processes not only promoted probiotic proliferation but also enhanced the bioavailability of phenolic compounds, including protocatechuic acid, gallic acid, catechol, and syringic acid, thereby improving antioxidant activity. Notably, different probiotic strains exhibited distinct growth responses depending on their metabolic capabilities and the specific blueberry components utilised, such as malvidin-3-O-glucoside, cyanidin-3-O-glucoside, rutin, glycosylated phenolic compounds, glucose, fructose and tannins
Keywords
Introduction
Blueberries (Vaccinium spp.), a diverse group of plants in the family Ericaceae, are well known for their rich nutrient profile and high content of bioactive compounds, particularly polyphenols. These compounds have been associated with various health benefits, including anti-inflammatory effects1–3 and reduced risk of chronic diseases.4,5 The rising interest in functional foods has led to growing attention toward the health-promoting properties of blueberries. 6 However, the bioavailability of their beneficial compound is limited, 7 and their high perishability necessitates the exploration of alternative forms of consumption such as fermented beverages.8,9 Fermentation, a traditional food preservation technique, has demonstrated significant potential for enhancing food safety 10 as well as improving the bio-accessibility and biological activity of plant-based compounds. 11 Fermented fruits have been utilised as functional, health-promoting beverages.12–14 Emerging evidence suggests that the gut microbiota is significantly modulated by dietary factors, including the intake of fermented foods.15–19 By modifying the composition and function of the gut microbiome, fermented blueberry products may exert beneficial effects on human health. Probiotic bacteria, such as Lactobacillus and Bifidobacterium species can metabolise blueberry polyphenols, leading to the production of health-related metabolites, including short-chain fatty acids (SCFAs).20–23 SCFAs such as acetic and butyric acids, have been shown to enhance intestinal health, reduce inflammation, 7 and potentially mitigate metabolic disorders such as obesity and type 2 diabetes.20,24 This systematic review highlights recent advances in understanding of how fermentation alters the biochemical composition of blueberries and in turn, influences the proliferation and metabolic activity of probiotic strains. Beyond antioxidant activity and polyphenol bioavailability, increasing evidence indicates that fermented blueberry products exert a wide range of biological activities, including anti-inflammatory, anti-obesity, antimicrobial, gut barrier–protective, metabolic regulatory, and enzyme-inhibitory effects. This review therefore evaluates fermented blueberries as multifunctional bioactive systems rather than solely antioxidant carriers. Gaining insights into these interactions may inform the development of blueberry-based functional foods and personalised probiotic therapies aimed at optimising the human gut microbiome and overall well-being.
Materials and methods
Database search strategy and eligibility criteria
This review was developed according to the Preferred Reporting Items for Systematic Reviews and Meta Analyses (PRISMA) guidelines.
25
The participants, intervention, comparators, outcomes, and study design (PICOS) criteria adopted in this review are shown in Table 1. Primary articles were identified from the following databases: Elsevier ( The impact of fermented blueberries on probiotic growth and metabolic activity. The impact of fermentation on the bioavailability of bioactive compounds in blueberries, as well as on the antioxidant activity of the fermented blueberry products.
Population, intervention comparators, outcomes, and study design (PICOS) criteria for study inclusion.

Exclusion criteria
Studies were excluded if they focused solely on non-probiotic or pathogenic microorganisms, investigated only non-fermented blueberries, failed to report on the growth dynamics or metabolic activity of probiotics or did not differentiate between the effects of blueberry fermentation from other variables. In addition, unpublished studies, duplicates, reviews, letters, book chapters, abstracts, case studies, and conference proceedings were excluded. Specific exclusion terms were individually applied to each database search to minimise false-positive results.
Data selection process
The following descriptors/keywords were used for the database searches: i) Blueberry fermentation, probiotic activity, and gut health, ii) Blueberry fermentation and polyphenols as well as other bioactive compounds, iii) Blueberry fermentation and organic acids (OAs) and short-chain fatty acids (SCFAs), iv) Blueberry fermentation and gamma-aminobutyric acid (GABA). Each article was initially screened based on its title, abstract and keywords. Full-text articles were then reviewed for eligibility according to the inclusion criteria. For all included studies, key data were extracted using standardised format, which captured the author(s) name, year of publication, intervention details, microorganisms used for fermentation, experimental methods and main findings (Table 2).
Summary of main characteristics of studies included in this review.
Results and discussion
Database search
A total of 5315 records were identified during the initial database search across Elsevier, Springer, PubMed, and Taylor and Francis. After removing 1946 duplicate entries, a total of 3369 records remained for screening. Title, abstract, and keyword screening resulted in the exclusion of 1475 records, leaving 72 articles for full-text review. Of these, 34 articles were excluded based on pre-defined inclusion criteria. An additional five relevant studies were identified through citation searching. Consequently, a total, 42 articles were included in this systematic review. The study selection process is summarised in the PRISMA flow diagram (Figure 1).
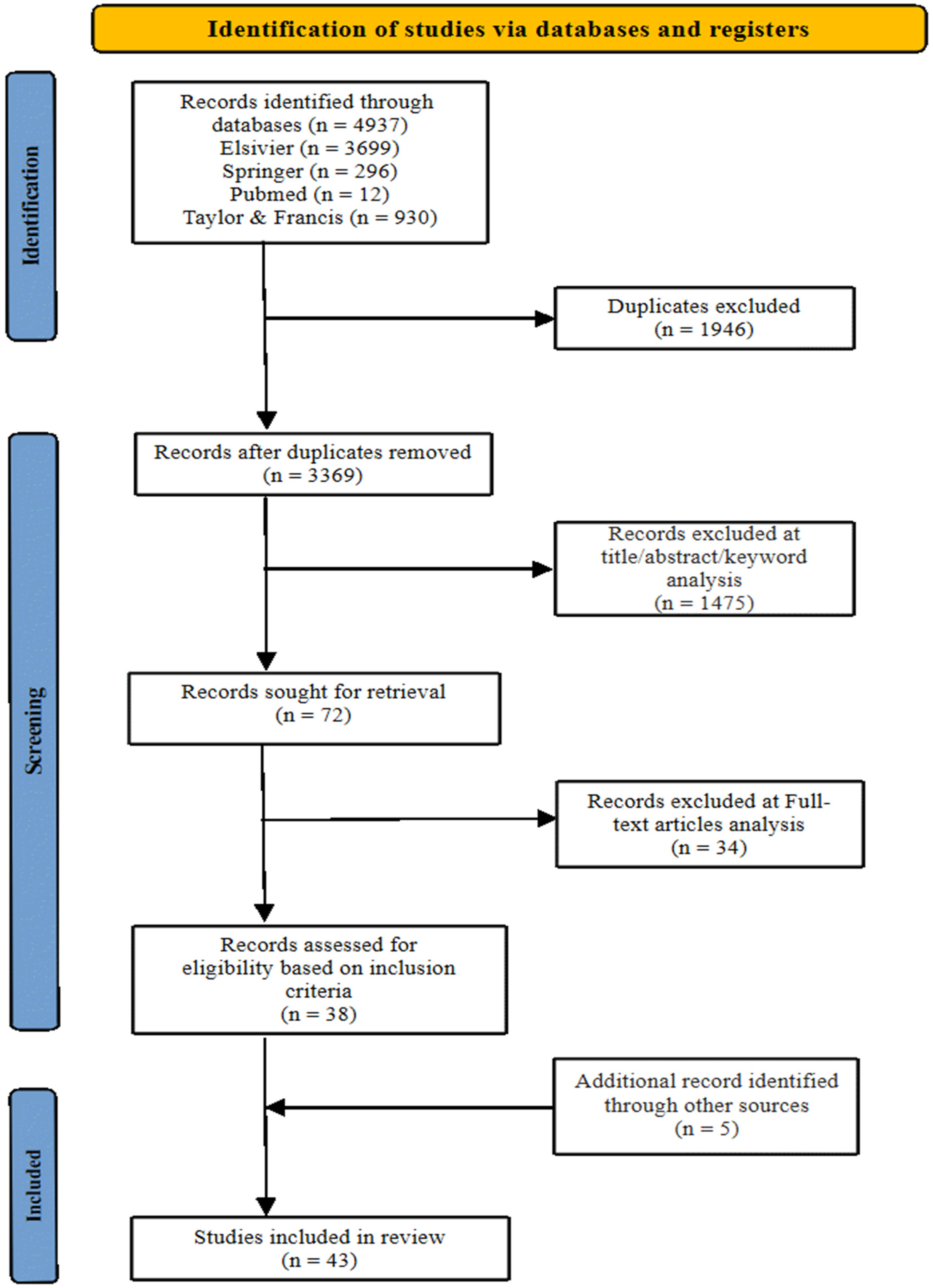
PRISMA flowchart depicting study systematic search and selection process.
Description of included studies
Publications from the years 2005 to 2025 were included in this review, although no studies were retrieved from 2008, 2010–2016. Most studies consisted of in vitro experiments, while eight studies involved animal models. Additionally, three studies employed hybrid methodologies, combining in vitro assays with animal experiments. Only one study included in this review conducted a pilot human trial. 39 The fermentation substrates varied across studies and included whole blueberries, blueberry juice, powder, pomace, and specific polyphenolic extracts. The outcomes assessed primarily focused on probiotic growth rates, metabolic activity, and changes in the bioavailability of polyphenols and antioxidant capacity. Despite some heterogeneity in experimental approaches, most studies consistently reported enhanced probiotic growth and increased polyphenol bioavailability post-fermentation. Fermentation durations varied significantly across the studies, reflecting differences in experimental designs and probiotic strains. Most in vitro studies used fermentation periods ranging from 24 to 72 h, with 48 h being the most common optimal duration for probiotic proliferation. A few studies extended the fermentation period up to seven days26,41 to evaluate the stability of bioactive compounds and to characterize physicochemical, antioxidant, antimicrobial, and enzyme inhibitory activities. The key characteristics of the included studies are summarised chronologically in Table 2.
Effect of blueberries on probiotic growth
Overall, the reviewed studies demonstrate that blueberry fermentation significantly influences the growth and viability of probiotics and supports improved gut microbiota in animal models fed blueberries. The strains used in the included studies belonged to the genera Lactobacillus, Bifidobacterium, Bacillus, Streptococcus, Starmerella, and yeast Saccharomyces, aligning with the most commonly used probiotics in functional foods and dietary supplements.58–60 A study indicated that the population of B. amyloliquefaciens and L. brevis remained stable during the first 72 h of blueberry fermentation, suggesting that these strains can persist in the fermentation medium for extended periods. 39 Similarly, L. rhamnosus and L. plantarum exhibited high survival rates under acidic conditions (pH 2.0) and in the presence of bile salts, with survival rates of 65.71% and 68.85% respectively. These strains also remained viable under simulated gastrointestinal conditions, which is critical for ensuring their effectiveness in functional foods and dietary applications over extended periods. 51 In agreement, a study found that microbial counts of the novel bacterium (S. vaccinii) increased from 5.80 log CFU/mL on day 0 to 9.11 log CFU/mL on day 1 of fermentation, with no significant decline observed over the subsequent 7 days. 26 This was further supported by another study, 45 who demonstrated that the probiotic strains L. plantarum, S. thermophilus, and B. bifidum increased by 0.4–0.7 log CFU/mL after 48 h of blueberry juice fermentation, with all strains reaching the recommended threshold of (>7.0 Log CFU/g).
The ability of probiotic strains to utilise blueberry components varies across species. For instance, L. plantarum NCU116 was observed to begin metabolising sucrose after 36 h, a capability not observed in L. casei NCU215 and L. acidophilus NCU402. 59 This finding suggests the strain-specific nature of metabolic activity, which can influence growth dynamics during fermentation. Different probiotic strains exhibited varying growth patterns in the blueberry growth medium. In another study, L. plantarum and B. bifidum achieved higher viable counts in fermented blueberry juice compared to S. thermophilus, potentially due to the optimal growth temperature of S. thermophilus and the selective effects of anthocyanins. 45 While many studies suggest that multi-strain fermentation yields better results than mono-strain fermentation, one study reported an exception. They found that the total viable bacteria in mixed-strain fermentations were slightly lower than in mono-strain fermentation, except for the combination of L. rhamnosus GG and L. plantarum-1, which maintained a higher viability. 34
Mechanisms and reasons for probiotics growth stimulation
Diverse Carbon and Nitrogen Sources
The stimulatory effect of blueberries on probiotic growth is driven by several complex nutritional and biochemical mechanisms. Blueberries are a rich source of fermentable carbohydrates, including monosaccharides (glucose, fructose), disaccharides, and oligosaccharides, which provide the essential carbon source for probiotic proliferation. 23 Probiotics predominantly utilize glucose and fructose found in blueberries as carbon sources. 61 Interestingly, certain strains like L. plantarum NCU116 can also initiate the utilization of sucrose after an initial fermentation period, facilitating extended growth. 61 Probiotics consume amino acids (such as arginine acid, aspartic acid, and glutamic acid) present in the fruit juice as nitrogen sources. Arginine, in particular, plays a vital role in helping these bacteria adapt to the acidic environment of the juice. 61
Enzymatic Biotransformation
Probiotics secrete specific enzymes that break down complex blueberry components into forms they can use for energy. Strains such as L. plantarum possess tannase, an enzyme that degrades complex tannins and plant cell walls. 62 This process releases simpler phenolic compounds and sugars that the bacteria use for energy. 62 Probiotics produce β-glucosidase and β-galactosidase, which cleave the sugar moieties from glycosylated anthocyanins. This provides the bacteria with a supplementary carbon source while transforming the remaining aglycones into more stable phenolic acids like gallic and syringic acid. 62
The “Antioxidant Shield” Effect
Oxidative stress is a primary cause of death for probiotic cells 41 Blueberries are rich in anthocyanins and other phenolics that act as powerful antioxidants, reducing the oxidative pressure in the fermentation medium. 63 This protective environment helps delay bacterial cell death and maintains high viability during processing and storage.41,63
Selective antimicrobial action
Several studies also reported a bidirectional relationship between the blueberry polyphenols and probiotics, promoting the growth of some microbes while inhibiting the growth of pathogenic microbes (Figure 2). For instance, a study reported that blueberry extracts promoted the growth of anaerobic microbes, particularly those from the phyla Firmicutes and Bacteroidetes, while suppressing the proliferation of Proteobacteria. 38 Another study reported that blueberry extracts supported the growth of L. rhamnosus and B. breve, while inhibiting the growth of pathogenic bacteria E. coli. 29 These outcomes indicate that blueberries modulate the gut environment by providing suitable substrates, particularly polyphenols, which probiotics metabolise into organic acid (Figure 2). This results in a reduction in gut pH, creating conditions favourable for probiotic proliferation while inhibiting pathogen growth.28,64
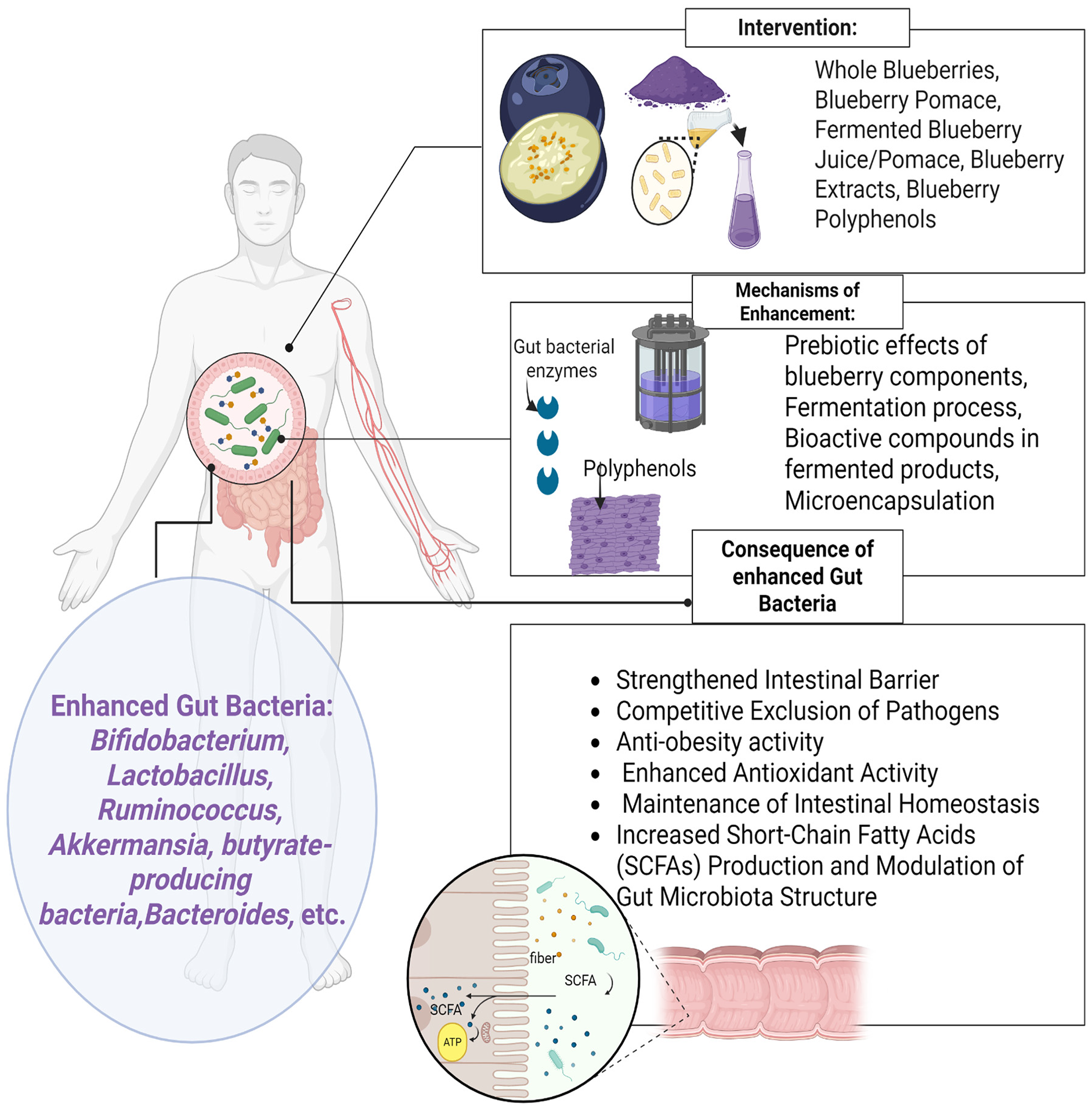
However, the pH can drop below optimal growth conditions, creating an excessively acidic environment. A study observed a significant decline in Bifidobacterium in the caecal content of rats fed Bifidobacterium DSM 15159 combined with blueberries, relative to other groups. 28 Similarly, research reported that feeding rats a combination of bifidobacterium and blueberries did not increase the viable counts of bifidobacterial in the rat caecum tissue. This reduction in viable counts in the caecal contents could be a result of LAB strains breaking weak molecular bonds, altering the three-dimensional structure, and resulting in less water retention in the caecum. 20
Effect of blueberries on probiotic metabolism
The metabolism of probiotics is primarily influenced by the availability of carbohydrates and bioactive compounds in blueberries. Fermentation by gut microbiota mainly produces organic acids and SCFAs.20,65,66 These metabolites can contribute to the distinct flavour, aroma, and nutritional properties of fermented blueberry products.49,67 The specific metabolites and their concentrations can vary depending on the probiotic strain, blueberry variety and fermentation conditions (e.g., time, temperature and pH). In this review, 14 studies reported on the production of organic acids and SCFAs, as summarised in Table 3.
Summary of impact of blueberries on probiotic metabolite(s) production.

Overall, the studies demonstrated that fermentation of blueberries, particularly lactic acid bacteria (LAB) strain, significantly enhances the production of organic acids and SCFAs. The most commonly reported organic acids were acetic acid, lactic acid and citric acid. These acids are typically the result of carbohydrate metabolism (Figure 3), where probiotics utilise the natural sugars in blueberries. 59 LAB fermentation in particular, has a substantial impact on the organic acid profile of blueberry juice, with lactic acid being predominant metabolite produced.5,45,49 These organic acids contribute to the sour taste of fermented products and also exhibit preservative effects by lowering the pH of the fermentation medium and inhibiting spoilage microorganisms. 49
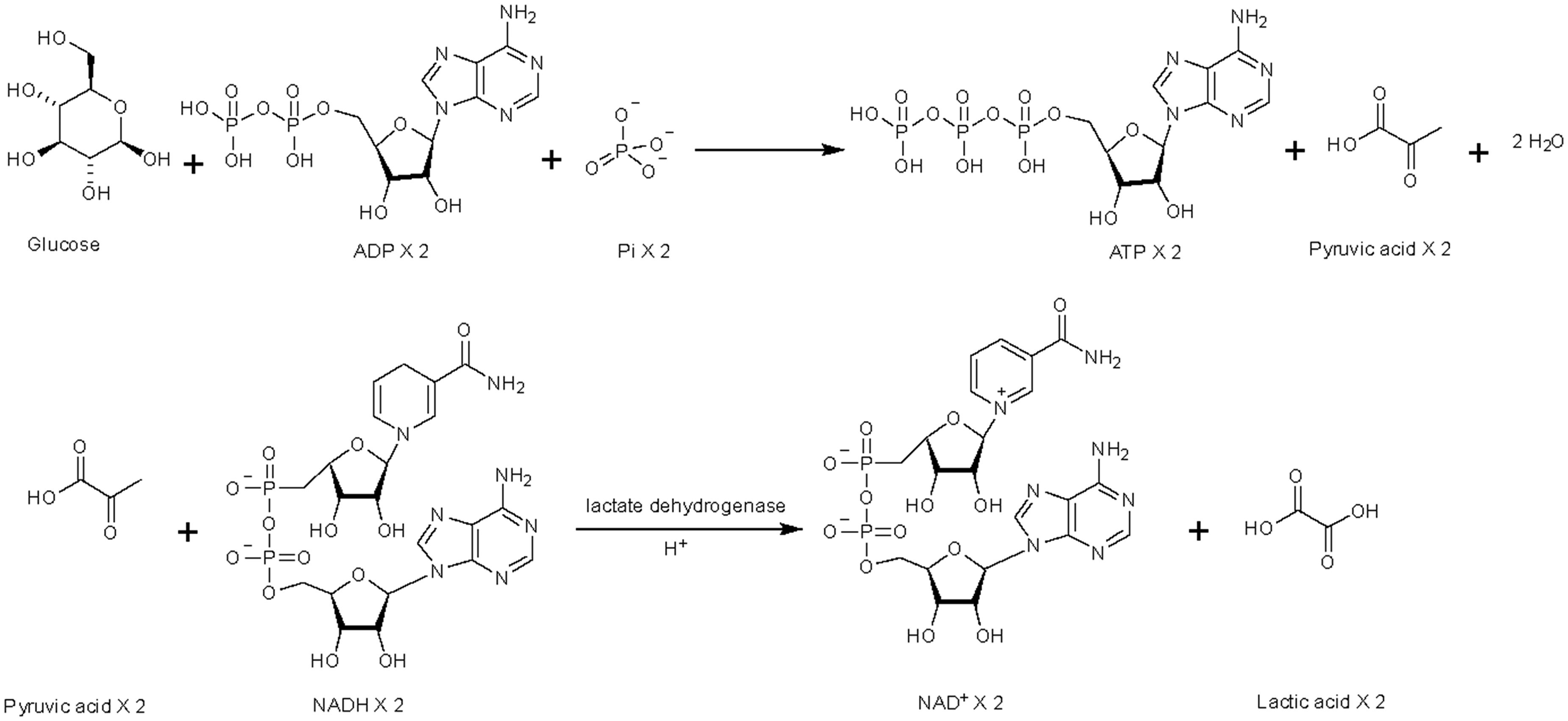
An overview of how certain lactic acid bacteria, such as Lactobacillus plantarum and Lactobacillus casei, utilize the Embden-Meyerhof-Parnas (EMP) pathway to metabolize sugars like glucose during fermentation, glucose is broken down through a series of enzymatic reactions into two molecules of pyruvic acid. Subsequently, pyruvic acid is reduced to lactic acid by the enzyme lactate dehydrogenase.
For instance, a study reported a significant increase in lactic acid in blueberry juice fermented with S. lactis. 35 Similarly, it was observed increased lactic acid levels and concurrent decreases in malic, citric, and pyruvic acids during the fermentation of blueberry and blackberry juice fermented with L. plantarum, S. thermophilus, and B. bifidum. 49 Another study also reported significant increases in lactic acid alongside a marked reduction in citric acid. 59 These shifts in organic acid profiles can be attributed to probiotic metabolic pathways. For instance, in homolactic fermentation (Figure 4), LAB strains metabolise glucose via the Embden-Meyerhof-Parnas pathway to primarily produce lactic acid (Figure 3). In contrast, heterofermentative LAB strains (Figure 4) utilise the pentose phosphate pathway, producing lactic acid along with other acids such as acetic acid.49,59 However, a different pattern was observed as the fermentation process progressed, acetic acid production exceeded lactic acid, with concentrations reaching 13.6 mg/mL for acetic acid compared to 6.3 mg/mL for lactic acid. Tartaric, oxalic, and citric acids were also detected in the fermentation liquid. 34
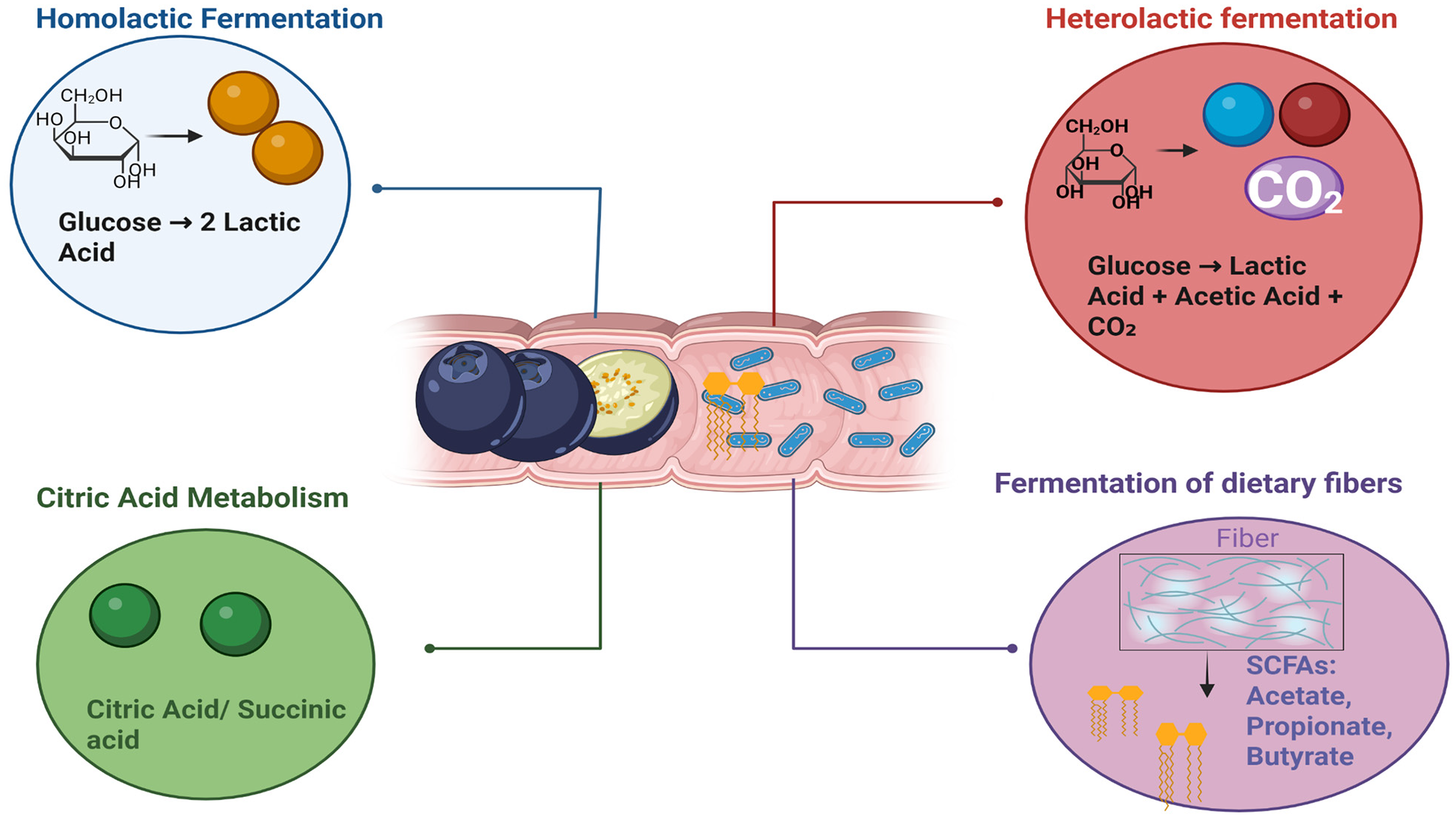
Overview of probiotic metabolism. Created in https://BioRender.com.
As discussed, probiotics involved in blueberry fermentation also produce SCFAs, including acetate, propionate, and butyrate. These SCFAs are commonly generated through the fermentation of dietary fibres such as pectin.5,53,68 A study found that total SCFAs concentrations in blueberry anthocyanin extracts increased from 2.98 mM to 78.06 mM, with acetate, propionate, and n-butyrate identified as the dominant metabolites. 54 Conversely, reductions in acetic acid and propionic acid levels in the caecal contents of rats supplemented with blueberries and either B. infantis DSM 15159 or L. plantarum DSM 15313 compared to the control groups. 28 The authors suggested that this decrease may result from improved mucosal barrier integrity, which could enhance SCFA absorption from the colonic lumen into systemic circulation. Supporting this, acetic acid (645–1394 μmol/L), propionic acid (22–77 μmol/L) and butyric acid (5–31 μmol/L), was identified as the predominant SCFAs in the portal blood of rats, along with minor levels of isobutyric, isovaleric and valeric acids. 20 However, SCFAs concentrations were lower in rats fed diets supplemented with blueberry husks compared to those on a control diet. 20
Bioavailability of polyphenols
Polyphenols are one of the largest classes of bioactive compounds and are among the most intensively studied in plant-based foods. 69 Blueberries are considered a rich source of polyphenols, including anthocyanins, phenolic acids, flavanols and tannins. 8 However, the bioavailability of polyphenols is generally low due to several factors that hinder their metabolism, such as poor solubility, high degrees of polymerization, complex chemical structures, and interactions with other molecules (Figure 5).35,70–72
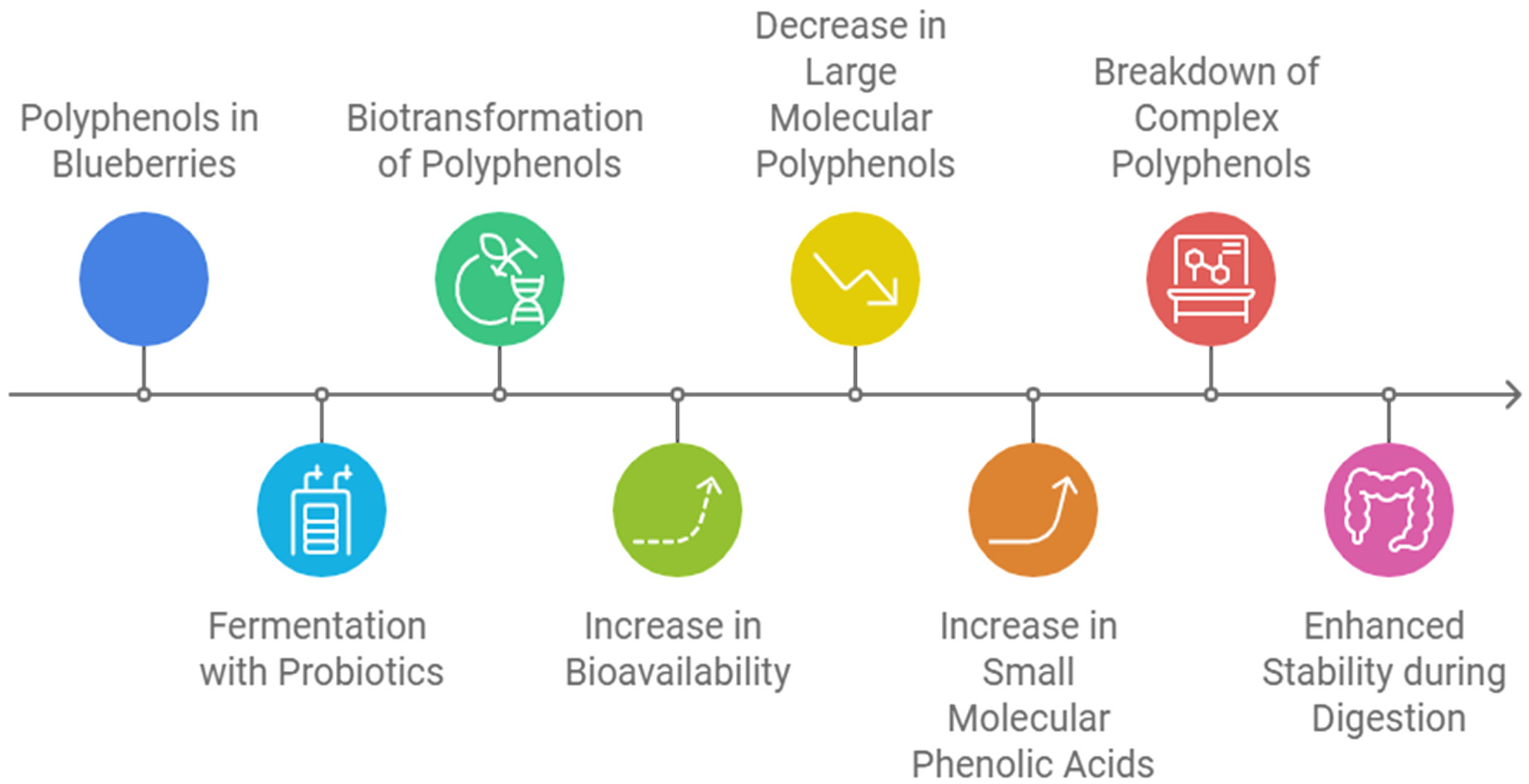
Summary of enhancing polyphenol bioavailability through fermentation.
The primary site of polyphenol biotransformation is the gastrointestinal tract, and it is estimated that between only 5–10% of polyphenols from the food matrix are absorbed and transported to the liver. 73 To better understand the bioavailability of polyphenols of blueberries, blueberry polyphenol-soy protein isolate (SPI) aggregates was prepared using spray drying. 27 The aggregates exhibited positive in vitro anti-inflammatory activity, including downregulation of inflammatory marker genes expression, which led to the inhibition of inflammatory factor production. Additionally, the PSI aggregates helped protect the stability of blueberry polyphenols by forming a thin layer around them for preservation and stabilisation purposes.27,73
The studies included in this review generally demonstrate that fermentation with probiotics leads to significant biotransformation of polyphenols in blueberries. For instance, a notable increase in total phenolic content in wild blueberry juice after one day of fermentation with a novel bacterium, S. vaccinii, isolated from the fruit microbiota. 26 This was evidenced by an increase of gallic acid concentration from 26.7 to 64.6 mg/ kg by day 3 of fermentation. Similarly, fermentation of blueberry juice with S. lactis increased polyphenol bioavailability by approximately 28.82%, while the concentration of bound phenols increased by 48.79%. 26 In contrast, a study by variable results were observed while levels of large molecular polyphenols, such as proanthocyanidins, cyanidin-3-O-glucoside, and malvidin-3-O-galactoside decreased, concentrations of smaller molecular phenolic acids increased. 38 Specifically, protocatechuic acid and caffeic acid levels increased from 19.12 mg/g and 11.02 mg/g to 32.01 mg/g and 24.27 mg/g, respectively. These findings are consistent with broader literature from other fruit fermentation studies who noted that LAB, particularly Lactobacillaceae, metabolize hydroxycinnamic and hydroxybenzoic acids into smaller, more bioavailable compounds through enzymes such as esterases, decarboxylases, and reductases, facilitating their absorption in the gastrointestinal tract (GIT). 73 For example, the conversion of hydroxycinnamic acids like ferulic acid into vinyl derivatives or dihydroferulic acid by LAB enhances their bioavailability and reduces antimicrobial activity, which may serve as a detoxification mechanism. 73 In another example, fermentation of red raspberries with W. anomalus significantly reduced the level of anthocyanins while markedly increasing the levels of total polyphenols and flavonoids. 27 Similarly, hydrolases produced by lactic acid bacteria contributed to the increased total flavonoid content in mulberry juice. 30 LAB fermentation also significantly enhanced the total phenolic and flavonoid contents of wolfberry–longan juice. 72 In another study, fermentation of guava juice with two strains of L. plantarum (PTCC 1896 and PTCC 1745) led to a marked increase in total phenolic content. 72 Notably, L. plantarum has been reported to exhibit a higher capacity for metabolising phenolic acids than other probiotic strains. 45
Fermentation facilitates the breakdown of complex, high-molecular-weight polyphenols into smaller oligomers, which are more readily absorbed by the body. These biotransformations enhance the bioavailability of free phenols. Fermentation with L. plantarum and L. sakei metabolised malvidin-3-O-glucosides into syringic acid, protocatechuic acid, and gallic acid. 74 These changes were attributed to glucosidases produced by L. plantarum, which play a key role in phenolic metabolism. A study emphasized that glycosyl hydrolases in lactobacilli, such as those in L. melliventris, target sugar molecules attached to flavonoids, releasing aglycons that are more bioavailable. 73 Additionally, methylation of the hydroxyl groups on certain phenols reduces their susceptibility to sulfation and glucuronidation, thereby enhancing their metabolic stability and transport across the intestinal barrier, thus improving their bioavailability. 35 Anthocyanins may also interact with proteins in juice matrices, potentially shielding them from degradation and increasing their stability during digestion. 45 Furthermore, pH changes resulting from LAB activity during fermentation can influence the structure and reactivity of phenolic compounds, 42 a finding corroborated by another study that demonstrated that the metabolic activity of LAB in fruit fermentations modifies the chemical environment, affecting the stability and reactivity of phenolic compounds. 73 With the exception of TanA, most lactobacilli enzymes acting on phenolic compounds are intracellular, requiring either transport of these compounds across the cytoplasmic membrane or cell lysis for substrate conversion. 75 Additionally, studies have highlighted the critical role of gut microbiota enzymes, specifically glucosidases and glucuronidases, in enhancing polyphenol bioavailability. 75 These enzymes hydrolyse O-glycosides and phase II-derived O-glucuronides, releasing aglycones for re-absorption. This microbial activity accounts for the biphasic peaks in polyphenol plasma concentrations, improving their bioavailability, extending their half-life, and increasing exposure to gut mucosa. Such effects are particularly significant for therapeutic applications, such as inflammatory bowel diseases (IBD). 75
However, not all studies have reported increased phenolic content following fermentation. For example, a significant reduction in both total anthocyanin content and total polyphenol content in fermented blueberry by-products. 76 This loss was attributed to the activity of polyphenol oxidase and other factors such as temperature and enzymatic degradation. Similarly, total phenolic content over a 96-h fermentation period using a co-fermentation of B. amyloliquefaciens, L. brevis, and S. bombicola, and reported a gradual decrease in phenolic content compared to the control. 31 Among the strains tested, B. amyloliquefaciens exhibited the most pronounced reduction, while S. bombicola and L. brevis showed comparable decreases after 96 H of fermentation. In addition to modulating probiotic metabolism, fermentation substantially alters the nutritional profile of blueberries. Microbial activity influences carbohydrate availability, organic acid composition, amino acid utilization, mineral bioaccessibility, and vitamin stability, thereby transforming blueberries from a raw phytochemical source into a nutritionally dynamic functional matrix.
Impact of fermentation on nutritional components
Fermentation by probiotic bacteria, particularly LAB profoundly transform the nutritional and chemical profile of blueberries. This process converts complex polymers into simpler, more bioavailable metabolites while simultaneously modulating sugars, organic acids, vitamins, and volatile compounds, thereby enhancing overall functional value.
Enhancement of Phenolic and Flavonoid Profiles
The most prominent nutritional benefit of fermentation is the substantial increase in total phenolic content (TPC) and total flavonoid content (TFC). LAB secrete enzymes such as cellulase, tannase, and esterase that degrade plant cell walls and disrupt cross-links between polymers, thereby releasing soluble conjugated or insoluble bound phenolic compounds. 72 Studies consistently report TPC increases ranging from 6.1% to 81.2% during lactic acid fermentation. 77 Complex tannins and flavonoids are further biotransformed into simpler, highly bioactive phenolic acids, including gallic acid, protocatechuic acid, catechol, ferulic acid, and syringic acid.62,78 For example, rutin can be hydrolyzed into its more bioactive aglycone form, quercetin. 79
Dynamic Changes in Anthocyanins
The effect on anthocyanins, the primary pigments responsible for blueberry color, is more complex, typically involving a net decrease in total content coupled with shifts in monomer stability and composition. Microorganisms utilize the sugar moieties of anthocyanins as an energy source, leading to reductions of over 30% in total anthocyanin levels.45,77 Probiotics produce β-glucosidase and β-galactosidase enzymes that cleave glucose or galactose bonds, degrading anthocyanins into anthocyanidins and corresponding phenolic acids. 45 However, the progressive acidification of the medium during fermentation (pH drop) can stabilize remaining anthocyanins by favouring their most stable flavylium cation form. 62
Macronutrient Utilization and Organic Acid Synthesis
Fermentation effectively “pre-digests” the fruit's carbohydrates and proteins to support microbial proliferation. Probiotics rapidly consume glucose and fructose, the main carbon sources in blueberries, while certain strains, such as L. plantarum NCU116, can utilize sucrose as a secondary carbon source later in the process. 61 Sugar metabolism results in a marked increase in lactic acid, alongside elevations in acetic, succinic, and oxalic acids; conversely, citric and malic acids often decrease as they are consumed in pathways such as the tricarboxylic acid (TCA) cycle.45,77 Free amino acids (e.g., arginine, glutamic acid, and leucine) universally decline as LAB utilize them as nitrogen sources and for adaptation to acidic stress. 61
Bioavailability and Bio-accessibility
Fermentation markedly enhances the bio-accessibility and bioavailability of blueberry nutrients, allowing a greater proportion of compounds to be absorbed and enter systemic circulation. 35 The conversion of large polyphenols into smaller molecular oligomers circumvents the poor absorption of high-molecular-weight polymers in the small intestine. 72 Additionally, LAB-mediated modifications such as methylation or acylation improve the metabolic stability and cellular transport of phenolic compounds. 35
Aroma and Volatile Organic Compounds (VOCs)
Nutritional transformations are accompanied by significant enhancements in flavour-active compounds. Total VOC content can increase dramatically (up to 13.6-fold), with enrichments in terpenes (e.g., linalool, geraniol) and alcohols (e.g., 1-hexanol) that contribute fruity, floral, and green notes. 61 Esterification reactions, driven by probiotics, produce esters such as ethyl acetate and ethyl lactate from alcohols and acids, further intensifying the mellow, fruity aroma profile.61,78 (Table 4).
Overview of the polyphenols found in the studies of this review and their health benefits.
Antioxidant activity of fermented blueberries
Antioxidant activity is another important aspect influenced by probiotic fermentation. The fermentation of blueberries with probiotics has been shown to significantly enhance their antioxidant and radical scavenging capacities through various mechanisms, including the stabilisation and biotransformation of phenolic compounds. The studies included in this review employ a range of assays to assess antioxidant activity, including the DPPH radical scavenging method, superoxide anion and hydroxy radical scavenging assays, 2,2`-azino-bis (3-ethylbenzothiazoline-6-sulfonic acid) (ABTS) assay, cupric-reducing antioxidant capacity (CUPRAC) assay, ferric-reducing ability power (FRAP) assay, α-tocopherol equivalent antioxidant capacity (αTEAC) assay and the thiobarbituric acid reactive substances (TBARS) assay. The majority of the reviewed studies reported an increase in antioxidant activity following fermentation. However, several factors including the specific probiotic strains used, fermentation conditions, and the type of blueberry matrix can influence the final outcome.
Studies reported increased antioxidant activity post-fermentation.26,30 These could be attributed to the increased total phenolics and alterations in the phenolic profile, as indicated by enhanced radical scavenging activity. 26 Similarly, another study demonstrated that probiotic-mediated fermentation significantly boosted the antioxidant activity of blueberry extracts, with fermented samples exhibiting higher ABTS and DPPH radical scavenging activity compared to non-fermented controls. 30 Isolated LAB strains exhibited strong antioxidant properties, with DPPH scavenging rates of 85.45% and 87.45% for L. rhamnosus and L. plantarum as seen in Figure 6. 51 A study reported significant improvements in DPPH, superoxide anion, and hydroxy radical scavenging activity in blueberry juice fermented by L. plantarum J26. 42 Likewise, the metabolism of phenolic compounds by L. plantarum, S. thermophilus, and B. bifidum led to an increase in ABTS radical scavenging activity by 40 ‒ 60%. 45
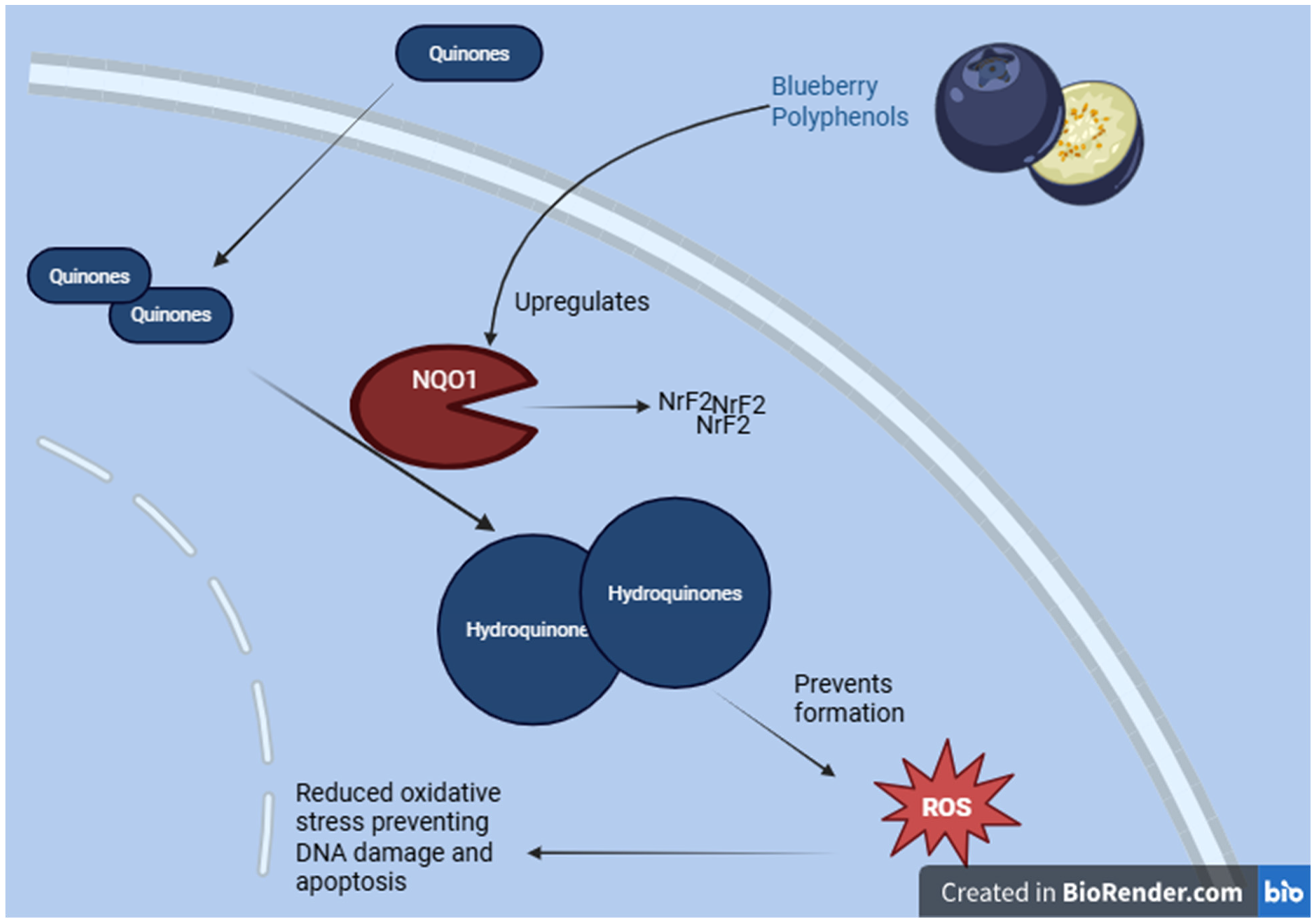
Schematic diagram, created using https://BioRender.com, illustrating the process by which Quinone Reductase (QR) detoxifies quinones, prevents ROS generation, and stabilizes antioxidant pathways, thereby protecting cells from oxidative stress.
This enhancement in antioxidant activity is primarily attributed to the biotransformation of polyphenols into metabolites with higher antioxidant potential. Fermenting wild blueberry juice with S. vaccinii resulted in a significant increase in both total phenolic content and radical scavenging activity after just one day of incubation. 26 They identified the formation of gallic acid, absent in non-fermented samples as a key indicator of this transformation, suggesting that the breakdown of hydrolysable tannins during fermentation contributed to the observed effect. Similarly, fermentation with L. plantarum led to a 43.42% increase in total phenolic content and enhanced DPPH, superoxide anion, and hydroxyl radical scavenging abilities. 42 Despite these findings, some studies have reported decreases in total phenolic content post-fermentation.35,78,82 These discrepancies are due to differences in microbial strains, fermentation substrates, and environmental conditions. For example, a study found that intact blueberry juice fermented with L. fermentum and L. plantarum had higher total phenolic content than juice fermented with S. thermophilus alone. 78 Conversely, although Kombucha-fermented blueberry juice contained more phenolic compounds than the non-fermented control, it exhibited lower antioxidant activity. 83
The reviewed studies also indicate that the combination of probiotic strains can influence the phenolic content and antioxidant activity of fermented blueberries. For instance, co-fermentation with L. fermentum and L. plantarum produced a synergistic effect, enhancing both the phenolic content and antioxidant capacity of blueberry juice compared to single-strain fermentation. 78 A similar study examined the effects of mixed fermentation of blueberry pomace with L. rhamnosus GG and L. plantarum-1.34,84 These results showed improved antioxidant activity, due to increased concentrations in total phenols and flavonoids.34,84 In contrast, combining S. thermophilus with either L. fermentum or L. plantarum did not enhance total phenolic content. 83
However, some studies report a decrease in antioxidant activity after fermentation. Lower antioxidant activity in fermented blueberry beverages compared to non-fermented controls, specifically in terms of DPPH radical scavenging activity (%), which showed only 16.64% inhibition. 83 This reduction was attributed to the alterations in anthocyanins during fermentation, likely due to a drop in the medium's pH. Different fermentation processes applied to blueberry pomace also showed that acetification significantly reduced total phenolic content, anthocyanin levels, and antioxidant activity, although some antioxidant properties and phenolic compounds were retained in the resulting vinegar. 76 In addition to free radical scavenging reduction (Table 5) and Chelation Reactions (Table 6), several studies have reported the modulation of antioxidant enzymes, assessing the effects of blueberry extracts on the activity of endogenous antioxidant enzymes in host tissue or serum.5,24
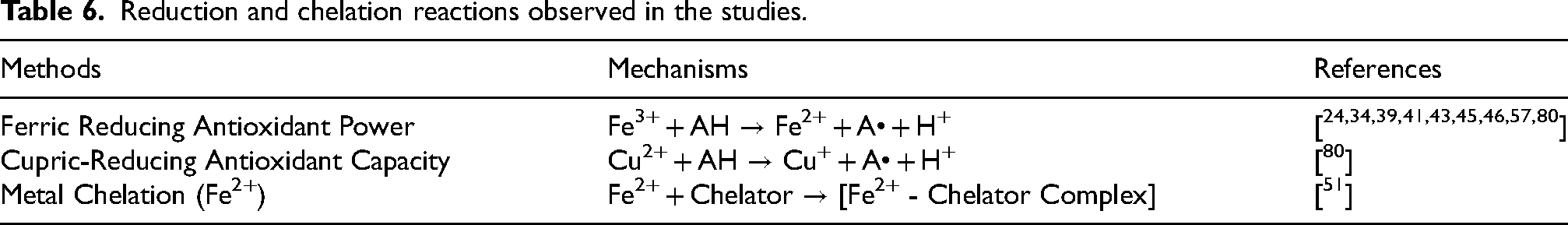
The studies primarily assessed the free radical scavenging blueberries, focusing on their ability to neutralize reactive species.
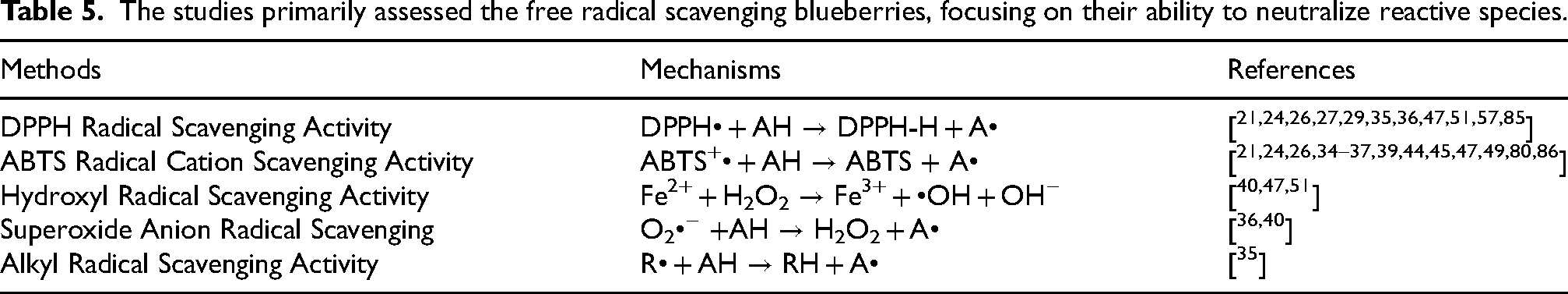
Reduction and chelation reactions observed in the studies.
Fermented blueberry pomace significantly enhances the activity of antioxidant enzymes in serum, including Superoxide Dismutase (SOD), Glutathione Peroxidase (GSH-Px), and Catalase (CAT), which subsequently reduced lipid peroxidation. Additionally fermented blueberry pomace elevated Glutathione (GSH) levels in the colon, indicating an attenuation of oxidative stress. Stimulation of Phase II detoxification enzymes, such as Quinone Reductase (QR). 22 Blueberry extracts rich in water-soluble phenolic compounds significantly increased QR activity compared to the negative control, suggesting Enhance protection against reactive oxygen species (ROS) (Figure 6).
A study investigated the effects of Lactobacillus casei-fermented blueberry pomace on colonic barrier function in high-fat diet mice. 24 The impact on colonic oxidative status was assessed by measuring the activities of Glutathione (GSH), Total Antioxidant Capacity (TAC), Catalase (CAT), and Superoxide Dismutase (SOD) in colonic tissues. Supplementation with fermented blueberries attenuated the decline in these enzyme activities. Overall, these the potential of studies highlight the potential microbial fermentation to modulate key enzymatic pathways, enhancing the therapeutic properties of blueberry-based beverages. 22
Additional biological activities of fermented blueberries
Fermented blueberries exhibit a wide range of health-promoting properties beyond their basic nutritional value, primarily due to enhanced bioavailability of bioactive compounds such as phenolic acids, anthocyanins, and postbiotic metabolites generated during fermentation. These activities span metabolic regulation, cardiovascular protection, anti-obesity effects, neuroprotection, and gastrointestinal health.
Metabolic and anti-diabetic activity
Fermented blueberries show considerable promise as anti-hyperglycemic agents. They inhibit key carbohydrate-hydrolysing enzymes, including α-glucosidase and α-amylase, functioning as natural “carbohydrate blockers” that attenuate rapid postprandial blood glucose spikes.41,87 Moreover, fermented blueberry juice improves insulin sensitivity, safeguards pancreatic β-cells against oxidative damage, and promotes the secretion of glucagon-like peptide-1 (GLP-1), which in turn suppresses hepatic gluconeogenesis. 41 In high-fat diet models, fermented products have also been shown to reduce fat mass, hepatic steatosis, and body weight gain by normalizing glucose and lipid metabolism. 47
Cardiovascular and vascular function
Fermented blueberries contribute to vascular homeostasis and overall cardiovascular health through multiple mechanisms. Chronic consumption is associated with reductions in both systolic and diastolic blood pressure. 41 The bioactive compounds in the fermented matrix enhance endothelial function by upregulating nitric oxide production and inhibiting low-density lipoprotein (LDL) oxidation, a critical step in atherogenesis. 41 Additionally, blueberries fermentation-enriched phenolic acids exhibit antithrombotic effects by disrupting platelet aggregation, thereby potentially lowering the risk of ischemic events. 41
Anti-obesity activity
Fermented blueberries counteract obesity by suppressing lipogenesis and reducing the size of lipid droplets. 41 They also modulate the gut-brain axis through regulation of satiety hormones, notably by inhibiting neuropeptide Y (NPY) to decrease appetite and stimulating leptin to enhance feelings of fullness. 41
Neuroprotective and Cognitive Health Fermentation facilitates the transport of blueberry metabolites across the blood-brain barrier, thereby amplifying their neuroprotective potential. 41 These compounds promote neurogenesis and neuroplasticity by elevating brain-derived neurotrophic factor (BDNF) levels. Furthermore, they offer protection against neurodegenerative disorders, such as Alzheimer's and Parkinson's diseases, by inhibiting amyloid-beta peptide aggregation and mitigating oxidative stress in neural cells. 41
Improvement of gastrointestinal barrier function
Fermented blueberry products enhance intestinal morphology and barrier integrity. 78 They increase ileal villus length and improve the villus-to-crypt ratio, thereby supporting better nutrient absorption. 78 These products also reinforce tight junctions by upregulating key proteins such as occludin and zonula occludens-1 (ZO-1), which help prevent “leaky gut”. 33 Additionally, they boost mucin (MUC2) production and goblet cell numbers, resulting in a thicker protective mucus layer. 78
Concluding remarks and future perspectives
Blueberry fermentation has been shown to positively influence probiotic growth, polyphenol bioavailability, probiotic metabolism, and antioxidant activity, making it a promising strategy for enhancing the health benefits of fermented foods. Blueberries, in their various forms (juice, pomace, powder, and extracts), provide an effective medium for the cultivation of diverse probiotic strains, including Lactobacillus, Bifidobacterium, Streptococcus, and Saccharomyces. The biotransformation of phenolic compounds during fermentation significantly improves their bioavailability, thereby enhancing antioxidant activity and promoting the production of health-beneficial metabolites such as SCFAs and organic acids. These transformations are complex and influenced by several factors, including the probiotic strain used, fermentation conditions, and the specific form of blueberry substrate. While the reviewed studies offer compelling evidence for the modulatory effects of blueberry fermentation on probiotic growth and metabolism, further research is needed to optimise fermentation parameters and fully elucidate the underlying mechanisms that govern metabolite production. Clinical trials in humans are particularly crucial to validate the beneficial effects observed in in vitro and animal studies, ultimately supporting the development of innovative blueberry beverages and personalised probiotic therapies with demonstrated health benefits. In recent years, polyphenols have garnered increasing attention, with multiple studies highlighting their role as key secondary metabolites and promising candidates for the next generation of prebiotics. 42 Preclinical evidence indicates that polyphenols can modulate the composition of beneficial gut bacteria and enhance the production of metabolites that promote gut homeostasis and a healthy microbiome. Findings from fermented blueberries are consistent with broader evidence from other fermented fruits and vegetables, including mulberry, wolfberry, guava, raspberry, persimmon, and longan-based fermentations. Across these systems, fermentation-driven biotransformation of polyphenols, organic acids, and microbial metabolites enhances biological efficacy, supporting the concept of food–medicine homology. These findings align with the emerging paradigm of food-medicine homology, which emphasizes that many foods possess therapeutic properties comparable to pharmaceuticals and can be utilized for both nutritional and medicinal purposes. 88 The comprehensive biotransformation processes occurring during probiotic fermentation of polyphenol-rich substrates such as those documented in blueberries and numerous other fruits and or vegetables represent a practical manifestation of this ancient concept in modern functional food science. The resulting products not only serve as conventional foods but also function as vehicles for delivering bioactive postbiotics, improved bioavailable polyphenols, and gut microbiome-modulating metabolites, blurring the traditional boundary between food and medicine.
To advance in this field, several cutting-edge and interdisciplinary approaches can be employed. For example, metagenomics, transcriptomics and metabolomics can provide a comprehensive understanding of microbial dynamics and metabolic pathways during fermentation, enabling identification of key genes and enzymes involved in polyphenol biotransformation and SCFA synthesis. The monitoring of multiple polyphenolic compounds during blueberry fermentation using a multi-source data deep fusion AI-based strategy, combined with non-destructive techniques such as near-infrared spectroscopy, can enhance our understanding of product quality, 75 safety, 76 and real-time detection of such chemometric analysis during fermentation.69,76 Additionally, AI can improve bioavailability of polyphenols, 31 support sustainable food design, identify emerging trends in the food industry, and optimise strategies for blueberry-based beverages. 78 AI-driven research on blueberry polyphenol bioavailability transformative potential to establish their role as the next generation prebiotics.33,82,83 Furthermore, precision blueberry fermentation integrating machine learning and real-time automation may offer a paradigm shift in the process, particularly in the design of probiotic strains, fermentation monitoring, anthocyanins bioavailability, and overall system control. 89
High-resolution techniques such as liquid chromatography-tandem mass spectrometry (LC-MS/MS) and nuclear magnetic resonance (NMR) spectroscopy can be used to profile blueberry-derived metabolites and track their transformations across different systems. Exploring next-generation probiotic strains, such as Akkermansia muciniphila or Faecalibacterium prausnitzii, may reveal novel interactions with blueberry substrate, given their unique metabolic capacities. Additionally, encapsulation techniques using blueberry-derived polyphenols or other biocompatible materials (e.g., alginate, chitosan) can improve probiotic survival under gastrointestinal conditions, as demonstrated by. 49 Nanoparticles (NPs) and nanozymes (NZs) also hold promise for enhancing the stability, targeted delivery, and bioavailability of bioactive compounds. 76
Future research should address several critical research gaps, including optimisation of fermentation conditions to maximise the production of health-promoting metabolites, elucidation of underlying mechanisms, and the integration of advanced technologies, such as gene editing and artificial intelligence, for the development of novel probiotic formulations in blueberry fermentation. Furthermore, a deeper understanding of the strain-specific efficacy of probiotics, the long-term consumption on systemic interactions, and targeted clinical interventions will pave the way for personalised probiotic therapies and innovative blueberry-based beverages aimed at restoring metabolic balance in the human gut microbiome. Together, these innovations can help bridge the gap between laboratory findings and clinical applications, driving the development of next-generation functional foods with improved efficiency and health benefits.
Footnotes
Acknowledgment
The authors express their gratitude to the Department of Biochemistry, Microbiology and Biotechnology, and the Science and Technology Division of MRS at the University of Namibia and the Department of Chemistry at the Cape Peninsula University of Technology in South Africa for providing financial support for the MSc research project and analysis conducted in this study.
Author contributions
Maggy M. Casey Nairenge: Writing – original draft, methodology, and data curation; Ahmad Cheikhyoussef: Writing – review and editing, supervision, project administration, conceptualization, methodology and validation; Ahmed A. Hussein: Writing – review and editing, supervision, and validation.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
