Abstract
Background
Although levodopa is the gold standard treatment for Parkinson's disease (PD), its chronic use is associated with levodopa-induced dyskinesia (LID), a motor complication that impacts prognosis, quality of life, and treatment costs. Most known LID-associated factors have been identified in European-descendant populations.
Objectives
To describe the epidemiology of LID in Latin American and Caribbean (LATAM) countries and assess the relevance of known and novel LID-associated factors in this population.
Methods
We conducted a cross-sectional study using data from the Latin American Research consortium on the Genetics of Parkinson's Disease (LARGE-PD). We included PD patients with information on LID status and levodopa use from eight LATAM countries. LID prevalence was calculated overall and by country. Countries were compared on demographic and clinical variables. Logistic regression was used to identify associations with LID.
Results
A total of 3695 PD patients (58.8% male) were included. Overall LID prevalence was 25.4% [95% CI: 24.06–26.87], ranging from 9.3% in Colombia to 45.1% in Puerto Rico. Prevalence increased progressively with longer disease duration. Country comparisons showed that not all known LID-associated factors explained prevalence differences. In logistic regression, fast disease progression was significantly associated with LID (OR: 1.55, 95%CI: 1.16–2.07), while sex was not (OR: 1.02, 95%CI: 0.87–1.18).
Conclusions
This is the largest study on LID epidemiology in LATAM. While some known risk factors remain relevant, others, like sex, do not, underscoring the need for population-specific studies. Future work should integrate environmental, clinical, and genetic data to better understand LID mechanisms.
Introduction
Treatments for Parkinson's disease (PD) are primarily symptomatic, as no disease-modifying therapies are currently available. Among these, levodopa, administered in combination with dopa decarboxylase inhibitors, is considered the gold standard and a first-line treatment for PD. 1 Levodopa is effective in controlling motor symptoms and improving survival.2,3 However, over time, its use is associated with the development of levodopa-induced dyskinesia (LID), an unpredictable motor complication that can range from mild to severe.1,2,4
LID is characterized by involuntary, discontinuous and arrhythmic movements that may affect various body regions. It is commonly classified into four main subtypes based on the timing of onset relative to levodopa administration (ON and OFF phases), anatomical distribution and movement patterns. 5
Regardless of type, LID can significantly impact the quality of life of people with PD and their families by increasing the risk of disability, balance impairments, and falls, which in turn raise treatment costs.1,6–8 Although certain advanced therapies, such as deep-brain stimulation (DBS) and infusion therapies, have proven effectiveness in alleviating LID, access to these interventions is limited, with only a minority of patients able to benefit from them. 8 This is particularly relevant in developing countries, where healthcare services are restricted to economic and technological constraints. 9
The prevalence of dyskinesia has been primarily reported in European populations, with more recent data emerging from Asian countries such as South Korea. Specifically, reported LID prevalence for European countries ranges from 14% to 30%,10–14 whereas in Asian countries it is approximately 40%. 15 However, LID prevalence has been shown to vary widely depending on the duration of PD, with rates gradually increasing over time for both populations.13–15
In addition to disease duration, other clinical factors have been associated with LID occurrence, including age at PD onset, disease severity, sex, body weight and the daily dose of levodopa therapy.7,14,16 However, as these findings are mainly derived from non-Latino populations, their relevance to Latino individuals remains unclear. This limits their applicability in clinical decision-making aimed at preventing or delaying the onset of LID.
Moreover, the limited literature on LID in Latino populations primarily comes from studies in Brazil9,17 and Mexico, 18 which often feature small sample sizes, thereby restricting the generalizability of their findings to other Latin American and the Caribbean (LATAM) countries.
This study aims to describe, for the first time, the epidemiology of LID across multiple LATAM countries. Additionally, we seek to assess the relevance of previously known LID-associated factors in this population and to explore other clinical variables that may influence the likelihood of developing LID. Understanding these factors is essential for optimizing therapeutic decisions and informing health policies across the region.
Methods
We conducted a cross-sectional study using data from the Latin American Research consortium on the Genetics of PD (LARGE-PD). 19 A total of 4069 participants with a PD diagnosis confirmed by a movement disorders specialist were recruited between April 2019 and April 2025 across Argentina, Brazil, Chile, Colombia, El Salvador, Mexico, Peru, and Puerto Rico. Only participants with available data on LID status (yes vs. no) and a documented history of levodopa use were included in the analysis. Data corresponds to the April 2025 extraction from the ongoing LARGE-PD study.
Demographic and clinical data were extracted from the LARGE-PD questionnaire administered at the time of recruitment. While UPDRS Part IV was available at one site through routine clinical care, it was not collected uniformly across all participating countries. Therefore, LID status was defined by a consistent description provided by participants and confirmed through the observation of dyskinetic movements during the study visit, allowing reliable case identification and comparable estimation of LID frequency across countries, in line with the epidemiological surveillance aims of our study.
Variables relevant to LID were selected for analysis upon previous studies, and include participants’ age, age at onset (AAO), sex, presence of tremor as a presenting motor symptom, dopaminergic treatment dosage and duration, as well as anxiety, a non-motor symptom also previously associated with increased LID risk. The full list of variables is presented in Table 1.
Characteristics of participants with and without Levodopa-induced dyskinesia (LID).
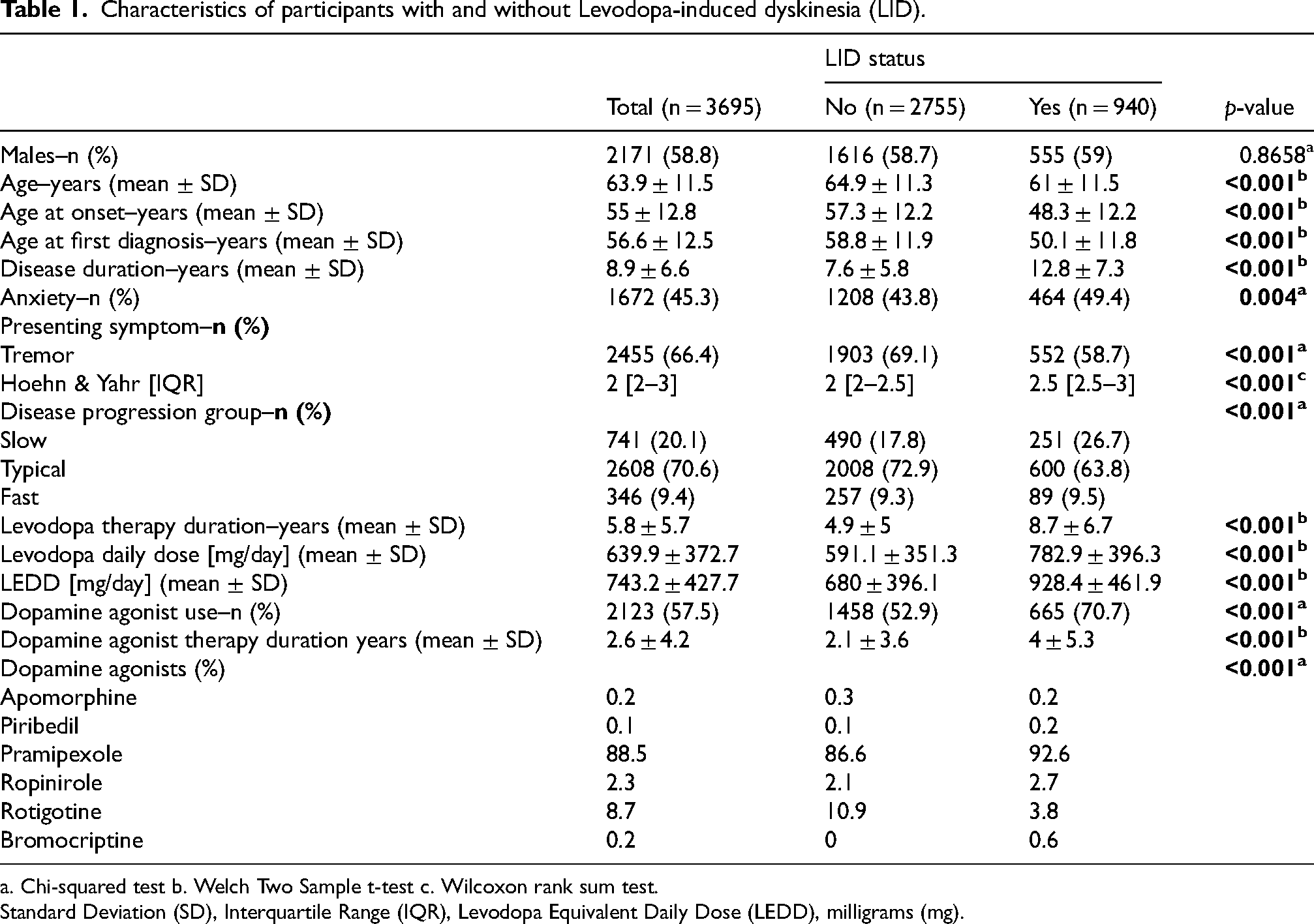
a. Chi-squared test b. Welch Two Sample t-test c. Wilcoxon rank sum test.
Standard Deviation (SD), Interquartile Range (IQR), Levodopa Equivalent Daily Dose (LEDD), milligrams (mg).
Participants with ≥20% missing data across variables were excluded. Simple imputation was applied for variables with <1% missingness, using the median for quantitative variables and the mode for qualitative variables. Variables with ≥1% but < 20% missingness were imputed using the Multivariate Imputation by Chained Equations (MICE) algorithm. 20 Sensitivity analyses were conducted using the dataset before imputation to ensure robustness of results.
Participants were categorized into two groups based on LID status (yes/no). Disease duration was defined as the number of years from the onset of the first motor symptom to the time of recruitment. For analysis, participants were categorized into five disease duration groups: 0–5 years, 6–10 years, 11–15 years, 16–20 years, or >20 years, following the classification used by Chang et al. 15
LID prevalence was calculated for the entire cohort and separately by country, stratified by disease duration groups. Prevalence estimates were accompanied by 95% confidence intervals. Additionally, prevalence was also analyzed by sex and AAO (<50 vs. ≥50 years old), following the cut-off for early-onset PD recommended by the Movement Disorders Society (MDS). 21
Disease progression was defined as a categorical variable using disease duration and Hoehn and Yahr (H&Y) stage at the time of recruitment. Based on the median time to reach H&Y stage 3 reported by Zhao et al. (∼8.9 years from stage 1), participants were classified as having “fast” progression if they reached H&Y ≥ 3 in less than 9 years, “slow” progression if they remained below stage 3 after 9 or more years, and “typical” otherwise. 22
The Levodopa Equivalent Daily Dose (LEDD) was calculated using the most recent formula proposed by Jost et al. 23 As the LARGE-PD questionnaire recorded daily doses only of levodopa and dopamine agonists, LEDD estimates were based solely on these medications. Considering the limited availability and affordability of other antiparkinsonian medications in Latin America, with levodopa being the most commonly available therapies in the region according with the World Health Organization Model Lists of Essential Medicines updated in 2025, 24 the calculated LEDD provides a reasonable approximation of dopaminergic load for patients with PD in our study and is suitable for epidemiological analyses within this healthcare context. 25
Numerical variables were summarized using means or medians, and categorical variables were reported as percentages. Differences between LID status groups were assessed using Chi-square tests for categorical variables and Wilcoxon or t-tests for numerical variables, as appropriate.
Differences in overall estimated LID prevalence between countries, as well as prevalence stratified by disease duration, were evaluated using pairwise Chi-square tests with Bonferroni adjustment. Similarly, average LEDD values, disease duration and AAO across countries were compared using Kruskal-Wallis test, followed by post hoc pairwise comparisons with Dunn's test and Bonferroni adjustment.
Simple logistic regression analyses were conducted to identify variables associated with a higher probability of developing LID. Variables that reached statistical significance were included in a multiple logistic regression model. Collinearity was assessed before model inclusion. Country of residence was included as a covariate to adjust for potential confounding due to between-country differences. The country with a LID prevalence closest to the cohort-wide average was selected as the reference group.
A significance threshold of p < 0.05 was used for all statistical tests. All analyses were completed using R version 4.3.1.
This study was approved by the Institutional Review Board of the Cleveland Clinic Foundation (IRB # 25- 081). In addition, each participating LARGE-PD site obtained approval from its local IRB in accordance with international ethical standards, and all participants provided written informed consent prior to enrollment.
Results
Demographics
A total of 3695 patients with PD (mean age 63.9 ± 11.5 years, 58.8% males) were included in this study. The average disease duration was 8.9 ± 6.6 years (Table 1).
The overall prevalence of LID in the entire group was 25.4%. When analyzed by country, Colombia had the lowest prevalence (9.3%) while Puerto Rico had the highest (45.1%) (Table 2).
Prevalence of Levodopa-induced dyskinesia (LID) among participants overall and by country.
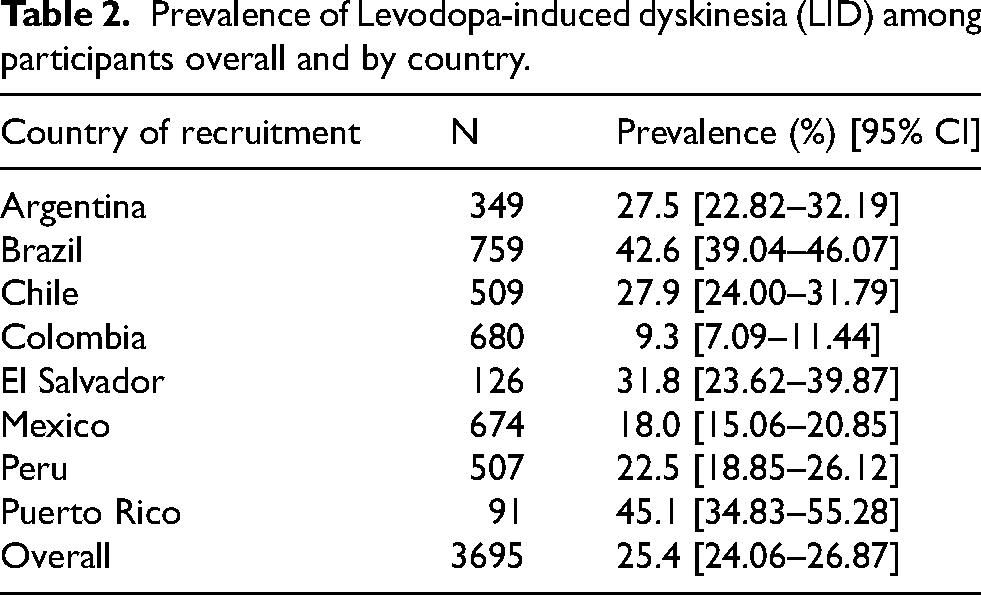
Statistically significant differences in overall LID prevalence were found between most countries, based on pairwise Chi-square tests with Bonferroni adjustment. Colombia was the only country with a significant lower prevalence compared to all others (Supplementary Figure 1). While differences in demographic and clinical characteristics were observed between some countries, these were not consistently present in all comparisons involving those with significantly different LID prevalence (Supplementary Figure 2).
LID-associated factors
Univariate model
Thirteen different factors were found to be significantly associated with LID in the univariate logistic regression analysis, including AAO, disease duration, LEDD, use of dopamine agonists, presence of tremor as an initial motor symptom, history of anxiety or panic as non-motor symptoms, and disease progression rate, among others. Interestingly, sex was not significantly associated with LID (Table 3).
Univariate and multivariable logistic regression analyses of factors associated with Levodopa-induced dyskinesia (LID).
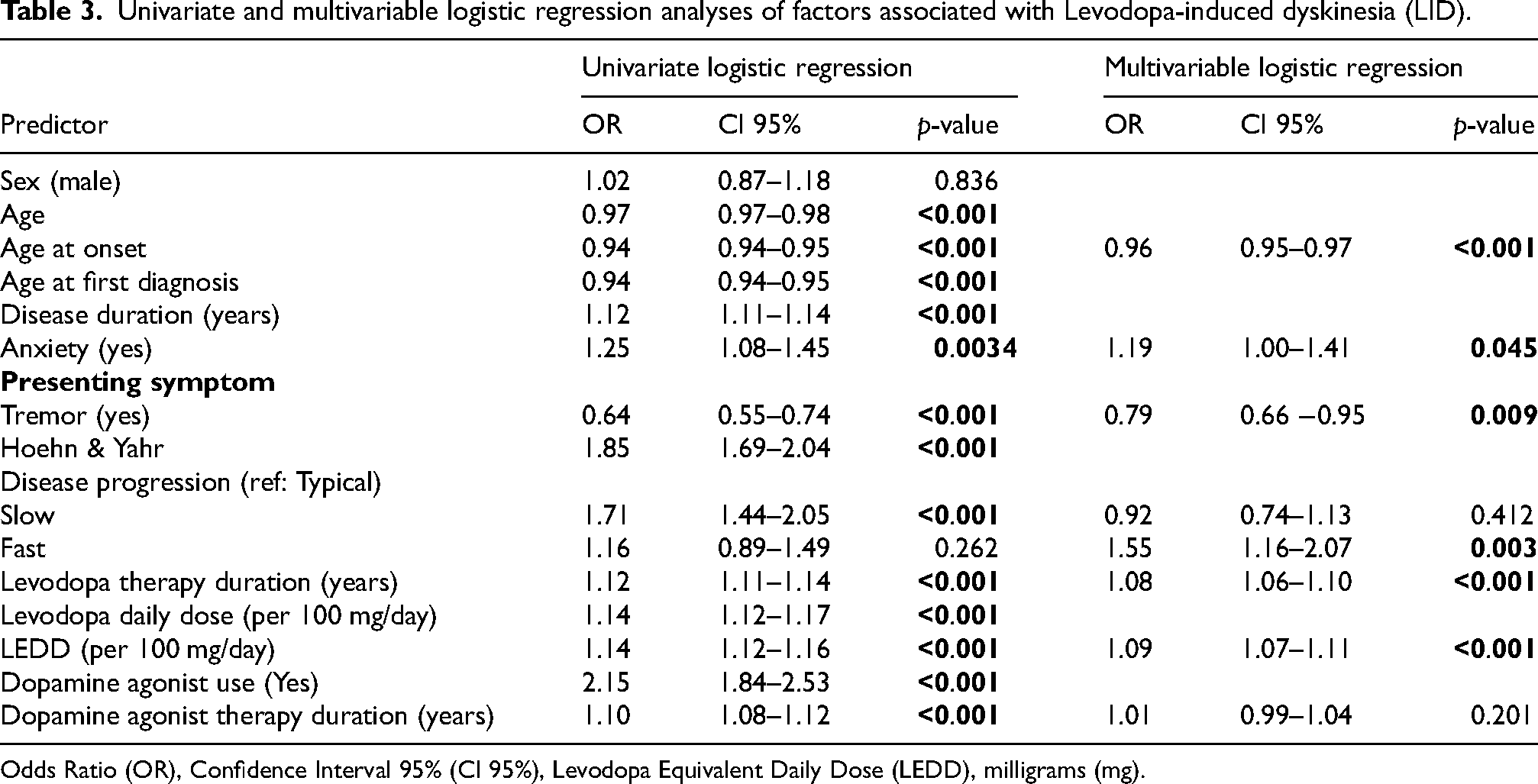
Odds Ratio (OR), Confidence Interval 95% (CI 95%), Levodopa Equivalent Daily Dose (LEDD), milligrams (mg).
Disease duration
A significant gradual increase in LID prevalence was observed across the defined disease duration groups (p < 0.05), with the most notable occurring between the first three groups (0–5 years; 6–10 years; 11–15 years). Pairwise comparisons using Chi-square tests with Bonferroni adjustment confirmed significant differences between adjacent groups up to the 16–20 years group, with a non-significant difference between the 16–20 and >20 years groups (Supplementary Table 1).
LID prevalence by disease duration group was also analyzed by country. This pattern was consistent across most countries, except for Peru and Puerto Rico (Figure 1).
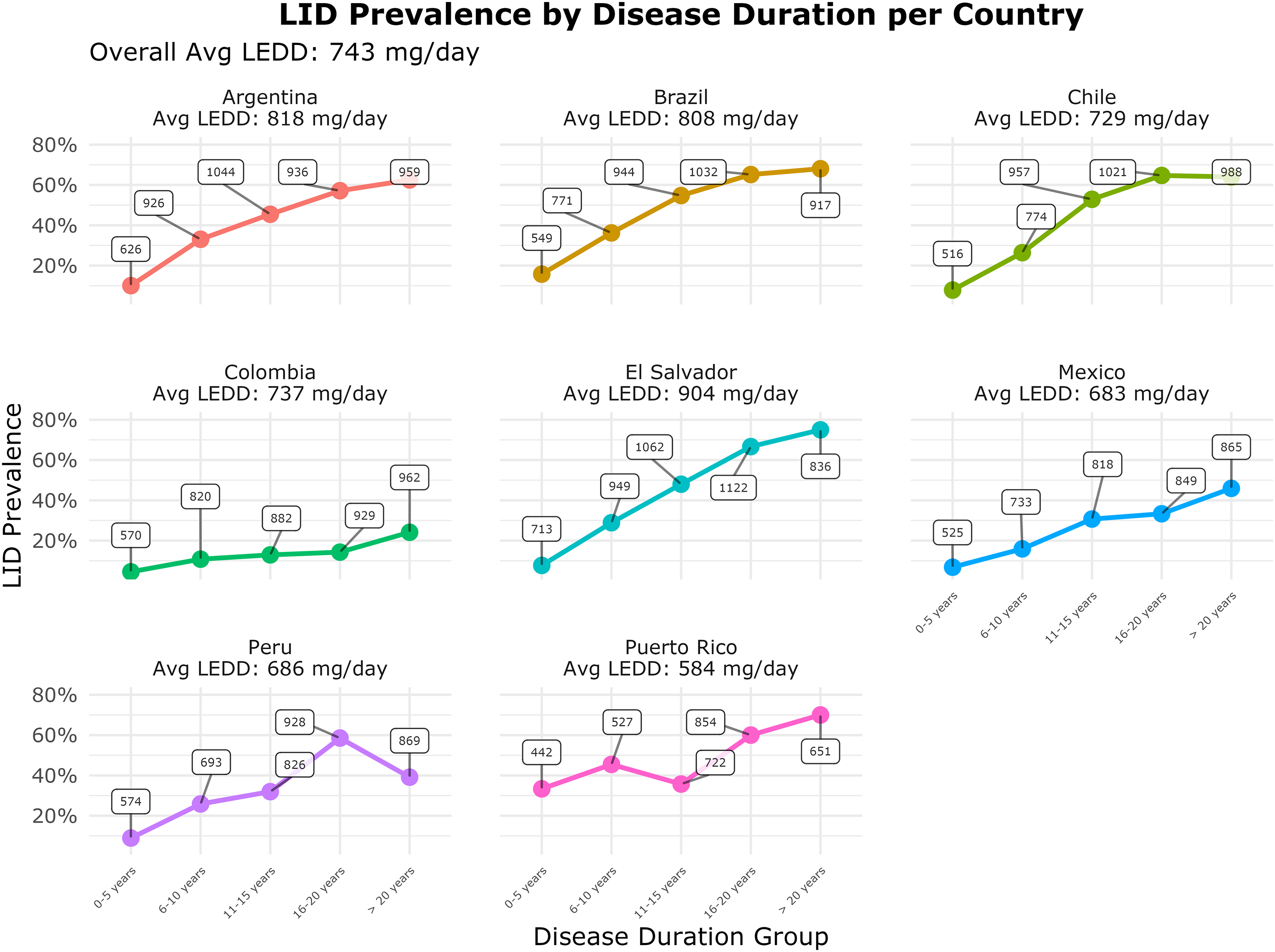
Levodopa-induced dyskinesia (LID) prevalence by Parkinson's disease duration and country, with average levodopa equivalent daily dose (Avg LEDD). Lines indicate country-specific LID trends; points show prevalence per duration group. White boxes and panel titles display Avg LEDD.
Age at onset
A similar analysis was conducted by AAO category (<50 vs. ≥50 years old). The overall prevalence of LID was 42.8% among participants with AAO <50 years, compared to 17.4% in those with onset ≥50 years. When stratified by disease duration, LID prevalence remained significantly higher in the younger-onset group across all categories (p < 0.001), except in the >20 years group, where the difference was not statistically significant (56.2% vs. 50.0%, p = 0.565; Supplementary Table 2).
In both AAO groups, LID prevalence increased with disease duration. Among participants with onset before age 50, prevalence rose from 16.8% in the 0–5 years group to 61.8% in the 16–20 years of disease duration group, followed by a slight, non-significant decline to 56.2% in those with >20 years (Chi-square test, p = 1.00; Supplementary Figure 3). In contrast, among individuals with onset at or after age 50, LID prevalence gradually increased from 7.0% in the 0–5 years group to 50% in the >20-year group (Figure 2).
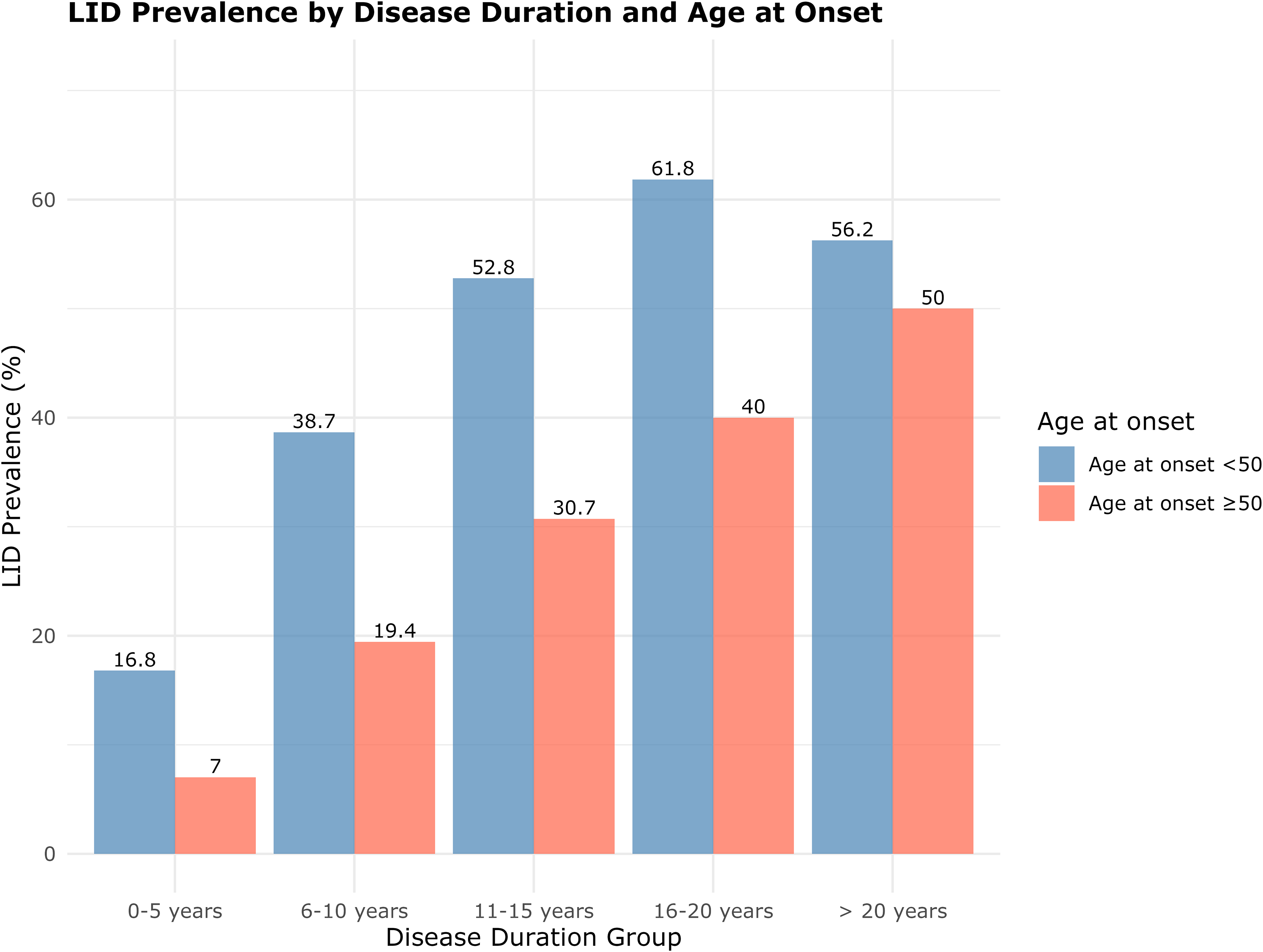
Levodopa-induced dyskinesia (LID) prevalence by disease duration and age at onset (AAO). Prevalence of LID (Y-axis) across different Parkinson's disease duration groups (X-axis), stratified by AAO (<50 vs. ≥50 years-old).
Levodopa equivalent daily dose (LEDD)
While Kruskal-Wallis test indicated a statistically significant difference in average LEDD across countries (p < 0.05), not all pairwise comparisons were significant. For example, average LEDD in Colombia was comparable to that in Brazil, Chile, Mexico, or Peru (adjusted p > 0.05; Supplementary Figure 2).
Multivariable model
A multiple logistic regression model, including all variables independently associated with LID and accounting for collinearity, identified several clinical factors that remained significantly associated after adjustment. Increased odds of LID were observed in participants with younger AAO, a history of anxiety or panic as non-motor symptoms, longer duration of levodopa therapy, higher LEDD, and faster disease progression, whereas tremor as a presenting symptom was associated with decreased odds (Table 3).
Discussion
To our knowledge, this is the largest study to analyze the epidemiology of LID in LATAM countries. We estimated an overall LID prevalence of 25.4%, ranging from 9.3% to 45.1% across participating sites, with Colombia and Mexico presenting the lowest, and Brazil and Puerto Rico the highest (Table 2).
Our overall LID prevalence of 25.4% was lower than estimates reported in other populations within and outside LATAM. For instance, Gandhi et al., in a prospective cohort of 3343 European-descendant participants, reported a LID frequency of 42.1% among those with 8–10 years of disease duration. 13 Notably, their study did not report an overall prevalence, as all values were stratified by disease duration. We therefore used the 8–10 years group for comparison, as it was closest to the mean disease duration in our cohort (8.9 years).
In a smaller retrospective sample of 279 patients in the United States, Turcano et al., found a LID prevalence of 30.1%, 10 while Chang et al. reported 43% in a larger sample of 2571 individuals from South Korea. 15 We did not identify additional previous reports in larger European samples that did not account for AAO or disease duration when reporting LID frequency.
When comparing prevalence by country, our estimate for Brazil (42.6%) fell between two previous reports, 34.7% and 54.9%, from independent LARGE-PD cohorts of 187 and 224 participants, respectively. The 34.7% estimate corresponded to a non-overlapping sub-sample relative to the present study, whereas the 54.9% was derived from a separate analysis with partial sample overlap. In both groups, LID was defined as a score ≥1 on item 4.1 of UPDRS Part IV. 9 Despite differences in LID assessment methods, the similar prevalence estimates support the consistency of our combined self-reported and clinician-observed approach.
A similar trend of lower LID prevalence estimates was observed when stratifying by disease duration. Although prevalence in the 0–5 years group was comparable between our cohort (8.8%) and that of Chang et al. (8.2%), differences became more pronounced in longer-duration groups. Specifically, compared with our cohort, Chang et al. reported higher LID prevalence across all subsequent categories: 40.7% vs. 25.1% for 6–10 years, 66% vs. 39% for 11–15 years, 74.6% vs. 52% for 16–20 years, and 83.2% vs. 55% for >20 years of disease duration. 15
When examining country-specific differences in clinical variables commonly associated with LID, such as disease duration and LEDD, we found statistically significant differences between some countries, but not all. For instance, Colombia, which had the lowest prevalence, did not differ significantly in disease duration from any country except Brazil, which had the second-highest prevalence. Conversely, when comparing Colombia's LEDD with the other countries, significant differences were observed with Argentina, El Salvador and Puerto Rico (Table 2; Supplementary Figure 2).
These findings raise the possibility that other factors beyond the well-known clinical variables associated with LID may influence its development differently across countries. Potential contributors could include population differences in genetic variations related to dopaminergic pathways, 26 timely access to healthcare, 27 and variability in medication availability or prescription practices, particularly the use of immediate-release versus controlled- or extended-release of levodopa formulations. This distinction is important, as non-immediate-release formulations may reduce the pulsatile stimulation associated with immediate-release levodopa, which has been implicated in the pathophysiology of LID. 28
Clinical factors previously associated with an increased odd of developing dyskinesia, such as earlier age at disease onset, longer disease and levodopa treatment duration, and higher LEDD, were confirmed in our cohort.
Although the calculated LEDD included only levodopa and dopamine agonist doses, LEDD remained the strongest predictor in the multivariable logistic regression model, suggesting that the primary dopaminergic therapies captured by the questionnaire account for a substantial proportion of the dopaminergic exposure relevant to LID risk. This is consistent with previous evidence in which LEDD has been described as one of the most important predictors of LID. 29
Tremor as a presenting symptom was found to be negatively associated with LID, consistent with prior reports.7,14,17 In addition, fast disease progression emerged as a potential novel factor associated with the development of LID.
From a biological standpoint, motor complications are primarily driven by the progressive loss of dopaminergic neurons, which diminishes the brain's capacity to buffer dopamine. This is facilitated by an increased dopamine availability, which is processed by serotonergic neurons. 28 This mechanism may help explain our observation that patients with faster disease progression, who experience these neurobiological changes more rapidly, develop LID more frequently than those with slower progression.
The association observed between AAO and LID in our analysis adds further evidence to the well-established link between the younger onset of PD and increased risk of LID. This may reflect a combination of treatment-related and intrinsic predisposition. Although early-onset PD is generally associated with slower motor progression, our findings, along with previous reports, 15 suggest that these patients remain at high risk for LID due to factors other than the accelerated neurodegeneration typically linked to faster disease progression. 28
One possible explanation is the longer disease duration, often accompanied by an early and rapid increase in dopaminergic dosage, likely driven by the need to control motor symptoms and maintain daily functioning, including occupational and family responsibilities. 30 In addition, genetic susceptibility to the development of LID may also be relevant. Previous evidence suggests that PD patients in the highest quartile of Polygenic Risk Scores (PRS), which aggregate PD-associated variants, have a higher risk of developing LID. 26 Moreover, a separate study has reported that higher PD-PRS is associated with earlier onset of LID. 13
Our findings propose that faster disease progression and younger AAO represent complementary risk profiles in Latin American populations, each independently increasing susceptibility to LID through distinct mechanisms.
Anxiety, a non-motor symptom of PD that also reflects dopaminergic dysfunction, has been proposed as a factor associated with LID. For instance, Dias et al., in a cohort of 348 participants from the Parkinson's Progression Markers Initiative (PPMI), reported that higher levels of trait anxiety preceded dyskinesia onset, as measured using the State-Trait Anxiety Inventory (STAI). 31 Similarly, Eusebi et al., also utilizing data from PPMI of 398 participants, found that higher trait anxiety, measured by the STAI-Trait subscore, was associated with increased risk of dyskinesia. 7
Complementary experimental evidence is provided by Kuan et al., who observed signs of LID improvement in rats with induced and controlled anxiety levels after treating them with citalopram. Importantly, the chronic use of citalopram nearly abolished 5-HT1B serotonin receptor expression in the striatum of rats showing this improvement. These findings suggest a potential biological link between anxiety and the serotonergic system in LID, as receptor expression was higher in rats with elevated basal anxiety compared with those with low anxiety levels. However, in Kuan et al. experiment, no association was observed between anxiety levels and LID occurrence. 32
Despite the findings reported by Dias et al., in which anxiety was described as preceding the onset of LID, aggregated evidence suggests that this causal association is not completely clear. In a systematic review by van der Velden et al., aimed at exploring the association between non-motor symptom fluctuations and motor fluctuations, anxiety was reported to be associated with the “off” state, the “on” state, or to fluctuate independently of motor state. As an example highlighted within this review, Menza et al. reported mood improvement in patients with PD during transitions from the “off” to the “on” state, followed by mood deterioration in the presence of dyskinesia. 33
In our study, although anxiety was self-reported by participants and not assessed using standardized instruments, we were still able to replicate its association with LID, even after adjusting for other variables in the multivariable model. This finding, aligned with previously published evidence, suggests that anxiety may be associated with dyskinesia. However, the cross-sectional nature of our study does not allow causal inference or temporal relationships between anxiety and LID, and therefore we cannot determine whether anxiety precedes dyskinesia or is related to its manifestation. Further investigations are needed to elucidate the biological mechanisms underlying the association between LID and anxiety.
While some studies in non-Latino populations have reported a higher risk of LID among females, we did not observe this association (Supplementary Table 3; Supplementary Figure 4). Our findings align with previous studies conducted in Latin American countries, specifically Mexico and Brazil9,17,18 where no association between LID and sex was identified. This suggests that sex-related differences in LID risk may vary across populations, probably due to genetic, environmental, or healthcare system-related factors.
Prior studies have shown that the risk of dyskinesia is more strongly related to LEDD per kilogram of body weight than to absolute levodopa dose. 34 In this context, the absence of body weight data in our study could influence the observed strength of associations between LID and other clinical factors, such as levodopa treatment duration and disease progression, by introducing residual confounding related to medication exposure per unit of body mass. Furthermore, the availability of body weight data could help explain some of the observed differences in LID prevalence across countries, as cross-population variation in nutritional status and body composition may affect effective dopaminergic exposure by modifying the pharmacokinetics of levodopa. 35
The association between dopamine agonist use and dyskinesia has been widely debated. More recent evidence suggests that when dopamine agonists are used concurrently with levodopa, they may exacerbate existing dyskinesias. However, this effect may not stem from dopamine agonists alone, but rather from the concomitant use of levodopa. Dopamine agonists do not extend their effect into or beyond levodopa's optimal therapeutic window and, on their own, are less likely to trigger LID. 6 Our findings align with this, suggesting that the total number of years on dopamine agonists, is not associated with LID after adjusting for LEDD and levodopa treatment duration.
A similar debate has been raised regarding the relevance of levodopa treatment duration and LID, with some studies stating that longer levodopa therapies increase the risk of developing LID, 14 while others discarding this association. 36 In our study, a longer duration of levodopa therapy appeared to be significantly associated with LID, independently and when adjusting for other covariates such as disease duration and LEDD (Table 3).
However, given the cross-sectional nature of our study and the ongoing debate on these topics, these findings should be interpreted with caution.
Our study has several limitations. First, LID occurrence was determined using a binary classification based on participants self-report and clinical observation at the study visit, rather than through a standardized assessment tool such as UPDRS Part IV. While UPDRS Part IV data were not available for the full cohort, we performed a concordance analysis in the subset of participants with both measures present. In this group (728 of 759 individuals in the Brazilian LARGE-PD cohort), we observed a high concordance (80.6%) between our questionnaire-based LID classification and UPDRS Part IV item 4.1, suggesting that our approach is appropriate for identifying the presence or absence of dyskinesias. Nevertheless, this cross-sectional and simplified assessment may introduce recall bias and does not allow detailed characterization of dyskinesia severity, impact on quality of life, time to onset (time-to-LID), or specific dyskinesia subtypes. Consequently, we were unable to evaluate factors associated with these additional clinically meaningful characteristics of dyskinesia.
Second, the calculated LEDD may underestimate the actual dopaminergic load, as data on medications other than levodopa and dopamine agonists were unavailable, as well as details on levodopa formulations (e.g., immediate- vs. extended-release). Although LEDD remained one of the strongest factors associated with LID, this underestimation could affect the magnitude and interpretation of associations observed for other variables in the multivariable model. Additionally, information on other LID-targeted therapies, such as DBS, amantadine, or clozapine, was not available, and the use of these therapies could significantly influence the observed prevalence of LID in a cross-sectional analysis.
As a future direction, following completion of this study, the LARGE-PD consortium has expanded the set of recorded LID-related variables to include body weight, time-to-LID, prior use of DBS, and a more comprehensive assessment of LEDD through the recording of all available antiparkinsonian medications. These additions aim to facilitate more detailed investigation of clinical and demographic factors associated with dyskinesia in LATAM populations.
In conclusion, our findings represent the largest study to date on the epidemiology of LID in LATAM countries. We demonstrate that several previously known dyskinesia-associated factors remain relevant in Latino populations, while also identifying potential new contributors, such as the clinical progression pattern of PD as defined in our cohort (fast, slow, or typical progressors). The absence of some expected associations, such as female sex, emphasizes the importance of population-specific research on LID and highlights the need for regionally tailored strategies for its prevention and management. Future studies incorporating more detailed clinical characterizations of dyskinesias, along with environmental, expositional, and genetic data, are needed to better understand the underlying mechanisms of LID.
Supplemental Material
sj-docx-1-pkn-10.1177_1877718X261431204 - Supplemental material for Levodopa-Induced dyskinesia in Latin America: Prevalence and associated clinical factors in the LARGE-PD cohort
Supplemental material, sj-docx-1-pkn-10.1177_1877718X261431204 for Levodopa-Induced dyskinesia in Latin America: Prevalence and associated clinical factors in the LARGE-PD cohort by Henry Mauricio Chaparro-Solano, Daniel Teixeira-dos-Santos, Emily Waldo, Thiago P Leal, Miguel Inca-Martinez, Sarael Alcauter, Alejandra Medina-Rivera, Alejandra E Ruiz-Contreras, Mario Cornejo-Olivas, Koni Mejia-Rojas, Cintia Armas, Pedro Chana-Cuevas, Natalia Rojas, Jorge L Orozco, Beatriz Munoz Ospina, David Aguillon, Omar Buritica, Sonia Moreno Masmela, Vitor Tumas, Artur FS Schuh, Carlos Roberto Rieder, Bruno Lopes Santos-Lobato, Juliana S Duarte, Susana L Pe🟍art쭥z, Mayela Rodr즵ez-Violante, Ana Jimena Hernez-Medrano, Emilia M Gatto, Gustavo Andres Da Prat De Magalhaes, Gonzalo Arboleda, Marcelo Andres Kauffman, Sergio Alejandro Rodriguez-Quiroga, Angel Vinuela, Alan Osvaldo Espinal Martinez, Pedro Braga-Neto, Sarah Camargos, Elias Fernandez-Toledo, Cesar Luis Avila, Francisco Eduardo Cardoso,Patricio Olguin, Vanderci Borges, María Valentina Müller, Marcelo Merello, Ana Rosso, Daniel Martinez-Ramirez, Dario Sergio Adamec, Marcela Susana Tela, Grace Helena Letro, Denise H Nicaretta, Ignacio F Mata, and on behalf of the Latin American Research consortium on the Genetics of Parkinson's Disease (LARGE-PD) in Journal of Parkinson's Disease
Footnotes
Acknowledgments
The authors thank Luis Aguilar, Alejandro León, and Jair García of the Laboratorio Nacional de Visualización Científica Avanzada at the Universidad Nacional Autónoma de México, as well as Carina Uribe Díaz, Christian Molina-Aguilar, and Alejandra Castillo Carbajal, for their technical support. We are also grateful to Denise Paredes from Clinic Sanatorio de la Trinidad. We acknowledge the DNA-Neurogenetics Bank of the Instituto Nacional de Ciencias Neurológicas (INCN) in Peru, the Biobank of Tissue and Fluids of the University of Chile, and Hospital Nacional San Rafael and Hospital Nacional Rosales in El Salvador, for their valuable support and collaboration in this study. Additional thanks to the Centro de Investigaciones Clínicas (CIC) from Fundación Valle del Lili, Colombia. Finally, we are deeply grateful to all participants in LARGE-PD, including people with Parkinson's disease and their families, whose involvement made this work possible.
Ethical considerations
This study was approved by the Institutional Review Board of the Cleveland Clinic Foundation (IRB # 25-081). In addition, each participating LARGE-PD site obtained approval from its local IRB in accordance with international ethical standards.
Consent to participate
All participants provided written informed consent before enrollment in the study.
Consent for publication
Not applicable
Author contributions
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the American Parkinson Disease Association (APDA, 1282087) [H.M.C-S., M.I-M., I.F.M.]. Additional sources of funding included the Michael J. Fox Foundation (MJFF-026283) [E.W., I.F.M.]; the Parkinson's Foundation (PDGENE-1333334) [M.I-M., I.F.M.]; the Alzheimer's Disease Sequencing Project (ADSP, 5U01AG076482-03) [E.W., M.I-M.]; the Veterans Affairs Puget Sound Healthcare System (5I01ABX005978-2) [T.P.L., I.F.M.]; the National Council for Scientific and Technological Development (CNPq, Brazil) [P.B-N., B.L.S-L., A.F.S.S.]; FAP UNIFESP [V.B.]; Universidad Nacional de Tucumán (PIUNT-D711) [C.L.A.]; the Programa de Apoyo a Proyectos de Investigación e Innovación Tecnológica-Universidad Nacional Autónoma de México (IN208622) [S.A.]; and the Secretaría de Ciencia, Humanidades, Tecnología e Innovación (Secihti, CF-2019/6390) [S.A.].
This project was also supported by the Global Parkinson's Genetics Program (GP2), which is funded by the Aligning Science Across Parkinson's (ASAP) initiative and implemented by The Michael J. Fox Foundation for Parkinson's Research (![]() ). A complete list of GP2 members is available at https://gp2.org
). A complete list of GP2 members is available at https://gp2.org
T.P.L., M.I-M., I.F.M. received funding from the National Institutes of Health (NIH, R01NS112499-01A1).
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: I.F.M. receives honoraria from the Parkinson's Foundation for his role as a member of the PDGENEration Steering Committee.
Data availability statement
The data that support the findings of this study are available from the corresponding author, I.F.M., upon reasonable request.
Supplemental material
Supplemental material for this article is available online.
