Abstract
Background
Levodopa is still the cornerstone of symptomatic treatment for people with Parkinson's disease (PwP). With disease progression and long-term use, PwP can develop motor complications such as wearing off and dyskinesia. There is limited practical guidance on the early identification of motor fluctuations and the optimal timing of adjunctive therapies in clinical practice.
Objective
To develop expert consensus on best practices for initiating and monitoring levodopa therapy, recognising early motor fluctuations, incorporating adjunctive therapies, and improving patient education in the management of Parkinson's disease (PD).
Methods
A modified Delphi methodology was used. A steering committee (SC) of nine clinicians with experience in managing PD drafted 59 consensus statements across four thematic domains. Statements were developed into an online survey and distributed to a panel of UK-based healthcare professionals (HCPs). Respondents rated their agreement using a four-point Likert scale. Consensus was defined as ≥75% agreement.
Results
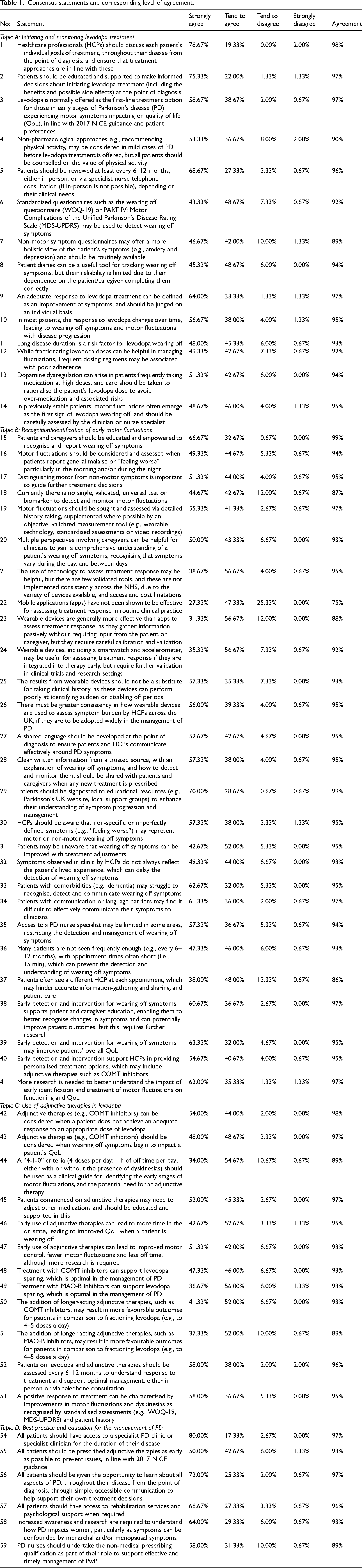
A total of 150 HCPs completed the survey, including neurologists, geriatricians, and PD nurse specialists. Consensus was achieved for all 59 statements (100%). Key areas of agreement included early alignment of treatment with individual patient goals, the need for routine monitoring using standardised tools, timely initiation of adjunctive therapies, and the importance of accessible, ongoing patient and caregiver education.
Conclusions
This Delphi consensus provides a set of expert-derived, clinically applicable recommendations to support proactive and individualised management of PD. Adoption of these practices may enhance the recognition and treatment of motor fluctuations, improve patient quality of life, and support better long-term outcomes.
Plain Language Summary Title
Experts agree on best practices for managing Parkinson's disease
Plain Language Summary
Why was the study done?
Levodopa is a key medicine for treating Parkinson's disease, helping with symptoms like stiffness and slow movement. Over time, though, some people notice changes in how well their medication works during the day. This can mean the effects wear off too soon or cause involuntary movements called dyskinesia.
Healthcare professionals do not have much practical guidance on how to spot these problems early, when to add new treatments, or how to best support people with Parkinson's and their caregivers.
This study brought experts together to create useful recommendations for better care.
What did the researchers do?
Nine UK experts who treat Parkinson's disease came up with 59 statements about different parts of care, like starting and monitoring levodopa, noticing early changes in movement, adding new medicines,and teaching patients and caregivers. They shared these statements with 150 healthcare professionals, including neurologists, geriatricians, and nurse specialists, who rated how much they agreed with each one. If 75% or more agreed, the statement was considered a consensus.
What did the researchers find?
All 59 statements reached full agreement from the group. The main recommendations were to start treatment based on each person's goals and needs, check symptoms regularly with standard tools, add new therapies at the right time to prevent problems, and make sure people with Parkinson's and their caregivers get ongoing, easy-to-understand education and support.
What do the findings mean?
The experts agreed on clear best practices to help healthcare professionals give more proactive and personalised care to people with Parkinson's disease. Using these recommendations may help spot movement problems sooner, improve symptom control, and boost patients' quality of life over time.
Introduction
The global number of people with Parkinson's disease (PwP) cases has been forecast to rise to 25.2 million by 2050, representing a 112% increase from 11.77 million in 2021.1,2 Population aging was projected to account for 89% of this growth. Despite the growing incidence of Parkinson's disease (PD), a disease-modifying therapy able to slow down its progression is still lacking. Current treatment strategies for PwP include symptomatic treatments aimed at increasing dopamine availability, thereby helping to control symptoms and improve quality of life (QoL).
The National Institute for Health and Care Excellence (NICE) recommends the use of levodopa as the first-line treatment for PD motor symptoms. 3 While motor symptoms are generally well controlled early in the disease, progression leads to the emergence of motor and non-motor fluctuations, including wearing off and early morning akinesia.4–7 These complications often appear earlier than recognised and can substantially impact daily functioning. 8
Strategies such as dose fractionation or increasing the total levodopa dose are commonly used but do not overcome the drug's short half-life and associated wearing off symptoms. 9 Adjunctive therapies such as monoamine oxidase type B (MAO-B) inhibitors, catechol-O-methyltransferase (COMT) inhibitors, and dopaminergic agonists (DAs) are recommended by NICE and the Movement Disorder Society (MDS) to optimise levodopa response.3,10 Evidence suggests that some adjunctive therapies, such as COMT inhibitors, reduce “off” time more effectively than levodopa dose modification alone.11,12
Despite these recommendations, current NICE and MDS guidelines provide limited practical direction on how and when to introduce adjunctive therapies, particularly in relation to early identification of wearing off, criteria for timely adjunct initiation, and structured monitoring of treatment response. This contributes to considerable variability in clinical practice.
This Delphi consensus sought to generate clear expert-driven recommendations on the use of adjunctive treatments, supported by pragmatic tools such as the 4-1-0 criteria to guide clinicians in recognising and managing levodopa wearing off complications. Additional aims included establishing consensus on other aspects of PD management, such as the initiation and monitoring of levodopa, early recognition of motor fluctuations, and best practices and education for the management of PD.
Methods
The process followed a modified Delphi methodology shown in Figure 1 and used ACCORD as a guideline for reporting the research. 13
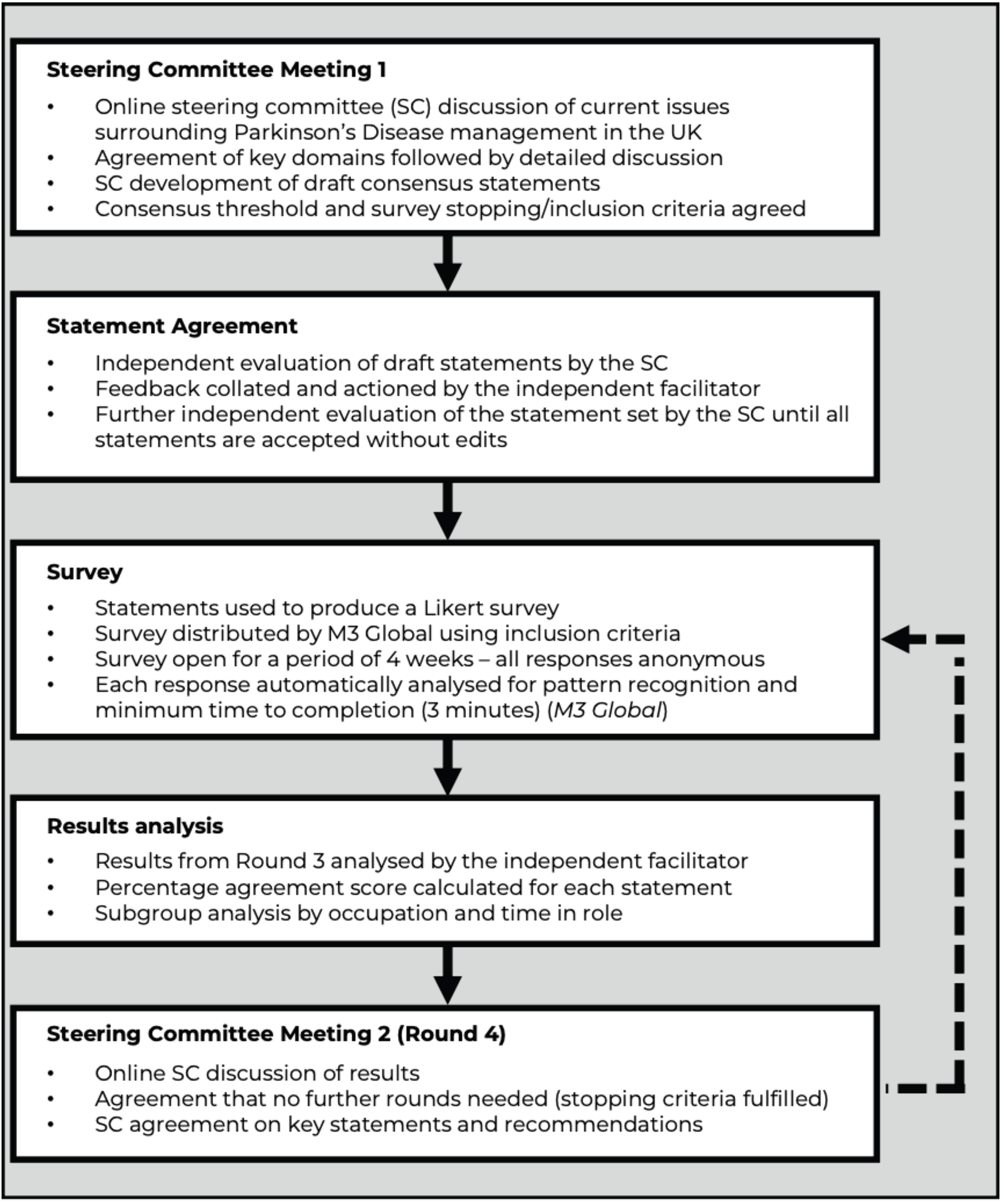
Modified Delphi study design.
In November 2024, a brief literature review on the topic of enhancing PD disease management was performed using the PubMed database, which identified 42 relevant papers published between 2015 and 2024. Search terms included but were not limited to: “[Parkinson's Disease]”, “[Earlier treatment]”, “[Levodopa]”, “[COMT]”, “[MAO-B]”, “[Guideline]”, “[Treatment goals]”.
Guided by an independent facilitator, a steering committee (SC) of UK PD specialists, including four neurologists, two geriatricians, and three PD nurse specialists, convened in January 2025. The SC were selected based on published research, clinical experience in PD, and to provide a variety of perspectives across roles and the UK.
The information gathered from the literature review was used to inform the meeting discussion. As part of the discussion, the SC agreed on four main domains of focus:
Initiating and monitoring levodopa treatment Recognition/identification of early motor fluctuations Use of adjunctive therapies in levodopa Best practices and education for management in PD
These domains were discussed in detail, and statements were suggested by the SC working collaboratively. Statements were independently reviewed by the SC and developed into a Likert survey. The survey was distributed by M3 Global Research, a market research company with a proprietary panel of verified healthcare professionals (HCPs). Eligible participants were neurologists, geriatricians, or PD nurse specialists based in the UK who were actively involved in the diagnosis and/or management of PwP. An equal distribution across the three professional roles was targeted to reflect a multidisciplinary perspective. Recruitment and dissemination were conducted entirely by M3 using their standard communication channels (e.g., email invitations and targeted outreach). In accordance with M3's policies, the number of HCPs invited and response-rate data were not disclosed to the study authors. Only completed responses were provided for analysis.
The identity of respondents was not known to the SC or the independent facilitator. The survey presented each statement along with a four-point Likert scale (‘strongly disagree’, ‘tend to disagree’, ‘tend to agree’, and ‘strongly agree’) to allow respondents to indicate their corresponding level of agreement. The survey also captured demographic data for further analysis, including occupation and time in role. All responses collected were included for the final analysis.
Stopping criteria were established a priori as a 4-week timeframe to collect responses, with a target of N = 150 responses, 90% of statements passing the threshold for consensus, with a threshold for consensus set at 75% (a widely accepted threshold). 14
A statement of consent was included at the start of the survey, and consent was implied by completion. As this study only collected the anonymous opinions of HCPs and no patient-specific data were captured, ethical approval was not sought.
Anonymity of responders was planned into the study design. All responder data were captured by M3 Global. No personal information beyond occupation, time in role, and country was captured or provided to the independent facilitator (Triducive Partners).
The third party provided an incentive payment of an appropriate honorarium to panellists on the completion of survey response.
Analysis of survey data was carried out in April 2025. Completed surveys were analysed using Microsoft Excel software to produce an arithmetic agreement score for each statement. The responses were aggregated to provide an overall agreement level (i.e., the number of responses in agreement as a percentage of the overall number of responses). The SC reconvened in May 2025 for analysis and discussion of results. After review of the results, the SC agreed that no further rounds of survey were required due to the high levels of consensus across all statements.
Data availability statement
All anonymised data is included in the Supplemental Materials.
Results
A total of 150 survey responses were received and included in the final analysis. The statements and corresponding agreement levels are shown in Table 1 and depicted in Figure 2. Respondent demographics by role and time in role are detailed in Tables 2 and 3, respectively. Breakdown of respondents’ agreement by role and region (country) is illustrated in Figures S1 and S2. Consensus was achieved for all 59 statements. As the stopping criteria were satisfied, no additional testing rounds were conducted. Distribution of consensus scores on the four-point Likert scale provided to respondents is represented in Figure S3.
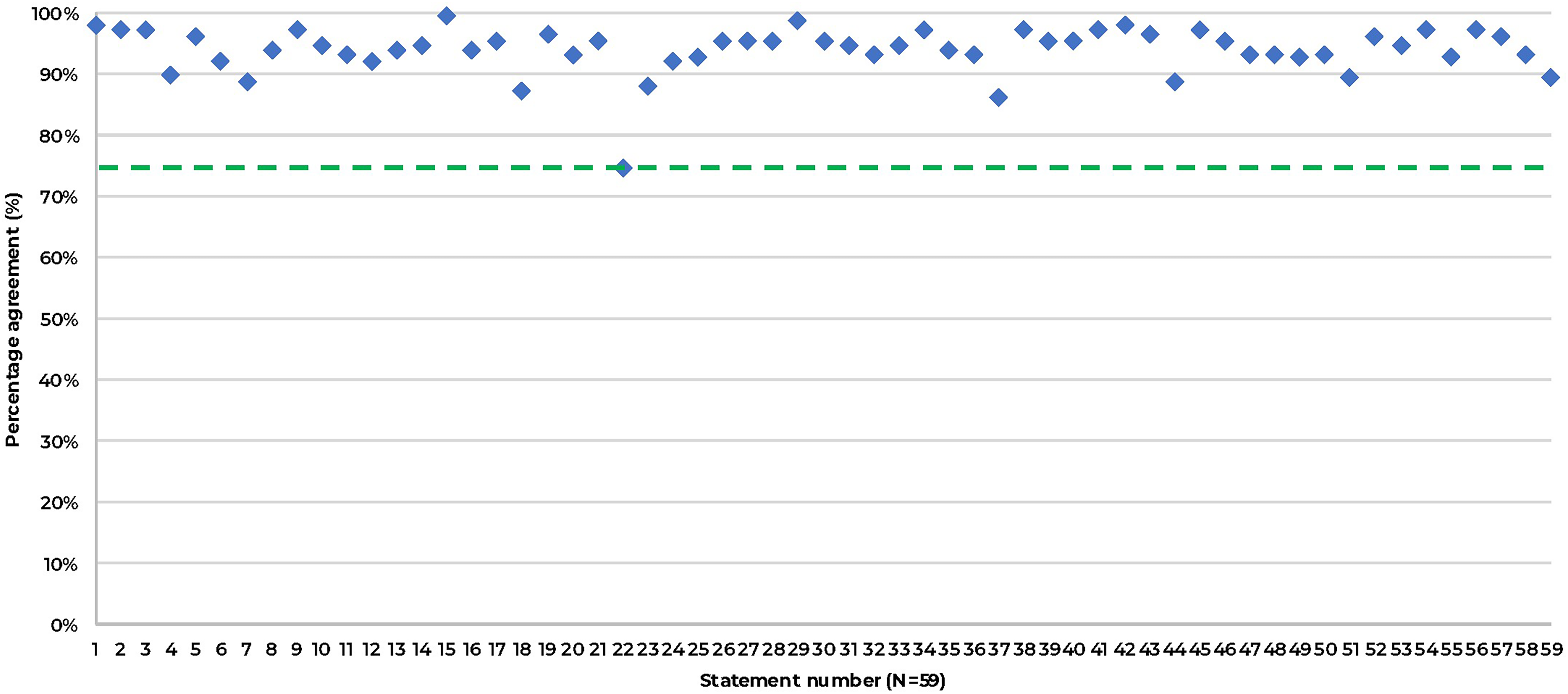
Consensus agreement levels by statement. The consensus threshold is depicted by the green line (75%).
Consensus statements and corresponding level of agreement.
Number of respondents by role.

Number of respondents by time in role.
Consensus recommendations and possible clinical implications.

The SC discussed the results and agreed on a set of recommendations for best practices in PD management.
Discussion
This Delphi consensus has provided a set of expert-derived, clinically applicable recommendations to support proactive and individualised management of PD (Table 4). Below, we discuss each recommendation and summarise the published literature that supports the consensus.
Treatment decisions in PD should be aligned with individual patient goals, and patients and caregivers should be educated about PD treatment and signs of wearing off from the point of diagnosis.
Evidence suggests that targeting therapy towards specific treatment goals leads to better physical function than standardised approaches. A randomised controlled trial (RCT) by Cabrera-Martos et al. demonstrated that PwP who received a goal-directed intervention achieved significantly greater improvements in both goal attainment and manual dexterity compared to those receiving standard therapy. 15 Several machine-learning-based tools have been tested to personalise medication strategies for patients.16–18 A recent study sought to advance personalised PD medicine through an algorithm that analyses sequential patient visit data and provides subsequent recommendations.19,20 It was concluded that consideration of personal medication history can significantly enhance decision-making for altering medication strategy. Furthermore, a secondary analysis of goal-setting interventions in PD found that treatment decisions should be aligned with patient-defined goals, which covered a diverse range of themes underscoring the multifaceted needs of PwP. 20
Poor concordance is closely linked to the complexity of dosing schedules and a lack of patient understanding. Empowering patients through targeted education and ensuring alignment of treatment goals with individual preferences has been shown to improve adherence and outcomes.
21
Education programmes, such as the Patient Education Programme Parkinson (PEPP), have demonstrated efficacy at improving health-related quality of life (HRQOL) for patients and carers through eight 90-min education sessions. A multicentre RCT by Chlond et al. involving 64 PwP and 46 caregivers evaluated its efficacy.
22
The intervention group participated in an 8-week programme and demonstrated significantly improved HRQOL at 3-month follow-up, measured using the Parkinson's disease quality of life questionnaire (PDQ-39). Similar effects were demonstrated by A’Campo et al., measured using the severity of dependence scale (SDS).
23
In scenarios where a formal programme may not be practical, these consensus results suggest that all patients should be signposted to external educational resources (e.g., Parkinson's UK website, local support groups) for additional guidance.
Levodopa should be offered as first-line therapy for PwP, alongside consideration of non-pharmacological approaches such as physical activity.
According to this Delphi consensus, levodopa remains the preferred first-line treatment option for patients with early-stage PD who experience motor symptoms impacting QoL, as recommended by NICE in 2017. 3 This consensus defined an adequate response to levodopa treatment as an improvement of motor symptoms, which should be judged on a case-by-case basis, given the heterogeneity of the disease. In most patients, the response to levodopa changes over time as disease progression occurs. This can lead to early wearing off in PwP whose disease state rapidly deteriorates. 24 Education on all aspects of levodopa treatment, including early wearing off, is therefore critical to empower patients to recognise and report early signs and facilitate assessment for levodopa adjustment or adjunctive treatments.
We strongly advocate that all PwP, regardless of disease stage, should be considered and educated on the value of physical activity. There is comprehensive RCT evidence supporting the long-term effectiveness of physical activity on outcome measures for PwP.25–27 As little as six months of aerobic exercise can attenuate off-state motor signs as measured by the Motor Complications of the Unified Parkinson’s Disease Rating Scale (MDS-UPDRS).
27
Furthermore, a 6-month intensive exercise programme was shown to increase dopamine transporter availability in the substantia nigra and putamen.
28
Finally, exercise has been shown to promote increased dopamine release and subsequently preserve motor and non-motor function.
29
Patients should be reviewed for motor and non-motor symptoms at least every 6–12 months (i.e., every appointment) to ensure the early detection of motor fluctuations and wearing off.
In addition to empowering patients to detect their own wearing off symptoms, regular clinical review enables HCPs to detect these changes early and adjust treatment accordingly. 30 This recommendation is consistent with current guidance from NICE and Parkinson's UK, which advocates for reviews every 6–12 months to monitor disease progression and treatment response.31,32
Evidence suggests that wearing off can emerge earlier in the disease course than previously assumed. One recent RCT defined ‘early’ wearing off as experiencing at least 1 h of daily “off” time for a minimum of 4 weeks, but for less than two years. 11 Similarly, the DEEP observational study reported that up to 41.8% of PwP with disease duration under 2.5 years experienced wearing off, assessed via the wearing off questionnaire (WOQ-19). 8
This consensus emphasised the critical role of early detection in optimising PD management. Early identification of wearing off allows HCPs to personalise treatment strategies, potentially improving symptom control and QoL. Respondents also agreed that early recognition supports patient and caregiver education by empowering them to better recognise changes in their symptoms. The authors note that further research is needed to clearly define the parameters of ‘early’ detection and confirm its impact on long-term outcomes.
33
Patients should be assessed using several evaluation tools, including validated questionnaires or standardised clinical questioning, detailed history-taking and an objective measurement tool where feasible.
Given the likelihood of developing motor complications, consistent review and monitoring of symptoms are critical. Standardised, validated questionnaires, such as the WOQ-19, the MDS-UPDRS, and non-motor symptom questionnaires, can be used during reviews to detect wearing off symptoms. However, the SC noted that these tools are applied inconsistently in clinical practice, with some HCPs, particularly PD nurses and physiotherapists, using tailored sets of questions developed and refined through clinical experience.
There is currently no single, validated objective measure or biomarker to detect and monitor motor fluctuations. Yet, findings indicate that incorporating one objective measurement into PD management enhances both motor and non-motor outcomes. 34 As such, this consensus explored alternative methods for the accurate tracking of wearing off symptoms. The SC agreed that detailed history-taking, which may include the use of patient diaries capturing multiple perspectives (e.g., from caregivers), should be supplemented by one or more objective measurement tools.
Wearable devices, including smartwatches and accelerometers, were deemed useful for contributing towards the clinical assessment of motor symptoms and treatment response if integrated into therapy early. Body-worn sensors could provide valuable data, with summarised scores for bradykinesia, dyskinesia, and tremor shown to be associated with suggested treatment modifications. 35 In this consensus, mobile applications were deemed less favourable due to the burden of regular symptom logging. This can be challenging for many patients, particularly those with cognitive impairment, and may limit the accuracy and completeness of data collection. Due to the multitude of devices and applications available, the SC stressed the need for consistency in their use and the analysis of data generated. Taken together, these findings highlight important unmet needs and priority areas for future research. The development and validation of reliable biomarkers, alongside standardised and user-friendly digital tools, will be critical to improving early detection and monitoring of motor fluctuations.
This consensus identified several additional barriers to the accurate assessment of symptoms in PD. Patients with comorbidities may struggle to recognise, detect and communicate wearing off signs and symptoms, while patients with communication or language barriers may find it difficult to effectively communicate their symptoms to clinicians. Reports of general malaise or ‘feeling worse’ should prompt clinicians to assess for motor fluctuation, as these may be indicative of wearing off.
It was also acknowledged that symptoms observed during clinic visits do not always reflect the patient's lived experience, potentially delaying recognition and management of wearing off symptoms. Patients may misinterpret wearing off symptoms as a sign that levodopa is no longer effective or that their disease is progressing, whilst being unaware that treatment adjustments can often alleviate such issues.
Infrastructural challenges further complicate symptom assessment. These include limited access to PD nurse specialists in some areas, irregular patient visits (e.g., >12 months), short appointment times (i.e., 15 min), and a lack of continuity of care when patients see different HCPs at each visit, which can hinder accurate information-gathering and sharing.
Adjunctive therapies should be initiated as early as clinically appropriate, with 4-1-0 criteria used as a clinical guide to support the management of levodopa wearing off complications.
Although adjustments in levodopa administration, such as fractionation, can be helpful to manage symptoms, frequent dosing regimens may be associated with poor medication concordance. 36 Additionally, dopamine dysregulation can arise in patients taking levodopa in high dosages, and so care must be taken to avoid over-medication and its associated risks. Levodopa sparing is an optimal strategy to mitigate these risks and can be supported with adjunctive therapies such as COMT or MAO-B inhibitors. This consensus confirmed views that the addition of adjunctive therapies may result in more favourable outcomes compared to fractioning levodopa.
NICE recommends that adjuvant therapy should be offered to patients who develop motor fluctuations or dyskinesia despite optimal levodopa therapy. 3 The results of this study reinforce this guidance by suggesting that adjunctive therapies should be considered as early as clinically appropriate, when a patient does not achieve an adequate response to an optimal dose of levodopa, and when wearing off symptoms begin to impact QoL. Clinician-perceived benefits of early adjunctive intervention were improved motor control and fewer fluctuations, leading to less time in the off state.
However, existing guidelines provide no specific criterion to determine when it is clinically appropriate to introduce adjunctive therapy. A previous Delphi consensus study defined the ‘5-2-1’ criteria for defining advanced PD, which have already been used to identify eligible subjects for device-aided therapies in longitudinal studies.37,38 Satisfying the criteria includes five levodopa doses per day, having at least 2 h of “off” time per day, or having at least 1 h of dyskinesia. 39 This criterion was used as a conceptual starting point by the SC to develop a novel ‘4-1-0’ rule of four levodopa doses per day, one hour of “off” time per day, with or without the presence of dyskinesias, as a clinical guide for identifying early stages of motor fluctuations and the potential need for adjunctive therapy. The 4-1-0 criteria were coined by the Steering Committee with the specific aim of providing a simple tool to be used by HCPs in clinical practice for the early identification of motor fluctuations in PD patients. While an 89% agreement was achieved among the wider expert panel, the criteria certainly needs a full validation in real-world clinical practice. However, they were formulated on a strong rationale and indirect evidence from published studies.
Five-times daily oral levodopa doses or more are already considered per se an indicator of advanced PD and eligibility for device-aided therapies. 37 It was, therefore, thought that the occurrence of “off” time despite four oral levodopa doses could be a valid criterion to identify people with early fluctuations who could potentially benefit from adjunctive therapies. Furthermore, a recent study has shown that in PD patients with early fluctuations on three to four intakes of levodopa, the adjunct of a long-acting COMT inhibitor was superior to an additional daily levodopa dose in reducing wearing off. 11
OFFELIA (OFF episode quality of life impact scale) has recently been proposed as a new measure of quality of life for “off” episodes in PD. 40 In the validation study, the duration of individual “off” periods was categorised and episodes lasting less than one hour were common (less than 15 min = 8.6%; between 15 and 30 min = 28.8%; between 30 and 45 min = 22%; between 45 min and 1 h = 27.2%), highlighting the need for proactive screening even for short-lasting fluctuations. In another large cohort of 722 PD patients, the vast majority (94%) of patients with “off” episodes experienced one to five hours of daily “off” time. 41 Interestingly, the presence of an “off” period significantly affected measures of quality of life regardless of the average hours of daily “off” time, implying that even one hour of “off” time could have a detectable impact on quality of life. Considering that all adjunctive drug classes effectively reduce “off” time by 0.8–1.5 h per day, an early introduction of these drugs could provide a full benefit and functional gain in these patients. 42
The proposed criterion of minimal or no dyskinesias is mechanistically rational, as the aim of the criteria is to identify patients with early motor fluctuations who could benefit from adjunctive therapy even before the occurrence of other motor complications. 42 Therefore, patients who fulfil the previous two criteria but have no or minimal dyskinesia are more likely to experience the greatest benefit from adjunctive therapy without the onset or worsening of dyskinesia.
Consensus also supported that longer-acting adjunctive therapies, such as COMT inhibitors and MAO-B inhibitors, may result in more favourable outcomes for patients compared to fractioning levodopa (e.g., to 4–5 doses per day). Data from various RCTs have demonstrated the superior efficacy of adjunctive COMT therapy versus levodopa fractionation.11,12 Therefore, longer-acting adjunctive therapies should be considered as early as clinically appropriate, rather than adjusting the dose of levodopa.
Despite these benefits, access to adjunctive therapies for PD in the UK and Europe remains inconsistent. This variability is influenced by multiple factors, including regional differences, the availability of pharmacological agents within each therapeutic class and prescribing practices of HCPs. 43 For example, the SC highlighted that although the current NICE guidance recommends a range of adjunctive options, local formularies and commissioning arrangements often restrict the availability of specific agents. In addition, there is inconsistent access to different COMT inhibitors across commissioners, resulting in regional variation in prescribing practices. Due to limited UK and international guidance on when and how to introduce adjunctive therapies into the treatment pathway, prescribing practices are often subjective and depend on clinicians’ attitudes and knowledge.
Addressing these disparities is necessary to ensure equitable care for individuals with PD. To achieve this, targeted training and case studies on the latest evidence surrounding adjunctive therapies should be provided to HCPs to support consistent adoption of evidence-based treatment strategies. Moreover, standardising guidance on when and how to introduce adjunctive therapies would promote consistent prescribing practices and reduce regional variation in care.
All patients with PD should have equitable access to appropriate specialist care, pharmacological treatments and multidisciplinary rehabilitation services. Further research is also required to understand sex-specific differences and the impact of PD on women.
According to Parkinson's UK, there is a shortage of approximately 100 PD specialist nurses across the UK, corroborating this consensus where respondents agreed that access to a PD nurse specialist may be limited in some areas, potentially restricting the detection and management of wearing off symptoms. 44 To ensure the provision of equitable care, this should be addressed.
Efforts should also be made to ensure equitable access to treatments and medications across all regions, so that all patients can benefit from holistic and individualised patient-centred care. A recent analysis of UK prescribing data from 2019–2024 showed that prescribing patterns for PD vary substantially. 45 While levodopa was found to be the most commonly prescribed drug in England, Scotland, and Wales, DAs were more frequently used in Northern Ireland. This disparity is thought to reflect differences in demographics, healthcare infrastructure, and economic factors. Therefore, addressing these disparities is an important step toward equitable access to holistic treatments for PD and improved patient-centred care.
In addition, all PwP should have access to appropriate multidisciplinary rehabilitation services, including disease-specific physiotherapy, occupational therapy, speech and language therapy and psychological support as necessary. Consistent with this consensus, an international statement concluded that a multidisciplinary rehabilitative approach is an essential component of comprehensive treatment of PD and should encompass discipline-specific support services across all PD stages. 46
Lastly, this consensus also highlighted that increased awareness and research are required to understand how PD impacts women, as symptoms can be confounded by menarcheal and/or menopausal symptoms. This represents an important yet often overlooked aspect of PD management that should be recognised and addressed.
Strengths and limitations
This study achieved consensus from a large sample of 150 specialist clinicians across multiple UK regions, thus making the findings strong and generalisable. A structured, modified Delphi methodology was used, ensuring rigour and reproducibility of the methods. Consensus was achieved for all statements, providing a strong evidence base for the given recommendations. This study only included one round of survey; however, this was deemed sufficient since the stopping criteria were met. Self-reported data can also be subject to recall bias and subjectivity. Additionally, the survey was only completed by HCPs, and the patient perspective may be particularly helpful in strengthening the utility of these findings. Finally, as with all Delphi processes, participants may have adjusted their responses to align with perceived group consensus, which could lead to bias.
Conclusions
This modified Delphi consensus achieved agreement from a panel of 150 HCPs involved in PD management for all 59 statements, offering a unified expert perspective in an area where practical guidance has been limited. The findings have clear implications for clinical practice, encouraging earlier recognition of motor fluctuations and more consistent assessment of symptoms, with an emphasis on the fact that wearing off often occurs sooner than traditionally assumed. Regularly scheduled reviews supported by validated questionnaires, detailed clinical questioning, and objective tools can help clinicians detect changes earlier and intervene more effectively. The consensus also supports earlier initiation of adjunctive therapies, guided by the 4-1-0 criteria, which offer a simple and actionable framework to reduce subjectivity and regional variability in prescribing decisions. By promoting timely intervention and more structured follow-up, these consensus recommendations help address existing gaps and support a more proactive, standardised approach that may improve long-term outcomes and quality of life for PwP.
Supplemental Material
sj-jpeg-1-pkn-10.1177_1877718X261427577 - Supplemental material for A modified Delphi consensus study to support and enhance Parkinson's disease management
Supplemental material, sj-jpeg-1-pkn-10.1177_1877718X261427577 for A modified Delphi consensus study to support and enhance Parkinson's disease management by N Pavese, J Evans, A Hand, EJ Newman, G Duncan, H Martin, L Ebenezer, B Mohamed, E Lassman, J Barnett and F Morgante in Journal of Parkinson's Disease
Supplemental Material
sj-jpeg-2-pkn-10.1177_1877718X261427577 - Supplemental material for A modified Delphi consensus study to support and enhance Parkinson's disease management
Supplemental material, sj-jpeg-2-pkn-10.1177_1877718X261427577 for A modified Delphi consensus study to support and enhance Parkinson's disease management by N Pavese, J Evans, A Hand, EJ Newman, G Duncan, H Martin, L Ebenezer, B Mohamed, E Lassman, J Barnett and F Morgante in Journal of Parkinson's Disease
Supplemental Material
sj-jpeg-3-pkn-10.1177_1877718X261427577 - Supplemental material for A modified Delphi consensus study to support and enhance Parkinson's disease management
Supplemental material, sj-jpeg-3-pkn-10.1177_1877718X261427577 for A modified Delphi consensus study to support and enhance Parkinson's disease management by N Pavese, J Evans, A Hand, EJ Newman, G Duncan, H Martin, L Ebenezer, B Mohamed, E Lassman, J Barnett and F Morgante in Journal of Parkinson's Disease
Footnotes
Acknowledgements
The authors wish to thank Emily Lassman and James Barnett from Triducive Partners Limited for their support in collating the data, analysing the results, writing the manuscript, and reviewing the final draft.
Authors’ contributions
All views expressed by the authors are those of the individual and do not represent those of their organisation/place of work. Contributors NP, JE, AH, EJN, GD, HM, LE, BM, and FM agreed on the design of the study, formulated and reviewed the statement set. Results of the study were discussed by all contributors as part of a formal Steering Group meeting, where commentary was agreed for development into the initial draft manuscript. NP, JE, AH, EJN, GD, HM, LE, BM, and FM took an equal role in reviewing the initial manuscript draft and providing comments, and all approved the final draft.
Ethics approval and consent to participate
Not applicable.
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Funding
The study was initiated and funded by BIAL. All authors received funding from BIAL while undertaking this study. BIAL commissioned Triducive Partners Limited to facilitate the project and analyse the responses to the consensus statements in line with the Delphi methodology. Triducive Partners Limited provided an incentive payment of an appropriate honorarium to panellists on the completion of survey response. After engaging Triducive Partners Limited, BIAL made no contribution to the design and development of the study outside of payment of honoraria. BIAL took no part in the writing, revision or editing of the manuscript except to check that the manuscript contained no promotion of specific medicines and that all recommendations were appropriate to drug label.
Declaration of conflicting interests
All authors received honoraria from BIAL while undertaking this study. BIAL commissioned Triducive Partners Limited to facilitate the project and analyse the responses to the consensus statements in line with the Delphi methodology.
The authors state the following conflicts of interest:
GD has received speaker fees from GE Healthcare, BIAL and AbbVie, and honoraria for advisory boards and consulting from BIAL and AbbVie.
HM has received honoraria for consultancy and educational meetings from BIAL, AbbVie, and Merz Pharma.
NP has received honoraria from BIAL, Britannia Pharmaceuticals, AbbVie, GE Healthcare, Boston Scientific, been a part of advisory boards for Hoffmann-La Roche, BIAL, AbbVie, Teva, Biohaven, and Teitur Trophics, and received grants from Independent Research Fund Denmark, Danish Parkinson's disease Association, Parkinson's UK, Center of Excellence in Neurodegeneration (CoEN) network award, GE Healthcare Grant, Multiple System Atrophy Trust, Weston Brain Institute, EU Joint Program Neurodegenerative Disease Research (JPND), EU Horizon 2020 research, The Michael J. Fox Foundation, F. Hoffmann-La Roche, Medtronic, and Symbyx.
FM has received consultancy fees from Boston Scientific and Medtronic, been a part of advisory boards for AbbVie, Boston Scientific, Merz Pharma, Medtronic and Roche, received speaking honoraria from AbbVie, Boston Scientific, Merz Pharma, Medtronic, Teva, Ipsen, International Parkinson's Disease and Movement Disorders Society, royalties from Springer, and research support from the National Institute for Health and Care Research, Innovate UK and Merz Pharma.
JE has received speaker and consultancy fees from BIAL, Zambon, AbbVie, Ipsen, Britannia Pharmaceuticals, and Kyowa Kirin, and conference subsistence support from BIAL, AbbVie, Ipsen, Britannia Pharmaceuticals, and UCB.
EJN has received travel bursaries or speaker fees from the following companies: AbbVie, BIAL, Britannia Pharmaceuticals, Ipsen, Medtronic, and Merz Pharma.
Availability of data and materials
Declarations
The study was not prospectively registered, as neither the assigned interventions nor the outcomes assessed were related to the health of participants.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
