Abstract
Background
The decision to undergo deep brain stimulation (DBS) surgery for patients with Parkinson's disease (PD) is often challenging and complex. Decisional outcomes may be improved by using decisional support tools that foster shared decision making.
Objective
The objective of this study was to develop a decision aid for PD patients considering DBS surgery and evaluate its acceptability.
Methods
The decision aid was developed using an evidence-based and systematic approach. The steps in development included a needs assessment, literature review, development of a decision aid prototype, and testing of the prototype with surveys for acceptability in a clinic setting.
Results
A total of 136 participants with PD participated in this study. The needs assessment included 57 participants who completed the decisional conflict scale with a mean score of 35.3, indicating high decisional conflict. After development, initial testing of the decision aid was completed by 22 participants (16 on paper version and 6 on interactive website version). Subsequently, 46 PD participants evaluated the decision aid for acceptability. Eighty seven percent of participants agreed or strongly agreed that they were satisfied with the quality of the decision aid. Most participants found the language, amount of information, length, balance, and risk and benefits section acceptable.
Conclusion
We determined that PD patients undergoing DBS evaluation experience high decisional conflict. We subsequently created a DBS decision aid to increase knowledge, manage expectations, clarify values, and facilitate shared decision making. The decision aid was acceptable to PD patients with and without DBS.
Plain language summary
Making the decision to have deep brain stimulation surgery for Parkinson's disease can be difficult. This study aimed to create a tool to help people with Parkinson's disease make this decision by providing up to date information and support discussions with their doctors. The researchers developed the tool by first understanding what patients needed, reviewing relevant research, creating a draft of the tool, and then testing it with patients in a clinic. A total of 136 people with Parkinson's disease took part in the study. Testing showed that many patients felt uncertain about their decision when considering deep brain stimulation surgery. After viewing the decision aid, most patients were satisfied with it. They found it helpful, clear, and well-balanced in terms of information, including the risks and benefits of surgery. The tool can make it easier for patients to understand their options and make informed decisions with their doctors.
Introduction
Deep Brain Stimulation (DBS) surgery is an advanced therapy option for patients with Parkinson's disease (PD) who experience motor fluctuations, bothersome dyskinesia, or medication resistant tremor. 1 While DBS can provide significant improvements in motor symptoms and quality of life, the decision to pursue this treatment is complex. 2 It requires careful consideration of potential benefits and risks, alongside an understanding of the patient's preferences, expectations, and values.3–5 Patients must weigh the risks and potential morbidities associated with an invasive neurosurgical procedure against the potential of meaningful improvement in PD symptoms. As more advanced therapy options for PD become available, shared decision making in the context of DBS has become increasingly important.
Shared decision making is a collaborative process in which clinicians and patients work together to make informed decisions about a patient's care. The approach involves weighing the best evidence for the risks and benefits of the available options while incorporating patients’ preferences and values.6,7 Prior studies have shown that while most PD patients desire active involvement in the decision-making process, they are often not fully informed about the available options.8,9 Shared decision making can be facilitated through the use of decision support tools, such as decision aids. These tools can help patients understand treatment options, consider their personal values in relation to potential benefits and risks, and foster active participation in the decision-making process. 10 A Cochrane review found that decision aids routinely improve knowledge and realistic expectations, reduce decisional conflict, and improve concordance between patient values and choice. 11 The International Patient Decision Aids Standards (IPDAS) collaborative developed a set of criteria to guide development of quality decision aids to better support patients in making informed, value-based decisions. 12
Currently, no decision aid exists for DBS surgery for PD. Therefore, the objective of this study was to develop a decision aid for patients with PD considering DBS surgery, aligned with IPDAS criteria, and to assess its acceptability.
Methods
Approvals and research protections
This study was approved by the Colorado Multiple Institutional Review Board (COMIRB) at the University of Colorado Anschutz Medical Campus. Participants were recruited through the University of Colorado Movement Disorders clinics, referral logs maintained by the DBS program manager, and the Center's research participant database.
Development team
The core development team included a movement disorder neurologist, a geriatrician and shared decision-making expert, a qualitative analyst, and a qualitative methodologist. A Steering Group, which included three Movement Disorder neurologists, two functional Neurosurgeons, a neuropsychologist, and a DBS Nurse Navigator, also provided input at several time points in development.
Literature review
We reviewed the literature to determine the best evidence for outcomes related to DBS surgery for PD. We searched the Embase, MEDLINE and CENTRAL databases for published evidence on symptoms treated, benefits, risks, quality of life and other advanced therapy options.13–23 The search strategy included the terms “deep brain stimulation” OR “DBS” combined with “Parkinson's disease.” In addition, the reference lists of all studies that described outcomes of DBS and relevant review articles were manually screened to identify additional studies. We used systematic reviews and meta-analyses whenever available, and the evidence was reviewed by the Steering Group. Symptomatic improvement percentages presented in the decision aid were derived from ranges reported in these studies,21–23 reflecting average motor improvement under optimized stimulation and medication conditions. Values were presented as approximate ranges rather than single point estimates to convey expected variability in response. Where available, percentages reflected mean improvement in validated motor scales rather than categorical responder thresholds.
Needs assessment
We completed the needs assessment by conducting semi-structured interviews with 33 PD patients who had undergone DBS evaluation within the past 3 years and decided to or not to undergo DBS surgery. We chose this timeframe to balance sufficient recruitment of both groups while trying to minimize recall bias. The results of this study are described elsewhere 2 and were used to inform the contents of this decision aid. In a separate cohort of patients who were referred for evaluation by a movement disorders neurologist and just beginning the DBS evaluation process, we measured decisional conflict using the Decisional Conflict Scale (DCS), one of the most frequently reported outcomes in decision aid interventions. 24 The DCS is a 16 item questionnaire that measures personal perceptions of uncertainty in choosing options, modifiable factors contributing to uncertainty, and effective decision making, on a 5-point Likert scale. A score greater than 37.5 is associated with decision delay and feeling unsure, while scores below 25 are associated with implementing decisions. We also assessed decisional readiness using a using a validated, single-item instrument in which participants were asked, “if you were to make a decision about undergoing DBS today, how ready would you be to make that decision?”. Potential answers are on a 5-point Likert scale and range from “not at all ready” to “very ready”. 24
Study design
We used the Ottawa Decision Support Framework 25 and the International Patient Decision Aid Standards (IPDAS) to guide the systematic development of the DBS decision aid. 12 The steps in development are shown in Figure 1, and include 1) a needs assessment, 2) literature review, 3) development of a decision aid prototype, 4) alpha testing of the prototype with PD participants, 5) review by the Steering Group composed of content and methodological experts, and 6) testing the finalized decision aid for acceptability.
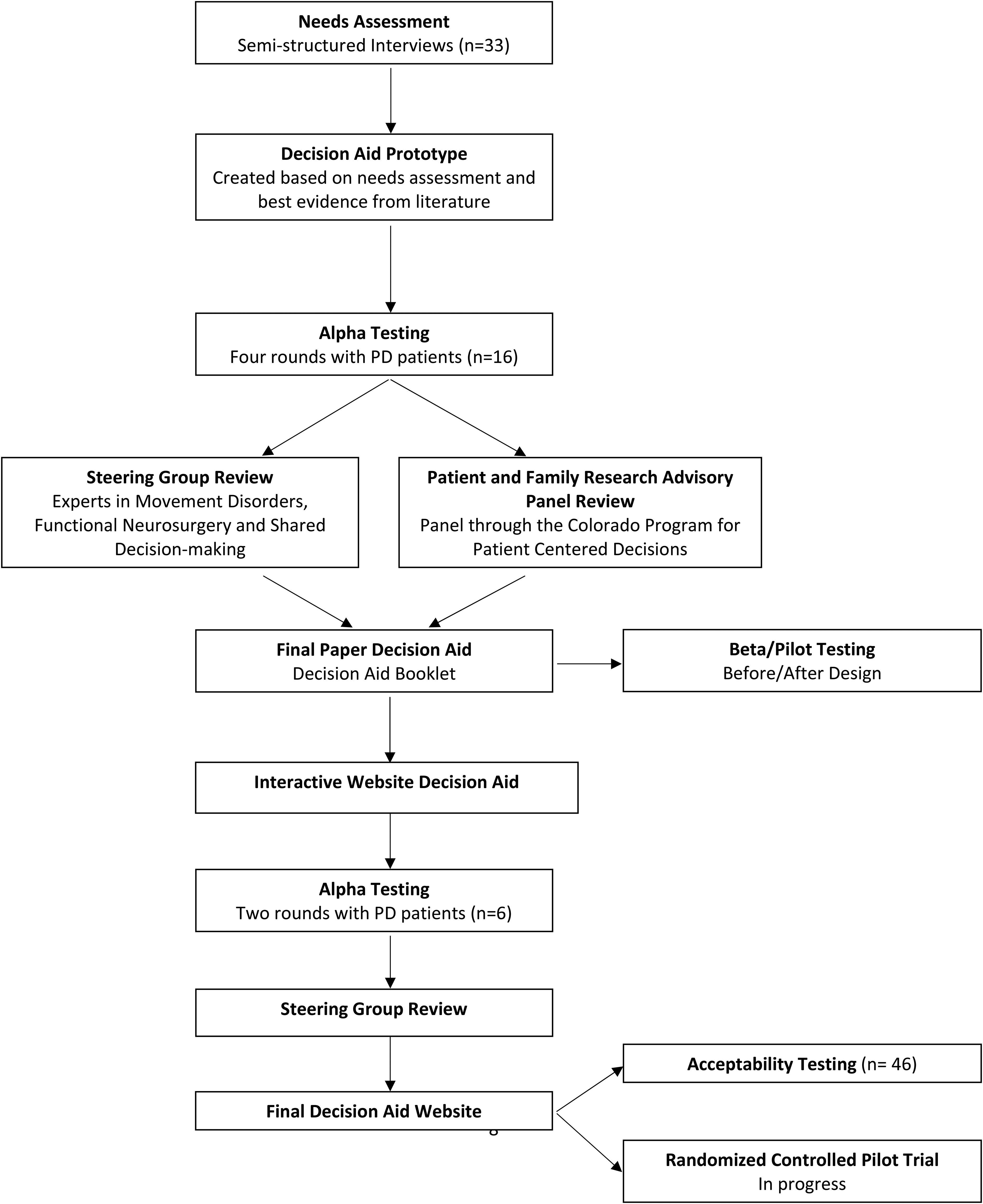
Development and testing of a DBS decision aid (adapted from coulter et al. 12 ). This flow chart outlines the step-by-step process followed in the development of the DBS decision aid, adhering to the International Patient Decision Aids Standards (IPDAS) and the Ottawa Decision Support Framework. A needs assessment was performed, followed by the creation of content based on the needs assessment and best evidence from the literature. The decision aid then underwent alpha-testing in which iterative feedback was obtained from patients and healthcare professionals. After addressing feedback, the decision aid was refined and finalized in a paper version. This paper version underwent pilot testing in a before/after design. An interactive website version was then created, which underwent additional alpha testing and steering group review. Acceptability of the decision aid was evaluated, and a randomized controlled pilot trial assessing decisional outcomes is currently underway.
Prototype development and alpha testing
The initial content of the decision aid combined our interview findings, the best evidence from the literature on risks, benefits and outcomes of surgery, and the guidance from IPDAS. Different versions of data displays were created for the risks and benefits, and a values clarification worksheet was included. The initial decision aid underwent multiple iterations with the development team, followed by alpha testing with 16 patients with PD who had undergone or were currently undergoing evaluation for DBS. Participants were asked to review the paper decision aid and then engage in a 30–60 min semi-structured interview based on the Ottawa Decision Support Framework to provide feedback on content, layout, graphics, understandability, balance, length, and format of the decision aid, as well as preferred timing for receiving the decision aid in the decision-making process. Four participants were interviewed per round of alpha testing, after which the development team met to discuss the feedback and to come to a consensus on how to incorporate it into the decision aid. All actionable feedback was documented in a spreadsheet to ensure systematic tracking. Alpha testing was concluded once a round was completed in which the study team agreed that no new actionable feedback was identified.
The decision aid was then reviewed by the Steering Group, as well as the Colorado Program for Patient Centered Decisions Patient and Family Research Advisory Panel. The development team met a final time to incorporate feedback and finalized the decision aid booklet. The decision aid booklet underwent pilot testing, which will be reported separately. Based on additional patient feedback, the decision aid booklet was adapted to a website version that included an interactive section. Patients check off the symptoms they experience and then receive a personalized symptom report with information on if, and by how much, their symptoms are expected to improve. We followed a similar process of alpha testing for the decision aid website, which included alpha testing with patients who had either undergone or were currently undergoing DBS evaluation, followed by review by the Steering Group, and then incorporation of feedback to finalize the decision aid website.
Acceptability evaluation
After the decision aid website was finalized, participants with a diagnosis of PD, who had and had not undergone DBS, were recruited to review the decision aid and complete an adapted version of the Ottawa Acceptability Questionnaire. 26 These participants were recruited from a movement disorders clinic and included individuals at any stage of Parkinson's disease. Enrollment did not require that participants be actively considering DBS at the time of the study. The Ottawa Acceptability Questionnaire is a common tool used to evaluate decision aids and consists of 10 items that assess different aspects of the decision aid, including amount of information, length, and balance. The final two questions are open-ended, allowing participants to provide narrative feedback on the decision aid.
Implementation
The decision aid is designed to be introduced at the start of the DBS evaluation process. In centers with a dedicated DBS nurse navigator, it may be provided by that individual, or alternatively, sent to patients electronically before their initial evaluation visit. Early access allows patients to review information in advance, enabling clinic visits to focus on addressing specific questions and assessing the suitability of DBS, rather than covering introductory material. For centers without nurse navigators, delivery can be managed by clinic staff involved in pre-visit planning or through automated EHR-based distribution prior to a DBS consultation. Core requirements include a defined trigger for deployment (e.g., referral for DBS evaluation), a delivery mechanism (e.g., patient portal, in-clinic tablet or printed copy), and a plan for incorporating decision aid outputs into the clinical discussion if desired. Brief standardized training materials or scripts can support consistent, unbiased use and facilitate scalable implementation across diverse DBS centers. The ongoing randomized controlled trial is evaluating preliminary implementation outcomes and examining both patient and clinician preferences regarding optimal timing and mode of delivery, as well as training needs for the decision aid administrator.
Results
Needs assessment and prototype development
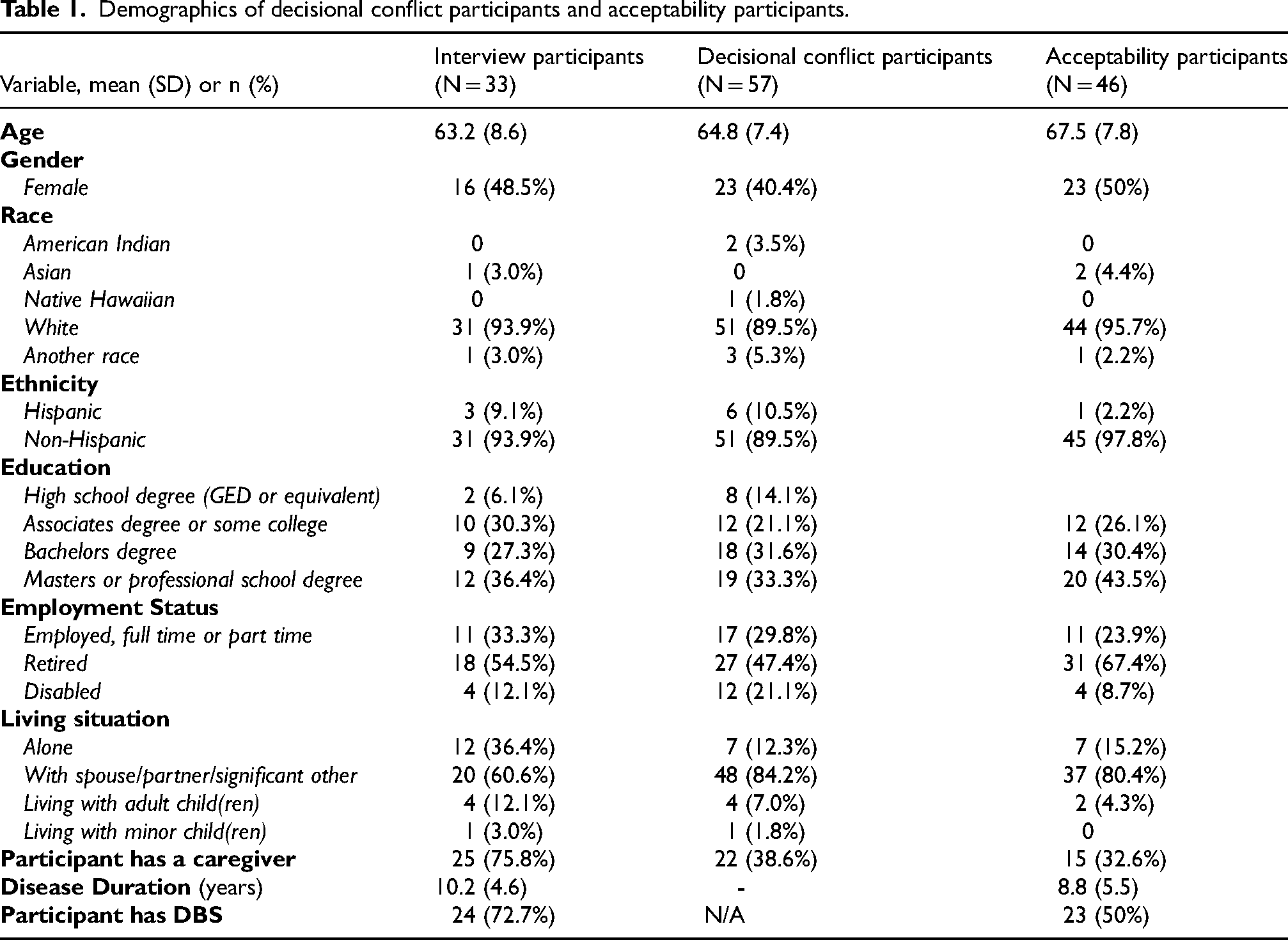
Fifty-seven participants with PD (23 women and 34 men) undergoing the DBS evaluation process completed the decisional conflict scale and the decisional readiness questionnaire. The mean age of participants was 64.8 years and mostly consisted of white and non-Hispanic individuals, with almost half being retired (Table 1). On average, the total decisional conflict score was high at 35.3 (Table 2). The highest sub scores were the uncertainty (45.8) and informed (39.9) sub scores. The only score below the cutoff associated with implementing decisions was the support sub score at 24.3. Regarding decisional readiness, 33% of the participants were “moderately ready” to make a decision about DBS, while half of the participants were “ready” or “very ready” to make a decision. There were no gender differences in the total decisional conflict score or subscores.
Demographics of decisional conflict participants and acceptability participants.
Decisional conflict and decisional readiness for participants undergoing DBS evaluation.

*Scores >37.5 are associated with decision delay and feeling unsure; scores <25 are associated with implementing decisions.
A total of 33 PD patients (16 women and 17 men) participated in semi-structured interviews. The results are published elsewhere. 2 Based on findings from the interviews, the final sections of the decision aid included an introduction to DBS and descriptions of: (1) symptoms that are treated, (2) who gets DBS, (3) risks and benefits, (4) the “ideal window” for DBS surgery, (5) other treatment options, (6) the evaluation process, (7) surgery and programming timeline, (8) support needed, and (9) how to choose a device. Additionally, the decision aid included a values worksheet and questions to ask the doctor and others with DBS. Gender differences were found in informational needs. Specifically, multiple women reported wanting more information about when and what kind of help they would need, as they more often lived alone and were less likely to have a caregiver. As such, we included a section on the support that one would need when going through the evaluation process and surgery. Finally, different pictorial displays were used to demonstrate benefits and a Cates plot was used to demonstrate the risks of surgery.
Alpha testing
Alpha testing was completed by 16 patients with PD. Four participants were included in each round, after which feedback was incorporated into the decision aid. Alpha testing for the decision aid website included 6 additional participants with PD who had either undergone or were undergoing DBS evaluation. Three patients participated in each round, and by the second round, no new feedback was obtained.
Acceptability evaluation
A total of 46 PD participants evaluated the decision aid for acceptability. As shown in Table 1, the mean age of participants was 67.5 years and 50% were women. The majority were white and non-Hispanic and had high levels of education. The mean disease duration was 8.8 years, and half of the participants had previously undergone DBS surgery. Of those who underwent DBS surgery, the mean time between surgery and completing the acceptability survey was 3.3 years, with a range of 6 months to 10 years. Most participants found the DBS decision aid to be acceptable (Table 3). Ninety-one percent either agreed or strongly agreed that the language was easy to follow, and 85% found that the amount of information was “about right”. Almost 9% thought it was a little less information than they wanted, and less than 5% thought it was more information than they wanted. The length was “just right” for 89% of participants and 70% thought it was well balanced. The other 30% thought it slanted towards DBS. Eighty-seven percent agreed or strongly agreed that the benefits and risks section was easy to follow. Seventy-eight percent thought that the decision aid would be helpful when deciding whether or not to get DBS. Most of those who chose “neutral” were participants who had not undergone
Acceptability items related to the DBS decision aid.

DBS and had a shorter disease duration. Eighty-seven percent of participants “agreed” or “strongly agreed” that they were satisfied with the quality of the decision aid and 11% were “neutral”. Ninety-four percent would recommend the decision aid to others. There were no gender differences in any of the acceptability ratings.
Forty-two participants provided narrative feedback on the decision aid. These comments highlighted strengths of the decision aid in four main categories: 1) information, 2) balance, 3) understandable, and 4) ease of use (Table 4). Many participants commented that the decision aid was comprehensive, easy to follow and easy to understand. They liked that all the information they needed was in one place. One participant noted, “it was thorough without being overwhelming with information.” Multiple participants appreciated the interactive section, including one who said, “I liked the ability to select symptoms and receive instant feedback on the impact DBS has on those symptoms. I liked the printable report.” Multiple participants commented that the decision aid was balanced and, “provided opportunities to explore alternatives to DBS”. Lastly, one participant noted that the decision aid “helps to set expectations of what will improve and what will be less likely to improve.”
Selected quotations from participant comments about the decision aid.

IPDAS checklist
The final decision aid was evaluated based on IPDAS criteria (Supplemental Table 1). All 6 qualifying criteria were met and all 6 of the applicable certification criteria were met. The DBS decision aid met almost all the specified quality criteria with several items related to testing the decision aid noted to be in progress.
Discussion
In this study, we found that decisional conflict was high among PD patients beginning the evaluation process for DBS, indicating that this population would likely benefit from a decision support intervention. High decisional conflict, which can stem from inadequate knowledge, unrealistic expectations, unclear values and insufficient support, can lead to low quality decisions, hesitation in decision making, and lower satisfaction with decisions. 25 To address this decisional conflict, we developed an evidence-based, patient-centered decision aid for people with PD considering DBS surgery and that followed the Ottawa Decision Support Framework and the IPDAS. The decision aid met almost all IPDAS criteria for a quality decision aid, with the criteria not currently met noted to be in progress (Supplemental Table 1). In addition, our prior study identified gender differences in informational needs, 2 so we incorporated women's specific informational needs. Acceptability testing demonstrated high acceptability of the decision aid among PD patients with and without DBS, and narrative feedback indicated that the decision aid was informative, balanced, understandable, and easy to use. An ongoing randomized controlled trial will determine the effectiveness of the decision aid in improving decisional outcomes.
One of the goals of developing this decision aid was to manage expectations for DBS. Prior studies have shown that expectations for DBS are associated with outcome satisfaction.27–29 Despite improvements in motor outcomes and quality of life, a proportion of patients consider DBS unsuccessful when it does not meet their expectations.27–29 In a qualitative study of 30 DBS patients, all experienced improved motor outcomes. 27 However, 27% reported an overall negative outcome, and these participants were more likely to have expected improvements in their mental state, socializing and partnership, which are not directly improved by DBS. Those who reported positive outcomes were more likely to have expected improvement in tremor and activities, which are more likely to improve with DBS. 27 The interactive section of our decision aid provides patients with immediate feedback on which of their symptoms are expected to improve and by approximately how much, similar to the DBS Edmonton application. 3 This exercise helps patients determine if DBS is anticipated to improve the symptoms that bother them the most, and it is anticipated that this will improve patient satisfaction with their decision. The report is printable, allowing patients to share it with family or the DBS care team if they choose.
In addition to managing expectations, a primary objective was to improve patients’ understanding of risk and to support clarification of personal values. The use of visual risk displays, including icon arrays, was informed by a substantial body of literature demonstrating that such formats enhance risk communication.30–32 Compared with numerical percentages alone, icon arrays and Cates plots have been shown to improve both accuracy of risk perception and overall comprehension.30,31 In parallel, values clarification exercises were incorporated based on evidence that they improve preparation for decision making and reduce subsequent decisional regret.33,34 Prior studies have demonstrated that explicit values clarification increase congruence between patient's stated values and their treatment decisions; for example, one randomized study of hormone replacement therapy found significantly greater value-choice congruence among participants who received a decision aid. 35 Additionally, a systematic review found that explicit values clarification methods reduce decisional conflict and decrease the likelihood of values-incongruent choices. 34 Together, these design features directly address core determinants of decision quality.
The decision aid was overall highly acceptable to PD patients with and without DBS. A lower proportion (78%) agreed or strongly agreed that the decision aid would be helpful when deciding whether to get DBS. However, 17% of participants were “neutral” on this topic, and this group had not undergone DBS and had a shorter disease duration. This suggests that the decision aid may not be as helpful if it is introduced early in the disease when participants are not considering DBS and are not experiencing symptoms, such as motor fluctuations or uncontrolled tremor, that often prompt patients to start considering DBS. Because decision aids are intended to support deliberation at a specific decision point, the acceptability of future iterations will be evaluated within clinical trials while patients are actively deciding whether or not to pursue DBS. Almost all participants would recommend the decision aid to others. In addition, the narrative feedback suggested that participants found the decision aid informative, balanced, understandable and easy to use. In acceptability testing, 70% found the decision aid balanced while 30% rated it as “slanted towards DBS”. Perceptions of imbalance may have been influenced by composition of the acceptability sample, as half of the respondents had already undergone DBS. While more of those who had DBS rated the decision aid as balanced compared to those who did not have DBS, the difference in the two groups was not statistically significant. Because the decision aid is designed to support decisions about whether or not to pursue DBS, alternative treatment options are addressed on a single dedicated page, with links to additional resources for patients who wish to explore these therapies further. Future iterations of the decision aid will expand content related to non-surgical management strategies and specifically elicit feedback on bias from patients, care partners and clinicians.
Although approximately half of the participants reported being “ready” or “very ready” to make a decision and one-third reported being “moderately ready”, the total decisional conflict scores and many of the sub scores were relatively high. This apparent discrepancy suggests that while many participants felt subjectively prepared to move forward with a decision, they still experienced uncertainty or unresolved concerns in specific domains captured by the decisional conflict scale (e.g., feeling uninformed, unclear about values, or unsupported). Thus, readiness to decide may reflect a general sense of willingness to proceed, whereas decisional conflict scores more precisely capture the quality and confidence in the decision-making process. We did find an inverse moderate correlation (r = -0.54) between decisional readiness and decisional conflict, suggesting that as decisional conflict decreases, decisional readiness increases.
One unique aspect of this decision aid is the incorporation of gender specific information. We learned from our needs assessment that there were gender differences in informational needs. Women in particular wanted information about the support they would need before and after surgery, as many did not have care partners and lived alone. We therefore included a section on the support required at each step of the evaluation process, during and after surgery, and with programming. We also aimed to include an equal number of women and men in every step of the development process to ensure the decision aid met the needs of both groups. By doing so, this decision aid fills an unmet need as set forth by the Parkinson Foundation for gender sensitive care in PD, including the use of decision support tools to improve decision making and empower women. 36
Strengths of this study include a systematic and evidenced-based development process that was patient-centered. In addition to including patients throughout the development process, we also received input from clinicians and experts in movement disorders, functional neurosurgery, and shared decision making. The decision aid has also been reviewed and shared by multiple Parkinson's disease organizations, aiding with dissemination. The online format allows us to update the decision aid as new data becomes available, ensuring that the decision aid does not become out of date.
There are several limitations of this study. Feedback on the decision aid, including alpha testing and acceptability testing, was obtained primarily from participants at a single academic center. Similar to clinical trial populations in PD, the population was mainly white, non-Hispanic and well-educated, and may not be representative of all PD populations. However, many of the decision aid's core elements are likely generalizable, including the information about DBS, general evaluation processes, comparison of treatment options, and presentations of risks and benefits. Elements that may require adaptations for different contexts include language and literacy, values clarification exercises that take into account cultural norms and family values, and outcome framing. Because participants in the qualitative study had all undergone DBS evaluation, perspectives from those unaware of or declining DBS earlier were not captured. Future work will assess the utility of introducing the decision aid earlier in the care trajectory, as it is possible that exposure earlier and at multiple time points may provide the greatest benefit. A randomized controlled trial is currently underway to evaluate the effectiveness of the decision aid in reducing decisional conflict and generate preliminary implementation outcomes. The trial is powered to detect a clinically meaningful 10-point reduction in decisional conflict, corresponding to a decrease from 35 (based on the current data) to 25, a threshold associated with readiness to implement decisions. Secondary outcomes include patient knowledge, decision quality, and satisfaction with the decision. In the future, we plan to test the decision aid in multiple centers across the US and translate the decision aid to several different languages to make it more widely accessible.
Conclusion
We created an evidence-based and novel decision aid for patients with PD considering DBS with the intent to increase knowledge, manage expectations, clarify values, and facilitate shared decision making for the decision to undergo DBS. The decision aid was found to be acceptable to PD patients with and without DBS. This study provides a framework to create similar tools for PD and other movement disorders.
Supplemental Material
sj-docx-1-pkn-10.1177_1877718X261425106 - Supplemental material for Development and acceptability of a deep brain stimulation surgery decision aid for Parkinson's disease
Supplemental material, sj-docx-1-pkn-10.1177_1877718X261425106 for Development and acceptability of a deep brain stimulation surgery decision aid for Parkinson's disease by Michelle E Fullard, Erika Shelton, Ashley Dafoe, Drew S Kern, Megan A Morris and Dan D Matlock in Journal of Parkinson's Disease
Supplemental Material
sj-docx-2-pkn-10.1177_1877718X261425106 - Supplemental material for Development and acceptability of a deep brain stimulation surgery decision aid for Parkinson's disease
Supplemental material, sj-docx-2-pkn-10.1177_1877718X261425106 for Development and acceptability of a deep brain stimulation surgery decision aid for Parkinson's disease by Michelle E Fullard, Erika Shelton, Ashley Dafoe, Drew S Kern, Megan A Morris and Dan D Matlock in Journal of Parkinson's Disease
Footnotes
Acknowledgements
We would like to thank all the patients, the Colorado Program for Patient Centered Decisions Patient and Family Research Advisory Panel, and the expert panel who participated in the development of this decision aid.
Ethical considerations
This study was approved by the Colorado Multiple Institutional Review Board (COMIRB) at the University of Colorado Anschutz Medical Campus.
Consent to participate
All participants provided consent prior to participating in the study. Interview participants provided verbal consent while survey participants provided written consent.
Consent for publication
Not applicable
Author contributions
M.F. was involved in the project conception, organization, execution, data analysis and interpretation and manuscript writing. A.D and E.K. were involved in project execution and manuscript review and critique. D.K. was involved in the project execution and manuscript review and critique. M.M. and D.M. were involved in project conception, organization, execution, interpretation of data and manuscript review and critique.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was funded by the Davis Phinney Foundation, the National Institute of Arthritis and Musculoskeletal and Skin Diseases BIRCWH K12AR084226, and the National Institute on Aging K76 Beeson K76AG083349-01A1.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability
The data that support the findings of this study are available from the corresponding author upon reasonable request.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
