Abstract
Deep brain stimulation (DBS), applying high-frequency electrical stimulation to deep brain structures, has now provided an effective therapeutic option for treatment of various neurological and psychiatric disorders. DBS targeting the internal segment of the globus pallidus, subthalamic nucleus, and thalamus is used to treat symptoms of movement disorders, such as Parkinson’s disease, dystonia, and tremor. However, the mechanism underlying the beneficial effects of DBS remains poorly understood and is still under debate: Does DBS inhibit or excite local neuronal elements? In this short review, we would like to introduce our recent work on the physiological mechanism of DBS and propose an alternative explanation: DBS dissociates input and output signals, resulting in the disruption of abnormal information flow through the stimulation site.
Keywords
Conventional stereotactic surgery, such as pallidotomy and thalamotomy that make small lesions in the globus pallidus and thalamus, went through a renaissance as treatment for Parkinson’s disease in early 1990s (Laitinen and others 1992). Around the same time, deep brain stimulation (DBS) that applies high-frequency electrical stimulation through chronically implanted electrodes into a specific target in the subcortical structures was put into practical use (Benabid and others 1991, 1994). DBS was soon found to be an effective and safe alternative to lesion therapy, because it was reversible and adjustable. DBS has now been widely accepted as an effective surgical treatment for movement disorders. DBS targeting the ventral intermediate nucleus of the thalamus dramatically reduces essential and resting tremor (Benabid and others 1991, 1996). DBS targeting the subthalamic nucleus (STN) and the internal segment of the globus pallidus (GPi) has been largely used for treatment of advanced Parkinson’s disease and dyskinesia, a major side effect of
However, despite clinical benefits of DBS, the exact mechanism underlying its effectiveness has remained to be clarified and there are still several controversies about its action mechanism: Does DBS inhibit or excite local neuronal elements? (Deniau and others 2010; Kringelbach and others 2007; Perlmutter and Mink 2006; Vitek 2008; Wichmann and Delong 2006). Since DBS brings about similar beneficial effects to those of lesion therapy, it was initially believed to inhibit local neuronal elements (“inhibition hypothesis”). Actually, STN-DBS and GPi-DBS inhibited firings of neighboring neurons. On the other hand, it is not surprising that DBS excites local neuronal elements just as single stimulation does (“excitation hypothesis”). STN-DBS and GPi-DBS excited their efferents and provided effects on the GPi and thalamus, respectively.
In this short review, first, we will summarize current concepts regarding the pathophysiology of Parkinson’s disease and other movement disorders, because DBS is considered to normalize, or at least change, the pathophysiological states of movement disorders. Second, we will critically review “inhibition hypothesis” and “excitation hypothesis” as the mechanism of DBS. Finally, we would like to introduce our recent work on the physiological mechanism of DBS and propose an alternative explanation: DBS dissociates input and output signals, resulting in the disruption of abnormal information flow through the stimulation site (“disruption hypothesis”) (Chiken and Nambu 2013).
Pathophysiology of Parkinson’s Disease
Parkinson’s disease is a neurodegenerative disorder characterized by the progressive loss of nigrostriatal dopaminergic neurons originating from the substantia nigra pars compacta. The loss of dopaminergic neurons induces severe motor and non-motor dysfunctions, such as akinesia, tremor, rigidity, postural instability, cognitive impairments and depression. Three models have been proposed to explain the pathophysiology of Parkinson’s disease.
Firing Rate Model
Dopamine provides tonic excitatory inputs to striatal direct pathway neurons projecting to the GPi and tonic inhibitory inputs to striatal indirect pathway neurons projecting to the external segment of the globus pallidus (GPe), and dopamine depletion reduces these tonic excitatory and inhibitory inputs (Albin and others 1989; DeLong 1990; Gerfen and others 1990; Mallet and others 2006). Both of these changes are thought to increase mean firing rates of GPi and substantia nigra pars reticulata (SNr) neurons by reduced inhibitory inputs through the striato-GPi/SNr direct pathway and increased excitatory inputs through the striato-GPe-STN-GPi/SNr indirect pathway. Such increased mean firing rates in the output nuclei of the basal ganglia seem to induce decreased activity in thalamic and cortical neurons, resulting in akinesia (“firing rate model”). These firing rate changes of the basal ganglia, that is, increased mean firing rates in the GPi and STN and decreased mean firing rates in the GPe, were confirmed in 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine (MPTP)–induced parkinsonian monkeys (Bergman and others 1994; Boraud and others 1996, 1998; Filion and Tremblay 1991; Heimer and others 2002; Miller and DeLong 1987; Soares and others 2004; Wichmann and others 2002). Moreover, lesioning of the STN or GPi, whose activity was increased, had beneficial effects on Parkinson’s disease (Baron and others 2000, 2002; Bergman and others 1990), supporting this “firing rate model.” This model seems to be applicable to hyperkinetic disorders, such as dystonia and hemiballism that exhibit involuntary movements, as well. It was reported that firing rates in the GPe and GPi were decreased in human patients of dystonia (Starr and others 2005; Tang and others 2007; Vitek and others 1999; Zhuang and others 2004) and an animal model of dystonia (Chiken and others 2008). The development of the involuntary movements can be explained as the result of reductions in inhibitory inputs to the thalamus from the GPi.
Firing Pattern Model
Dopamine depletion enhances connections between the GPe and STN, and promotes oscillatory activity in the basal ganglia. Oscillatory and/or synchronized firings of the basal ganglia disable individual neurons to process and relay motor-related information, resulting in the failure of appropriate movements (“firing pattern model”) (Bergman and others 1998). Abnormal firing patterns, such as bursts and oscillations, were recorded in the GPe, GPi and STN of parkinsonian monkeys (Bergman and others 1994; Heimer and others 2002, 2006; Raz and others 2000; Tachibana and others 2011; Wichmann and Soares 2006) and parkinsonian patients (Levy and others 2000). Oscillatory local filed potentials (LFPs), especially those in the beta frequency band, were also observed in parkinsonian patients using DBS electrodes (Brown 2003; Brown and others 2001; Brown and Williams 2005; Gatev and others 2006; Hammond and others 2007).
Dynamic Activity Model
In the normal state, signals through the cortico-STN-GPi/SNr hyperdirect, cortico-striato-GPi/SNr direct, and cortico-striato-GPe-STN-GPi/SNr indirect pathways cause dynamic activity changes in the GPi (see Fig. 3C) and release only a selected motor program at a selected timing with a clear boundary between the selected and other unnecessary competing motor programs (Nambu 2008; Nambu and others 2015). In Parkinson’s disease, dopamine depletion reduces movement-related GPi inhibition through the direct pathway and facilitates movement-related GPi excitation through the hyperdirect and indirect pathways (Boraud and others 2000; Degos and others 2005; Kita and Kita 2011; Leblois and others 2006). These changes shorten and narrow movement-related GPi inhibition, which leads to the reduction of disinhibition in the thalamus and cortex, resulting in akinesia (“dynamic activity model”). In hyperkinetic disorders, movement-related inhibition in the GPi through the direct pathway is enhanced, and GPi excitation through the hyperdirect and indirect pathways is reduced. These dynamic changes induce excessive, uncontrolled disinhibition in the thalamus and cortex, leading to involuntary movements (Chiken and others 2008; Nambu and others 2011; Nishibayashi and others 2011).
“Inhibition Hypothesis”: DBS Inhibits Local Neuronal Elements
Both DBS and lesion therapy have similar beneficial effects on the alleviation of symptoms. STN-DBS (Benazzouz and others 1993; Benabid and others 1994; Limousin and others 1995) showed similar effects on parkinsonian motor signs to STN-lesion (Aziz and others 1991; Bergman and others 1990; Levy and others 2001) or STN-blockade (Luo and others 2002). Thus, DBS was initially believed to inhibit local neuronal elements (“inhibition hypothesis”). Actually, the most common effects of STN-DBS and GPi-DBS are the reduction of the firing rates of neighboring neurons. Suppression of neuronal activity was recorded around the stimulating sites of STN-DBS in parkinsonian patients (Filali and others 2004; Welter and others 2004), parkinsonian monkeys (Meissner and others 2005; Moran and others 2011) and parkinsonian rats (Shi and others 2006; Tai and others 2003). However, a limited number of STN neurons showed complete cessation of firings, and other STN neurons exhibited residual neuronal activity during STN-DBS (Meissner and others 2005; Tai and others 2003; Welter and others 2004). Inhibitory effects of GPi-DBS on firings of neighboring neurons were also reported in parkinsonian patients (Dostrovsky and others 2000; Lafreniere-Roula and others 2010; Wu and others 2001), parkinsonian monkeys (Boraud and others 1996) and normal monkeys (Fig. 1A) (Chiken and Nambu 2013). GPi-DBS induced complete inhibition of local neuronal firings more commonly than STN-DBS.
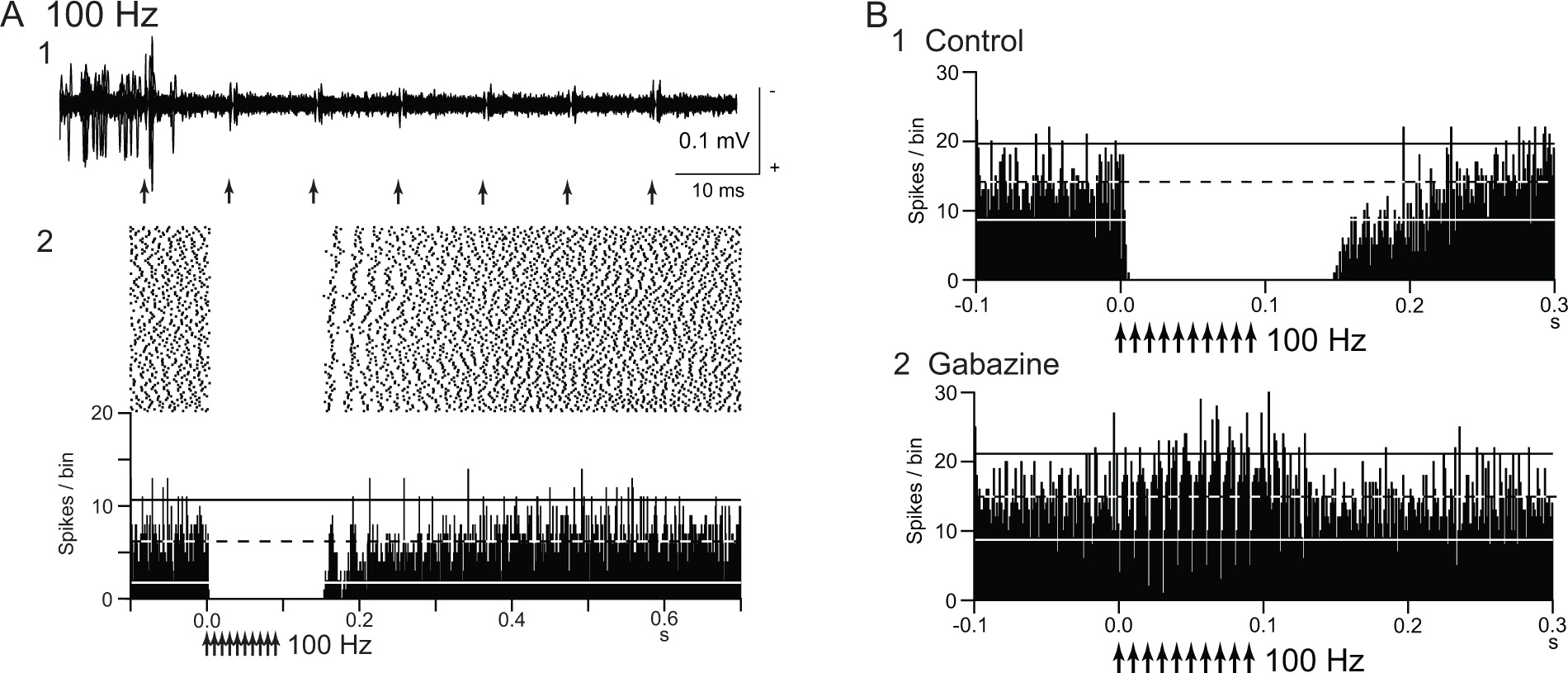
Deep brain stimulation (DBS) inhibits local neuronal firings. (A) Responses of an internal pallidal (GPi) neuron to local repetitive high-frequency stimulation (GPi-DBS; 30 µA, 100 Hz, 10 pulses; arrows) in a normal monkey. Raw traces of spike discharges after removing the stimulus artifacts (1) and raster and peristimulus time histograms (PSTHs; 100 trials; binwidth, 1 ms) (2) are shown. Spontaneous discharge of the GPi neuron was completely inhibited by GPi-DBS. (B) Effect of local injection of gabazine (GABAA receptor antagonist) in the vicinity of the recorded GPi neuron. The inhibition induced by GPi-DBS (1) was abolished after gabazine injection (2). Modified from Chiken and Nambu (2013).
The “inhibition hypothesis” fits well with the “firing rate model” and “firing pattern model” of movement disorders. DBS reduces abnormally increased firings or abnormal firing patterns in the STN and GPi and ameliorates parkinsonian motor symptoms. However, it seems to be difficult to explain why GPi-DBS can treat dystonic symptoms, in which the GPi shows low activity.
Several possible mechanisms can account for the inhibitory responses during DBS: (1) depolarization block, (2) inactivation of voltage-gated currents (Beurrier and others 2001; Do and Bean 2003; Shin and others 2007), and (3) activation of inhibitory afferents (Boraud and others 1996; Chiken and Nambu 2013; Deniau and others 2010; Dostrovsky and others 2000; Dostrovsky and Lozano 2002; Johnson and McIntyre 2008; Liu and others 2008; Meissner and others 2005). Our recent study (Chiken and Nambu 2013) confirmed that inhibitory responses induced by GPi-DBS were mediated by GABA receptors (Fig. 1B). The GPi receives inhibitory GABAergic inputs from the striatum and GPe (Shink and Smith 1995; Smith and others 1994), and these inhibitory GABAergic afferents are considered to be activated by GPi-DBS. The GPi also receives excitatory glutamatergic inputs from the STN, and these afferents should also be activated. However, GABAergic terminals are predominant (Shink and Smith 1995), and thus, GABAergic inhibition probably overwhelms glutamatergic excitation. GPi-stimulation induced directly-evoked spikes, which are characterized by a short and constant latency (Fig. 2A). GPi-DBS also suppressed such directly evoked spikes by strong GABAergic inhibition (Fig. 2B). In contrast to the GPi, GPe-DBS induced complex responses composed of excitation and inhibition in neighboring GPe neurons (Chiken and Nambu 2013). Since GABAergic terminals on GPe neurons are less dense than those on GPi neurons (Shink and Smith, 1995), glutamatergic excitation can be observed in the GPe. Similarly, STN-DBS generated both excitatory and inhibitory postsynaptic potentials in STN neurons through activation of both glutamatergic and GABAergic afferents (Lee and others 2004). Thus, DBS activates afferent axons in the stimulated nucleus, and the net effects vary depending on the composition of the inhibitory and excitatory axon terminals.
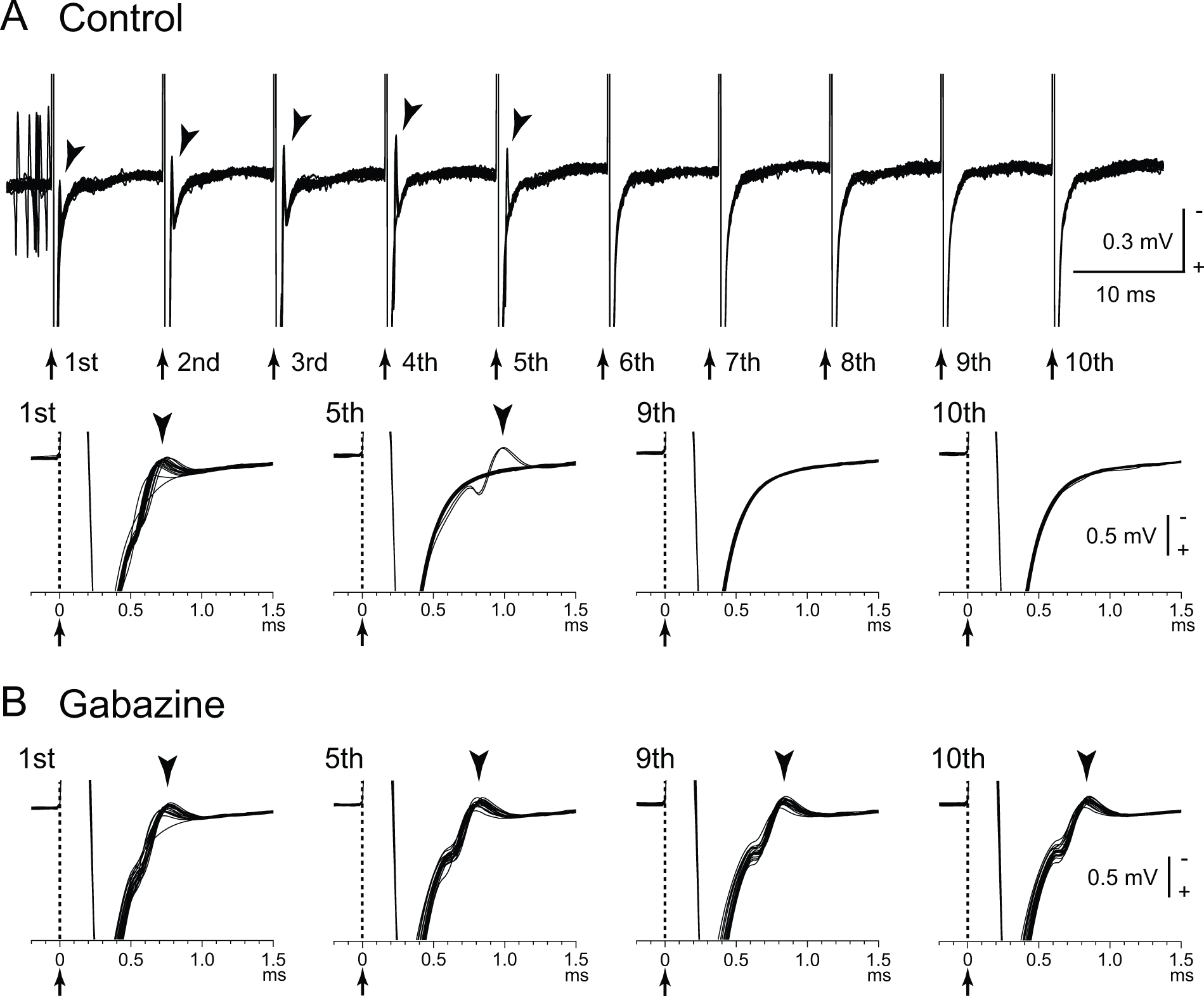
Directly evoked spikes of internal pallidal (GPi) neurons are inhibited during GPi–deep brain stimulation (DBS). (A) Raw traces showing directly evoked spikes (arrowheads) of a GPi neuron by GPi-DBS (40 µA, 100 Hz, 10 pulses; arrows with dotted lines) in a normal monkey. Traces with long (top) and short (bottom) time scales are shown. GPi-DBS failed to evoke spikes (from 6th to 10th stimuli). (B) Effects of local gabazine injection on the inhibition of directly evoked GPi responses. Gabazine injection decreased the failure rate, and each stimulus successfully evoked spikes (5th, 9th, and 10th stimuli). Modified from Chiken and Nambu (2013).
“Excitation Hypothesis”: DBS Excites Local Neuronal Elements
It is not surprising that DBS excites local neuronal elements just as single stimulation does (“excitation hypothesis”). Directly evoked spikes were induced by GPi-DBS in GPi neurons (Johnson and McIntyre 2008; McCairn and Turner 2009). GPi-DBS reduced firings in thalamic neurons of parkinsonian monkeys (Anderson and others 2003) and dystonia patients (Pralong and others 2003; Montgomery 2006) through the inhibitory GPi-thalamic projections. STN-DBS increased firings in GPi neurons of parkinsonian monkeys (Hashimoto and others 2003) and GPi/GPe neurons (Reese and others 2011) and SNr neurons (Galati and others 2006) of parkinsonian patients through the excitatory STN-GPi/SNr/GPe projections. A modeling study has suggested that subthreshold DBS suppresses intrinsic firings in the cell bodies, while suprathreshold DBS induces spikes at the stimulus frequency in the axons without corresponding firings in the cell bodies (McIntyre and others 2004). Thus, although stimulation may fail to activate the cell bodies due to strong GABAergic inhibition, it can still excite the efferent axons and provide spikes to the target nucleus at the stimulus frequency. Other studies showed that GPi-DBS induced multiphasic responses consisting of excitation and inhibition in the GPi of parkinsonian monkeys (Bar-Gad and others 2004; Erez and others 2009; McCairn and Turner 2009) and dystonia hamsters (Leblois and others 2010). It was recently reported that GPe-DBS changed firing patterns in STN, GPi and thalamic neurons of parkinsonian monkeys and improved motor signs, suggesting the GPe as a potential target for DBS (Vitek and others 2012).
Deep brain stimulation also excites afferent axons antidromically. STN-DBS activated GPi neurons antidromically in parkinsonian monkeys (Moran and others 2011), probably by current spread to the lenticular fasciculus, a part of GPi-thalamic fibers (Miocinovic and others 2006). GPi-DBS activated thalamic neurons antidromically in dystonia patients, probably by activation of thalamic axons passing in the vicinity of GPi-DBS electrodes (Montgomery 2006). STN-DBS with low intensity induced GABAergic inhibition in the SNr through antidromic activation of GPe neurons projecting to both the STN and SNr, while STN-DBS with higher intensity induced glutamatergic excitation in the SNr through the STN-SNr projections (Deniau and others 2010; Maurice and others 2003). STN-DBS activated neurons in the motor cortex antidromically (Degos and others 2013; Q. Li and others 2012, S. Li and others 2007). Recent development of optogenetic study showed that selective stimulation of cortico-STN afferent axons without activation of STN efferent axons ameliorated the symptoms of parkinsonian mice (Gradinaru and others 2009).
The “excitation hypothesis” fits well with the “firing pattern model” of movement disorders, but not with the “firing rate model”. Excitation and/or excitation-inhibition reach the target nucleus along efferent pathways, or antidromic activation reaches the original region along afferent pathways. These activity changes may alter the firing rates and patterns, and normalize or suppress abnormal firings of target nucleus (Anderson and others 2003; Degos and others 2013; Deniau and others 2010; Hammond and others 2007; Hashimoto and others 2003; Johnson and McIntyre 2008; Q. Li and others 2012; S. Li and others 2007; Vitek 2008). However, the precise mechanism how DBS normalizes firing patterns remains to be elucidated.
“Disruption Hypothesis”: DBS Disrupts Abnormal Information Flow
We recently examined the effects of GPi-DBS on the responses of GPi neurons evoked by motor cortical stimulation in normal monkeys (Fig. 3) (Chiken and Nambu 2013). Cortical stimulation induces a triphasic response composed of early excitation, inhibition and late excitation in the GPi (Fig. 3A), which are mediated by the hyperdirect, direct, and indirect pathways, respectively (Nambu and others 2000, 2002) (Fig. 3C). GPi-DBS completely inhibited both cortically evoked responses and spontaneous discharges by strong GABAergic inhibition (Fig. 3B), suggesting that it blocks information flow through the GPi (“disruption hypothesis”) (Fig. 4). STN-DBS may similarly block transmission of signals through the STN: Maurice and others (2003) examined the effects of STN-DBS on cortically evoked responses in SNr neurons of normal rats. Cortically evoked early and late excitation was abolished or largely reduced during STN-DBS, whereas cortically evoked inhibition was preserved, suggesting that information flow through the hyperdirect and indirect pathways was blocked by STN-DBS without interrupting the direct pathway.
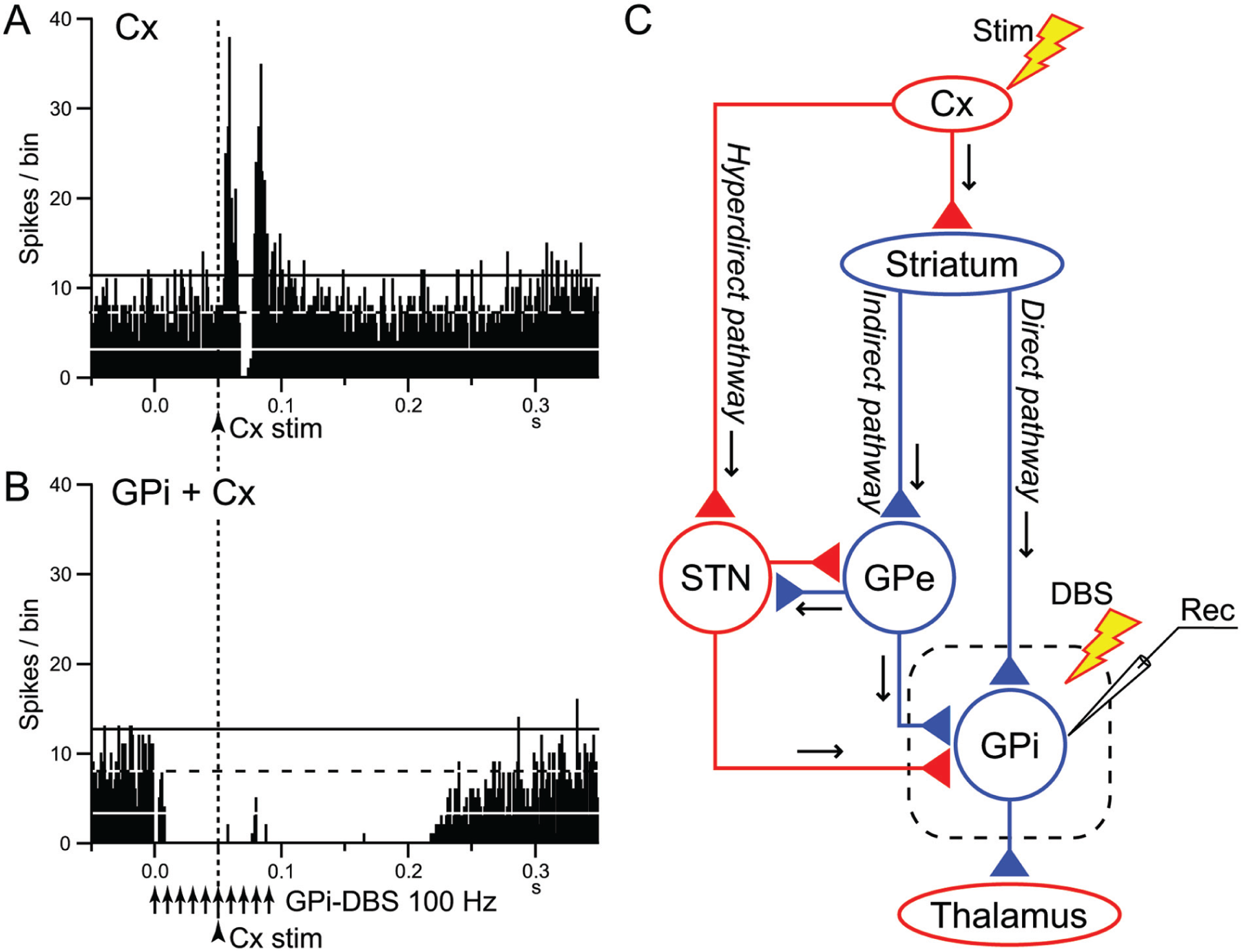
Deep brain stimulation (DBS) of the internal segment of the globus pallidus (GPi) disrupts information flow through the GPi. (A, B) Effects of local GPi-DBS on cortically evoked responses of a GPi neuron in a normal monkey. PSTHs in response to a single-pulse stimulation of the primary motor cortex (Cx) (arrowhead with dotted line) without (A) and with GPi-DBS (arrows) (B) are shown. In (B), cortical stimulation was applied 50 ms after the initiation of GPi-DBS. The cortically evoked responses were entirely inhibited during GPi-DBS. (C) Schematic diagram showing the cortico-basal ganglia pathways and stimulating (Stim and DBS) and recording (Rec) sites. Cortically evoked early excitation, inhibition and late excitation in (A) are mediated by the hyperdirect, direct, and indirect pathways, respectively. Cx, cerebral cortex; GPe, external segment of the globus pallidus; STN, subthalamic nucleus. Red and blue triangles represent glutamatergic excitatory and GABAergic inhibitory terminals, respectively. Modified from Chiken and Nambu (2013).
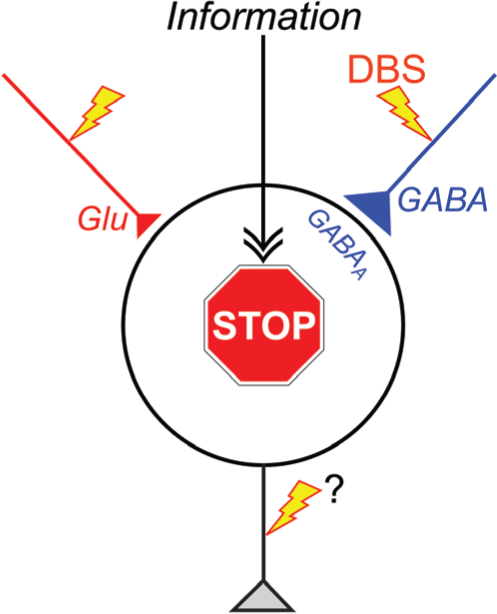
“Disruption hypothesis” explaining the mechanism underlying the effectiveness of deep brain stimulation (DBS). DBS activates axon terminals in the stimulated nucleus, induces extensive release of neurotransmitters, such as GABA and glutamate (Glu), and dissociates inputs and outputs in the stimulated nucleus. Thus, DBS results in disruption of the abnormal information flow through the cortico-basal ganglia loop in the pathological conditions. GABAA, GABAA receptors.
The “disruption hypothesis” fits well with the “firing rate model,” “firing pattern model,” and “dynamic activity model.” Since abnormally increased firings, abnormal firing patterns, or abnormal dynamic activity changes in the basal ganglia are transmitted to the thalamus and motor cortex, and finally induce motor symptoms, disrupting such abnormal information flow through the GPi and STN can suppress the expression of motor symptoms. The GPi is an output nucleus of the basal ganglia, and thus GPi-DBS disrupts all information outflow from the basal ganglia. On the other hand, the GPe-STN reciprocal connections produce abnormal firing patterns in Parkinson’s disease (“firing pattern model”), and interruption of information flow through the STN reduces them. In addition, the hyperactivity along the hyperdirect and indirect pathways are suggested in Parkinson’s disease (“firing rate model” and “dynamic activity model”), and interruption of information flow through the STN blocks such hyperactivity. The “disruption hypothesis” can explain the long-standing paradox as well: DBS produces similar therapeutic effects to lesion therapy because both DBS and lesions interrupt abnormal information flow. Another paradox is that GPi-DBS has therapeutic effects to both Parkinson’s disease and dystonia. Parkinsonian symptoms are induced by increased firing rates (“firing rate model”), abnormal firing patterns (“firing pattern model”) or reduced movement-related inhibition (“dynamic activity model”) in the GPi. In the case of dystonia, signals through the hyperdirect, direct, and indirect pathways may induce a sequence of bursts and pauses in the GPi, and subsequent inhibition and rebound bursts in the thalamus and cortex, leading to the manifestation of involuntary movements (“dynamic activity model”). GPi-DBS blocks abnormal information flow responsible for motor symptoms in both diseases. Other research groups have also proposed similar ideas of functional disconnection of the stimulated elements (Anderson and others 2006; Deniau and others 2010; Moran and others 2011).
Concluding Remarks
Deep brain stimulation has a variety of effects on neurons in the stimulated nucleus of the cortico-basal ganglia loop through orthodromic activation of efferent axons, antidromic and orthodrimic activation of afferent axons. The total effects may vary depending on the composition of neuronal elements in the stimulated nucleus. Here, we have suggested a common key mechanism of DBS: DBS dissociates input and output signals in the stimulated nucleus and disrupts abnormal information flow through the cortico-basal ganglia loop in the pathological conditions (“disruption hypothesis”) (Fig. 4). Understanding the exact mechanism of DBS will lead us to better therapeutic options, toward improvements and upgrading of DBS.
Footnotes
Acknowledgements
The authors thank Shigeki Sato, Kanako Awamura, and Hitomi Isogai for technical assistance.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research authorship, and/or publication of this article: This work was supported by CREST and Strategic Japanese-German Cooperative Programme from Japan Science and Technology Agency, and a Grant-in-Aid for Scientific Research (A) (26250009) from the Ministry of Education, Culture, Sports, Science and Technology (MEXT) of Japan.
