Abstract
Background
Parkinson's disease is (PD) a progressive neurodegenerative disorder. This study investigated the effects of an individualized nutritional intervention based on the Ketoflex 12/3 protocol, in addition to standard medical treatment, on motor and non-motor symptoms in PD.
Methods
40 individuals with PD were included in the study, and participants were randomly assigned to intervention and control. All individuals were classified according to inflammatory, glycotoxic, toxic, and vascular biotypes. The intervention group was assigned a plant-rich diet with a low glycemic index, consistent with ketogenic principles, free of inflammatory effects, and including intermittent fasting. The primary endpoint was the change in motor symptoms measured by the Unified Parkinson's Disease Rating Scale part III (UPDRS-III) from baseline to six months. Secondary endpoints included apathy (Starkstein Apathy Scale), activities of daily living (ADL/IADL), and gastrointestinal function (Bristol stool scale).
Results
Compared with the control group, the intervention group showed a significantly greater improvement in motor symptoms as measured by UPDRS-III (−11.0 vs +2.1 points from baseline to six months; p < 0.001). Significant between-group differences were also observed for secondary endpoints, including apathy (Starkstein Apathy Scale), activities of daily living (ADL/IADL), and gastrointestinal function (Bristol stool scale), all favoring the intervention group. Spearman correlation analyses revealed significant negative correlations with ADL and UPDRS-III scores, particularly in individuals with an inflammatory phenotype.
Conclusions
The findings suggest an individualized nutritional approach may contribute to improvement in both motor and non-motor symptoms in PD. Larger, multi-center trials with extended follow-up are needed.
Plain language summary
Parkinson's disease is a complex condition that affects both movement and non-motor functions such as digestion, sleep, and mood. In this study, we examined whether a personalized, plant-rich eating approach, in which participants ate their daily meals within a fixed time window, could improve selected motor and non-motor symptoms in people with Parkinson's disease. Participants followed an individualized diet plan based on their metabolic characteristics, including guidance on what and when to eat, and received regular support over a six-month period.
The results suggest that this nutritional approach was feasible in daily life and associated with improvements in motor symptoms and some non-motor features. However, as this was a pilot study with a relatively small number of participants, the findings should be interpreted with caution. Larger, well-controlled studies are needed to confirm these results and to better understand how personalized nutrition may support the management of Parkinson's disease.
Keywords
Background
Parkinson's disease (PD) is a progressive neurodegenerative disorder. Pharmacological treatments remain central to conventional PD management; however, their limited efficacy has increased interest in supportive and complementary strategies, particularly the growing focus on the role of nutrition in modifying disease mechanisms. 1
Current treatment approaches are based on dopaminergic pharmacotherapy. Although these therapies remain central to PD management and provide meaningful symptomatic relief, they offer limited support for the metabolic, inflammatory, and gastrointestinal contributors that underlie many non-motor symptoms. This unmet need has intensified interest in holistic approaches that address the systemic nature of PD, including structured nutritional interventions. This situation highlights the need for holistic interventions that go beyond symptomatic control and target systemic effects in the management of PD. In recent years, ketogenic-based dietary approaches have begun to be investigated for their therapeutic potential in neurodegenerative diseases2–8. Modern dietary patterns, typically characterized by high glycemic load, low fiber intake, and widespread consumption of ultra-processed foods, are associated with systemic inflammation and gut dysbiosis. These characteristics may be particularly relevant for Parkinson's disease, where chronic inflammation, impaired metabolic flexibility, and gastrointestinal dysfunction play central roles in disease progression9–13. Nutrition emerges as a factor that directly affects the functionality of nerve cells. Mechanisms involved in the pathophysiology of PD can be modulated by diets.14,15 Dietary approaches such as the Mediterranean diet, intermittent fasting, and ketogenic-based interventions have been increasingly explored for their potential metabolic and anti-inflammatory effects in neurodegenerative conditions. This growing body of work has encouraged interest in structured nutritional frameworks that integrate complementary dietary principles.
The Ketoflex 12/3 protocol was implemented according to its core scheduling and dietary principles. Participants were instructed to stop eating at least three hours before bedtime (‘3’) and maintain a 12-h overnight fasting window (‘12’), resulting in a feeding period of approximately 12 h each day. The dietary composition emphasized a plant-rich, low-glycemic, structurally aligned with ketogenic principles and designed to promote anti-inflammatory metabolic effects16–18. This approach aims to support synaptic function, neuroplasticity, and cellular regeneration. While the Ketoflex 12/3 protocol has shown promising results in particularly Alzheimer's disease, systematically evaluated data in PD is quite limited. 17
Methods
This trial was conducted as an assessor-blinded randomized controlled design. The clinical diagnosis of PD was established by the neurologist (L.H.). All dietary counseling sessions, nutritional education, and protocol follow-up were performed by the study nutritionist (B.T.). Due to the nature of the dietary intervention, participants and the study nutritionist were aware of group allocation, whereas clinicians performing outcome assessments remained blinded to treatment assignment. Allocation concealment was not applied during randomization. The study is designed to evaluate the effects of a six-month application of the Ketoflex 12/3 protocol, added to routine medical treatment, on motor and non-motor symptoms in individuals with PD. Participants were randomly assigned to the intervention or control group using a computer-generated simple randomization sequence with a 1:1 allocation ratio. All participants completed the study with no dropouts. The intervention group received a plant-rich ketogenic dietary based on the Ketoflex 12/3 protocol in addition to their standard treatment. Participants in the control group continued conventional PD care. They were seen by their treating neurologist approximately every three months for routine clinical follow-up and medication management, with pharmacological adjustments made as clinically indicated. No dietary counseling, nutritional education, fasting instructions, or Ketoflex 12/3 protocol -related materials were provided to the control group. Apart from the scheduled baseline and 6-month study assessments, the control group received no additional study-related contact. In both groups, all primary and secondary outcomes were evaluated at baseline and at the end of the 6-month study period using standardized clinical assessments.
The study was approved by the Istanbul Medipol University Non-Interventional Clinical Research Ethics Committee (Approval No: E-10840098-202.3.02-1820, March 6, 2025) and conducted in accordance with the Declaration of Helsinki and its subsequent revisions. (ClinicalTrials.gov ID:NCT06892418).
Forty individuals aged 40–85 years who met the Movement Disorder Society (MDS) Clinical Diagnostic Criteria for Parkinson's Disease (2015) were enrolled 19 after providing informed consent. Participants were required to be on stable dopaminergic treatment, defined as no changes in medication type or dosage for at least two months prior to enrollment. Exclusion criteria included a history of alcohol or substance dependence, chronic renal failure, neuromuscular disease, or the presence of amalgam fillings, due to their potential to confound metabolic and detoxification-related outcomes. No specific score thresholds were applied as inclusion criteria. Forty participants were then randomized in a 1:1 ratio to the intervention (n = 20) or control group (n = 20) through computer-generated simple randomization. Levodopa equivalent daily doses (LEDD) were calculated for all participants. 20
Firstly, a comprehensive nutritional and lifestyle history was obtained through a structured clinical interview rather than a formal validated questionnaire. This included a detailed assessment of habitual dietary intake, meal timing, physical activity patterns, sleep routines, and metabolic risk factors. The baseline lifestyle history focused primarily on habitual nutritional patterns, including meal timing, food choices, and dietary routines, as these domains were directly relevant to the intervention. Based on the data obtained, each individual was classified into one of four basic pathophysiological subtypes based on clinical and biochemical indicators: inflammatory, glycotoxic, toxic, and vascular. This biotype classification was exploratory in nature and has not yet been formally validated. The inflammatory type was defined by elevated hs-CRP (>0.9 mg/L). The glycotoxic type was characterized by metabolic syndrome, including BMI >30 kg/m2, HOMA-IR >2.5, and fasting insulin >10 μIU/mL. The toxic type was characterized by heavy metal accumulation, chemical sensitivity, or relevant exposure history. The vascular type was characterized by hypertension, atherosclerosis, or a history of cardiovascular disease. Participants may meet multiple subtypes simultaneously, necessitating a layered, integrated, and flexible intervention approach.
Participants in the intervention group received structured dietary counseling delivered by the study nutritionist (B.T.) according to a predefined schedule: weekly sessions during the first month, monthly sessions through month three, and a final follow-up session at month six. Each participant was provided with written Ketoflex 12/3 protocol nutritional guidelines. The personalized six-month program emphasized a plant-rich, low-carbohydrate dietary pattern; elimination of refined carbohydrates; and selection of nutrient-dense, anti-inflammatory foods. Participants were instructed to stop eating at least three hours before bedtime and maintain a 12-h overnight fasting window. All meals were self-prepared by participants; no pre-prepared meals or meal delivery were provided. Adherence was monitored at every online or in-person counseling session through structured 24-h dietary recalls and detailed menu reviews, with some participants additionally submitting meal photographs or brief food diaries. No participants used laxatives or other constipation-related medications during the study period. Adherence to the dietary protocol was assessed qualitatively during regular counseling sessions using structured 24-h dietary recalls and detailed menu reviews.
Taking into account pathophysiological characteristics specific to each metabolic biotype, the plant-rich ketogenic diet and intermittent fasting intervention was delivered within an individualized framework. Within the Ketoflex 12/3 framework, dietary counseling was tailored to address dominant metabolic features, including inflammation, insulin resistance, detoxification capacity, and vascular risk. Accordingly, participants classified as inflammatory biotypes received guidance emphasizing the avoidance of pro-inflammatory foods; glycotoxic biotypes received guidance focused on glycemic load modulation and metabolic balance; toxic biotypes were counseled on minimizing exposure to potential dietary toxins and prioritizing antioxidant-rich foods; and vascular biotypes received targeted recommendations regarding salt intake and fat quality. Across all biotypes, foods were organized into standardized categories, with individualized “prohibitions” and “replacements” defined according to each participant's baseline dietary pattern and clinical context. All participants were instructed to maintain a daily fasting period of at least 12 h and to avoid food intake within three hours before bedtime.
The primary outcome of the study was the change in motor symptom severity measured by the Movement Disorder Society–Unified Parkinson's Disease Rating Scale, part III (MDS-UPDRS III), 21 from baseline to the end of the 6-month intervention period. Secondary outcomes included changes in selected non-motor symptoms and functional measures, assessed using the Mini Mental State Examination (MMSE), 22 the Geriatric Depression Scale (GDS), 23 the Starkstein Apathy Scale (SAS), 24 the Impulsive-Compulsive Behavior Scale (ICBS), 25 the Epworth Sleepiness Scale, 26 the Bristol Stool Scale, 27 the Activities of Daily Living and Instrumental Activities of Daily Living (ADL/IADL).28,29
Data analysis was performed using Jamovi 2.4.8. Normality of continuous variables was assessed using the Shapiro–Wilk test, and homogeneity of variances was evaluated with Levene's test. For variables meeting parametric assumptions, paired-samples t-tests or repeated-measures ANOVA were applied. When ANOVA indicated significance, post-hoc pairwise comparisons were conducted using Bonferroni correction. Nonparametric or ordinal variables were analyzed with the Wilcoxon signed-rank test. Statistical significance was defined as p < 0.05.
Results
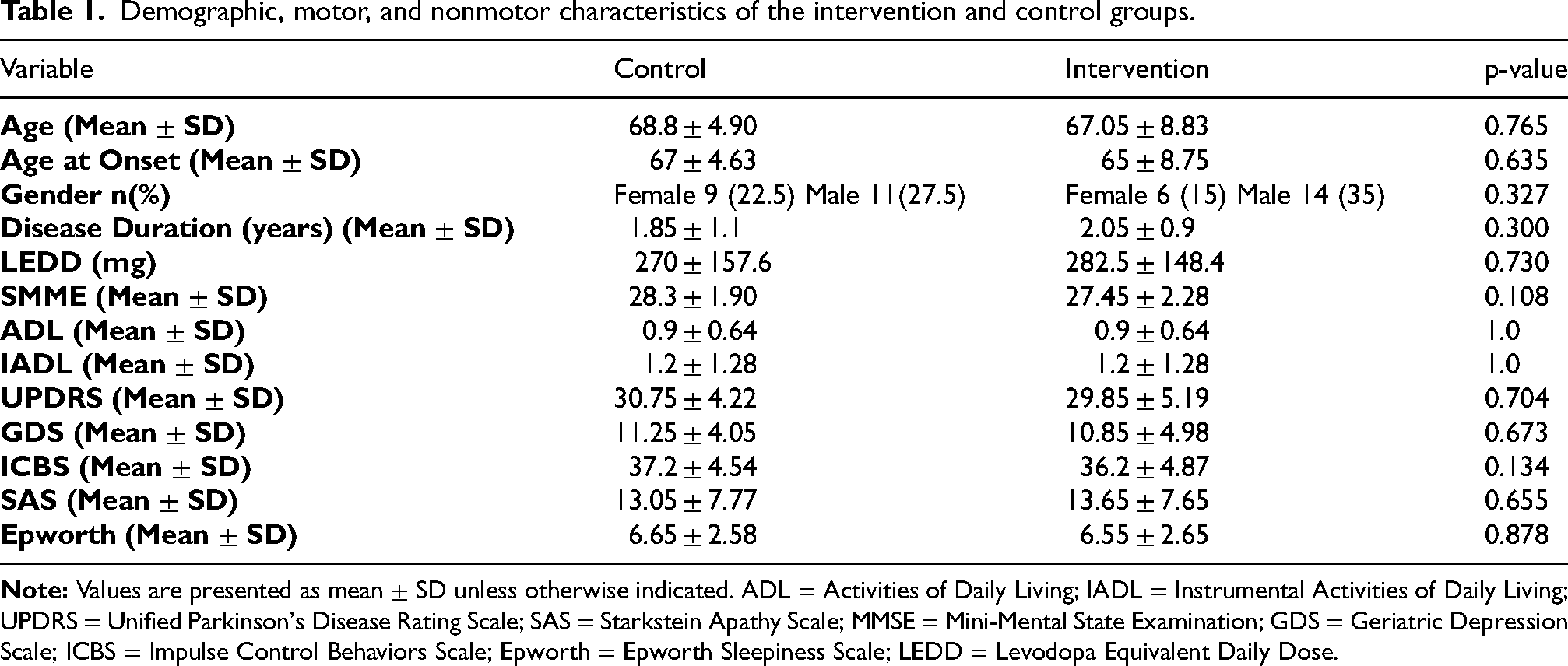
The mean age of individuals in the intervention group was 67.0 (SD = 8.83), while the mean age of individuals in the control was 68.8 (SD = 4.89) (Table 1).
Demographic, motor, and nonmotor characteristics of the intervention and control groups.
Frequency analysis showed that the inflammatory type comprised 60% of the sample—42.5% in the intervention group and 17.5% in the control group. The glycotoxic type accounted for 35% (15% intervention, 20% control), the toxic type for 30% (7.5% intervention, 22.5% control), and the vascular type for 12.5% (7.5% intervention, 5% control).
At baseline, there was no significant difference between the intervention and control groups in UPDRS-III scores. Over the 6-month intervention period, the intervention group showed a marked improvement in motor symptoms, whereas the control group showed a worsening of UPDRS-III scores. The between-group difference in change over time was highly significant (p < 0.001), indicating a substantially greater improvement in the intervention group. On the UPDRS-III scale, the main effect of time was first found to be statistically significant (F (1, 38) = 30.30, p < 0.001, η2 = 0.444). This result indicates that, when evaluated independently of the groups, there was a significant change in the UPDRS-III scores of all participants over time. However, the direction and extent of this change is meaningful not only in terms of the passage of time, but also in terms of group differences.
The most striking finding is that the interaction between time and group was significant (F (1, 38) = 66.20, p < 0.001, η2 = 0.635). The strong interaction effect shows that the intervention and control groups changed in significantly different ways over time. Specifically, a significant decrease in UPDRS-III scores was observed in the intervention group, while no similar level of improvement was detected in the control group. This suggests that the improvement in motor symptoms observed in the intervention group is related to the direct effect of the intervention, rather than simply a natural progression over time. Furthermore, the group main effect was also statistically significant (F (1, 38) = 64.10, p < 0.001, η2 = 0.628), indicating a significant overall difference in UPDRS-III scores between the two groups. The overall UPDRS-III means for the intervention group were lower than those for the control group.
Post-hoc comparisons supporting these results also revealed that the intervention group's post-intervention UPDRS-III scores were statistically significantly lower than both their own baseline levels and the control group's post-test scores (t (38) = 16.09, p < 0.001). The pronounced post-test decline in the intervention group indicates that the effect is attributable to the intervention itself rather than to time alone.
Over the six-month period, basic activities of daily living (ADL) improved significantly in the intervention group, whereas no meaningful improvement was observed in the control group. The between-group difference in ADL change was statistically significant, indicating a greater functional benefit in the intervention group. Repeated measures on ADL scores revealed a significant interaction between time and group factors according to the analysis of variance, F (1, 38) = 38.38, p < 0.001, η2 = 0.503. The main effect of time was not statistically significant, F (1, 38) = 0.384, p = 0.539, η2 = 0.010. The main effect between groups was found to be significant, F (1, 38) = 5.63, p = .023, η2 = 0.129. According to the Levene test results, variance homogeneity was achieved for both the pre-test (p = 1.000) and the post-test (p = .165).
In post-hoc comparisons, a statistically significant difference was observed between the intervention and control groups in the posttest period (p < 0.001). The posttest mean of the intervention group was significantly lower than that of the control group. This finding indicates that changes in ADL scores over time varied by group.
Instrumental activities of daily living (IADL) showed a significant improvement within the intervention group over the six-month period (p < 0.001), whereas no significant change was observed in the control group. Although post-intervention IADL scores were numerically lower in the intervention group, the between-group difference did not reach statistical significance. Repeated measures on IADL scores assessing the complex functions of daily living revealed a significant interaction between time and group factors according to the results of the analysis of variance, F (1, 38) = 24.43, p < 0.001, η2 = 0.391. The main effect of time was not statistically significant, F (1, 38) = 2.71, p = 0.108, η2 = 0.067. The main effect of groups was also not significant, F (1, 38) = 1.04, p = 0.315, η2 = 0.027.
In post-hoc comparisons, the intervention group's IADL scores in the posttest period showed a significant difference compared to the control group (p < 0.001). This finding suggests that the intervention program positively influenced participants’ independent living activities.
Apathy scores improved significantly within the intervention group over the six-month period (p < 0.001), whereas no significant change was observed in the control group (p = 0.866). However, the between-group difference in SAS scores at post-intervention was not statistically significant (p = 0.333). Repeated-measures analysis using the SAS indicated that both the main effect of time and the time–group interaction were statistically significant. The time effect was found to be significant (F (1, 38) = 61.70, p < 0.001, η2 = 0.619), indicating that participants experienced a significant change in their overall SAS scores over time. More importantly, the interaction between time and group was also highly significant (F (1, 38) = 99.30, p < 0.001, η2 = 0.723). This interaction effect indicates that the change demonstrated by the intervention group over time was statistically significantly different compared to the control group, reflecting a specific effect of the intervention.
Although the overall difference between groups was not found to be significant (F (1, 38) = 0.71, p = 0.404, η2 = 0.018), post-hoc comparisons showed a significant decrease in SAS scores in the intervention group. The post-test SAS scores of the intervention group were significantly lower than both their own pre-test scores and the post-test levels of the control group (t (38) = 12.60, p < 0.001).
The Wilcoxon signed-rank test comparing pre- and post-intervention MMSE scores revealed no significant change in cognitive function.
The Wilcoxon signed-rank test assessing changes in geriatric depression levels indicated that the intervention did not significantly affect depressive symptoms.
The Wilcoxon signed-rank test conducted to assess the level of impulsive-compulsive behaviors did not reveal a statistically significant difference between pre- and post-intervention ICBS scores (W = 17, p = 0.198). However, the calculated effect size value (r = 0.619) indicates a medium-high level of effect. Although statistical significance was not reached, this finding suggests that the intervention may have a clinically meaningful effect on impulsive–compulsive symptoms.
The results of the analysis of the Epworth showed no significant difference between Epworth scores before and after the intervention (Wilcoxon W = 37.0, p = 0.904).
Gastrointestinal function, assessed by the Bristol stool scale, improved significantly within the intervention group over the six-month period (p = 0.002), whereas no significant change was observed in the control group. However, the between-group difference in post-intervention Bristol scores was not statistically significant. Data obtained from the Bristol revealed a statistically significant difference between pre- and post-intervention assessments. The paired-sample t-test showed a significant decrease in Bristol scores over time (t(39) = 3.01, p = 0.005). This result indicates that there was a significant change in participants’ stool consistency after the intervention. The effect size was found to be moderate (Cohen's d = 0.476), suggesting that the observed change may be clinically meaningful (Table 2)
Comparison of intervention and control groups on clinical outcomes at baseline and post-test.
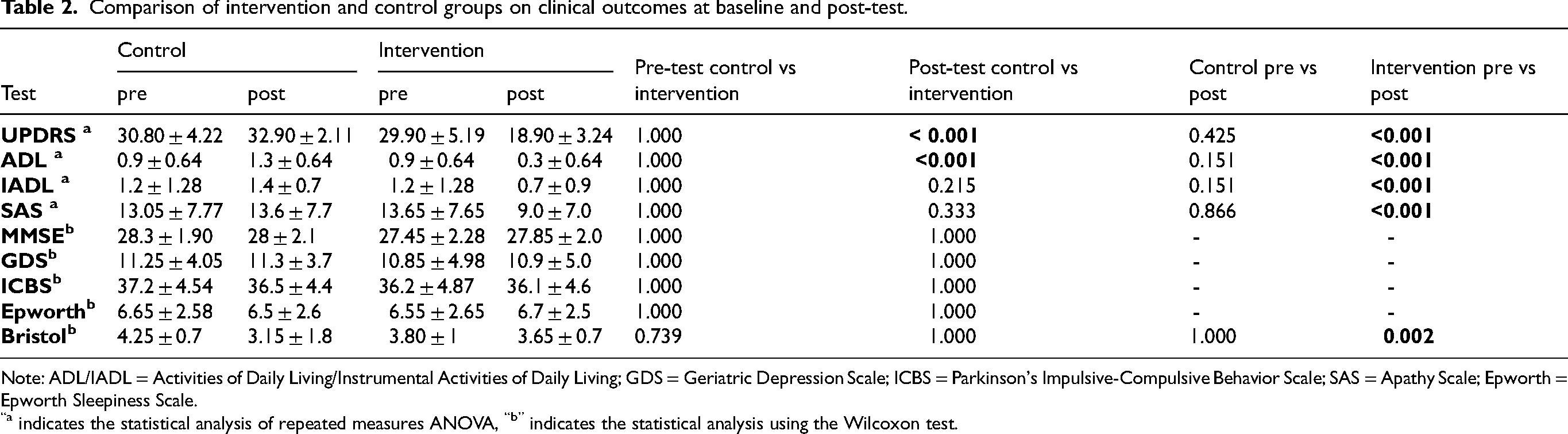
Note: ADL/IADL = Activities of Daily Living/Instrumental Activities of Daily Living; GDS = Geriatric Depression Scale; ICBS = Parkinson's Impulsive-Compulsive Behavior Scale; SAS = Apathy Scale; Epworth = Epworth Sleepiness Scale.
“a indicates the statistical analysis of repeated measures ANOVA, “b” indicates the statistical analysis using the Wilcoxon test.
Spearman correlation analysis was used to assess associations between post-intervention clinical and functional variables in PD patients and their inflammatory, glycotoxic, toxic, and vascular phenotypes. The results showed significant correlations between the inflammatory phenotype and specific clinical parameters (Table 3) (Figure 1).
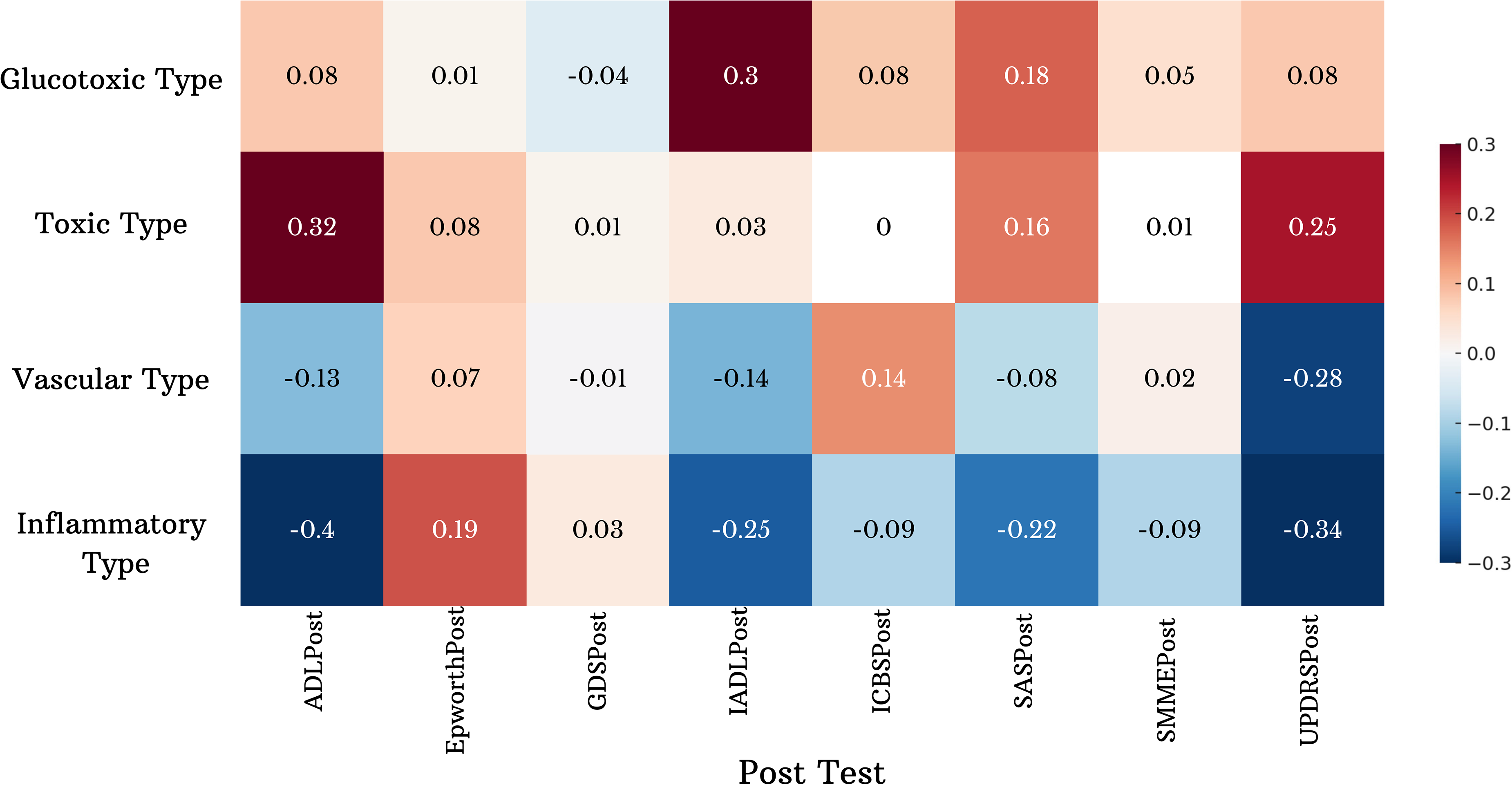
Heat map of correlation between clinical scales and biotypes.
Correlations between biotype subgroups and clinical scales.
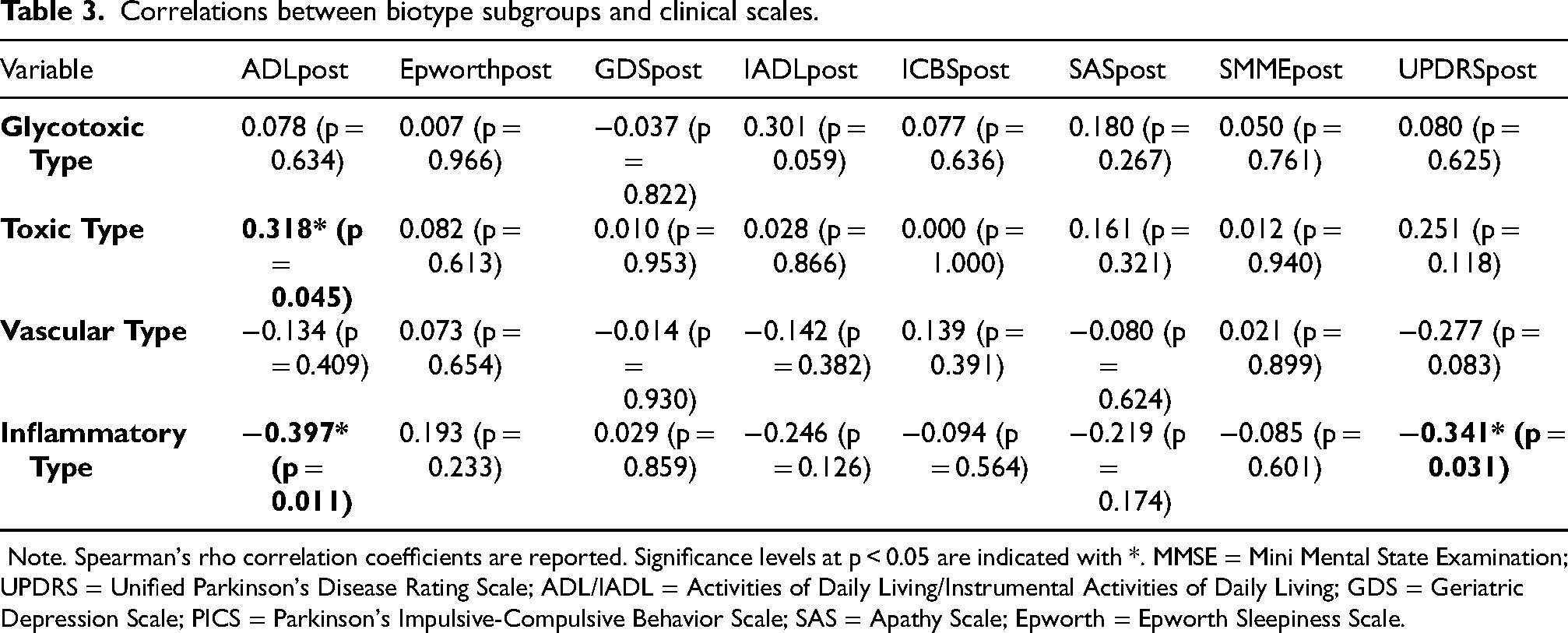
Note. Spearman's rho correlation coefficients are reported. Significance levels at p < 0.05 are indicated with *. MMSE = Mini Mental State Examination; UPDRS = Unified Parkinson's Disease Rating Scale; ADL/IADL = Activities of Daily Living/Instrumental Activities of Daily Living; GDS = Geriatric Depression Scale; PICS = Parkinson's Impulsive-Compulsive Behavior Scale; SAS = Apathy Scale; Epworth = Epworth Sleepiness Scale.
The inflammatory type showed a significant negative correlation with UPDRSpost (r = −0.341, p = 0.031). This finding suggests that individuals with the inflammatory phenotype showed more pronounced improvement in motor symptoms after intervention. Similarly, the inflammatory type showed a significant negative correlation with ADLpost scores (r = −0.397, p = 0.011). This indicates that individuals in this group were able to maintain greater independence in their daily living activities.
A positive and significant relationship was found between the toxic phenotype and ADLpost (r = 0.318, p = 0.045), suggesting that the toxic phenotype may have a certain effect on daily living functionality. However, the direction and clinical significance of this relationship should be carefully evaluated.
Conclusions
In this study, Ketoflex 12/3 protocol, was added to standard treatment in PD, resulting in clinically significant improvements in both motor and non-motor symptoms.5,30,31
Current evidence indicates that combining ketogenic principles with time-restricted eating may support neurodegenerative conditions by enhancing mitochondrial energy production and reducing inflammatory stress pathways. 32 Plant-rich ketogenic approaches, unlike the classic ketogenic diet, may offer advantages in modulating systemic inflammation. In this context, the Ketoflex 12/3 protocol used in our study presents a multidimensional effect profile consistent with current biological evidence.
The Ketoflex 12/3 protocol aims for metabolic flexibility through an intermittent fasting approach. Ketogenic diet applications in PD have been conducted in the literature mostly through animal-based models. For example, Phillips et al. (2018) reported that the animal-based classic ketogenic diet improved UPDRS-III scores but had limitations in terms of gastrointestinal tolerability and long-term sustainability. 33 However, systematic interventions combining intermittent fasting and a plant-rich ketogenic diet are quite limited. In this regard, the study is one of the first clinically evaluated examples of the Ketoflex 12/3 protocol in PD.
In the literature, ketogenic diet applications are often animal-based, and studies evaluating them in conjunction with intermittent fasting remain limited. While Phillips et al. demonstrated motor benefits with a classical, animal-based ketogenic diet, tolerability issues restricted its clinical applicability. 33 Similarly, Krikorian et al. reported cognitive improvements with a ketosis-inducing dietary pattern, yet their approach relied on more restrictive macronutrient targets. In contrast, the broader non-motor improvements and high tolerability observed in our individualized, plant-rich, time-restricted intervention suggest that metabolically oriented dietary strategies can be both clinically meaningful and more sustainable for people with Parkinson's disease. 34
The Ketoflex 12/3 protocol may differ from other nutritional interventions in three main ways: it comprehensively addresses the fundamental mechanisms of neurodegeneration by providing metabolic flexibility; it suppresses inflammation with its plant-rich and non-glycotoxic composition; and it supports mitochondrial function with its intermittent fasting component. 17 This multidimensional structure may distinguish the protocol from approaches such as the current Mediterranean diet or the classic ketogenic diet.
The outcome measures analyzed below were selected to capture symptom domains that are among the most prevalent and clinically impactful in Parkinson's disease, and that may plausibly be influenced by dietary and metabolic factors.
Statistically significant improvement was observed in the UPDRS-III Part 3 scores, which assess motor performance, in the intervention group (p < 0.05). The neuroprotective effects of the ketogenic diet are explained by mechanisms such as the use of ketone bodies in energy metabolism, support for mitochondrial function, and reduction of oxidative stress. Krikorian et al. (2019) reported that ketogenic nutrition corrected metabolic imbalance in motor circuits in PD. 34 Similarly, Norwitz et al. (2020) suggested that ketone use may protect dopaminergic functions in the substantia nigra. 35 These mechanisms explain the biological basis of the motor improvement observed in our study.
Apathy, which is common in PD, has been linked not only to mesocortical dopaminergic degeneration but also to metabolic and inflammatory pathways. The modest improvements observed in our study are consistent with the potential influence of nutritional and fasting-based interventions on these pathways, warranting further mechanistic investigation. The fiber- and polyphenol-rich Ketoflex 12/3 protocol may alleviate systemic inflammation by restoring microbial balance and enhance motivational circuit function through ketone-supported mitochondrial metabolism. Taken together, these mechanisms suggest that the observed reduction in apathy may reflect not only dopaminergic modulation but also the broader systemic effects of a comprehensive nutritional model36–41.
According to SAS results, apathy levels significantly decreased in the intervention group (p < 0.05). The ketogenic diet's ability to suppress neuroinflammation and elevate BDNF levels may modulate neurochemical pathways involved in motivation, such as those underlying apathy. Apathy is known to be particularly sensitive to dopamine levels within mesocorticolimbic circuits, notably the connections among the ventral tegmental area (VTA), nucleus accumbens, and prefrontal cortex. Ketone bodies stabilize cellular energy balance by increasing ATP production in dopaminergic neurons, while also supporting dopamine production and release by reducing oxidative stress. 42 In addition, the ketogenic diet has been shown to increase BDNF expression in hippocampal and cortical regions. 43 BDNF plays a critical role in maintaining the plasticity of dopaminergic synapses and circuit integrity between the VTA and prefrontal cortex. This neurotrophic support may contribute to the reduction of apathy symptoms by facilitating the reorganization of motivational circuits. Furthermore, suppressing inflammation and reducing microglial activity may limit the inhibitory effect of proinflammatory cytokines (IL-6, TNF-α) on motivational centers. 44 All these mechanisms provide important clues in explaining the multi-level effect of Ketoflex 12/3 protocol on the biological infrastructure of apathy. Mattson (2019) showed that time-restricted feeding supports prefrontal cortex functions and positively affects motivational processes by increasing BDNF levels 45 . In this context, the Ketoflex 12/3 protocol approach can be considered an effective intervention tool not only at the metabolic level but also at the behavioral level.46,47
Significant improvement was also observed in the intervention group on ADL and IADL scales assessing functional independence (p < 0.05). This positive effect on daily living activities may be related not only to improvement in motor skills but also to the regulation of systemic energy use and improved neuromuscular coordination. The metabolic flexibility provided by the ketogenic diet through the use of ketones instead of glucose in energy metabolism increases efficiency in nerve-muscle transmission and supports muscle performance. 43 It is known that intermittent fasting also contributes positively to muscle plasticity and neuromuscular integrity via the GH/IGF-1 axis 45 . These biological effects may have contributed to strengthening neuromuscular coordination, which is critical for daily life activities such as fine motor skills, balance, and agility, beyond motor functions. Furthermore, there is evidence that microbiota modulation via the gut-brain axis may also affect cortical motor planning and execution processes. 48 Therefore, the improvement in ADL and IADL scores demonstrates that the Ketoflex 12/3 protocol exerts a multidimensional effect not only on the central motor system but also through the peripheral energy system and microbial interactions. 49
Significant improvement was also observed in gastrointestinal function as assessed by the Bristol Scale (p < 0.05). Although both Type 3 and 4 fall within the clinically normal range, the slight improvement within this range suggests enhanced gastrointestinal regularity and reduced constipation tendency. The positive effect of a fiber-rich plant-based ketogenic diet on intestinal motility may explain this result. Moreover, prebiotic components that promote gut microbiota balance, together with the digestive rest afforded by intermittent fasting, may have contributed to the improvement in constipation. 50
The onset of gastrointestinal symptoms before motor symptoms in PD highlights the importance of the gut-brain axis in its pathogenesis. 51 Central inflammation can be initiated through vagal signaling along this axis. 52 The fiber- and polyphenol-rich ketogenic diet, combined with the metabolic rest afforded by intermittent fasting, may synergistically suppress systemic inflammation by modulating gut microbiota. 53 Concurrently, ketone-induced mitochondrial biogenesis and reduced oxidative stress may help restore cellular function in the substantia nigra. 54 Together, these effects enable targeting both symptoms and underlying disease mechanisms.
The Ketoflex 12/3 protocol offers a plant-rich structure that is rich in fiber, contains fermentable prebiotic components, and is free from inflammatory foods. Fiber-rich foods stabilize the composition of the microbiota by increasing the production of short-chain fatty acids that support the intestinal mucosa. The increase in butyrate production is particularly important in providing energy to intestinal epithelial cells and suppressing inflammation. Additionally, intermittent fasting may help restore peristaltic rhythm by allowing periodic rest of the digestive system. The literature shows that time-restricted feeding increases microbiota diversity, reduces intestinal permeability, and modulates vagal tone. These effects contribute to suppressing central neuroinflammation by reducing peripheral inflammatory load45,55–57. Therefore, the beneficial effect of Ketoflex 12/3 protocol on the Bristol score is not limited to the gastrointestinal system; it may also be a systemic regulatory factor underlying improvements in UPDRS-III, ADL, and SAS scores.
Individuals were classified according to inflammatory, glycotoxic, toxic, and vascular subtypes. According to the correlation results obtained, the inflammatory type stood out as the group that responded better to the intervention. Significant negative correlations (p < 0.05) were observed between the inflammatory subtype and UPDRSpost and ADLpost, indicating greater improvement in motor function and activities of daily living. Similar trends were observed in the glycototoxic and toxic subtypes, but these did not reach statistical significance. These trends may indicate effects that could reach statistical significance in studies with larger subgroup sample sizes. Findings emphasize the importance of considering biomarker-based subtype differentiation when creating individualized protocols.
The inflammatory subtype characterizes individuals with elevated levels of proinflammatory markers such as systemic C-reactive protein, interleukin-6 (IL-6), and TNF-α, impaired mitochondrial function, and increased oxidative stress load. In this profile, neuronal damage extends beyond dopaminergic loss, involving glial activation, cytokine-driven synaptic dysfunction, and gut dysbiosis. The Ketoflex 12/3 protocol exerts anti-inflammatory effects through ketone-mediated NF-κB inhibition and microbiota restoration via plant-based polyphenols and fiber. In inflammatory type, these mechanisms may have enhanced clinical outcomes by reducing systemic inflammation, improving vagal tone, and boosting mitochondrial metabolism. These effects help explain the biological basis underlying improvements in functional indicators such as UPDRS-III and ADL. The literature reports that anti-inflammatory diets are particularly effective in individuals with high CRP and IL-6 levels58–60.
The marked improvements seen in the inflammatory subgroup reinforce the connection between systemic inflammation and neurological symptoms. In individuals with high levels of proinflammatory cytokines, the effect of ketogenic and anti-inflammatory components may become more pronounced. This indicates that designing nutritional interventions based on “biotypes” may increase their effectiveness. Future studies comparing inflammatory markers may provide biochemical support for these observations.
In the study, no significant changes were observed in areas such as the MMSE, GDS, ICBS, and Epworth Scale. This can be explained by the systemic biological mechanisms targeted by the Ketoflex 12/3 protocol. Symptoms such as cognitive function and daytime sleepiness are more closely related to cortical or cholinergic degeneration in PD. Moreover, although the gut–brain axis primarily influences motivation, energy, and motor systems, cortical cognitive measures and scales like the Epworth are less affected by it.61,62 Therefore, the fact that these symptoms are not affected by the protocol biologically strengthens the results of the study.
These findings once again demonstrate that PD has a systemic and multidimensional pathophysiology. The increasingly recognized brain–gut axis underscores the contribution of gastrointestinal symptoms to PD pathogenesis, as gut microbiota disturbances can provoke central neuroinflammation through the vagus nerve. 63
The ketogenic diet's stimulation of mitochondrial biogenesis addresses mitochondrial dysfunction. The ketone bodies provided by the Ketoflex 12/3 protocol support energy production in dopaminergic neurons while reducing the accumulation of reactive oxygen species and limiting oxidative damage. 64 This approach, when combined with intermittent fasting, activates autophagy mechanisms, supporting cellular cleanup processes and providing systemic protection against neurodegeneration. 65
In summary, this randomized controlled trial provides early evidence that a personalized, plant-rich, time-restricted nutritional intervention may offer meaningful improvements in motor and selected non-motor symptoms in Parkinson's disease. Beyond symptomatic effects, the study contributes novel insight by demonstrating the feasibility of applying a structured metabolic approach within distinct patient biotypes—an area not previously examined in PD. These findings highlight nutrition as a promising complementary strategy alongside conventional care. As a real-world dietary intervention, several factors could not be fully controlled. Nutritional and lifestyle information was collected through structured clinical interviews rather than validated questionnaires, which may have introduced reporting bias. Adherence relied largely on participants’ self-report, meal photographs were not consistently provided, and all meals were self-prepared rather than standardized by the study team. Because this protocol was not designed as a weight-loss intervention, no caloric restriction was prescribed, and participants were encouraged to avoid perceiving the intervention as a diet aimed at reducing body weight. Although formal weight measurements were not collected, participants consistently reported no concerning or unintended weight loss during follow-up. We acknowledge these as methodological limitations. A primary limitation of this study is the lack of mechanistic biomarker validation, as no biological samples were collected to directly assess proposed pathways such as ketogenesis, inflammatory modulation, insulin resistance, or gut microbiota changes. Future studies could benefit from incorporating more detailed gastrointestinal assessments to further elucidate diet-related effects on non-motor symptoms. Together with recent reviews and randomized trials,66–70 our results contribute to a growing body of evidence supporting the evaluation of dietary interventions in Parkinson's disease, highlighting the need for future multi-center studies with larger samples and mechanistic evaluations to assess their potential integration into routine clinical practice.
Footnotes
Funding
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors. All costs were covered by the authors, and the dietary counseling sessions were provided free of charge.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
The data supporting the findings of this study are available on request from the corresponding author. The data are not publicly available due to privacy or ethical restrictions.
Consent statement
All participants provided written informed consent prior to enrollment. Ethical approval was granted by the Istanbul Medipol University Non-Interventional Clinical Research Ethics Committee (Approval No: E-10840098-202.3.02-1820). All procedures adhered to the Declaration of Helsinki.
