Abstract
Background
Proteomic studies have identified cerebrospinal fluid (CSF) DOPA decarboxylase (DDC) as a promising biomarker candidate for Parkinson's disease (PD). The aim of this study was to develop an immunoassay for CSF DDC quantification and gain further insight into its potential as a biomarker for PD.
Methods
We validated our DDC immunoassay by quantifying CSF DDC levels in the Parkinson's Progression Markers Initiative cohort, including healthy controls (n = 29), dopaminergic drug-naïve PD patients (n = 27), and patients with scans without evidence for dopaminergic deficit (SWEDD) (n = 18).
Results
Our DDC assay detected elevated levels in CSF from dopaminergic drug-naïve PD patients and discriminated them against SWEDD patients and controls with high sensitivity and specificity. There was an inverse correlation between DDC levels and ioflupane-[123I]-single-photon emission computed tomography-based dopamine transporter (DaT-SPECT) striatal binding ratios (SBRs) from the putamen and caudate nucleus. CSF DDC levels demonstrated prognostic potential for Movement Disorder Society Unified Parkinson's Disease Rating Scale total score change five to eight years post-diagnosis. DDC levels were further increased at the three-year follow-up visit in PD patients and positively correlated with the L-DOPA equivalent daily dose. There was a strong correlation between the relative CSF DDC levels determined by a proprietary immune-based proximity extension assay and absolute levels determined with our assay.
Conclusions
Our assay provided further insight into the potential of CSF DDC as a diagnostic and prognostic biomarker for PD. The unchanged levels in SWEDD patients and inverse correlation with DaT-SPECT SBRs suggest that CSF DDC levels are connected to dopaminergic deficit.
Plain language summary title
Advancing Parkinson's disease diagnosis and prognosis: a novel assay for DOPA decarboxylase in cerebrospinal fluid
Plain language summary
Novel biomarkers for Parkinson's disease (PD) are needed to enable early diagnosis and inform clinical trials. Previous studies have identified that a protein called DOPA decarboxylase (DDC) might be a useful indicator for PD when measured in the cerebrospinal fluid (CSF) surrounding the brain and spinal cord. Our study aimed to develop a simple and robust method to measure DDC levels in CSF, facilitating studies on DDC as a potential biomarker for PD. Our newly developed assay confirmed that PD patients who have not yet taken any PD medication had higher DDC levels in CSF compared to healthy controls. This was an important finding, since we also showed that DDC levels are influenced by medication alleviating PD motor symptoms. For the first time, we also showed that DDC is elevated compared to so-called SWEDD patients which are often misdiagnosed as PD but do not have a loss of brain cells producing dopamine, i.e., dopaminergic loss, that is characteristic for PD. Thus, DDC could be used for more accurate diagnosis of PD patients. We also compared DDC levels with imaging of dopamine transporter in the brain as a measure for dopaminergic loss and confirmed that higher CSF DDC levels were associated with higher dopaminergic loss. Finally, our data suggest that DDC levels at baseline could help to predict the severity of PD symptoms in the future, five to eight years later. Thus, the DDC biomarker could help design more effective clinical trials for therapies aiming to slow the worsening of motor symptoms over time. DDC levels measured with our new assay matched closely with results from another proprietary immune-based proximity extension assay. In conclusion, our new assay for measuring DDC levels in CSF shows promise for diagnosis of PD and predicting the worsening of PD symptoms over time.
Background
Parkinson's disease (PD) is the second most prevalent neurodegenerative disorder, and its incidence and public health burden continue to rise.1,2 The diagnosis of PD remains based on clinical assessment. Ancillary tests, such as imaging or genetic tests, are generally reserved for individuals with an atypical clinical presentation.3,4 Research is also being conducted on tissue and fluid biomarkers for PD, with α-synuclein seed amplification assays (SAAs) in cerebrospinal fluid (CSF) and skin samples demonstrating high performance in differentiating patients with PD from healthy controls (Ctrl) and detecting individuals in the prodromal phase.5–8 However, the timely diagnosis of PD remains challenging, highlighting the need to develop further supportive diagnostic biomarkers. 9 Moreover, striking heterogeneity has been observed among patients with PD with regards to their disease etiology, clinical presentation, progression rate, and treatment response, emphasizing the significance of identifying prognostic biomarkers and implementing a precision medicine approach.3,9
DOPA decarboxylase (DDC) is implicated in dopamine biosynthesis.10,11 It has recently emerged as a potential biomarker for PD, demonstrating elevated levels in CSF of patients with PD, including those who are dopaminergic drug-naïve or in the prodromal phase.12–17 In addition, increased DDC levels have been detected in CSF from patients with atypical Parkinsonian syndromes, including dementia with Lewy bodies.13,15,16 Potential reasons for the elevation of CSF DDC have been investigated and an association with both dopaminergic treatment and dopamine transporter imaging have been reported.12,13,17 However, the interpretation of these findings is complicated by the interconnected nature of dopaminergic deficit, dopaminergic treatment, and disease severity. Therefore, further studies are needed to better understand the reasons for increased CSF DDC levels, its changes with disease progression, and its ability to serve as a prognostic biomarker.
Most previous publications have relied on a proprietary proximity extension assay (PEA) for DDC detection.12–18 A broadly accessible, sensitive, and quantitative immunoassay for CSF DDC assessment is needed to confirm the previous findings and accelerate research on DDC as a biofluidic biomarker.
Here, we describe the development and validation of a sensitive immunoassay for absolute DDC quantification in CSF using commercially available monoclonal antibodies. Using this assay, we quantified CSF DDC levels in healthy controls, patients with PD, and patients with scans without evidence for dopaminergic deficit (SWEDD) from the Parkinson's Progression Markers Initiative (PPMI) cohort at baseline and three-year follow-up to gain further insight into the potential of DDC as a diagnostic and prognostic biomarker for PD.
Methods
Study participants
Assay validation cohort
Assay validation was performed in a cohort including CSF samples from 10 patients with PD and 10 Ctrl. Their demographic characteristics are presented in Table S1. The samples were obtained from PrecisionMed, LLC (Carlsbad, CA, USA). CSF samples were diluted 1:10, and all conditions were tested in duplicates.
Parkinson's progression markers initiative (PPMI) cohort
CSF samples from individuals from the PPMI cohort were included in the study for DDC level quantification. 19 Baseline and three-year follow-up CSF samples from 27 dopaminergic drug-naïve patients with PD, 18 dopaminergic drug-naïve patients with SWEDD and, 29 Ctrl were analyzed. Three-year follow-up CSF samples from the same PD and Ctrl were analyzed. It is noteworthy that 26 out of 27 PD patients were under dopaminergic treatment at this time point (Figure S7c).
Several other measures and characteristics that were available for the study participants from the PPMI cohort were also used in the study. Ioflupane [123I]-single-photon emission computed tomography (SPECT)-based dopamine transporter imaging (DaT-SPECT) striatal binding ratios (SBRs) from the study participants were available at baseline. 4 The L-DOPA equivalent daily dose (LEDD) was considered at the three-year follow-up visit.20–22 Yearly Movement Disorder Society Unified Parkinson's Disease Rating Scale (MDS-UPDRS) score changes for years 1 through 8 were also incorporated in the study. 23 For the MDS-UPDRS score changes, in addition to the total score OFF, the subscores for Part I, Non-Motor Aspects of Experiences of Daily Living; Part II, Motor Aspects of Experiences of Daily Living; and Part III OFF, Motor Examination were also considered. A previous analysis of MDS-UPDRS subscores in the PPMI cohort revealed a continual measured progression of motor and non-motor symptoms during the initial five years following PD diagnosis. 24
Identification and screening of antibodies for a DDC immunoassay on the mesoscale discovery (MSD) platform
We searched the literature for commercially available anti-DDC antibodies. Seven monoclonal antibodies targeting different DDC epitopes and one polyclonal antibody raised against the full-length DDC protein were selected for further analysis (Table S2). The antibody buffer was exchanged to phosphate-buffered saline (PBS) in all instances using a desalting column (Thermo Fisher Scientific, 87764). Three recombinant DDC proteins from different expression systems (bacterial, insect, and human cells) were tested as calibrators (Table S3). The antibodies and calibrators were quality controlled using NanoDrop and SDS-PAGE. Next, 70 µg and 15 µg of antibodies were used for biotin (Thermo Fisher Scientific, 90407) and for SULFO-TAG labeling (Meso Scale Discovery, R91AO), respectively. Calibrators were tested at 1 ng/mL. CSF samples diluted 1:5 and 1:20, plasma samples diluted 1:10 and 1:40, eight antibodies and, three standard proteins were analyzed in LowCross-Buffer MILD (Candor; 101 500). MSD GOLD 96-well Small Spot Streptavidin SECTOR plates were used (Meso Scale Discovery, L45SA). A 3.5 µg/mL capture antibody was incubated for 1 h at room temperature and 700 rpm in Diluent 100 (Meso Scale Discovery, R50AA). The samples were then incubated for 2 h at room temperature and 700 rpm. Next, a 0.75 µg/mL detection antibody was added and incubated for 1 h at room temperature and 700 rpm in LowCross-Buffer MILD. Finally, MSD GOLD Read Buffer B (Meso Scale Discovery, R60AM) was added. The signal-to-noise (S/N) ratio was used to screen for an optimal combination of a capture antibody, detection antibody, and calibrator protein.
Development of an ultrasensitive single molecule counting (SMC) immunoassay
The antibodies, samples, and calibrator were diluted in LowCross Buffer® MILD (Candor; 101 500). Anti-human DDC mouse monoclonal 413911 antibody (R&D Systems; MAB3564) at 17.5 µg of antibody/mg of beads (Merck; 03-0077-02) was used as the capture antibody. Recombinant human DDC (rhDDC) protein (R&D Systems; 3564-DC-010) was employed as a calibrator. Capture antibody-coated beads (10 mg of beads per mL) were mixed at 1250 rpm until all beads were resuspended and diluted 200-fold. Next, 100 µL of diluted beads and 100 µL of samples (human CSF samples diluted 1:10) or calibrators were added to each well of Axygen Assay Plates (Axygen; P-96-450V-C), which were incubated for 2 h at 25 °C with shaking (Jitterbug; setting Mix 5). The plates were washed with Erenna System buffer (Merck, 02-0111-03) using a Tecan Hydroflex washer. After aspiration, 20 μL of a filtered fluorescently labeled (Merck; 03-0076-02) anti-human DDC mouse monoclonal A17096A antibody (BioLegend; 869501) at 937.5 ng/mL, which served as the detection antibody, were added to each well and incubated for 1 h at 25°C with shaking (Jitterburg, setting Mix 5). The plates were washed as described previously. The beads were transferred to a fresh Axygen 96-well plate, into 200 μL of Erenna System buffer. After final aspiration, 9.5 μL of Elution Buffer B (Merck; 02-0297-00) were added to each well of the assay plate, and the plates were incubated for 20 min at 25°C with shaking (Jitterburg, setting Mix 5). The eluate was magnetically separated from the beads, transferred into a 384-well plate (Thermo Fisher Scientific; 264573), and neutralized immediately with 9.5 µL/well of Buffer D (Merck, 02-0368-00). After heat-sealing (4titude; 4ti-0531), the plate was analyzed using the Singulex Erenna instrument.
In vitro validation of the SMC DDC assay
The newly developed assay was analytically validated for its sensitivity, parallelism, spike-in recovery, and dilution linearity. In all instances, the validation criteria were ±30% for recovery and coefficient of variability (CV). To determine the assay's limits of quantification, the rhDDC protein was tested in concentrations of 0.316–10,000 pg/mL. A five-parameter logistic regression, weight by 1/Y2 was performed, and the lower limit of quantitation (LLOQ) and upper limit of quantitation (ULOQ) were determined (corresponding to the lowest and highest concentration of the calibration standards that fulfilled the validation criteria). To assess the parallelism of the assay, samples pooled from 5–6 individuals were tested. The spike-in recovery of rhDDC in human CSF was tested by measuring the recovered DDC concentrations for 0, 10, 100, and 1000 pg/mL DDC calibrator protein spiked into biological matrix. Both the mean of four samples per spike concentration and 75% of individual samples per spike concentration had to match the recovery validation criterion. The dilution linearity of rhDDC in human CSF was assessed for concentrations of 0.06–2000 pg/mL.
DDC assay conditions for the PPMI cohort (Project 274)
All samples from the PPMI cohort were run on the same day on four plates with four quality control samples (CSF pools) on each plate. Because the CV of the quality control samples was low (<6%) (Figure S1), no data correction was applied across the plates. Finally, the correlation of DDC levels quantified with the newly developed SMC assay and DDC levels determined with the proteomic Olink Explore assay (cardiometabolic panel) was explored in a subset of the samples.
Analysis of the prognostic potential of DDC CSF levels in patients with PD
Baseline DDC levels in CSF from dopaminergic drug-naïve patients with PD were split at the median into low-DDC (13 patients) and high-DDC (14 patients) groups. Subsequently, changes in the MDS-UPDRS scores in the low-DDC and high-DDC groups over time as well as correlations between DDC levels and MDS-UPDRS scores were evaluated. A cutoff of 8 years ensured a minimum of 14 patients with available MDS-UPDRS scores and baseline DDC levels in both the low-DDC and high-DDC groups.
Statistical analysis
Numerical data are presented as mean ± standard deviation (SD) or median with 95% confidence interval (CI). A receiver operating characteristic (ROC) curve was plotted, and the area under the curve (AUC) and the sensitivity and specificity of the DDC assay in the validation PPMI cohorts were calculated. For statistical group comparisons, the methods used are thoroughly described in the corresponding figure legends. Spearman's rank correlation was used to investigate the correlation between two parameters.
Results
Development and validation of an ultrasensitive SMC assay for DDC quantification in CSF
Initially, we screened anti-DDC antibodies in an MSD assay format. However, for CSF samples, all combinations of potential capture and detection antibodies with acceptable signal to noise (S/N > 2) ratios included at least one polyclonal antibody (Figure S2). Polyclonal antibodies are a mixture of antibodies that could recognize multiple epitopes on the same antigen that can lead to cross-reactivity with other proteins, causing non-specific binding. Moreover, they can show variability between production batches, affecting the consistency and reliability of the assay results over time. Therefore, we selected the most promising combination of monoclonal antibodies (capture Ab = clone 413911; detection Ab = clone A17096A; Table S2) and calibrator (R&D Systems; Table S3) and set out to develop a novel SMC assay.
Analytical validation of the SMC DDC assay using rhDDC protein revealed an estimated LLOQ of 1 pg/mL and estimated ULOQ of 1000 pg/mL (Figure 1(a)). For human CSF samples, parallelism was observed for 1:4–1:64 dilutions (Figure 1(b)). In the spike-in recovery analysis, the high and medium concentration of spiked DDC fell in the acceptance range (Figure 1(c)). For the low concentration of spiked DDC (10 pg/mL), the mean of four samples met the ±30% acceptance criterion, but only one out of the four individual samples did (Figure 1(c)). The dilution linearity of rhDDC in human CSF met the acceptance criteria (Figure 1(d)).
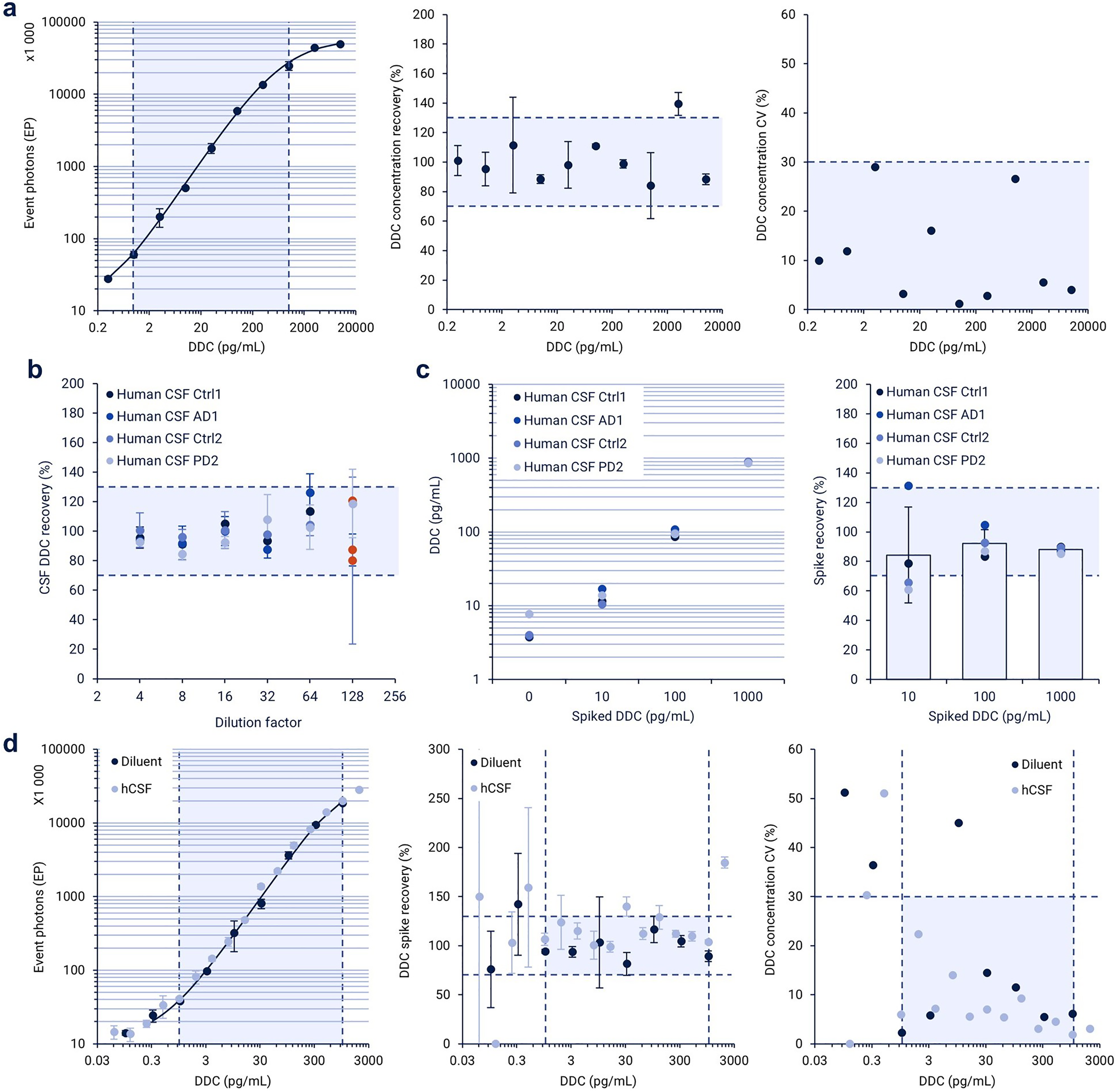
In vitro validation of the SMC DDC assay. Limits of quantification (
We used our validated SMC assay to quantify DDC in CSF samples from a small pilot cohort of patients with PD (n = 10) and Ctrl (n = 10) (Table S1). DDC levels were significantly higher in patients with PD than in the control group (p = 0.0001) (Figure S3).
The new DDC SMC assay has high discriminatory power for dopaminergic drug-naïve PD patients
Next, CSF DDC levels were quantified in a subset of the PPMI cohort, including Ctrl (n = 29) and dopaminergic drug-naïve patients with PD (n = 27) or with SWEDD (n = 18). The demographic baseline characteristics of the participants are presented in Table 1.
Demographic characteristics of the tested PPMI cohort at baseline.

Ctrl: healthy control; PD: Parkinson's disease; SWEDD: scans without evidence for dopaminergic deficit. aKruskal-Wallis test; bChi-squared test.
At baseline, CSF DDC levels were higher in dopaminergic drug-naïve patients with PD than in Ctrl (1.62-fold; p < 0.0001) and patients with SWEDD (1.64-fold; p < 0.0001). No significant difference was detected between the CSF DDC levels of Ctrl and patients with SWEDD (p = 0.5040) (Figure 2(a)). There were no significant age differences among the groups (Figure S4a) as well as no significant correlation between CSF DDC levels and age (Figure S4b). No significant differences in DDC levels were observed between males and females in each group (Figure S4c). There was no significant correlation between CSF DDC levels and hemoglobin contamination levels (Figure S4d; PPMI project 134). Because DDC is involved in several metabolic pathways, 25 we looked at but did not observe significant correlation between CSF DDC levels and body mass index (Figure S4e).
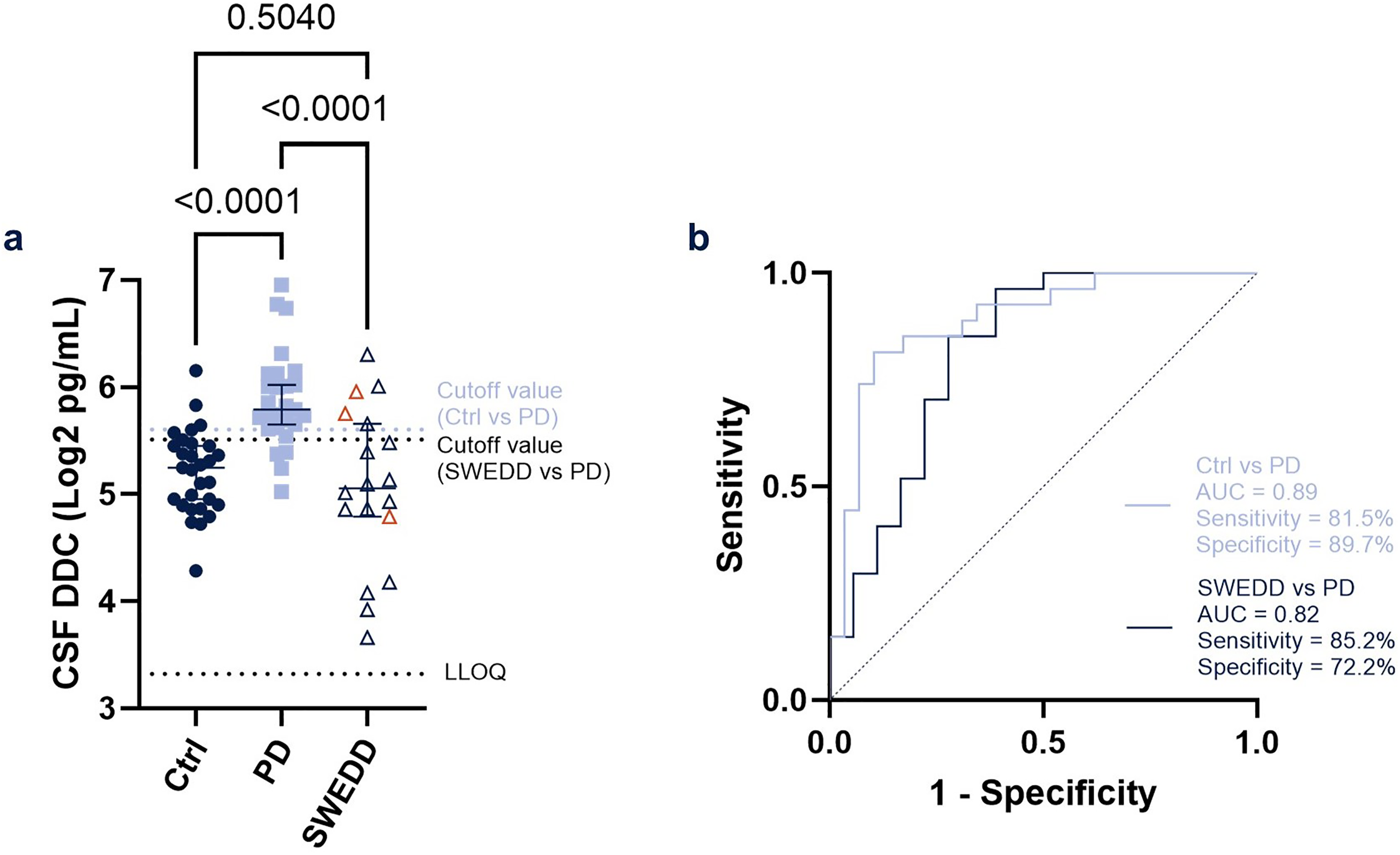
CSF DDC levels of dopaminergic drug-naïve patients with PD and SWEDD and Ctrl.
The discriminatory performance of DDC for dopaminergic drug-naïve patients with PD was assessed using a ROC curve analysis. It revealed an AUC of 0.89 (p < 0.0001) and 81.5% sensitivity and 89.7% specificity to discriminate dopaminergic drug-naïve patients with PD from Ctrl and an AUC of 0.82 (p = 0.0004) and 85.2% sensitivity and 72.2% specificity to discriminate dopaminergic drug-naïve patients with PD from patients with SWEDD (Figure 2(b)).
In addition, there was a strong correlation between CSF DDC levels determined with a proprietary PEA in two different studies and the SMC assay developed in the current study (r = 0.76; p = 0.0002 and r = 0.71; p < 0.0001) (Figure S5).
CSF DDC levels in all patients correlate with DaT-SPECT SBRs
At baseline, CSF DDC levels showed a significant inverse correlation with the DaT-SPECT SBRs of the contralateral caudate and putamen (Spearman r = −0.51; p < 0.0001 and r = −0.54; p < 0.0001, respectively) when analyzing all dopaminergic drug-naïve disease groups (Figure 3). However, no significant correlation was observed when analyzing only the PD group (data not shown).
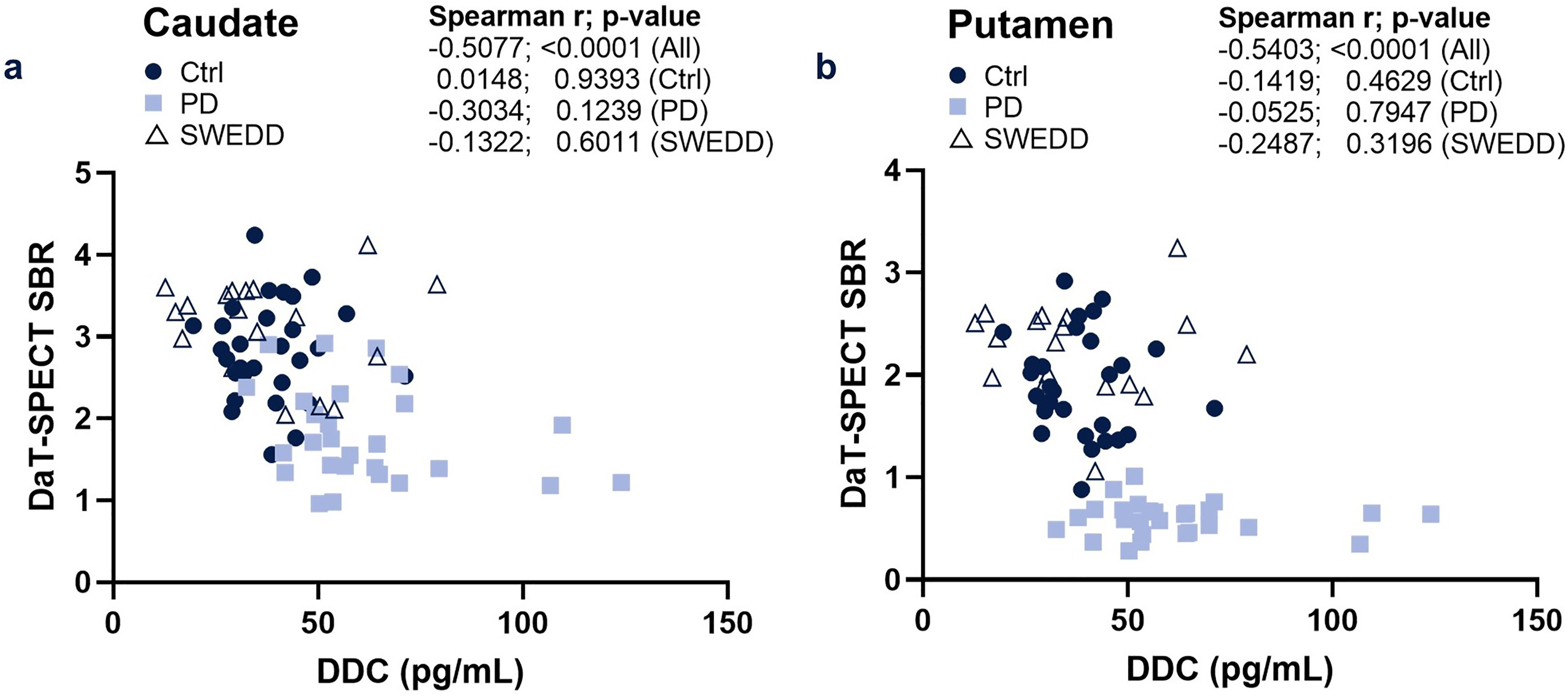
Correlation of CSF DDC levels with DaT-SPECT SBRs from the contralateral caudate (
Prognostic potential of CSF DDC levels in dopaminergic drug-naïve PD patients
To gain insight into the potential of CSF DDC levels as a prognostic biomarker in PD patients, MDS-UPDRS total score OFF changes over time were compared in patients with low and high baseline DDC levels after splitting at the median. The increase of MDS-UPDRS total scores OFF over time was more pronounced in PD patients with high baseline CSF DDC levels (Figure 4(a)). In addition, the correlation of baseline CSF DDC levels with MDS-UPDRS total score OFF changes for year 1 through year 8 was evaluated. A significant correlation was first detected at year 5 and persisted through year 8, suggesting DDC may have promise as a prognostic biomarker (Figure 4(b)).
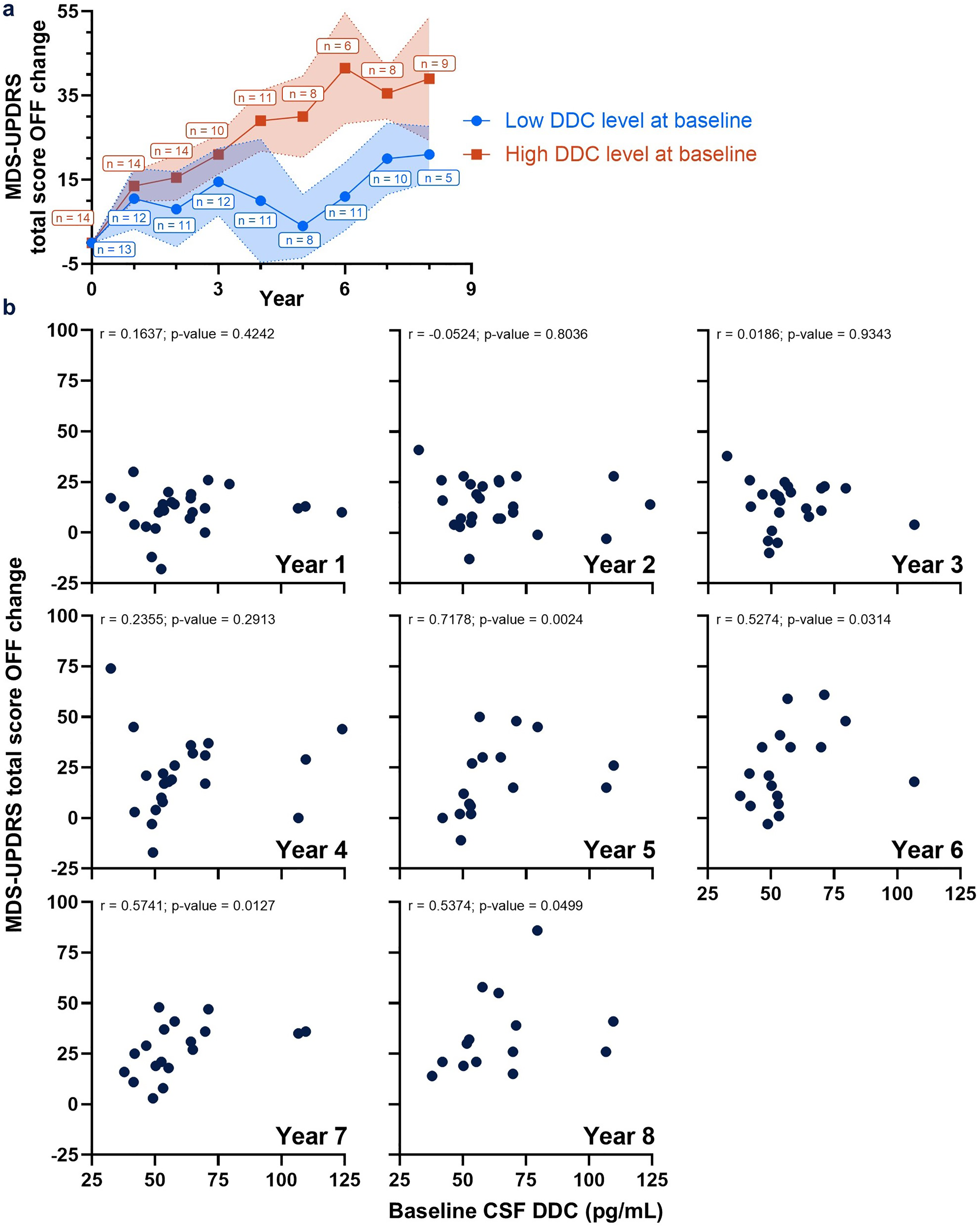
Prognostic potential of CSF DDC levels in dopaminergic drug-naïve PD patients.
The correlation of baseline CSF DDC levels with MDS-UPDRS part I, MDS-UPDRS part II and MDS-UPDRS part III OFF sub score changes at years 1 through 8 post diagnosis was also investigated. Baseline CSF DDC levels correlated significantly with MDS-UPDRS part II scores at years 5, 6, and 8 as well as with MDS-UPDRS part III scores OFF at years 5 through 7 (Figure S6).
Changes in CSF DDC levels with PD progression and their correlation with the LEDD
At the 3-year follow-up visit, the CSF DDC levels were 2.17-fold higher in patients with PD in comparison to Ctrl (p < 0.0001). While DDC levels in the Ctrl group did not change from baseline to the 3-year follow-up (p = 0.3304), DDC levels in the PD group increased 1.42-fold (p < 0.0001) (Figure S7a). In addition, at the 3-year follow-up, CSF DDC levels were significantly higher (1.57-fold) in males than in females in the PD group (p = 0.0145), while there was no significant sex difference in DDC levels among controls (Figure S7b).
However, LEDD levels were also significantly higher in males than in females (p = 0.0223) (Figure S7c). The relationship between CSF DDC levels and the LEDD was assessed at the 3-year follow-up, and a significant correlation was detected (Spearman r = 0.45, p = 0.0198) (Figure S7d). The correlation was stronger in females than in males (Figure S7d).
Discussion
In the current study, we developed and validated a novel ultrasensitive immunoassay for quantification of DDC levels in human CSF. The assay discriminated dopaminergic drug-naïve PD patients from Ctrl and patients with SWEDD with high sensitivity and specificity. At baseline, DDC levels in human CSF were inversely correlated with DaT-SPECT SBRs when all study participants were included for analysis. In addition, the study suggested that DDC has potential as a prognostic biomarker in patients with PD. With disease progression, CSF DDC levels increased further but were positively correlated with the LEDD.
Previous investigations have relied on proteomic technologies to determine DDC levels in human CSF.12–16 However, these approaches are currently limited to relative quantification and associated with high costs. Different proteomics technologies for detection of DDC in CSF have shown differences in diagnostic performance 14 highlighting that DDC detection reagents need to be optimized towards the most relevant DDC proteoforms in biofluids. Upon extensive screening of antibody combinations (Figure S2), we developed and validated a DDC immunoassay using highly sensitive SMC technology. 26 Our assay is based on commercially available antibodies and protein calibrator, rendering it accessible to other researchers in the field. Its AUC in a ROC analysis (AUC = 0.89) and its sensitivity and specificity for discriminating dopaminergic drug-naïve PD patients from Ctrl were comparable to those described previously for drug-naïve PD and Lewy body disorders using a proprietary PEA (AUC ranging from 0.79 to 0.92).12,14,16 Indeed, our DDC SMC assay correlated moderately with PEA data for CSF samples (Figure S5). Notably, our DDC SMC assay could discriminate dopaminergic drug-naïve patients with PD not only from Ctrl but also from dopaminergic drug-naïve patients with SWEDD. To our knowledge, this is the first study showing that CSF DDC could be used for differential diagnosis of patients with PD versus SWEDD, which are commonly misdiagnosed based on a clinical assessment.27–29 While a commercially available assay for DDC quantification in a clinical setting is still needed, our assay provides a number of advantages over existing assays: (1) It relies exclusively on monoclonal antibodies, reducing lot to lot variability and preserving assay consistency and reproducibility; (2) The assay performance has been extensively validated (limits of quantification, parallelism, spike-in recovery and dilution linearity); (3) The assay reproduced previously published data on PD; (4) Because it relies solely on commercially available reagents, the assay is easily adoptable by the research community and can be applied to gain further insight into DDC.
DaT-SPECT imaging of the dopamine transporter can detect dopaminergic deficit in patient brain and facilitate the differential diagnosis of nigrostriatal neurodegenerative Parkinsonian syndromes and parkinsonism that is not caused by nigrostriatal neurodegeneration.4,30 In the current study, an inverse correlation was detected between CSF DDC levels and DaT-SPECT SBRs at baseline when analyzing all groups combined. These data, in parallel with unchanged CSF DDC levels in patients with SWEDD, support the hypothesis that the elevation of CSF DDC levels in PD patients is connected to dopaminergic deficit. However, no significant correlation was observed between CSF DDC levels and DaT-SPECT SBRs in the PD group only. This observation agrees with a previous study on dopaminergic treatment-naïve PD patients from two different cohorts. 17 It has been hypothesized that the elevation of CSF DDC may be the consequence of a compensatory reaction to dopaminergic cell loss.12–14,16,17 However, the spatial and temporal relationship between these two processes has not been elucidated. Further work is needed to study the relationship between CSF DDC level and dopaminergic imaging, especially within the most affected sub-region of the putamen 31 - the post-commissural putamen - and also between stage 2A and 2B patients within the Neuronal alpha-Synuclein Disease Integrated Staging System 32 (CSF SAA + without or with dopaminergic neuron dysfunction, respectively). Additionally, studies employing longitudinal DaT-SPECT and fluorodopa positron emission tomography as well as CSF sampling could help inform on the relationship between dopaminergic cell loss and DDC levels. Finally, it would be interesting to investigate the potential of CSF DDC for other dopaminergic deficiencies, including restless legs syndrome, depression, schizophrenia, and attention deficit hyperactivity disorder.33–36
CSF DDC levels in patients with PD were further elevated at the three-year follow-up visit. The reasons underlying the increase of DDC in CSF of patients with PD have not been definitively elucidated. Several potential mechanisms, including the result of neuronal loss, a compensatory reaction to degeneration of dopaminergic neurons, and a possible dopaminergic treatment effect, have been discussed.12,15,17 Our current study was not designed to elucidate mechanistic questions. To the best of our knowledge, no study has observed a direct increase of CSF DDC level in untreated PD patients over time compared to baseline. Only the use of a linear mixed effects model suggested a significant increase of CSF DDC over time in PD participants, but this was still confounded with LEDD. 14 Similarly, only one patient from our study remains dopaminergic drug-naïve at the three-year follow-up visit preventing unconfounded evaluation of CSF DDC longitudinally. Nevertheless, we observed a weak correlation between CSF DDC levels in PD patients three years post diagnosis and the LEDD. This finding aligns with other studies reporting a weak positive correlation or no significant correlation.12,13,17 In contrast to CSF, elevated DDC levels in plasma are strongly driven by dopaminergic treatment. 12 Since DDC levels in plasma are much higher than in CSF (approximately 150-fold using our assay; data not shown) and proteins cross the blood-CSF barrier to a small extent, 37 it is possible that DDC levels in CSF are at least partially influenced by increased levels in plasma. An interventional study with biofluid sampling before and after the onset of dopaminergic treatment could elucidate to what extent it influences DDC levels.
While at baseline there were no significant differences in CSF DDC levels between males and females in any of the groups in the current study, at three years post diagnosis there was a 1.56-fold increase of CSF DDC levels in male compared to female patients with PD (p = 0.0145). Even though the underlying mechanism has not been elucidated, sex differences in the dysfunction of the dopaminergic system in PD have been reported, with a predisposition to nigrostriatal derangement in males and mesolimbic system changes in females. 38 In our study, males had a higher LEDD than females which could be a confounding factor (Figure S7d).
Most interestingly, PD patients with high baseline CSF DDC levels were more likely to have a higher change in the MDS-UPDRS total score (OFF) with disease progression than PD patients with low baseline CSF DDC levels (Figure 4(a)). Baseline CSF DDC levels were correlated with changes in the MDS-UPDRS total score (OFF) at five to eight years post diagnosis. An analysis of MDS-UPDRS part II and III (OFF) revealed a correlation of CSF DDC levels with motor symptoms. These data suggest that DDC may have potential as a prognostic biomarker for the progression of motor symptoms in patients with PD. A recent publication by Sturchio et al. has also shown prognostic potential of CSF DDC levels to predict another clinical feature, the cognitive decline in PD patients, 39 evaluated by the Montreal Cognitive Assessment. 40 Unfortunately, only five patients from our study showed cognitive decline up to eight years after baseline using the same cognitive assessment, rendering confirmation analysis with our assay statistically impossible. However, it is noteworthy that 4 out of 5 patients showing cognitive decline are in the subgroup of PD patients with high level of DDC at baseline (Table S4). The sample size was relatively small in both studies, and further investigations in larger cohorts are needed to assess the usefulness of DDC for prognosis.
The current study focused on DDC levels in CSF, which is in direct contact with the central nervous system and thus the most proximal source of biomarkers for neurodegenerative diseases. However, more easily accessible biofluids like plasma are of high interest. Recent studies using a proprietary PEA for DDC showed that plasma DDC is not elevated in dopaminergic drug-naïve patients with PD.12,14,18 Although our DDC SMC assay correlated moderately with PEA data for CSF samples (Figure S5), it is unclear whether the same is true for plasma. Since it is possible that different assays detect different proteoforms of DDC, it would be highly interesting to test our new DDC SMC assay in plasma samples from drug-naïve patients with PD.
This study has several strengths. First, a new immunoassay for quantification of DDC that can be readily used by other researchers was validated and employed. Second, analysis of the well-characterized PPMI cohort enabled us to investigate the association of DDC levels in CSF with dopaminergic treatment, functional assessments, and DaT-SPECT imaging. Third, our study included for the first time patients with SWEDD.
However, we acknowledge that our study also has certain limitations. First, the analyzed cohort did not allow to distinguish between changes in CSF DDC levels caused by disease progression versus dopaminergic treatment. Second, the sample size of the investigated cohorts was relatively small, especially for addressing the potential prognostic value of CSF DDC. In the future, we are aiming to assess plasma and CSF DDC levels cross-sectionally and longitudinally in larger cohorts of drug-naïve prodromal individuals with and without dopaminergic deficit observed by imaging.
Conclusions
In conclusion, our study on CSF DDC using a novel SMC assay highlighted its potential as diagnostic biomarker for PD and as prognostic biomarker for progression of motor symptoms. Our findings that CSF DDC levels inversely correlate with DaT-SPECT SBRs and are unchanged in patients with SWEDD support the hypothesis that elevated CSF DDC levels are connected to dopaminergic deficit in the brain.
Supplemental Material
sj-docx-1-pkn-10.1177_1877718X261421740 - Supplemental material for A monoclonal antibody-based immunoassay reinforces DOPA decarboxylase in cerebrospinal fluid as a diagnostic biomarker for Parkinson's disease with potential prognostic value
Supplemental material, sj-docx-1-pkn-10.1177_1877718X261421740 for A monoclonal antibody-based immunoassay reinforces DOPA decarboxylase in cerebrospinal fluid as a diagnostic biomarker for Parkinson's disease with potential prognostic value by Hubert Aviolat, Jennifer Mollon, Simone Giaisi, Stefan Barghorn and Roland G Heym in Journal of Parkinson's Disease
Footnotes
Abbreviations
Acknowledgements
We thank the participants for their generous donation of samples. We thank Dr Zoya Marinova (ZMedBio, LLC) for manuscript preparation and Dr Samantha Hutten for her assistance. Data used in the preparation of this article were obtained on (2024-07-29) from the Parkinson's Progression Markers Initiative (PPMI) database (www.ppmi-info.org/access-data-specimens/download-data), RRID:SCR_006431. For up-to-date information on the study, visit ![]() .
.
Ethics approval and consent to participate
Details on sample collection were provided by the vendors PrecisionMed Inc. (Solana Beach, CA, USA). The PPMI study was approved by the Institutional Review Boards of each PPMI site. Informed written consent was obtained from all subjects at each site.
Authors’ contributions
HA and RGH conceptualized the study. HA designed, performed and analyzed experiments. HA and JEM performed statistical analysis. HA, JEM, SG, SB and RGH interpreted data. HA and RGH obtained the human sample cohort. HA and RGH wrote the final manuscript draft. All authors critically reviewed, provided feedback, and approved the final manuscript.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The design, study conduct, and financial support for this research were provided by AbbVie. AbbVie participated in the interpretation of data, review, and approval of the publication.
PPMI – a public-private partnership – is funded by the Michael J. Fox Foundation for Parkinson's Research and funding partners, including 4D Pharma, Abbvie, AcureX, Allergan, Amathus Therapeutics, Aligning Science Across Parkinson's, AskBio, Avid Radiopharmaceuticals, BIAL, BioArctic, Biogen, Biohaven, BioLegend, BlueRock Therapeutics, Bristol-Myers Squibb, Calico Labs, Capsida Biotherapeutics, Celgene, Cerevel Therapeutics, Coave Therapeutics, DaCapo Brainscience, Denali, Edmond J. Safra Foundation, Eli Lilly, Gain Therapeutics, GE HealthCare, Genentech, GSK, Golub Capital, Handl Therapeutics, Insitro, Jazz Pharmaceuticals, Johnson & Johnson Innovative Medicine, Lundbeck, Merck, Meso Scale Discovery, Mission Therapeutics, Neurocrine Biosciences, Neuron23, Neuropore, Pfizer, Piramal, Prevail Therapeutics, Roche, Sanofi, Servier, Sun Pharma Advanced Research Company, Takeda, Teva, UCB, Vanqua Bio, Verily, Voyager Therapeutics, the Weston Family Foundation and Yumanity Therapeutics.
Competing interests
HA, JM, SG, SB, & RGH are employees of AbbVie. SB and RGH are stockholders of AbbVie.
Availability of data and materials
All data generated and analyzed during the current study are included in this published article and its supplementary file. CSF DDC measurements are available in the PPMI database (project 274).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
