Abstract
Background
Despite substantial research efforts, advances in Parkinson's disease therapeutics remain largely confined to the management of motor symptoms, with comparatively limited progress in addressing non-motor features, and with no proven success in disease-modifying therapies to date.
Objectives
To describe recent trends in Parkinson's disease therapeutic trials and to characterize the experimental compounds and targets investigated in drug development programs.
Methods
We conducted a cross-sectional analysis of Parkinson's disease therapeutic clinical trials registered in ClinicalTrials.gov, EUCTR, and CTIS since 2013. For the subset of commercially sponsored medicinal product trials, we further described therapeutic objectives and pharmacological targets.
Results
We identified 1855 trials, of which 29% were ongoing. Commercial trials predominantly investigated drugs and devices, whereas non-commercial trials more often focused on non-pharmacological interventions. Among 294 commercial medicinal product trials, 166 distinct products representing 146 active compounds were identified, mapped to 52 pharmacological targets, with additional compounds acting through multiple or unclear mechanisms. Dopaminergic approaches dominated (dopamine receptor agents, 18%; dopamine replacement, 15%), followed by aSyn-targeted strategies (10%). Advanced therapies, including cell and gene therapies, were investigated in 8%. Motor symptoms, particularly motor fluctuations, were the most frequent objectives (45%), whereas non-motor symptoms were rarely addressed (8%).
Conclusions
The landscape of Parkinson's disease trials has expanded over the past decade, but progress has largely been limited to incremental improvements in dopaminergic therapies. The continued lack of effective treatments for non-motor symptoms and disease modification suggests a need to rethink current approaches to drug development in Parkinson's disease.
Plain Language Summary
Over the past decade, progress in treatments for Parkinson's disease has been modest. Most newly approved therapies represent new formulations of existing drugs rather than true breakthroughs, and no intervention has yet been shown to slow or halt disease progression. We analyzed clinical trials registered since 2013 in the three largest public trial registries used in Europe and the United States, including therapeutic trials in patients with Parkinson's disease. Trial characteristics were reviewed, and for drug trials sponsored by pharmaceutical companies, we evaluated study objectives and biological targets in greater detail.
In total, we identified nearly 1900 trials. Commercial trials, primarily sponsored by pharmaceutical companies, focused largely on drugs, whereas academic trials more often investigated devices or behavioral interventions. Among commercial trials, dopamine-based therapies remained the predominant focus, although new biological targets and other approaches, such as cell and gene therapies, were also explored. Despite these initiatives, most efforts continued to target motor symptoms, while non-motor problems, such as dementia, psychosis, fatigue, and sleep disorders remained underrepresented.
Overall, our findings indicate that although therapeutic research activity in Parkinson's disease has expanded, genuine therapeutic innovation has been limited and demands innovation.
Introduction
Parkinson's disease (PD) is the second most common age-related neurodegenerative disorder, affecting over 8 million people worldwide. The global prevalence of PD is expected to double by 2050, driven by aging populations and demographic shifts. 1
Several symptomatic therapies exist for PD, particularly dopaminergic agents. However, no treatment has been shown to alter the underlying course of the disease thus far. 2 Levodopa continues to be the most efficacious therapeutic drug for PD treatment, but since PD is a multifaceted condition, including a panoply of additional elements such as dementia, falls, levodopa-induced dyskinesia, psychosis, among others, the current treatment toolbox is not entirely satisfactory. Although the number of compounds for treating PD continues to expand, the number of compounds licensed in the past 10 years has been sparse. 3 In fact, only a few new therapies have reached approval for PD or its complications, such as MAO-B and COMT inhibitors, adenosine A2A receptor antagonists, novel levodopa formulations and delivery systems, and treatments for dyskinesia and psychosis.4,5 At the same time, investigational programs have broadened beyond dopaminergic pathways, with active development targeting alpha-synuclein (aSyn), LRRK2, GCase and other novel mechanisms, reflecting both diversification of approaches and ongoing challenges in bringing new therapies to patients. 6 Increasing emphasis is now placed on therapies directed at specific genotypes, particularly those associated with GBA and LRRK2 variants, illustrating the growing role of precision medicine in PD drug development. 7
Clinical trial registries have become a fundamental tool in ensuring transparency in drug development. Since 2005, the International Committee of Medical Journal Editors (ICMJE) has required trial registration as a condition for publication and, in 2006, the U.S. Food and Drug Administration (FDA) mandated the registration of all trials.8,9 In 2000, Clinicaltrials.gov was made publicly available, and is the largest clinical trials registry, representing most of the available trials in World Health Organization (WHO) International Clinical Trials Search Portal (ICTRP). 10 In the European Union, the European Union Clinical Trials Register (EUCTR) was the main registry until it was gradually replaced by the Clinical Trials Information System (CTIS), which became mandatory for all new clinical trial applications as of January 2023. Together, these registries capture the majority of ongoing and recent interventional studies and can offer insights into global drug development, although publication rules and regional registration practices mean that early-phase trials (particularly phase 1) and studies conducted exclusively outside the US/EU are likely underrepresented.
Previous research has provided insight into the PD drug development pipeline. Trends have been described since 1999, highlighting the predominance of repurposing strategies and a moderate overall success rate, with approvals largely restricted to dopaminergic therapies and motor symptoms. 3 More recently, PD clinical trials registered in ClinicalTrials.gov as of January 2024 have been reviewed. In this effort, 136 active interventional studies evaluating 107 distinct agents, were identified, with approximately half directed at symptomatic therapies and half at disease-modifying approaches. Only three disease-modifying agents had advanced to phase 3, underscoring the limited late-stage development of novel mechanisms. This analysis was restricted to ongoing trials and to a single registry, which limits the ability to evaluate longer-term trends and the full extent of development activity. 11
Here, we analyzed clinical trial data from three major registries in order to characterize therapeutic clinical trials in PD registered since 2013. Using an approach that combined conventional registry analysis with large language model (LLM)-assisted screening, we examined trial design features, therapeutic objectives, and the range of experimental products and pharmacological targets. Our aim was to provide an updated overview of the drug development landscape in PD and to identify emerging trends over the past decade.
Methods
We conducted a cross-sectional data analysis of all clinical trials with a therapeutic objective performed in patients with PD, and registered in CTIS, EUCTR, and ClinicalTrials.gov since 2013. The search was performed on 9 February 2025 using the term “Parkinson” in any field for CTIS and EUCTR, and “Parkinson's disease” in the “Medical Condition” field with the “Interventional Studies” filter for ClinicalTrials.gov.
Trials were eligible for inclusion if they were first registered on or after 1 January 2013 and involved participants with PD. We focused exclusively on interventional studies with a therapeutic objective, and excluded studies with diagnostic, imaging, or other non-therapeutic objectives, as well as those with a registration status of “withdrawn” or “not authorized.” Trials registered before 2013 were excluded to minimize heterogeneity introduced by evolving regulatory requirements and registration practices.
For a subset analysis of commercially sponsored medicinal product trials, we applied additional criteria: trials had to investigate interventions classified as drug, biological, genetic, or combination products; have a commercial sponsor; and not be phase 4 studies. Additional manual exclusions were applied to ensure alignment with these criteria.
We employed a GPT-4-based LLM to support the screening process by identifying trials that, despite matching the initial search terms, were unlikely to involve PD patients. For each record, the model analyzed the text from the trial's title, objectives, endpoints, and inclusion criteria, and flagged potentially ineligible studies. These flagged records were then reviewed by an author (GSD) to determine whether they should be excluded from the dataset. The titles of the remaining records were manually screened (TM) to identify potentially ineligible trials.
Data was retrieved directly from the public portals of those clinical trial registries, using R (version 4.4.2), and ctrdata package (versions 1.20.0) functions. 12 Duplicate trial records were identified and resolved using ctrdata functions, based on matching trial identifiers. When duplicates were found across registries, the record from the preferred source was retained, following the order: ClinicalTrials.gov, then CTIS, and finally EUCTR. For each selected trial record, we extracted key structured fields from the public registry data, including trial identification number, registry source, title, approval and start dates, trial phase, sponsor type, recruitment status, number of participants, randomization status, blinding, brief summary, primary objective, primary endpoint, and whether the intervention was classified as a drug, biological, genetic, or combination product. The date variable was derived primarily from the reported start date, or from the approval date when the start date was unavailable.
For the subset of commercial medicinal product trials, we performed additional manual classification of each trial's therapeutic objective, interventions and pharmacological targets. Each trial was assigned a specific therapeutic indication, based primarily on the stated primary objective and endpoint. These indications were then grouped into broader therapeutic focus categories: motor symptoms (parkinsonism, dyskinesia, motor fluctuations, falls), non-motor symptoms (dementia, depression, psychosis, orthostatic hypotension, sleep disorders), disease-modifying therapies, and other or unclear indications. We extracted information on the experimental drugs being studied. Drug names were harmonized across trials through manual verification using online resources, including the European Union Telematics Controlled Terms and other publicly available sources, to ensure that different denominations of the same substance were consolidated. 13 We distinguished between “products,” defined as specific formulations under investigation, and “active substances”, defined at the drug level. Only experimental products were considered. Each active substance was then manually classified according to its pharmacological target.
We performed descriptive analyses of the collected data. Trial characteristics were summarized descriptively using counts and percentages for categorical variables and median and interquartile range (IQR) for continuous variables. We used R for data analyses and visualizations.
Research ethics approval was not required as all used data is publicly available, and only metadata was used.
Results
Our initial search query yielded 3853 trial records. After the exclusion of duplicate records, trials registered before 2013 and those with status of “withdrawn” or “not authorized”, 1963 remained (Figure 1). An LLM-based filter was then used to identify non-PD or non-therapeutic trials, flagging 77 records for potential exclusion, of which 72 were confirmed and 5 were retained after manual review. An additional 36 studies were excluded during manual screening, resulting in a total of 108 exclusions. In total, 1855 trials were included. The vast majority were registered in Clinicaltrials.gov (1770 trials, 95%). Of these, 394 had commercial sponsors, and 1461 had non-commercial sponsors (Table 1). From the commercial trials, we selected a subset of 294 commercially sponsored trials studying medicinal products trials (Table 2).
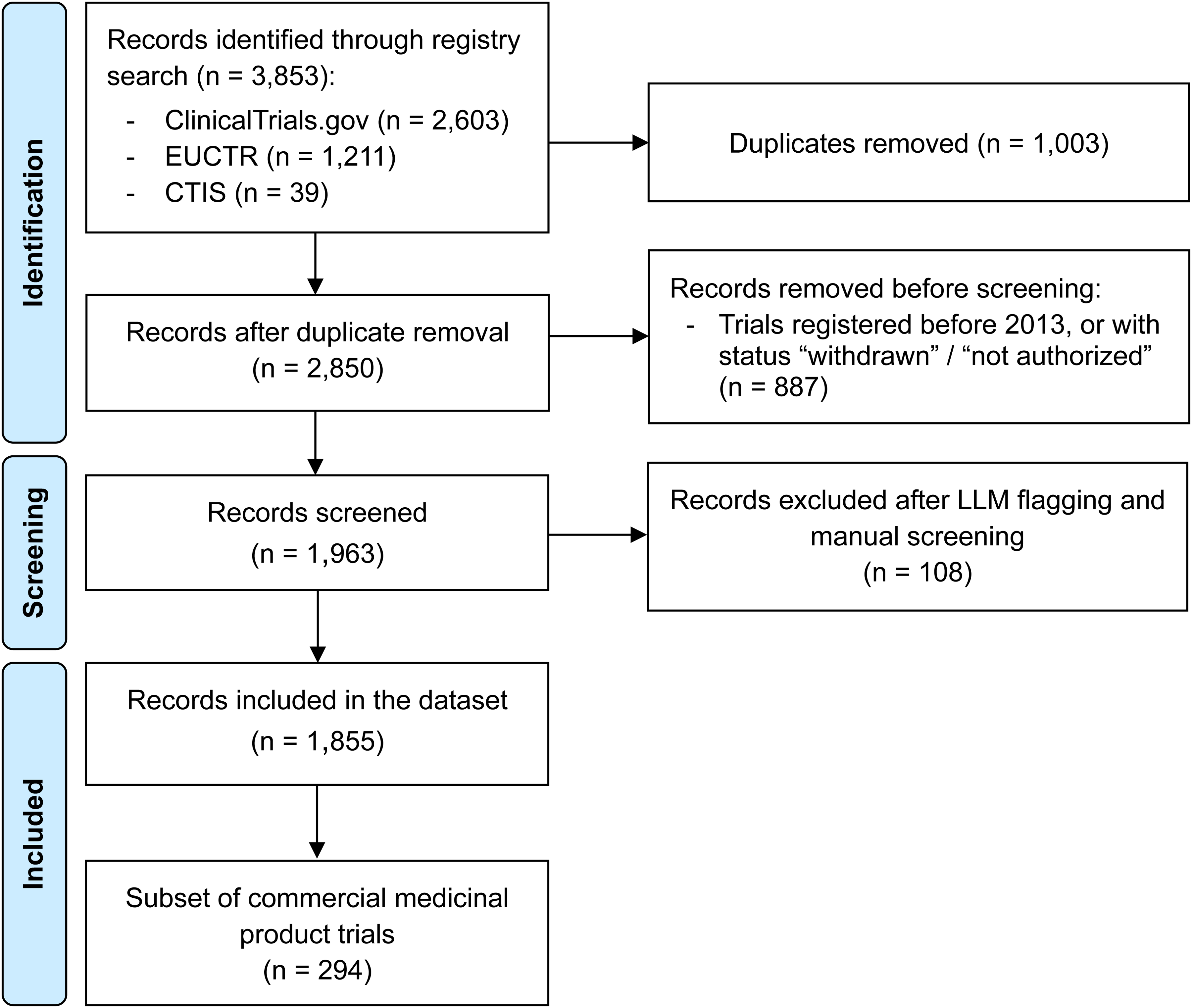
Flow diagram of the trial identification and selection strategy used in this study.
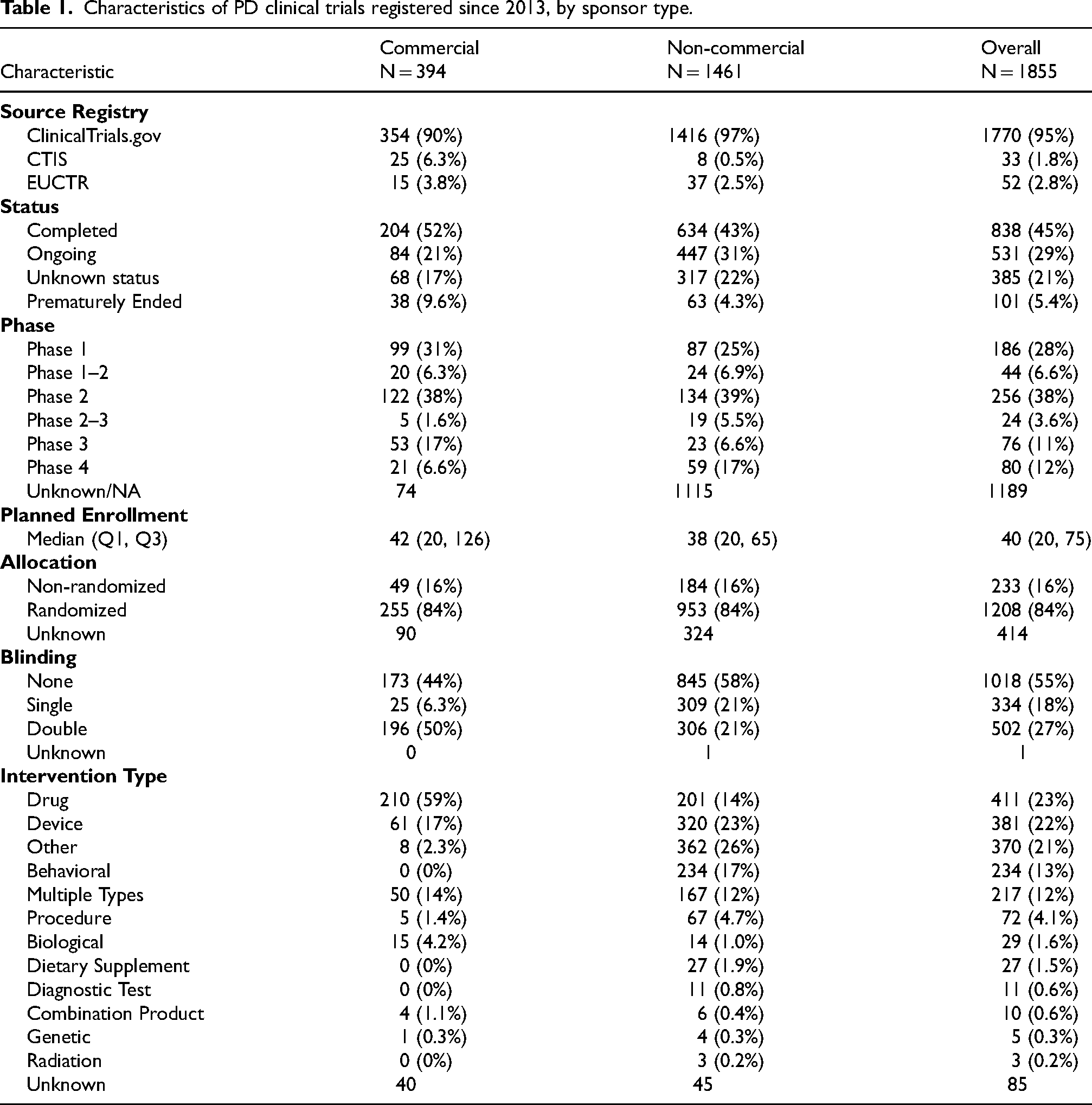
Characteristics of PD clinical trials registered since 2013, by sponsor type.
Characteristics of commercial medicinal product trials in PD.

The characteristics of all trials included are summarized in Table 1. Nearly half were listed as completed (838 trials, 45%), while 531 (29%) were ongoing and 385 (21%) had an unknown status. Overall, the number of trials registered per year has been steadily increasing, reaching a peak of 188 new trials registered in 2024 (Figure 2).
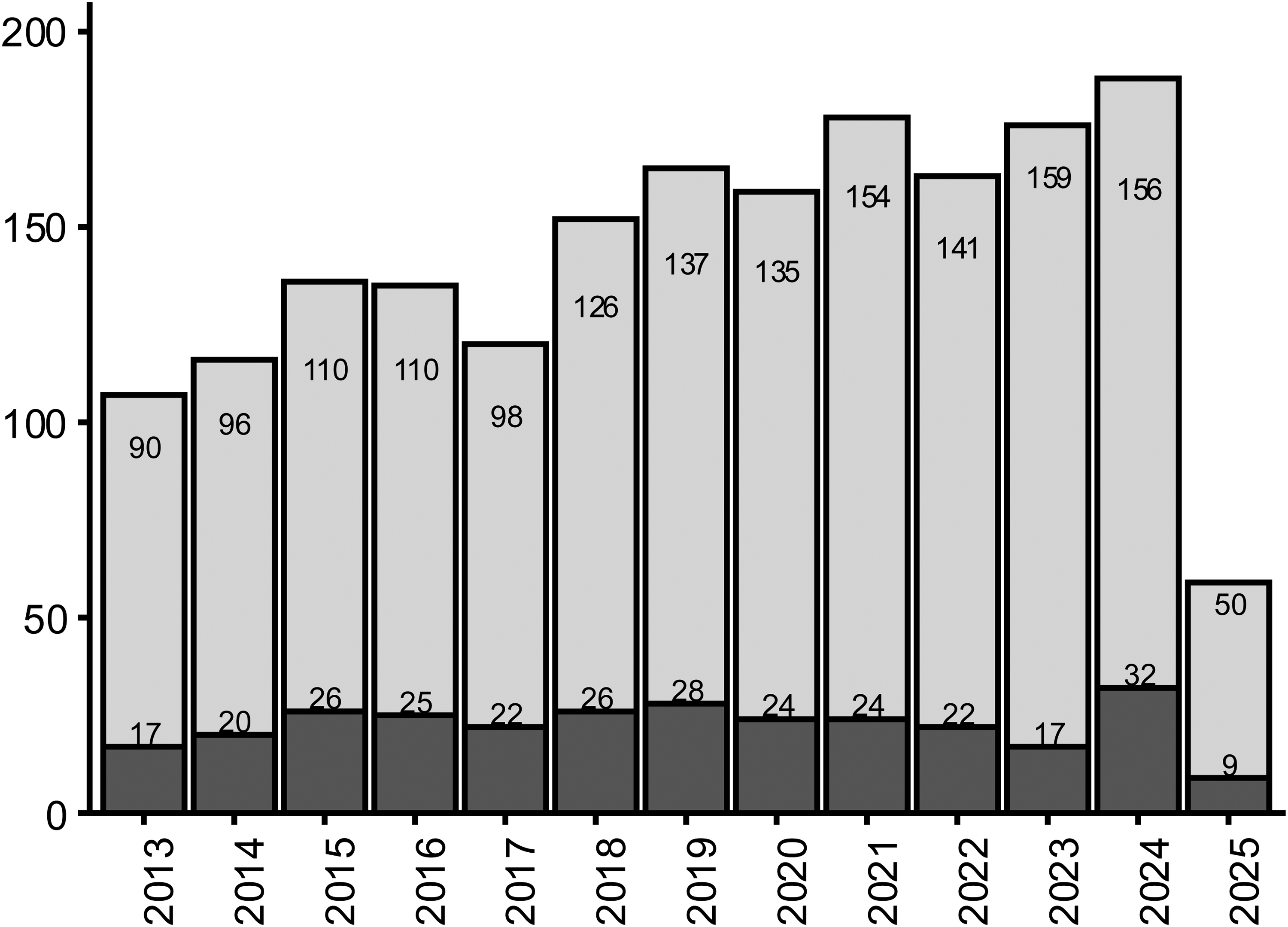
Annual Distribution of PD Clinical Trials. Light grey bars represent all included trials; dark grey bars represent the subset of commercial medicinal product trials. Y-axis: number of trials; X-axis: year. Data for 2025 are still incomplete.
Trial phase was not reported or not applicable in 1189 of 1855 trials (64%), the vast majority of which were non-commercial (94%). Among trials with a specified phase, 186 (28%) were phase 1, 256 (38%) phase 2, 76 (11%) phase 3, and 80 (12%) phase 4; in addition, 68 trials (10%) were registered as multi-phase (phase 1–2 or 2–3). Commercial and non-commercial trials showed notable differences: phase 3 trials accounted for 17% of commercial studies but only 7% of non-commercial studies, whereas phase 4 trials were more common among non-commercial trials (17% vs. 7%).
Planned enrolment was generally modest, with a median of 40 participants across all trials (IQR 20–75) and did not differ meaningfully between commercial and non-commercial sponsors. Most trials (1208; 84%) were randomized controlled trials. Regarding blinding, half of all commercial trials were double-blind (196 trials), while only around 20% of non-commercial trials were double-blind.
The types of interventions were noticeably different between commercial and non-commercial trials. The most common interventions in commercial trials were drugs (210 trials, 59%), medical devices (61 trials, 17%), and studies testing multiple intervention types (50 trials, 14%). In contrast, non-commercial trials often evaluated non-pharmacological interventions, most commonly listed as “other” (362 trials, 26%), medical devices (320 trials, 23%), and behavioral interventions (234 trials, 17%).
From the 394 commercial trials, we selected the 294 that investigated medicinal products, excluding phase 4 studies and non-drug interventions. This subset was chosen as it best reflects ongoing innovation in drug development. The characteristics of these trials are summarized in Table 2. Just over half were completed (160, 54%), while 56 (19%) were ongoing, 30 (10%) had been terminated early, and 48 (16%) had an unknown status. More than half were completed (160, 54%), while 56 (19%) were ongoing, 30 (10%) had been terminated early, and 48 (16%) had an unknown status. Regarding trial phase, 96 (33%) were phase 1, 121 (41%) phase 2, and 53 (18%) phase 3. An additional 22 trials (7%) were registered as multi-phase (17 phase 1–2 and 5 phase 2–3). Most trials were randomized (203, 83%) and more than half were double-blind (171, 58%).
The main therapeutic objectives across commercial trials were related to motor symptoms (133 trials, 45%) and disease modification (53 trials, 18%). Trials addressing non-motor symptoms were less common (22 trials, 8%). For 86 trials (29%), the therapeutic objective was unknown or not applicable. Among trials focusing on motor symptoms, most targeted motor fluctuations (77 trials, 58%), followed by parkinsonism (31 trials, 23%) and dyskinesia (24 trials, 18%). Trials addressing non-motor symptoms, most often targeted dementia associated with PD (10 trials, 3%) and sleep disorders (5 trials, 2%).
More than half of the clinical trials with objectives related to motor symptoms, were regarding motor fluctuations (77 trials, 26%), while the next most common therapeutic indications related to motor symptoms were parkinsonism (31 trials, 11%). Regarding trials with therapeutic objectives regarding non-motor symptoms, the most common indications were dementia associated with PD (10 trials, 45%) and sleep disorders (5 trials, 23%).
To explore the mechanisms of action and pharmacological targets being researched, we manually mapped each investigational product to its presumed target. Across the 294 commercial medicinal product trials, we identified 166 distinct products corresponding to 146 distinct active substances. These included 52 different pharmacological targets or mechanisms, and 23 active substances (studied in 32 trials) that could not be clearly assigned to a single target, either because they act through multiple targets or because their mechanism of action is not sufficiently characterized. The most frequently investigated were dopamine-related therapies: dopamine receptors were the target of 52 trials (18%), involving 12 distinct active substances, and dopamine replacement strategies (here including levodopa/carbidopa and fos-levodopa) were the focus of 45 trials (15%), covering 16 different products. Strategies targeting aSyn were also common, appearing in 28 trials (10%) and including monoclonal antibodies, peptide vaccines, siRNA therapies, and other approaches. Of note, 23 trials (8%) investigated advanced therapy medicinal products, most commonly stem cell transplantation and gene therapies. The full distribution of identified pharmacological targets is provided in Table 3.
Pharmacological targets in commercial medicinal product trials.

Discussion
In this cross-sectional analysis of clinical trial registries, we found that the number of PD trials has steadily increased over the past decade. Commercial trials were more often focused on pharmacological interventions, while non-commercial trials encompassed a wider range of approaches, including behavioral and device-based strategies. Within the subset of commercial medicinal product trials, dopaminergic therapies, particularly dopamine receptor agents and levodopa formulations, remained the most frequently studied. However, a considerable share of development targeted novel pathways. Overall, the therapeutic landscape included a broad spectrum of strategies, from reformulations of established compounds to repurposing efforts and novel mechanisms of action. Most trials pursued motor symptoms, especially motor fluctuations, whereas comparatively fewer addressed non-motor complications such as dementia, sleep disorders, or psychosis.
Notably, the pipeline is still dominated by incremental advances in symptomatic motor therapies, especially dopaminergic strategies, even though other mechanisms are being explored. Disease-modifying approaches, such as those targeting aSyn or LRRK2, along with cell and gene therapies, account for a visible share of activity, but so far these approaches have not translated into regulatory approvals. The 52 distinct pharmacological targets identified, not including the 23 active substances with multiple or unknown targets, show considerable diversification across development programs, especially given the relatively small number of trials analyzed. The contrast between commercial and non-commercial activity points to complementary priorities: commercial programs continue to drive drug development, whereas academic sponsors often focus on needs that are less likely to attract industry investment.
We examined indicators of drug development programs in PD by using the data available in publicly available trial registries. In drug research, especially when so many compounds are repurposed, these may not necessarily go through each development phase. This non-linearity may be a contributor to the smaller number of phase 1 trials. However, more importantly, transparency policies restrict the visibility of early-phase trial activity. In the US, most phase 1 drug trials are not required to be publicly registered in ClinicalTrials.gov under FDAAA/Final Rule. 14 Similarly, in the EU/EEA, phase 1 trials conducted solely in adults were generally not made public until the implementation of CTIS and entering in effect of the Clinical Trials Regulation in 2022, which mandated public disclosure of all clinical trials.15,16 As a result, phase 1 trials are likely underrepresented in our dataset.
Despite the broad spectrum of motor and non-motor features in PD, drug approvals in the US and EU over the past decade have focused almost exclusively on parkinsonism, motor fluctuations, or select non-motor complications. Safinamide and opicapone (new MAO-B and COMT inhibitors) were introduced as adjuncts to levodopa, while novel symptomatic mechanisms include istradefylline, an adenosine A2A receptor antagonist for “off” episodes, and pimavanserin for PD psychosis. New formulations and delivery systems of levodopa, such as extended-release formulations, inhaled rescue doses, and subcutaneous infusion pumps, have aimed to stabilize dopaminergic stimulation. New formulations of amantadine extended/sustained-release for dyskinesias were also approved.4,5 While these broaden the therapeutic arsenal, they largely represent incremental advances in motor manifestations rather than disease-modifying innovation. The lack of progress in non-motor manifestations likely reflects limited preclinical evidence, absence of validated animal models, and lack of regulatory guidance, whereas motor outcomes benefit from the established benchmark of levodopa. Non-motor symptoms, however, affect the vast majority of patients and represent a major source of disability and impaired quality of life, underscoring the need for targeted therapeutic development in this domain. 17
Although our study provides a novel systematic overview of ongoing trials in PD, we acknowledge it has several limitations. First, the analysis was restricted to trials with publicly available records. Registration and disclosure requirements differ across jurisdictions and have evolved over the study period. As discussed above, transparency rules exempting most phase 1 trials from public registration likely contributed to underrepresentation of early-phase activity. As a result, compounds that fail to progress beyond early stages may not be captured. Second, although the registries examined capture a large share of global activity, trials conducted solely in regions outside the EU/EEA and US may not be included. Third, the classification of therapeutic objectives, active substances, and pharmacological targets necessarily entails oversimplification, and also required manual review, which may result in misclassification. In addition, while the LLM-assisted screening step improved the efficiency of identifying eligible trials, it did not replace manual screening, indicating that the approach should be regarded as complementary rather than substitutive. Fourth, the analyses of therapeutic objectives, experimental drugs and targets was limited to commercially sponsored medicinal product trials. Non-commercial studies, in contrast, more frequently investigated devices, behavioral, or other interventions. Therefore, while our analysis highlights patterns in drug development, some findings do not generalize to the complete trial landscape and may miss important aspects of non-commercial research, including non-pharmacological interventions and repurposing efforts. Finally, while originally this study also aimed to calculate success or approval rates of PD drug development programs, this was deemed not feasible, as it is difficult to delineate distinct development programs from registry data, especially when multiple trials test different formulations or related compounds, and when many programs are ongoing or incompletely reported.
Despite the growing volume of clinical trials, the development of new pharmacological therapies for PD has brought few regulatory approvals in the past decade. Most progress has come from incremental advances in dopaminergic treatments, while disease-modifying strategies have so far failed to deliver effective therapies. This gap raises the possibility that current efforts are not addressing the right biological targets or pathways and highlights the need for better understanding of disease mechanisms to guide future drug development in PD.
Footnotes
Acknowledgements
Not applicable
Author contributions
To be included later at the proofing stage.
Ethical considerations
Research ethics approval was not required as all used data is publicly available, and only metadata was used.
Consent to participate
Not applicable
Consent for publication
Not applicable
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interest
Tiago Machado, Gonçalo S. Duarte, and Tiago F. Outeiro declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Joaquim J. Ferreira has provided consultancy to Bial, Biogen, AbbVie, Sunovion Pharmaceuticals, Roche, Stada, Neuroderm, Lundbeck, Affiris, Lilly and Organon, received speaker fees from Bial, Biogen, AbbVie, Sunovion Pharmaceuticals, Infucure, Zambon, Roche, Stada, ONO Pharma, Britannia, SK Chemicals, Major Pharma and has received grants from AbbVie, Bial, Novartis, GlaxoSmithKline, Medtronic and Angelini.
Data availability
All used data is publicly available from the original sources. The used datasets are available upon request.
