Abstract
Background
Patients with Parkinson's disease (PD) frequently experience a progressive decline in their activities of daily living (ADL), necessitating caregiver support. Discrepancies between patient and caregiver ADL ratings are common and may hinder optimal care.
Objectives
To quantify patient-caregiver differences in ADL ratings and identify the clinical factors associated with these discrepancies.
Methods
We conducted a cross-sectional study involving 217 patients with PD and their primary caregivers. Both groups independently completed the Activities of Daily Living Questionnaire (ADLQ). Discrepancy (dADLQ) was defined by subtracting the caregiver-rated score from the patient-rated score; its absolute value (δADLQ) reflected the degree of disagreement. Associations with clinical variables, including motor and non-motor symptoms (cognition, mood, sleep, and autonomic function), were examined using multivariate regression.
Results
The mean ADLQ scores were similar between patients and caregivers, but individual differences varied. The no-discrepancy group was characterized by younger age, shorter disease duration, and lower caregiver burden than those with any discrepancy. Larger δADLQ was associated with more severe motor and non-motor symptoms, as well as increased caregiver burden and depression. The dADLQ scores showed directional associations with patient mood and caregiver burden. Multivariate analysis revealed that δADLQ was independently predicted by greater motor symptom severity and gastrointestinal dysfunction.
Conclusions
Discrepancies in ADL ratings were common in patients with PD and increased with disease severity. They were associated with motor and non-motor symptoms, particularly gastrointestinal dysfunction and mood factors. These findings underscore the importance of integrating both perspectives in clinical assessment, particularly for advanced diseases.
Plain language summary title
Understanding the Gap Between Patients with Parkinson's Disease and Their Caregivers in Daily Life Abilities
Plain language summary
People with Parkinson's disease often need help with everyday tasks like eating, dressing, or bathing as their condition worsens. In this study, we looked at how differently patients and their caregivers rate these daily abilities. Sometimes patients think they are doing better than their caregivers believe, or vice versa.
We asked 217 people with Parkinson's disease and their main caregivers to fill out the same questionnaire about the patient's daily abilities. We found that their answers often didn't match, especially when the patient had more severe symptoms, such as problems with movement, memory, or digestion.
We also found that when caregivers were more stressed or depressed, they were more likely to rate the patient's abilities as worse. These mismatches were more common in older patients or those who had lived with the disease for a longer time.
This finding highlights the importance of hearing from both the patient and the caregiver when evaluating how someone with Parkinson's disease is managing daily life. Especially in more advanced stages of the disease, considering both perspectives can help clinicians better understand the patient's real needs and provide more appropriate support and treatment.
Keywords
Introduction
Parkinson's disease (PD) is a progressive neurodegenerative disorder characterized by typical motor and various non-motor symptoms. As PD advances, patients become increasingly vulnerable to difficulties in activities of daily living (ADL), such as bathing, dressing, walking, and eating. Assessing ADL function is critical for evaluating disease impact and treatment efficacy. 1 Caregivers play an increasingly important role in supporting the daily lives of people with moderate to advanced stages of the disease. 2 Alongside this growing involvement, caregivers develop their own perceptions of the patient's functional abilities and needs. These perceptions may not always align with the patients’ self-assessments. It is often observed anecdotally that some patients with PD underestimate their difficulties (or overestimate their independence), while in other cases, patients may perceive themselves as more disabled than what their caregivers believe.3–6 Such patient-caregiver discrepancies in reporting ADL can have important implications; they might lead to under- or over-estimation of care needs, affect clinical decision-making, and even contribute to caregiver strain if a caregiver feels that the patient lacks insight.
Despite these implications, the extent and determinants of discrepancies between patient and caregiver reports on ADLs in PD have not been fully elucidated. Previous research on self vs. informant ratings has yielded mixed results. Some earlier studies have suggested a relatively good agreement between patients and caregivers. For instance, one study reported moderate agreement between patient and care partner subjective reports of cognitive deficits, despite discrepancies between these reports and objective neuropsychological evaluations. 5 However, that study was limited by a small sample size of 42 PD patients and by the fact that patient and care partner reports were not collected independently. Conversely, other studies have found significant divergence. For example, in the context of motor symptoms, one study noted that patients with PD and their communication partners both tended to misjudge the severity of certain symptoms (such as rest tremor) compared to clinical ratings, indicating possible perceptual deficits in both groups. 7 Although this study also had a relatively small sample size of only 20 PD patients, it nonetheless demonstrated a contrasting pattern of perceptual discrepancy. More recently, Becker et al. examined patient vs. informant ratings of ADL using the 10-item Functional Activities Questionnaire and, in a study involving a comparatively larger sample of 150 PD patients, further analyzed ADL performance based on patient's cognitive status, ultimately reporting an overall fair to moderate agreement between patient and informant ratings. 6 In that study, greater discrepancies were observed in patients with cognitive impairment, and factors such as motor severity, non-motor symptom burden, and depression influenced both patient and informant ADL scores. These findings suggest that disease severity and certain symptoms (particularly cognitive deficits) can widen the gap between patient and caregiver perceptions. However, which specific motor or non-motor features are most associated with differing views of daily disability and how caregiver-related factors (such as caregiver mood or burden) contribute to these differences remain unclear.
In this study, we aimed to investigate the difference between patient- and caregiver-reported ADL abilities in a cohort of PD patient-caregiver pairs using the Activities of Daily Living Questionnaire (ADLQ). 8 Our objectives were: (1) to characterize the distribution of patient-caregiver ADL score discrepancies, and (2) to identify clinical correlates of greater discrepancies through the application of a comprehensive battery of standardized clinical assessments. We hypothesized that more complex and severe diseases (both motor and non-motor aspects) would yield greater patient-caregiver differences in perceived ADL performance and that psychological distress in either the patient or caregiver might further exacerbate these differences.
Methods
Study population
This cross-sectional study included 217 patients with PD and their primary caregivers, who were recruited from the movement disorders clinic at the referral hospital. Eligible patients met the MDS clinical diagnostic criteria for PD and had a dedicated primary caregiver (family member or cohabitant) who was responsible for daily care. 9 We included patient-caregiver pairs if the caregiver lived with the patient or saw the patient daily, so as to be very familiar with the patient's daily functioning. All the patients were in a stable medication state and provided informed consent along with their caregivers. The regional institutional review board approved the study protocol, and all procedures were conducted in accordance with the Declaration of Helsinki.
Clinical assessments
Basic demographic and clinical data were collected, including patient age, sex, disease duration, PD medication usage, and caregiver age, sex, and relationship with the patient (e.g., spouse, adult children). PD severity was assessed by the modified Hoehn and Yahr (HY) stage and the Movement Disorder Society Unified Parkinson's Disease Rating Scale (MDS-UPDRS). 10 We administered the Korean version of the Mini-Mental Status Examination (K-MMSE) and Montreal Cognitive Assessment (K-MoCA) to evaluate global cognitive status.11,12
Activities of daily living questionnaire (ADLQ)
The ADLQ is a PD-specific ADL questionnaire and contains 20 items covering various daily activities (e.g., feeding, dressing, bathing, mobility, handwriting, social activities), each rated by the respondent on a difficulty scale from 0 (no difficulty) to 5 (cannot perform at all or total dependence). 8 The total score ranges from 0 to 100, with higher scores indicating greater overall disability in ADLs. The ADLQ was originally designed for patient self-assessment; in this study, both the patient and caregiver independently completed the ADLQ (caregivers rated the patient's level of difficulty for each activity). Neither the patient nor the caregiver saw the others’ responses. We defined the raw difference in scores as follows: dADLQ = (patient's ADLQ score) – (caregiver's ADLQ score). A negative dADLQ, designated as the “UE” (underestimation) group, indicates that the patient rated themselves as less impaired than the caregiver did, reflecting underestimation of disability by the patient. Conversely, a positive dADLQ, designated as the “OE” (overestimation) group, indicates that the patient reported more disability than the caregiver perceived, reflecting overestimation by the patient. Cases with no difference between patient and caregiver ratings were classified as the “CON” (concordant) group. The absolute difference was defined as δADLQ = |dADLQ|, reflecting the magnitude of patient-caregiver discrepancy regardless of direction. Based on the distribution of δADLQ values, participants were categorized into three groups: ZERO (δADLQ = 0), MILD (0 < δADLQ < 7), and LARGE (δADLQ ≥ 7). A cutoff score of 7 was empirically selected, as it corresponded approximately to both the median value and the 66th percentile of the distribution, thereby allowing for a balanced three-way stratification of participants. This threshold served as a data-driven and exploratory reference, given the absence of established cutoffs in previous literature.
Non-motor symptoms
We evaluated a broad spectrum of non-motor symptoms using the Non-Motor Symptoms Scale (NMSS), which yields a total score (0–360) across nine domains of non-motor symptoms in PD. 13 We also evaluated symptoms of autonomic dysfunction using the comprehensive symptom survey. 14 In addition, excessive daytime sleepiness was measured using the Epworth Sleepiness Scale (ESS). 15
Mood and psychosocial measures
Patients completed the Beck Depression Inventory (BDI) and the Beck Anxiety Inventory (BAI).16,17 Caregivers were asked to complete the BDI with reference to their mood state. Caregiver burden was assessed using the Zarit Burden Interview (ZBI), a 22-item questionnaire that gauges the caregiver's perceived strain in caring for the patient (score range 0–88, with higher scores indicating a greater burden). 18
Health-related quality of life
We administered the Parkinson's Disease Questionnaire-39 (PDQ-39) to patients as a measure of PD-specific quality of life. 19 We did not have a formal caregiver quality-of-life instrument, but aspects of caregiver well-being were indirectly captured by mood and burden scales.
Statistical analysis
Categorical variables are presented as frequencies and percentages, and continuous variables are presented as mean ± standard deviation (SD). Differences in the ADLQ scores evaluated by the patients and caregivers were compared using the paired t-test or Wilcoxon signed-rank test, as appropriate, for paired data. Pearson's correlation coefficient was used to assess the association between continuous clinical variables and ADLQ discrepancies. For group comparisons, categorical variables were analyzed using the chi-squared test or Fisher's exact test. Continuous variables were compared between the two groups using the independent t-test or Mann-Whitney U test, and across more than two groups using one-way analysis of variance (ANOVA) with Scheffe's post-hoc test or the Kruskal-Wallis test with Dunn's post-hoc test, as appropriate. To identify independent predictors of dADLQ and δADLQ, a multivariate linear regression analysis was conducted. Variables were selected using backward elimination with a significance threshold of α = 0.05. All statistical analyses were performed using SPSS (version 26.0; IBM Corp., Armonk, NY, USA) and the R software (version 4.5.0; R Foundation for Statistical Computing, Vienna, Austria). P values less than 0.05 were considered statistically significant.
Results
Participant characteristics
A total of 217 patient-caregiver pairs were analyzed (Table 1). The patients with PD had a mean age of 66.1 years (± 8.5, range 45–85) and disease duration of 2.3 years (± 2.7). More than half (57.6%) of the patients were male. The majority of patients were at early to mid-stage disease (HY stage 2–3), and the mean MDS-UPDRS total score was 46.3 (± 16.5). Cognitive screening scores indicated that most patients were cognitively intact or only mildly impaired (mean K-MMSE 26.6 ± 3.6; 13.9% scored < 25). Regarding non-motor symptoms, the mean NMSS total score was 31.8 (± 30.0). The mean BDI score for patients was 14.4 (± 7.6), indicating mild depressive symptoms on average, and the mean BAI was 31.1 (± 8.0). Approximately one-third of patients (34.0%) met the criteria for at least mild depression (BDI ≥17).
Demographics and clinical characteristics of patients and caregivers.
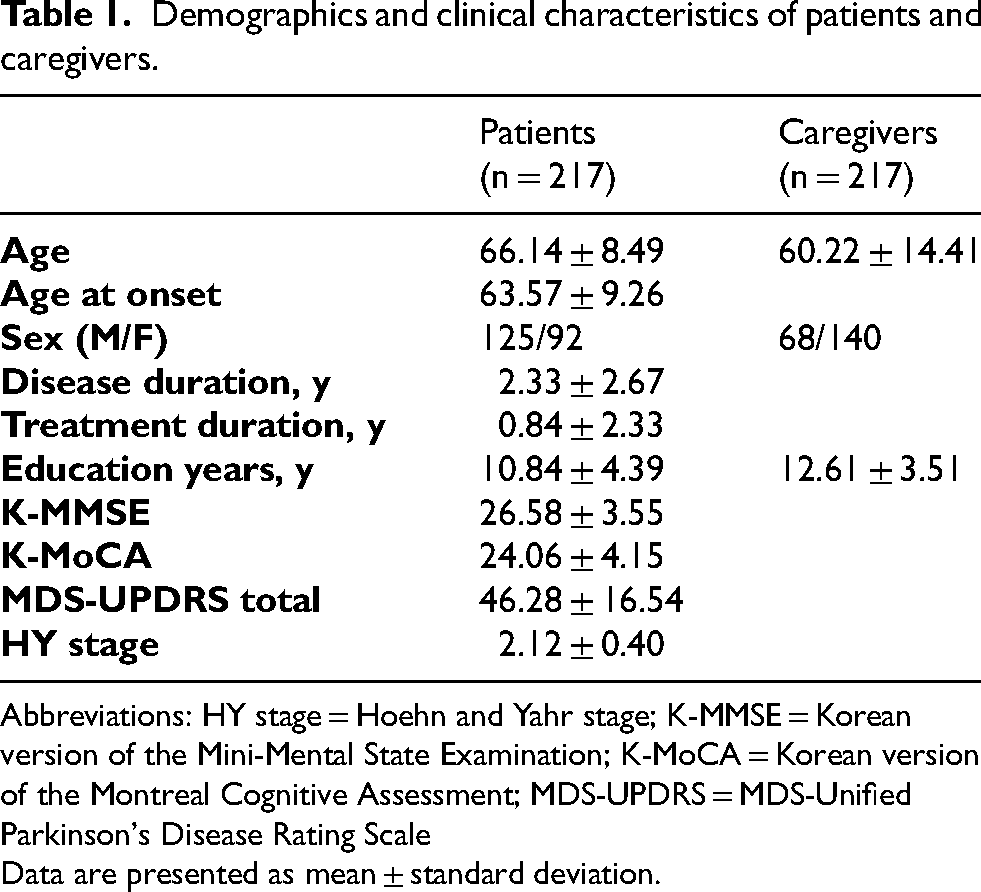
Abbreviations: HY stage = Hoehn and Yahr stage; K-MMSE = Korean version of the Mini-Mental State Examination; K-MoCA = Korean version of the Montreal Cognitive Assessment; MDS-UPDRS = MDS-Unified Parkinson's Disease Rating Scale
Data are presented as mean ± standard deviation.
The caregivers had a mean age of 60.2 years (± 14.4), and 67.3% were female. Most caregivers were the patient's spouse (78.8%), with 21.2% being adult children and the remaining being other relatives or paid caretakers. Caregiver burden scores varied widely (range 0–76), with a mean of 15.3 (± 14.0). Caregivers’ own mood assessments showed a mean BDI of 10.4 (± 7.6); 18.3% of caregivers had BDI scores in the mild depression range or higher.
Patient vs. Caregiver ADL Ratings
The mean patient ADLQ score was 9.5 (± 15.1) and the mean caregiver ADLQ score was 9.7 (± 14.7), with no statistically significant difference between the two (p = 0.727). However, discrepancies were common among individual patient-caregiver pairs. Approximately one-third of the patients (n = 71, 32.7%) rated themselves as less disabled than their caregivers (underestimation group, UE), another third (n = 75, 34.6%) rated themselves as more disabled (overestimation group, OE), and the remaining third (n = 71, 32.7%) showed no difference between patient and caregiver ratings (concordant group, CON). The range of dADLQ was from −47 to +50. The δADLQ, reflecting the magnitude of discrepancy regardless of direction, ranged from 0 to 50, with a mean of 6.5 (± 9.5). Based on the distribution of δADLQ, a total of 71 pairs (32.7%) showed perfect concordance (ZERO group; δADLQ = 0), 75 pairs (34.6%) exhibited mild discrepancy (MILD group; 0 < δADLQ < 7), and 71 pairs (32.7%) demonstrated large discrepancy (LARGE group; δADLQ ≥7), using 7 as an approximate median-based threshold.
We explored whether any characteristics distinguished pairs with little or no differences from those with any differences in either direction (Table 2). Patients in the CON group (dADL = 0) tended to be younger (63.3 ± 7.8) than those in the UE (68.9 ± 7.8) and OE (66.2 ± 9.0) (p < 0.001) groups. They also had shorter disease duration (1.5 ± 1.4 vs. UE: 2.7 ± 3.4, OE: 2.8 ± 2.7; p = 0.002), better quality of life (PDQ-39: 10.0 ± 12.3 vs. UE: 30.0 ± 25.4, OE: 33.5 ± 27.7; p < 0.001), and milder motor and non-motor symptoms (MDS-UPDRS total: 36.0 ± 12.9 vs. UE: 50.8 ± 16.9, OE: 51.4 ± 15.0, NMSS total: 14.7 ± 15.4 vs. UE: 38.3 ± 29.1, OE: 41.6 ± 34.3; both p < 0.001). Caregivers in the CON group showed lower depression scores than those in groups with any discrepancy (7.3 ± 5.7 vs. UE: 12.8 ± 7.9, OE: 11.2 ± 7.9; p < 0.001), and caregiver burden was highest in the UE group, followed by the OE group, and lowest in the CON group (22.2 ± 14.4 vs. OE: 16.5 ± 14.8, CON: 7.1 ± 6.9; p < 0.001). However, there were no significant differences in motor or non-motor symptoms between the UE and OE groups.
Baseline characteristics by direction of patient-caregiver discrepancy in ADLQ scores (dADLQ).
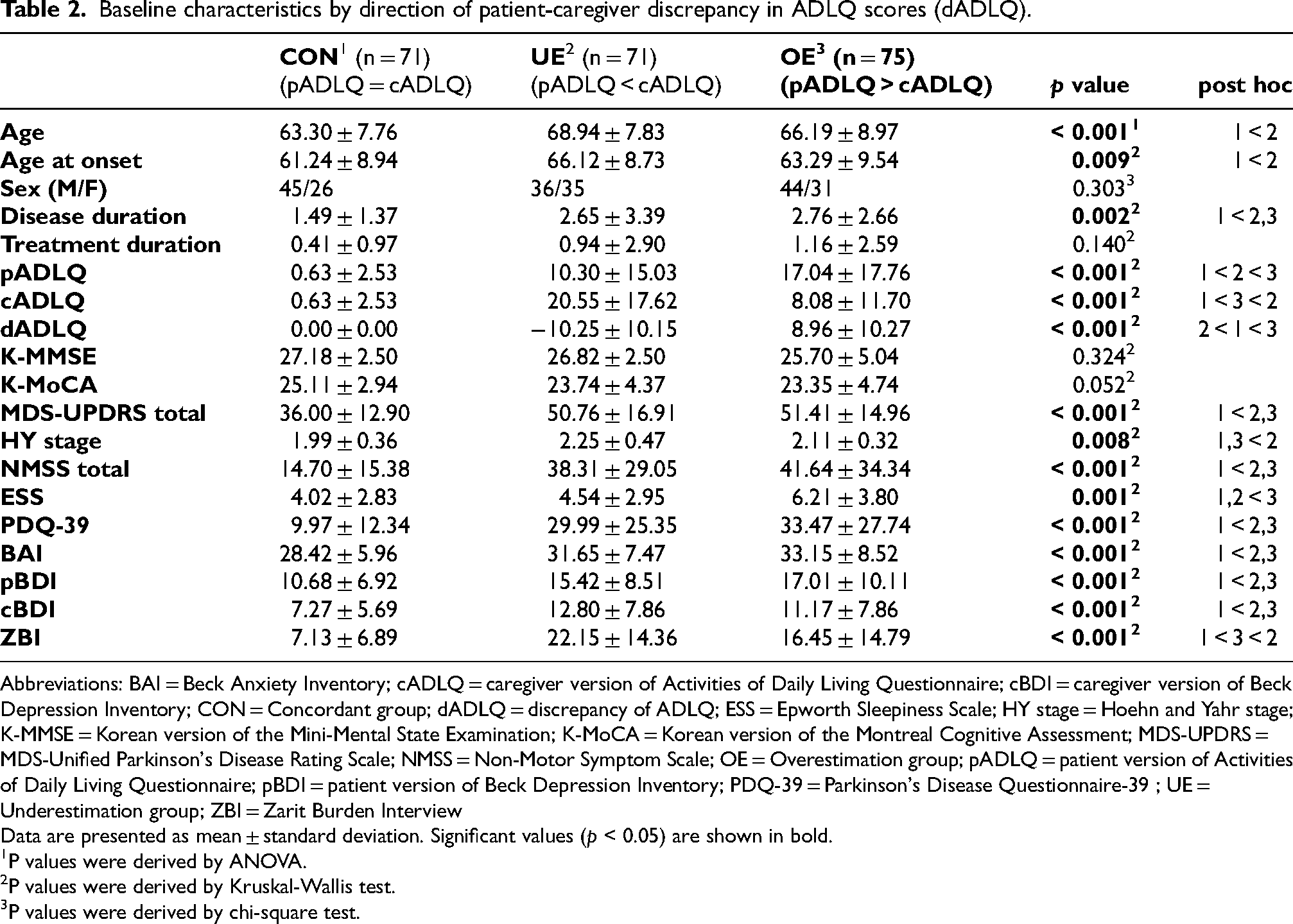
Abbreviations: BAI = Beck Anxiety Inventory; cADLQ = caregiver version of Activities of Daily Living Questionnaire; cBDI = caregiver version of Beck Depression Inventory; CON = Concordant group; dADLQ = discrepancy of ADLQ; ESS = Epworth Sleepiness Scale; HY stage = Hoehn and Yahr stage; K-MMSE = Korean version of the Mini-Mental State Examination; K-MoCA = Korean version of the Montreal Cognitive Assessment; MDS-UPDRS = MDS-Unified Parkinson's Disease Rating Scale; NMSS = Non-Motor Symptom Scale; OE = Overestimation group; pADLQ = patient version of Activities of Daily Living Questionnaire; pBDI = patient version of Beck Depression Inventory; PDQ-39 = Parkinson's Disease Questionnaire-39 ; UE = Underestimation group; ZBI = Zarit Burden Interview
Data are presented as mean ± standard deviation. Significant values (p < 0.05) are shown in bold.
P values were derived by ANOVA.
P values were derived by Kruskal-Wallis test.
P values were derived by chi-square test.
Then, we compared the characteristics of groups stratified by δADLQ (Table 3). The ZERO group was the youngest (63.3 ± 7.8), followed by the MILD (66.8 ± 9.3) and LARGE (68.3 ± 7.6) (p = 0.002). They also had younger age of onset (61.2 ± 8.9 vs. MILD: 64.2 ± 9.9, LARGE: 65.2 ± 8.5; p = 0.030) and shorter duration of disease (1.5 ± 1.4 vs. MILD: 2.6 ± 3.1, LARGE: 2.8 ± 3.0; p = 0.002). Clinical characteristic of patients (motor and non-motor symptoms and quality of life) were most severe in the LARGE group (MDS-UPDRS total: 54.8 ± 15.2; NMSS total: 46.9 ± 34.1; PDQ-39:39.1 ± 28.8), followed by the MILD group (MDS-UPDRS total: 47.6 ± 15.9; NMSS total: 33.5 ± 28.2; PDQ-39:24.8 ± 22.3), and least in the ZERO group (MDS-UPDRS total: 36.0 ± 12.9; NMSS total: 14.7 ± 15.4; PDQ-39:10.0 ± 12.3) (all p < 0.001). Caregiver depression scores and caregiver burden were highest in the LARGE group (cBDI: 13.6 ± 8.7; ZBI: 22.5 ± 14.8), followed by the MILD group (cBDI: 10.4 ± 6.8; ZBI: 16.2 ± 14.2), and lowest in the ZERO group (cBDI: 7.3 ± 5.7; ZBI: 7.1 ± 6.9) (both p < 0.001) .
Baseline characteristics by degree of patient-caregiver discrepancy in ADLQ scores (δADLQ).
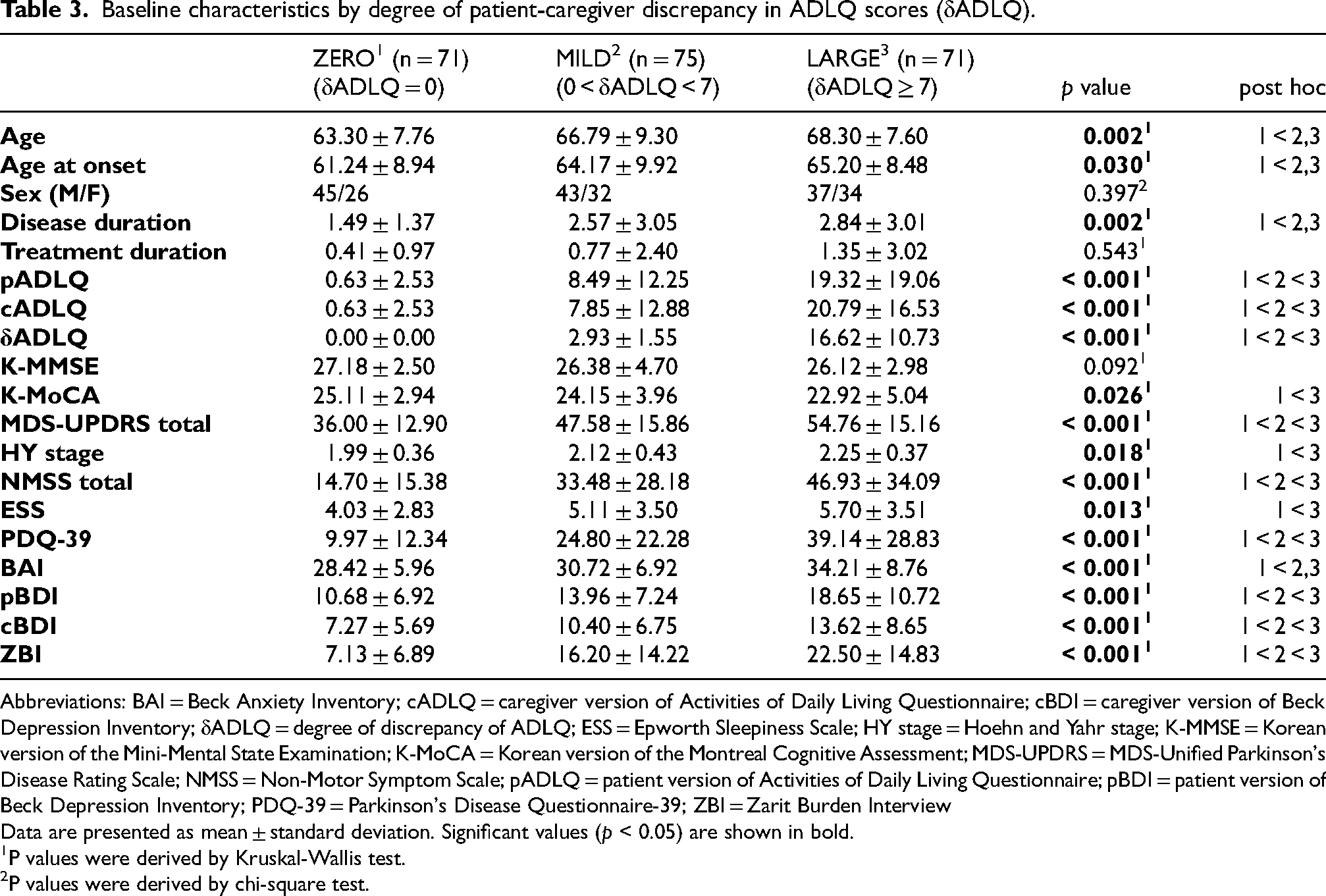
Abbreviations: BAI = Beck Anxiety Inventory; cADLQ = caregiver version of Activities of Daily Living Questionnaire; cBDI = caregiver version of Beck Depression Inventory; δADLQ = degree of discrepancy of ADLQ; ESS = Epworth Sleepiness Scale; HY stage = Hoehn and Yahr stage; K-MMSE = Korean version of the Mini-Mental State Examination; K-MoCA = Korean version of the Montreal Cognitive Assessment; MDS-UPDRS = MDS-Unified Parkinson's Disease Rating Scale; NMSS = Non-Motor Symptom Scale; pADLQ = patient version of Activities of Daily Living Questionnaire; pBDI = patient version of Beck Depression Inventory; PDQ-39 = Parkinson's Disease Questionnaire-39; ZBI = Zarit Burden Interview
Data are presented as mean ± standard deviation. Significant values (p < 0.05) are shown in bold.
P values were derived by Kruskal-Wallis test.
P values were derived by chi-square test.
Multivariate linear regression analysis of clinical predictors independently associated with dADLQ and δADLQ scores.
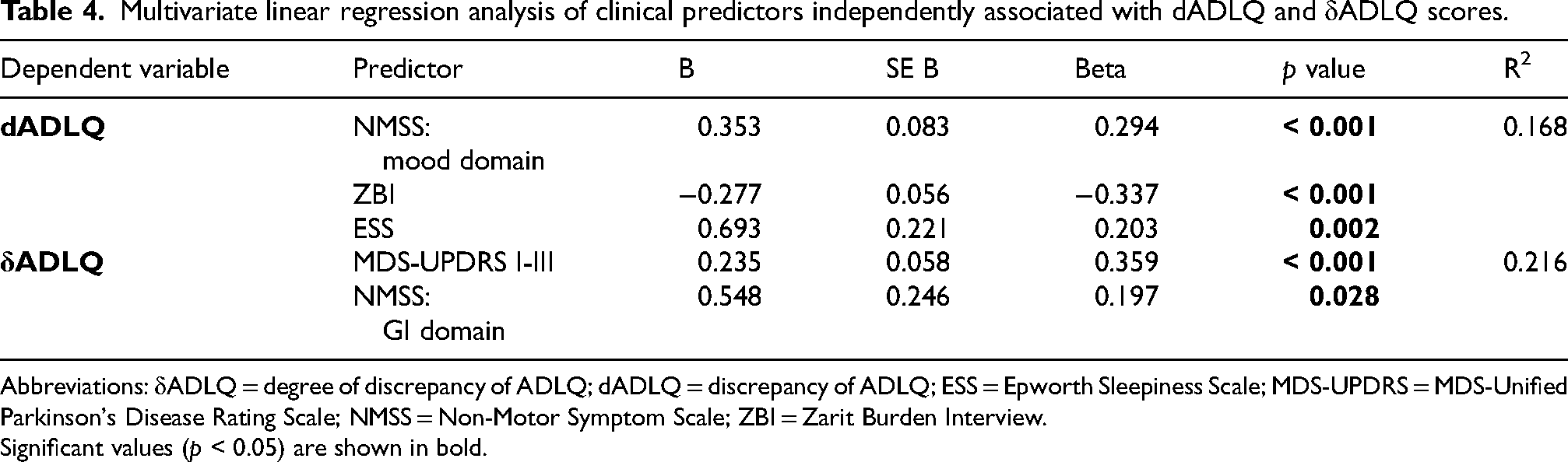
Abbreviations: δADLQ = degree of discrepancy of ADLQ; dADLQ = discrepancy of ADLQ; ESS = Epworth Sleepiness Scale; MDS-UPDRS = MDS-Unified Parkinson's Disease Rating Scale; NMSS = Non-Motor Symptom Scale; ZBI = Zarit Burden Interview.
Significant values (p < 0.05) are shown in bold.
Correlates of ADL rating discrepancies
We performed univariate regression analyses to examine associations between individual clinical characteristics and the discrepancy groups defined by dADLQ and δADLQ (Figure 1). The dADLQ showed a significant positive association with patients’ mood (β = 0.230, p = 0.001) and daytime sleepiness (β = 0.207, p = 0.002) and a negative association with patients’ cognition (β = -0.144, p = 0.034) and caregiver burden (β = -0.189, p = 0.005). However, higher δADLQ was significantly correlated with greater motor symptom severity (β = 0.400, p < 0.001) and with all non-motor symptom severity domains, except for the miscellaneous domain (all p < 0.001). In addition, δADLQ was negatively correlated with patient cognitive function (β = -0.231, p = 0.001), as well as with years of education for both patients (β = -0.159, p = 0.020) and caregivers (β = -0.211, p = 0.002). On the caregiver side, δADLQ was significantly associated with caregiver's own depression (β = 0.273, p < 0.001) and caregiver burden (β = 0.304, p < 0.001).
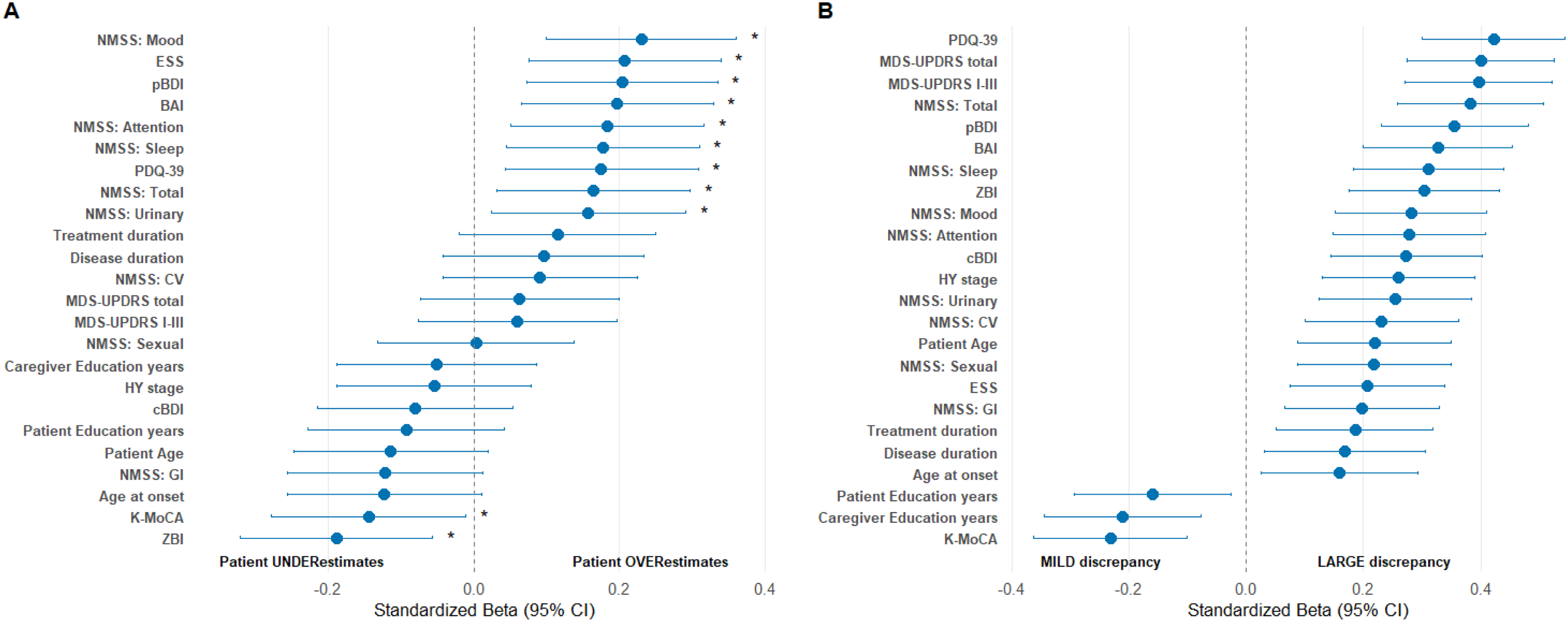
Standardized Beta coefficients from univariate linear regression analyses for dADLQ (A) and δADLQ (B)
In the multivariate linear regression analysis, a higher score on the NMSS mood domain was independently associated with a greater dADLQ (β = 0.294, p < 0.001), whereas increased caregiver burden was negatively associated (β = -0.337, p < 0.001) (Table 4). For δADLQ, the final model identified two patient-related factors as independent predictors: motor symptom severity, as measured by the MDS-UPDRS part I-III (β = 0.359, p < 0.001), and the gastrointestinal (GI) domain score of the NMSS (β = 0.197, p = 0.028).
Discussion
In this cross-sectional study of 217 PD patient-caregiver pairs, we found that discrepancies in reporting ADL abilities were prevalent and clinically meaningful. At the group level, patients and caregivers had similar average ADL scores, and approximately two-thirds of the individual pairs demonstrated some degree of discrepancy. These results highlight that a patient's self-assessment of daily functions often does not perfectly match that of their caregiver, even when using the same questionnaire framework. Importantly, we identified specific factors associated with greater patient-caregiver discrepancies. The δADLQ tended to increase in the context of more advanced disease. In milder cases, patients and caregivers may easily agree that the patient is largely independent of minor issues, whereas in more severe cases, they may diverge in their perceptions of how much assistance is required and how well certain tasks are performed. Our findings are in concordance with those of Becker et al., who reported that PD motor severity had the greatest influence on both patient and informant ADL ratings, contributing to the discordance. 6
In addition, we found that non-motor symptom burden was associated with ADL rating discrepancies. In particular, GI symptoms emerged as an independent contributor to rating divergence. To the best of our knowledge, this is a novel observation; prior studies on patient-caregiver rating differences in PD have largely focused on cognitive impairment and global function, but not specifically on GI issues. GI symptoms are among the most prevalent non-motor features of PD, and they can significantly impact the patients’ quality of life. 20 They may contribute to discrepancies in several ways. For example, symptoms such as constipation or abdominal discomfort may be underrecognized by caregivers. Conversely, caregivers may observe outward manifestations (e.g., choking episodes or excessive drooling) that patients might downplay. Dysphagia, in particular, has been shown to negatively affect caregiver well-being, which could heighten a caregiver's perception of disability even if the patient feels otherwise. 21 The association we found between the NMSS GI domain and δADLQ suggests that clinicians should pay close attention to non-motor symptoms, especially GI and potentially other autonomic issues, when evaluating discrepancies between patient and caregiver reports.
Mood and neuropsychiatric factors also influenced patient-caregiver discrepancies. Patients with more depressive and anxiety symptoms tended to have greater divergence from their caregivers. Depression may influence patients’ perceptions of their own abilities to be more negative, although our data suggested effects in both directions. In some cases, mood symptoms may lead to underreporting. In contrast, caregiver burden and mood were more consistently associated with a unidirectional discrepancy; caregivers who reported higher burden tended to rate patients as more impaired than what the patients rated themselves. This is consistent with prior literature showing that caregiver burden in PD is strongly driven by the patient's loss of independence and neuropsychiatric complications.22,23 Our findings suggest a bidirectional relationship: greater patient-caregiver discrepancy may itself be a source of strain, but, more plausibly, disease severity and associated burden lead to both caregiver stress and divergent reporting. This aligns with findings in mild dementia, where both patient insight and caregiver perspectives are influenced by mood. 24 Another prior multicenter study, conducted in a moderately sized cohort of patients with predominantly moderate disease severity, examined discrepancies in UPDRS-ADL ratings among patients, caregivers, and physicians. 25 The study reported generally good concordance across the three perspectives, which contrasts with our findings showing greater variability between patient and caregiver reports. However, despite the overall agreement, patients with depression in that study also tended to rate their ADL functioning as poorer, suggesting that mood symptoms may still influence self-perceived disability. Likewise, other studies investigating rater concordance in ADL or disability scales have generally shown good agreement, with depression exerting little or no impact on rating differences. 26 Taken together, these findings suggest that the influence of mood on ADL rating discrepancies may vary depending on cohort composition, disease stage, and other clinical characteristics.
Cognitive impairment has been highlighted as a key factor in patient-caregiver discordance in previous studies. 27 In our cohort, cognitive status (MoCA) was correlated with δADLQ in univariate analysis, but it did not remain an independent predictor. This may be attributed to the relatively low proportion of patients with cognitive dysfunction in our sample. It is plausible that the inclusion of a greater number of patients with PD-mild cognitive impairment or PD dementia could have yielded different findings, potentially identifying cognitive dysfunction as a more salient predictor of patient-caregiver discrepancy. Thus, our results do not contradict the importance of cognition; rather, they suggest that in patients without dementia, other factors such as motor and non-motor symptom severity may play a more prominent role in shaping perceptual discrepancies between patients and caregivers.
A previous study explored discrepancy from a different perspective by comparing patients who attended clinical visits with or without caregivers
Our findings have several clinically relevant implications. First, they emphasize the importance of incorporating both patient and caregiver perspectives when assessing daily functional abilities. Relying on either source alone may yield an incomplete understanding. In cases with minimal patient-caregiver discrepancies, patients tended to be younger and at earlier disease stage, where insight was largely preserved and caregiver strain was relatively low. However, as disease complexity increases, a dual perspective assessment becomes crucial. Clinicians should recognize that ADL is a clinically meaningful construct—not only for dementia diagnosis but also for optimizing medication adjustments and individualized care planning. Importantly, discrepancies between patient and caregiver perceptions of ADL were not uncommon in our cohort and were evenly distributed. This underscores the need for clinicians to remain attentive to such perceptual gaps and, whenever feasible, to obtain independent ADL reports from both patients and caregivers during routine visits. Second, our findings highlighted specific symptom domains that warrant attention for potential discrepancies. Motor examinations and non-motor assessments, particularly GI assessments, should not be overlooked. The fact that GI symptoms independently predicted disagreement in our study indicates the broader impact of autonomic dysfunction on patient well-being. Effectively managing such symptoms may not only improve patients’ actual functioning, but also help caregivers appreciate the patients’ daily challenges, potentially reducing discordance. Third, greater discrepancies were associated with greater caregiver burden. Recognizing and addressing these discrepancies may therefore serve as a novel intervention target. Programs that support caregivers, improve communication, and enhance mutual understanding between patients and caregivers may foster better insight and collaboration, ultimately improving care outcomes.
Our study had some limitations. First, our sample predominantly comprised patients with mild disease and relatively preserved cognitive function, which may limit the generalizability of our findings to advanced stages of PD. Second, the cross-sectional design captured associations but could not prove causation in the relationships between motor and non-motor symptoms and ADL discrepancies. Longitudinal studies would be valuable in determining whether increasing motor or GI symptoms within the same patient lead to growing patient-caregiver discrepancies over time. Third, although this study attempted to quantify several relevant factors, we did not measure intangible aspects, such as the quality of the patient-caregiver relationship, communication patterns, and cultural attitudes, which may contribute to reporting discrepancies. Qualitative interviews could complement our findings by exploring why certain differences arise. Finally, although the ADLQ was used to assess ADL abilities via caregiver report, it was originally developed for patient self-report and has not been formally validated for proxy use.
Conclusions
Discrepancies between patients with PD and their caregivers in assessing patients’ daily functional abilities are common and tend to widen with greater disease severity and complexity. Using a patient-centered ADL questionnaire, we found that the absolute patient-caregiver difference in ADL scores increased most significantly in the presence of advanced motor impairment and burdensome non-motor symptoms, particularly GI issues. Mood factors in both the patients and caregivers also influenced these perceptions. These findings emphasize the importance of a dual-perspective approach in PD care. Clinicians should routinely seek and reconcile both patient and caregiver reports, especially in advanced-stage PD. By recognizing and addressing the factors that drive patient-caregiver discrepancies, we may improve not only the objective patient outcomes, but also the alignment of patient and caregiver expectations and understanding. Ultimately, better agreement and communication between patients and caregivers can facilitate appropriate care plans, enhance safety, and reduce frustration in managing the daily challenges of Parkinson's disease.
Footnotes
Ethical considerations
Each participant provided written, informed consent. The study received ethical approval from the Ethics Committee of the Dong-A University Hospital.
Author contribution
BRJ contributed to data curation, formal analysis, and drafting of the original manuscript. SMC conceptualized and supervised the study, contributed to methodology and investigation, revised the manuscript, and acquired funding.
Funding
This study was supported by Dong-A University Research Fund.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability
The data that support the findings of ths study are available from the corresponding author upon reasonable request.
