Abstract
Background
Parkinson's disease (PD) is associated with a high prevalence of cardiovascular dysfunction, a leading cause of mortality in these patients. Autonomic dysfunction, including cardiac autonomic dysfunction (CAD), is increasingly recognized as a significant non-motor symptom in PD and may contribute to adverse cardiac outcomes.
Objective
To investigate echocardiographic alterations in PD patients with CAD and evaluate their diagnostic utility for CAD detection.
Methods
Participants were categorized into PD-CAD and PD-nCAD groups based on Cardiovascular Autonomic Reflex Tests. Echocardiographic assessments included standard transthoracic echocardiography and two-dimensional speckle-tracking strain imaging. Multivariable regression was used to identify predictors of PD-CAD. Receiver operating characteristic curves, integrated discrimination improvement
Results
A total of 78 participants were included, with a median Hoehn and Yahr stage of 2.00 [IQR 2.00, 2.50]. Among them, 33.33% were classified as having PD-CAD. Impaired systolic function characterized by decreasing left ventricular global longitudinal strain and lower systolic mitral annular velocity were found in PD-CAD group. A model combining LV-GLS, s’, and LVMI predicted CAD with AUC = 0.737 (95% CI:0.624–0.850), comparable to conventional autonomic markers.
Conclusion
Subclinical systolic dysfunction (LV-GLS, s’) reflects autonomic-mediated myocardial injury in PD and demonstrates diagnostic potential for CAD identification. Echocardiography may bridge autonomic dysfunction and cardiovascular risk in PD.
Plain Language Summary: Heart Ultrasound Detects Early Autonomic-Related Heart Changes in Parkinson's Disease
Parkinson's disease (PD) is mainly recognized for its movement problems, but it may also be linked to a higher risk of heart complications. One important factor is cardiovascular autonomic dysfunction (CAD), which is common in people with PD. Previous studies have reported heart changes in PD patients, but the connection between CAD and these changes is not fully clear. In this study, we used echocardiography, a heart ultrasound that can detect early heart damage, to evaluate PD patients with and without CAD. We found that patients with CAD showed impaired systolic function, even after accounting for age, sex, medication, and PD severity. This suggests that CAD itself may play an important role in heart problems related to PD. We further tested whether ultrasound measures could help identify CAD. A combination of three echocardiographic markers showed similar diagnostic performance to traditional tests. These findings suggest that echocardiography may be a useful, non-invasive tool to detect CAD in PD, helping doctors identify PD patients at higher risk of heart complications.
Keywords
Introduction
Parkinson's disease (PD) is the second most common neurodegenerative disorder worldwide and contributes substantially to morbidity and mortality.1,2 Among non-motor complications, cardiovascular dysfunction has emerged as a leading cause of death in PD. 3 Although the conclusions remain inconclusive,4,5 several studies have reported that PD patients are at increased risk of cardiovascular complications compared with age-matched healthy controls.6–10 Notably, most cardiovascular abnormalities in PD are related to cardiovascular autonomic dysfunction (CAD), a major non-motor manifestation that affects up to 80% of patients and typically presents as orthostatic hypotension, supine hypertension, and abnormal nocturnal blood pressure patterns.11–13
While PD-related cardiovascular abnormalities are increasingly recognized as key determinants of prognosis, the underlying pathophysiological mechanisms linking PD-CAD and cardiac dysfunction remain poorly understood. Recent studies suggest that myocardial structural remodeling and functional impairment observed in PD patients may represent a mechanistic bridge between autonomic dysfunction and adverse cardiovascular outcomes.14–16 However, a knowledge gap remains between PD-CAD and specific echocardiographic alterations.
Cardiovascular autonomic reflex tests (CARTs), widely regarded as the gold standard for assessing cardiovascular autonomic dysfunction,17–21 have been extensively applied in PD-related research.22–24 In parallel, echocardiography has emerged as a sensitive and accessible imaging modality for detecting subclinical myocardial dysfunction. Integrating echocardiographic assessments with CARTs-defined CAD classification may provide valuable insight into the interplay between autonomic dysfunction and cardiac remodeling in PD.
Given the potential relationship between PD-CAD and cardiovascular abnormalities, early detection of PD-CAD is of clinical importance. However, many PD patients remain asymptomatic for long periods despite a high prevalence of cardiovascular dysfunction, highlighting the limitations of current diagnostic tools. Although MIBG cardiac scintigraphy offers high diagnostic specificity,25,26 its clinical utility is constrained by cost, radiation exposure, and limited availability. Likewise, although CARTs remain the diagnostic gold standard, their widespread clinical adoption is hindered by procedural complexity and time requirements. Additionally, markers such as orthostatic hypotension and reverse blood pressure dipping, though indicative of dysautonomia, tend to emerge in more advanced stages of CAD.22–24
This study aims to investigate echocardiographic alterations in PD patients with and without CAD, and to explore their associations with autonomic dysfunction. We hypothesize that echocardiography-derived parameters could serve a dual role: (1) as potential mechanistic markers linking autonomic impairment (PD-CAD) to myocardial injury (a precursor of cardiovascular adverse outcome); and (2) as practical, non-invasive clinical tools for the detection of PD-CAD.
Methods
Study design and populations
This cross-sectional study was conducted at Guangdong Provincial People's Hospital between November 2020 and August 2023. The enrolled PD patients constituted a subset of the Prospective Assessments with Neuroimaging and fluid Data Analysis in PD (PANDA) cohort, 27 and the inclusion criteria were as follows: (1) diagnosis by two or more experienced neurologists according to the MDS diagnostic criteria,25,28 (2) age between 40 and 80 years, (3) onset of the first motor symptoms after the age of 40. The exclusion criteria were as follows: (1) presence of cardiovascular diseases, including ischemic heart disease, cardiomyopathy, or atrial fibrillation; (2) morbid obesity (BMI ≥ 28 kg/m²); (3) comorbidities affecting autonomic function, such as diabetes, epilepsy, stroke, thyroid dysfunction, chronic kidney disease, severe anxiety, or depression; 4) use of medications that may influence autonomic function; (5) contraindications to cardiovascular autonomic reflex tests (CARTs), such as a history of proliferative retinopathy; and (6) inability to cooperate with or complete CARTs for any reason. Following CARTs evaluation, PD patients were classified into two groups: PD with cardiac autonomic dysfunction (PD-CAD) and PD without cardiac autonomic dysfunction (PD-nCAD). A total of 78 participants were ultimately enrolled in the study (Figure S1). None of the participants exhibited clinical features suggestive of atypical or familial Parkinson's disease. All evaluations were conducted during the participants’ usual medication state. A flowchart of participants included and study design is shown in Figure 1.
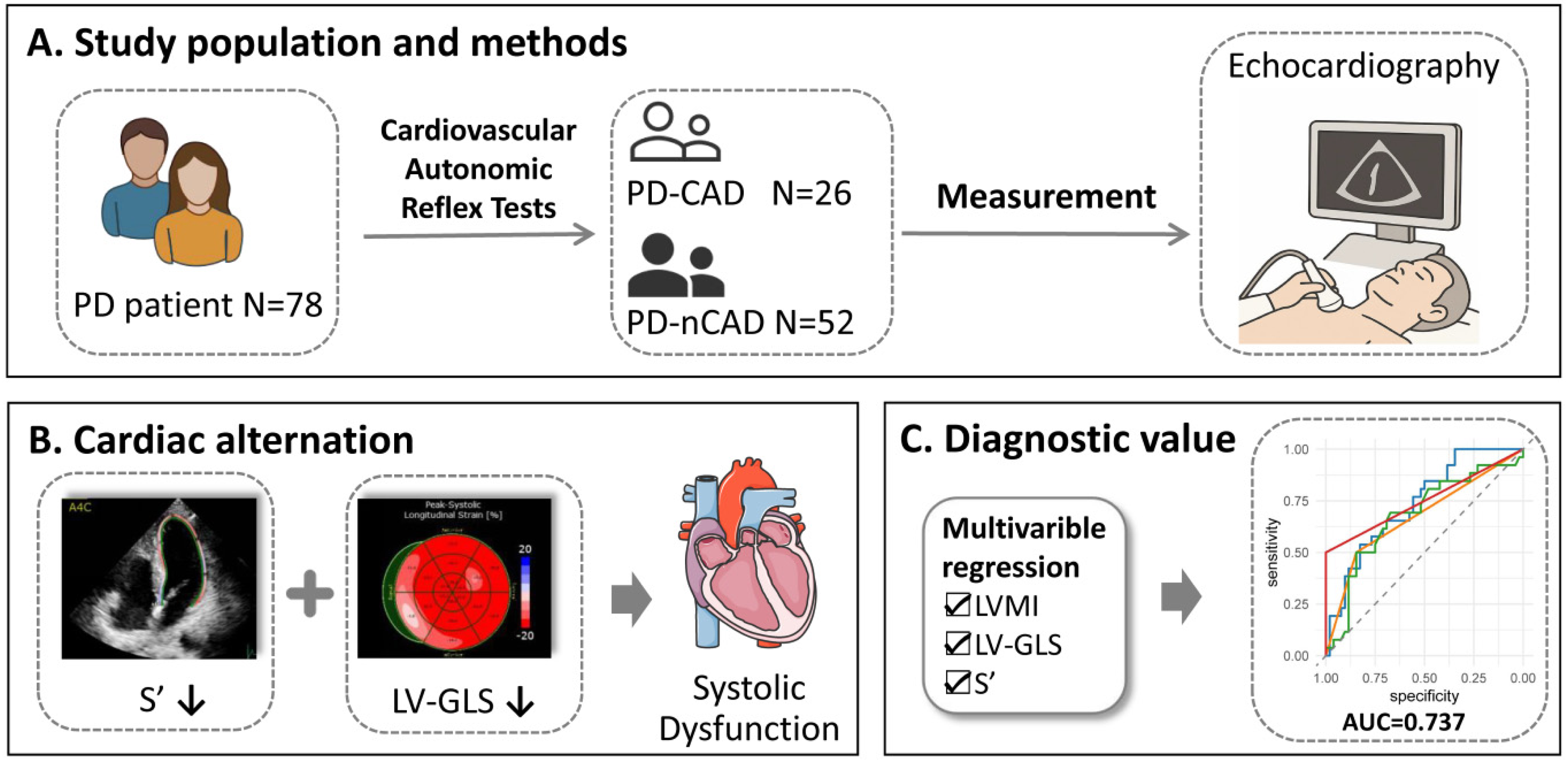
Flowchart of study design.
Written informed consent was obtained from all participants or their legal guardians before enrollment. The study protocol was approved by the Ethics Committee of Guangdong Provincial People's Hospital (No. GDREC2020187H [R1]). Additionally, the study was registered with the Chinese Clinical Trial Registry (ChiCTR2000039475).
PD patients were classified into the PD with cardiac autonomic dysfunction group (PD-CAD) based on cardiovascular autonomic reflex tests. Standard transthoracic echocardiography and two-dimensional speckle-tracking strain were performed to evaluate cardiac alterations in PD patients. Multivariable regression models were applied to analyze group differences and identify predictors of PD-CAD. Receiver operating characteristic curve analysis was used to assess the diagnostic performance of these predictors.
Clinical evaluation
Demographic and clinical data (e.g., age, sex, past medical history, body mass index) were recorded at study entry according to the study protocol. The Movement Disorder Society Unified Parkinson's Disease Rating Scale (MDS-UPDRS) part III and the Hoehn and Yahr (H-Y) stage were used to assess the motor symptoms and severity of Parkinson's disease.29,30 Levodopa equivalent daily dose (LEDD) was calculated according to standard protocols. 31 The Scales for Outcomes in Parkinson's Disease-Autonomic Questionnaire (SCOPA-AUT), Drooling Rating Scale (DRS), Swallowing Disturbance Questionnaire (SDQ) and Cleveland Clinic Incontinence Score (Wexner) were used to evaluate the autonomic symptoms. 32
Cardiovascular autonomic evaluation
Cardiovascular autonomic reflex tests
Cardiovascular autonomic function was assessed using CARTs, the current gold standard in clinical autonomic evaluation. CARTs were performed following a standard protocol and included assessments of parasympathetic function (e.g., heart rate responses to the Valsalva maneuver, deep breathing, and postural change from lying to standing) and sympathetic function (e.g., blood pressure response to standing).18,33,34 Detailed procedures are provided in Supplemental Table 1. Each test was scored as follows: 0 for normal, 1 for borderline, and 2 for abnormal. The total score of four parts was thereby the CARTs score. CAD was diagnosed when CARTs score ≥4 or abnormal result of orthostatic hypotension test was present. OH was defined as a reduction of more than 20 mm Hg in systolic and/or more than 10 mm Hg in diastolic blood pressure during the active standing test. 35
Ambulatory blood pressure monitoring
Ambulatory blood pressure monitoring was performed for 24 consecutive hours using an automatic portable device (Spacelabs 90217, Redmond, WA, USA) with readings collected every 30 min during the day (06:00–22:00) and every 60 min during the night (22:00–06:00).
Heart rate variability (HRV) analysis
A continuous 24-h ambulatory electrocardiogram was recorded using an automatic portable ECG recorder [DMS300-4AL, DM Systems (Beijing) Co, DMS]. Time-domain HRV indexes were subjected to analysis.
Echocardiographic evaluation
Standard transthoracic echocardiographic images were obtained using EPIQ 7C echocardiograph with a 2.5–3.5 MHz transducer, S5-1 (Philips Ultrasound, Bothell, WA, USA). Two-dimensional speckle-tracking strain was analyzed offline using automatic tracking of the left ventricle (LV) with QLAB (Version 13, Philips Healthcare, Andover, MA, USA) in standard apical 4-, 3- and 2-chamber views. 36 The endocardial borders were traced automatically on a frame-to-frame basis with minor artificial modification of the unsatisfied tracing.
Key measurements included the Left Ventricular Mass Index (LVMI), Left Ventricular Ejection Fraction (LVEF), Left Atrial Volume Index (LAVI), etc. Details of the calculation methods and clinical significance for these indices are outlined in sTable 2. All measurements were performed in accordance with the recommendations of the American Society of Echocardiography and the European Association of Cardiovascular Imaging. 37
Statistical analysis
All statistical analyses were performed using R software (version 4.3.3). Continuous variables were expressed as the mean ± standard deviation (SD) for normally distributed data or the median [interquartile range, IQR] for non-normally distributed data and compared using Student's t-test or the Mann-Whitney U test, as appropriate. The normality of continuous data was assessed using the Shapiro-Wilk test. Categorical variables were presented as frequencies (percentages) and were compared using χ² tests or Fisher's exact test, as appropriate. Missing data were imputed using the “MICE” package in R. The proportion of missing data for all variables was below 20%, except for LAVI (Figure S2). A two-tailed p-values < 0.05 was considered statistically significant.
Association between PD-CAD, motor symptom, LEDD and cardiac alterations
Partial correlation analysis was performed to examine the association between cardiovascular autonomic parameters and echocardiographic alterations, adjusting for age, sex. Multivariable regression analysis was performed to examine the association between cardiovascular autonomic dysfunction, orthostatic hypotension, motor symptoms (evaluated using the H-Y stage and MDS-UPDRS III scores), LEDD, and echocardiographic parameters, adjusting for age, sex, and mean SBP. As a sensitivity analysis, we additionally adjusted for the use of antihypertensive medications (Supplemental Table 4).
Diagnostic value of echocardiography-derived parameters for PD-CAD
Furthermore, we investigated the diagnostic value of echocardiography-derived parameters for PD-CAD. To identify significant echocardiographic predictors of risk of PD-CAD, candidate predictors were selected from echocardiographic parameters with p-values below 0.10 during univariate logistic regression. These candidate variables were incorporated as potential covariates in multivariable logistic regression models.
To ensure model efficiency and prevent overfitting, a stepwise bidirectional selection approach was applied, and analyses were conducted on 1000 bootstrapped datasets of the same size as the original dataset. Multicollinearity among candidate variables was assessed using variance inflation factor (VIF) analysis and correlation analysis. All VIF values were below 5.
To evaluate the diagnostic accuracy of these predictors, we utilized Receiver Operating Characteristic (ROC) curves, considering an AUC value above 0.70 as indicative of good diagnostic performance. The DeLong test was employed to compare differences between the ROC curves.
To further evaluate the diagnostic performance of these predictors, we compared them to other established markers of CAD, including reverse dipping, the standard deviation of all normal-to-normal intervals (SDNN), and orthostatic hypotension (OH), which were selected based on previous literature and a stepwise bidirectional selection strategy in the multivariable logistic regression model.24,38–40 The overall model fit was compared using the Akaike Information Criterion (AIC). Discrimination ability was assessed using the C-statistic and integrated discrimination improvement (IDI), while reclassification performance was evaluated using both the continuous and categorical net reclassification index (NRI). Risk thresholds for the outcome were set at <33.33% and ≥33.33%, corresponding to the prevalence of PD-CAD in our study cohort and previous research findings.
Results
Demographic and clinical characteristics
A total of 78 participants were included in this study, with a median Hoehn and Yahr stage of 2.00 [IQR 2.00, 2.50] and a median LEDD of 212.50 mg [IQR 81.25, 568.00]; among them, 26 (33.3%) were diagnosed with PD-CAD. Compared to those without CAD, patients in the PD-CAD group were older and had a later disease onset, whereas the duration of PD did not differ significantly between groups. Additionally, there was no significant difference in the prevalence of hypertension between the two groups. The PD-CAD group exhibited significantly higher SCOPA-AUT and Wexner scores, indicating more severe autonomic dysfunction and constipation symptoms. However, no significant differences were observed in MDS-UPDRS III scores or Hoehn-Yahr stage. Detailed characteristics of the participants are presented in Table 1.
Basic characteristics and cardiovascular evaluation of study population.
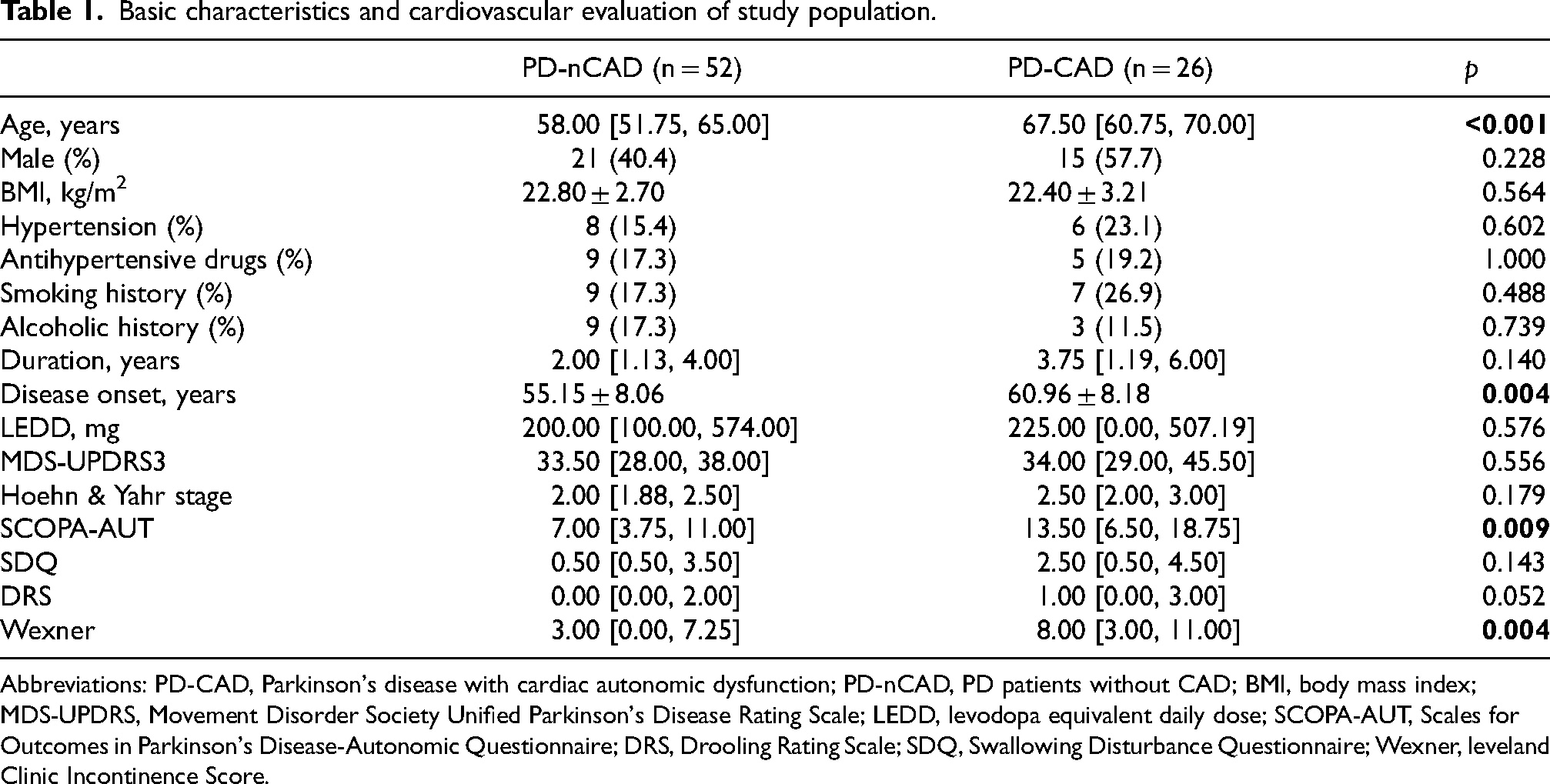
Abbreviations: PD-CAD, Parkinson's disease with cardiac autonomic dysfunction; PD-nCAD, PD patients without CAD; BMI, body mass index; MDS-UPDRS, Movement Disorder Society Unified Parkinson's Disease Rating Scale; LEDD, levodopa equivalent daily dose; SCOPA-AUT, Scales for Outcomes in Parkinson's Disease-Autonomic Questionnaire; DRS, Drooling Rating Scale; SDQ, Swallowing Disturbance Questionnaire; Wexner, leveland Clinic Incontinence Score.
Characteristics of cardiovascular autonomic evaluation
Detailed characteristics of cardiovascular autonomic evaluation in the study population are presented in Table 2. Compared to participants in the PD-nCAD group, those in the PD-CAD group exhibited lower Valsalva ratio, heart rate response to deep breathing, and 30 s/15 s ratio, as well as a higher systolic blood pressure response to orthostatic change during the CARTs test. Ambulatory blood pressure monitoring revealed a significantly higher nocturnal SBP in the PD-CAD group. Furthermore, analysis of blood pressure patterns showed a significantly greater proportion of reverse dippers among patients with PD-CAD. Regarding heart rate variability, the PD-CAD group exhibited significantly lower SDNN, SDANN, and SDNN index, whereas rMSSD and pNN50 did not differ significantly between groups.
Cardiovascular evaluation of study population.

Abbreviations: PD-CAD, Parkinson's disease with cardiac autonomic dysfunction; PD-nCAD, PD patients without CAD; ΔHR, Heart response to deep breathing; 30 s/15 s ratio, the ratio of the longest RR interval in the (30 ± 5)th heartbeat to the shortest RR interval in the (15 ± 5)th heartbeat after standing; ΔSBP, SBP response to orthostatic change; OH, orthostatic hypotension; DND: day-night difference; SDNN, SD of all normal-to-normal intervals; SDANN, SD of the average NN intervals calculated over 5-min periods of the entire recording; rMSSD, the root mean square successive difference of the RR interval; pNN50, percentage of adjacent RR intervals with a difference of duration greater 50 ms.
Association between PD-CAD and cardiac alterations
Partial correlation analysis showed that PD-CAD and OH were negatively correlated with systolic mitral annular velocity (s’), and positively correlated with left ventricular end-diastolic volume index (LVEDVI) and global longitudinal strain (LV-GLS). Mean and daytime blood pressure were negatively associated with LVEDVI, left ventricular end-systolic volume index (LVESVI), and early diastolic mitral annular velocity (e′), but positively associated with the ratio of early diastolic mitral inflow velocity to annular velocity (E/e′) (Supplemental Figure 4).
The association between PD-CAD and cardiac structural and functional parameters in the study population are summarized in Figure 2 and Supplemental Table 3 and 4. Regarding structural parameters, patients with PD-CAD exhibited a higher left ventricular mass index (LVMI) and lower LVEDVI compared to those in the PD-nCAD group. However, these differences lost statistical significance after multivariate adjustment for age, sex, and mean SBP.
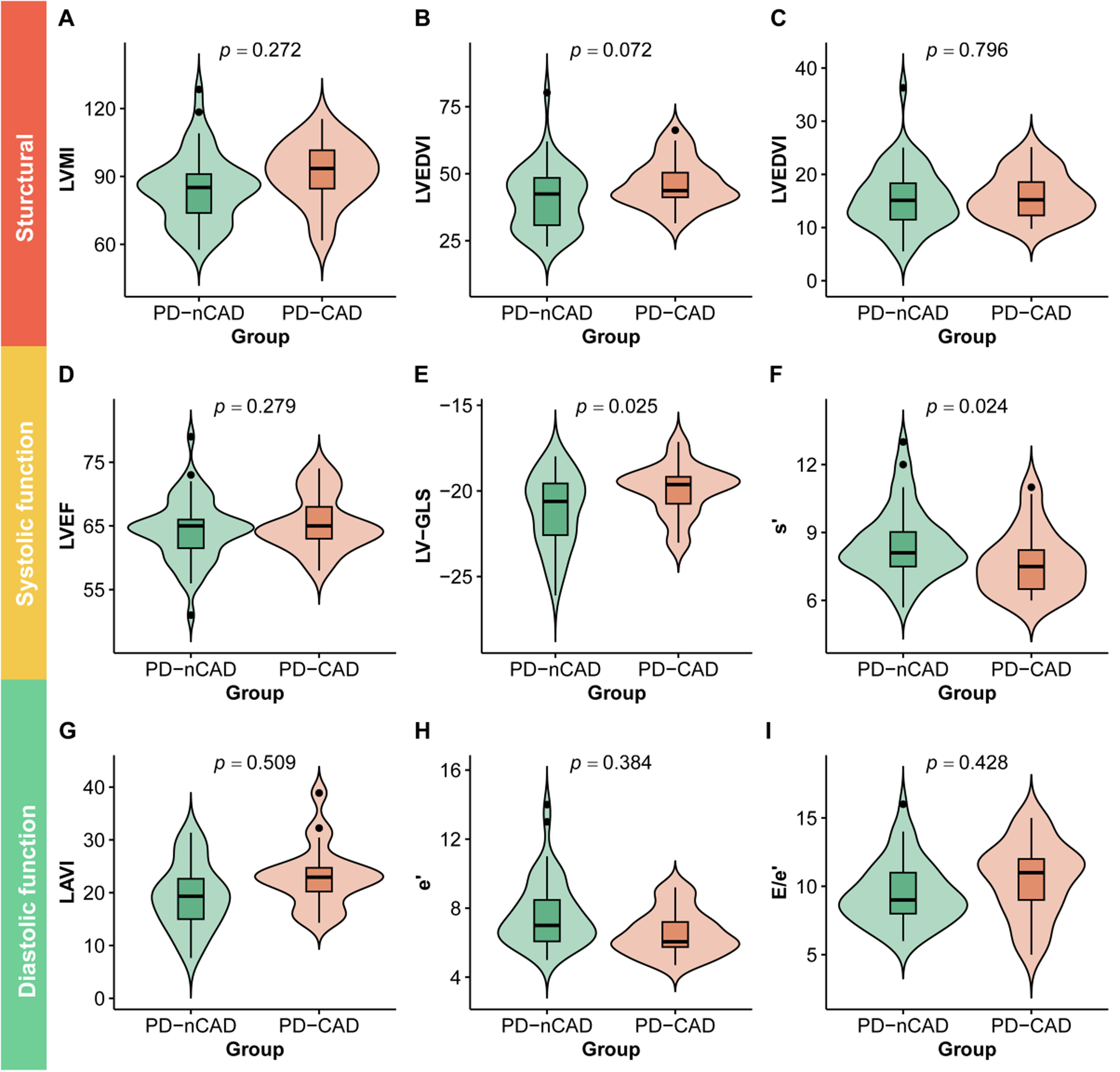
Association between PD-CAD and cardiac alterations.
In the assessment of left ventricular systolic function, there was no significant difference in left ventricular ejection fraction (LVEF) between two groups. However, patients with PD-CAD exhibited a significantly reduced LV-GLS and slower s’, both of which remained statistically significant after multivariate analysis.
Regarding left ventricular diastolic function, compared to the PD-nCAD group, patients with PD-CAD demonstrated a reduced early diastolic mitral annular velocity (e’), higher left atrial volume index (LAVI) and higher ratio of early diastolic mitral velocity to early diastolic mitral annular velocity (E/e’). However, these differences were no longer statistically significant after multivariate adjustment.
Additionally, we analysis the echocardiographic parameters of PD-CAD patients with and without orthostatic hypotension (OH). As shown in Supplemental Figure 4, the PD-CAD with OH group exhibited a lower LVEF compared to those without OH.
Association between motor symptom, LEDD and cardiac alterations
The associations between PD motor symptoms, LEDD, and cardiac structural and functional parameters are summarized in Table 3 and Supplemental Table 4. Neither motor symptom severity, as assessed by the H-Y stage and MDS-UPDRS III, nor LEDD showed a significant association with cardiac alterations.
Association between motor symptoms, LEDD and cardiac alterations.
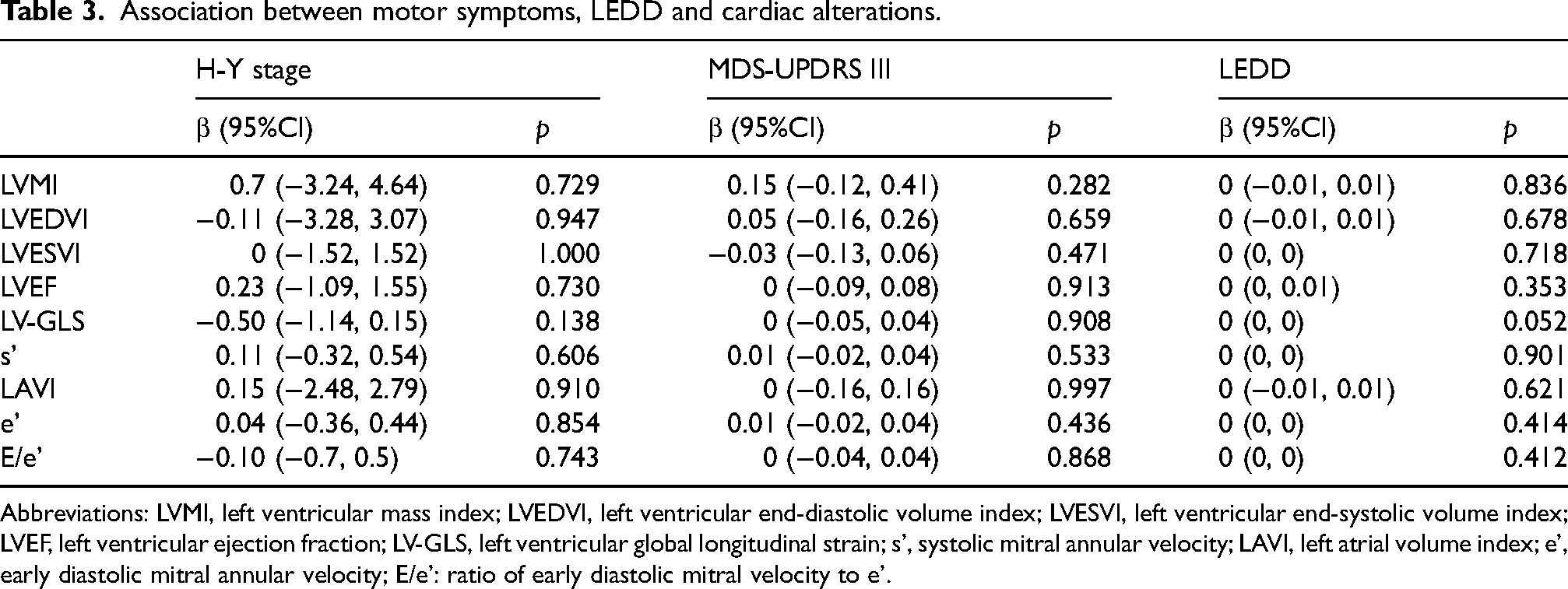
Abbreviations: LVMI, left ventricular mass index; LVEDVI, left ventricular end-diastolic volume index; LVESVI, left ventricular end-systolic volume index; LVEF, left ventricular ejection fraction; LV-GLS, left ventricular global longitudinal strain; s’, systolic mitral annular velocity; LAVI, left atrial volume index; e’, early diastolic mitral annular velocity; E/e': ratio of early diastolic mitral velocity to e’.
Diagnostic value of echocardiography-derived parameters for PD-CAD
Following multivariate analysis and assessment of multicollinearity, three echocardiography-derived parameters were identified as significant predictors of an increased risk of PD-CAD: reduced LV-GLS, slower s’, and higher LVMI. These parameters collectively explained 22.4% of the variance (R²= 0.224) in PD-CAD risk. Receiver operating characteristic curve analysis demonstrated that these three parameters had a C-index of 0.737 (95% CI, 0.624 to 0.850), indicating good discriminatory ability (Figure 3). Furthermore, when compared to other predictive parameters for PD-CAD, these echocardiographic markers exhibited comparable diagnostic performance, as reflected by similar C-index values, integrated discrimination improvement (IDI), and net reclassification improvement (NRI) (Table 4 and Supplemental Tabled 5 to 8).
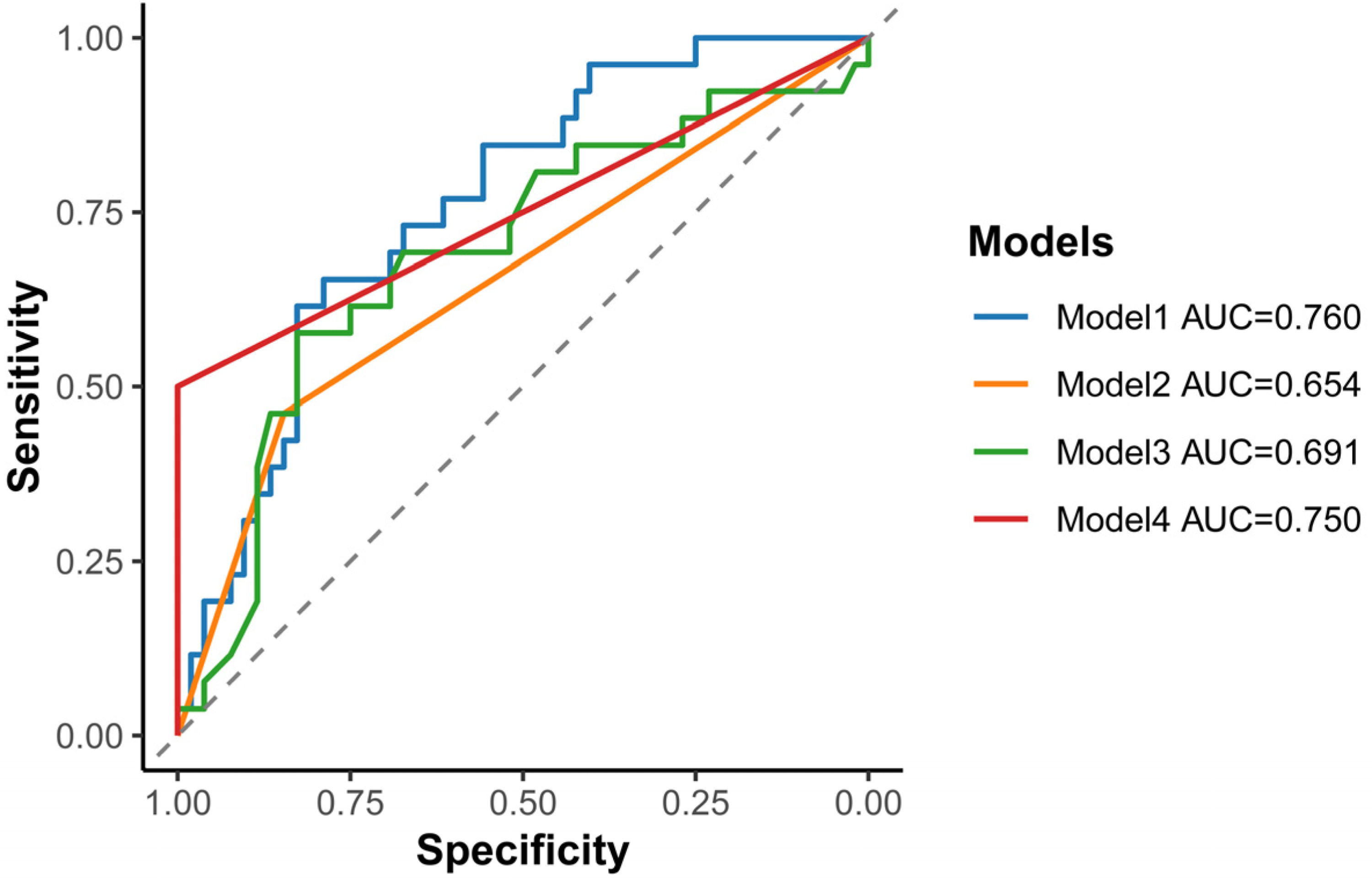
Diagnostic value of echocardiography-derived parameters for PD-CAD.
Diagnostic performance for PD-CAD.
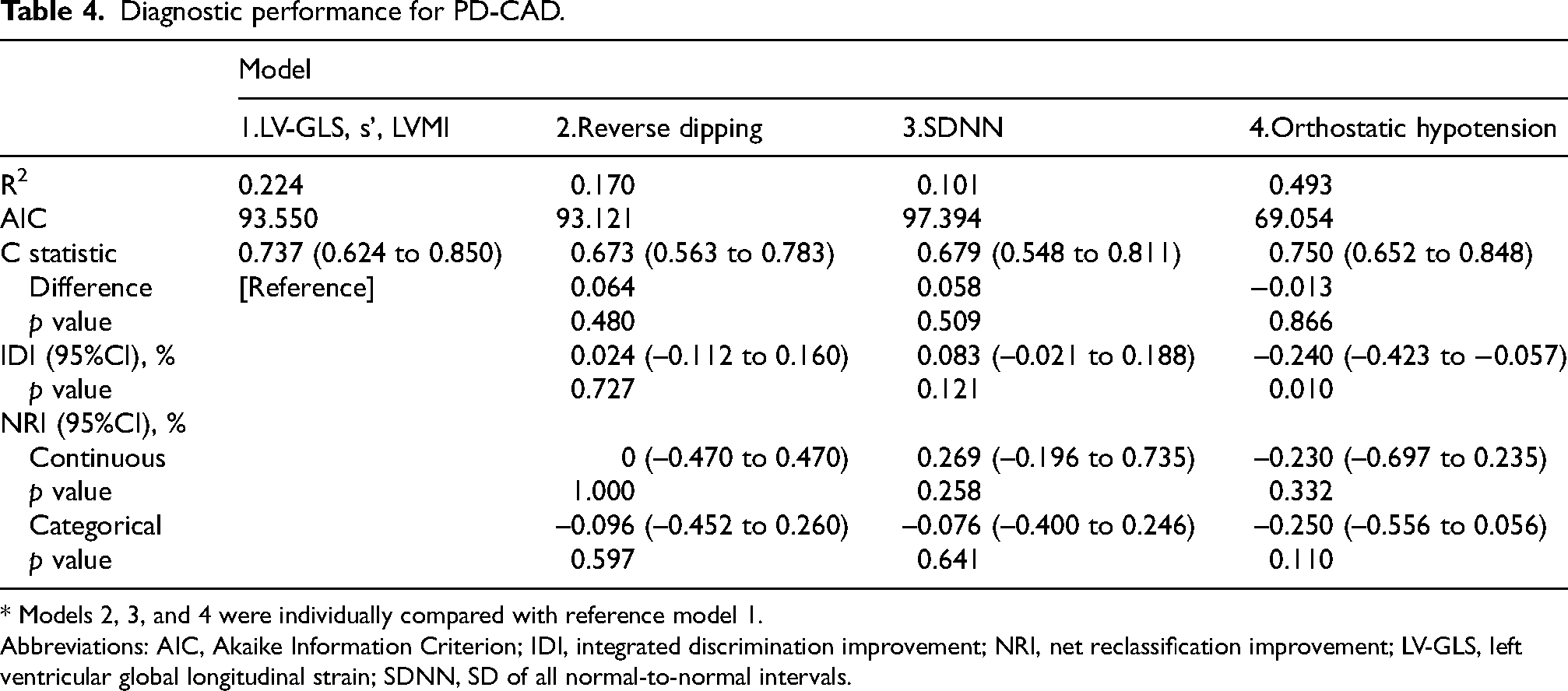
* Models 2, 3, and 4 were individually compared with reference model 1.
Abbreviations: AIC, Akaike Information Criterion; IDI, integrated discrimination improvement; NRI, net reclassification improvement; LV-GLS, left ventricular global longitudinal strain; SDNN, SD of all normal-to-normal intervals.
Discussion
This study aimed to elucidate the relationship between PD-CAD and myocardial structural and functional alterations as evaluated by echocardiography. Furthermore, we evaluated the diagnostic utility of echocardiographic parameters in detecting PD-CAD. Our findings support two key hypotheses: (1) echocardiographic measures reflect autonomic-related myocardial injury, potentially serving as mechanistic links between PD and adverse cardiovascular outcomes; and (2) these parameters may have potential value in future cardiovascular assessment frameworks.
Our analysis of 78 PD patients revealed that patients with PD-CAD exhibited impairments in left ventricular systolic function as evidenced by reduced left ventricular global longitudinal strain (LV-GLS) and lower systolic mitral annular velocity (s’), independent of traditional cardiovascular risk factors. Although an initial increase in the left ventricular mass index (LVMI) was observed in PD-CAD patients, consistent with the results of previous studies,14–16 this difference became non-significant after adjusting for confounders such as age, sex, and mean SBP. Importantly, no significant associations were observed between cardiac changes and motor symptom severity, as measured by H-Y stage or MDS-UPDRS III, nor with LEDD, suggesting that myocardial dysfunction may occur independently of motor progression or medication exposure.
Prior studies have reported left ventricular systolic dysfunction in PD patients. 41 Our findings further bridge this observation with cardiovascular autonomic dysfunction, as evidenced by reduced LV-GLS and lower s’, sensitive markers for detecting early myocardial impairment and related to cardiac adverse events, even when LVEF remains within the normal range.42–45 Sympathetic overactivity and parasympathetic withdrawal, frequently seen in early-stage of PD, may drive myocardial remodeling via mechanisms such as increased catecholamine exposure, oxidative stress, and microvascular dysfunction.46–50 Early sympathetic hyperactivation can lead to excessive norepinephrine release, triggering both structural and functional myocardial alterations. Of note, our exploratory subgroup analysis revealed that orthostatic hypotension, a clinical marker of sympathetic dysfunction typically observed in later disease stages, was associated with reduced LVEF among PD-CAD patients. This finding suggests a possible transition from early-stage compensatory sympathetic activation to overt sympathetic failure, which may further exacerbate systolic dysfunction. These pathophysiological processes mirror those seen in other forms of autonomic dysfunction and hypertensive cardiomyopathy. Notably, the absence of differences in hypertension history and the persistence of associations after adjustment for key covariates suggest a shared mechanistic pathway, predisposing PD patients to cardiovascular complications.
The lack of a significant difference in LVMI between PD-CAD and PD-nCAD groups after multivariable adjustment warrants further discussion. LVMI elevation is commonly linked to chronic pressure overload and adverse remodeling, its non-significance in our cohort may reflect the early stage of myocardial remodeling in these patients. Most participants were at earlier PD stages with relatively short disease duration, potentially precluding the development of overt structural changes. In this context, functional indices such as LV-GLS and s’ may be more sensitive than structural metrics in detecting early myocardial involvement.
Although there were trends toward altered diastolic parameters (e.g., lower early diastolic mitral annular velocity and higher left atrial volume index) in the PD-CAD group, these differences were not statistically significant after adjustment. While Piqueras-Flores et al. reported diastolic left ventricular dysfunction in PD patients, 15 none of our participants met the diagnostic criteria for left ventricular diastolic dysfunction based on the 2016 ASE recommendations. 51 This finding aligns with results from other studies employing rigorous exclusion criteria. 14 Taken together, this may indicate that diastolic dysfunction emerges later in the course of autonomic-induced remodeling or may have been mitigated by our exclusion of patients with overt cardiovascular disease. These observations underscore the need for longitudinal studies to delineate the temporal evolution from subclinical systolic dysfunction to overt diastolic impairment in PD.
Our study corroborates and extends previous reports linking PD with cardiovascular autonomic dysfunction. 41 Previous studies have identified autonomic dysfunction, including reduced heart rate variability and orthostatic hypotension, as risk factors for cardiovascular morbidity and mortality. Building on this knowledge, our study is among the first to combine echocardiographic measures with CARTs-defined autonomic profiles in PD, providing a more comprehensive evaluation of the interplay between autonomic impairment and myocardial performance. Furthermore, our study incorporated a comprehensive cardiovascular evaluation including ambulatory blood pressure monitoring and heart rate variability analysis, which provided a detailed view of the autonomic profile of these patients. Notably, the PD-CAD group exhibited higher nocturnal SBP, a greater prevalence of reverse dipping, and reduced heart rate variability, all of which are linked to elevated cardiovascular risk.
Previous evidence has suggested, although not widely conclusive, an increased cardiovascular risk in patients with PD.6–10 Consistent with these observations, our findings support an association between PD-CAD and adverse subclinical cardiac alterations in PD, highlighting the need to enhance clinical awareness and promote comprehensive assessment of cardiovascular autonomic dysfunction in this population. Through multivariable regression analysis and logistic modeling, we identified reduced LV-GLS, lower s’, and higher LVMI as significant predictors of PD-CAD. These three echocardiographic parameters yielded a C-index of 0.737, indicating diagnostic performance comparable to established predictive markers. It should be noted that they explained only 22.4% of the variance, suggesting that other unmeasured factors may also contribute to CAD. Nevertheless, their non-invasive nature and accessibility suggest a potential role in future multimodal diagnostic frameworks. These results not only highlight the potential clinical utility of echocardiographic markers for evaluating PD-CAD, but also underscores the value of combining echocardiographic and autonomic assessments to capture the multifaceted nature of cardiovascular involvement in PD.
To our knowledge, this study is the first to assess the diagnostic potential of echocardiographic markers for PD-CAD. The predictor variables included in these diagnostic models have been previously proved to be associated with PD-CAD. Importantly, reduced LV-GLS may not only aid in diagnosis but also serve as a marker of cardiovascular risk stratification, mirroring its prognostic role in broader cardiovascular diseases. These parameters are easily accessible during hospitalization or outpatient consultations, thereby facilitating their integration into comprehensive assessment of cardiovascular autonomic dysfunction in PD. Identification of such parameters could help guide prognostic assessment and subsequent clinical follow-up. Previous studies have demonstrated the prognostic value of GLS and explored the potential benefits of early interventions (e.g., β-blockers, ACE inhibitors) based on GLS in populations with cancer therapeutics-related cardiac dysfunction.52–54 However, specific research in the PD population is still lacking.
This study has several limitations. First, although LV-GLS and s’ have been associated with adverse cardiovascular events in the general population,42,55 their predictive value specifically in PD patients remains uncertain and requires further validation. Longitudinal analysis in the present study is limited, as the first follow-up of the PANDA cohort is still ongoing. Consequently, the cross-sectional findings may not suggest the causal relationships between autonomic dysfunction, cardiac alterations, and adverse cardiovascular outcomes. Second, the strict inclusion criteria, while minimizing confounding factors that might obscure the interpretation of disease-specific pathophysiological mechanisms, may have introduced selection bias, limiting the generalizability of our results to PD patients with comorbid cardiovascular diseases. Although age was adjusted for in the multivariable analyses, this cannot completely eliminate the potential contribution of physiological aging or other non-specific factors. Therefore, future studies in larger and more representative populations are warranted to validate these findings. Additionally, the absence of direct measurements of catecholamine levels or other biomarkers of autonomic activity limits our ability to mechanistically link the observed echocardiographic changes to the underlying pathophysiological processes. The inclusion of a healthy control group in future studies would also provide a more comprehensive understanding of these relationships. Finally, although antihypertensive medications were considered in sensitivity analyses, the potential influence of other medications with cardiovascular effects, such as nonsteroidal anti-inflammatory drugs (NSAIDs) or certain antibiotics, was not systematically assessed, which may have introduced residual confounding. Since medication state was not standardized during assessments, minor effects of dopaminergic medication cannot be entirely excluded.
Conclusions
In conclusion, our study revealed that PD patients with cardiovascular autonomic dysfunction exhibit subclinical myocardial dysfunction, as evidenced by reduced LV-GLS and slower s’, despite preserved LVEF. These echocardiographic alterations, alongside other autonomic measures, underscore the complex interplay between autonomic dysregulation and myocardial remodeling in PD. Additionally, echocardiography has the potential to be a valuable tool for identification of PD-CAD.
Supplemental Material
sj-docx-1-pkn-10.1177_1877718X251405816 - Supplemental material for Echocardiographic insights into cardiovascular autonomic dysfunction in Parkinson's disease
Supplemental material, sj-docx-1-pkn-10.1177_1877718X251405816 for Echocardiographic insights into cardiovascular autonomic dysfunction in Parkinson's disease by Zihao Li, Xiaohong Li, Wenlin Huang, Ziqi Gao, Piao Zhang, Chentao He, Siming Rong, Mengfei Cai, Zhenzhen Chen, Yan Li, Ruixue Xu, Lijuan Wang, Zhichao Zheng, Hongwen Fei and Yuhu Zhang in Journal of Parkinson's Disease
Footnotes
Acknowledgments
This work was supported by National Natural Science Foundation of China (No.82071419, No.82301663, No.82301420); Key Research and Development Program of Guangzhou (No.202206010086); Science and Technology Planning Project of Guangzhou (No.202201000005); China Postdoctoral Science Foundation (No.2023M730742). The funder played no role in study design, data collection, analysis and interpretation of data, or the writing of this manuscript. We thank Ms. Yunlian Xue for her professional advice on statistical analysis and all patients for their cooperation in this study.
Author's Contributions
Xiaohong Li, Zihao Li, Yuhu Zhang designed and organized this study.
Wenlin Huang, Ziqi Gao, Chentao He, Zhenzhen Chen assisted with patient enrollment and clinical data collection.
Piao Zhang, Yan Li assisted with statistical analysis.
Ruixue Xu, Hongwen Fei, Zhichao Zheng were responsible for echocardiographic image collection, processing and analysis.
Zihao Li, Xiaohong Li wrote the first draft.
Yuhu Zhang, Siming Rong, Mengfei Cai revised the manuscript.
Yuhu Zhang, Lijuan Wang approved the final submission of the article.
All authors contributed to the article and approved the submitted version.
Zihao Li and Xiaohong Li contributed equally to this work.
Yuhu Zhang, Hongwen Fei and Zhichao Zheng contributed equally to this work.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Natural Science Foundation of China, (grant number No.82071419, No.82301663, No.82301420); Key Research and Development Program of Guangzhou (No.202206010086); Science and Technology Planning Project of Guangzhou (No.202201000005); China Postdoctoral Science Foundation (No.2023M730742).
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability
The datasets analysed during the current study are available from the corresponding author on reasonable request.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
