Abstract
Bradykinesia and delayed step initiation are central features of Parkinson's disease (PD) and have been linked to reduced movement vigor. Evidence suggests that externally imposed urgency may upregulate vigor and improve motor speed. Previous home-based cueing interventions without time limits improved gait but failed to reduce reaction times, indicating that time constraints may be critical. We conducted a randomized controlled trial to evaluate whether time-constrained, visually guided reaching and stepping exercises enhance response speed in PD. Sixty participants with idiopathic PD (Hoehn & Yahr I—III) completed 24 supervised, home-based sessions using a set of small interactive devices that provided visual and auditory cues and recorded response times. The experimental group trained with time windows adapted during each session to maintain ∼80% success, receiving immediate success/failure feedback, while the control group performed identical tasks without time restrictions. Response times were assessed before and after training with choice stepping and reaching reaction time tasks in a laboratory setting, alongside gait and mobility tests. Repeated measures ANOVA revealed significant group × time interactions favoring the experimental group for both reaching (p < 0.05) and stepping (p < 0.001) reaction times, with medium-to-large effect sizes, whereas controls showed no change. Gait speed improved in both groups, with no additional between-group differences. These findings demonstrate that introducing temporal urgency during reactive training produces meaningful improvements in bradykinesia-related response times in individuals with PD, supporting the integration of time-constrained cueing into scalable home-based rehabilitation. ClinicalTrials.gov: NCT05829915
Plain language summary
People with Parkinson's disease often move slowly and have trouble starting actions such as reaching for an object or taking the first step. One reason may be that the brain does not create enough urgency to move quickly. Setting short time limits can create this urgency and encourage faster movement. In this study, we tested a home-based exercise program using small devices that gave light and sound signals. When a light appeared, participants had to touch it with their hand or step toward it. Everyone trained several times a week. The key difference was that in one group the time allowed to touch the target was shortened during training so that people succeeded about 80% of the time. The other group did the same exercises without time pressure. We found that participants who trained with time limits became faster at both reaching with the hand and starting a step. Those who trained without time limits did not improve movement speed. These results show that adding short, achievable time limits to simple exercises can help people with Parkinson's disease move faster. This approach is practical, safe, and can be done at home, making it a promising tool for rehabilitation and everyday life.
Introduction
Bradykinesia, defined as the slowness of voluntary movement, is one of the cardinal motor signs of Parkinson's disease (PD). 1 Traditionally, it has been characterized by slowness combined with a progressive reduction in movement amplitude or speed during repetitive actions–the sequence effect. 2 This slowness of movement manifests itself in both distal and proximal muscles, affecting basic movements such as reaching and stepping.3,4
Beyond pure motor execution limitations, several studies suggest that bradykinesia is in part a deficit in the modulation of movement vigor–that is, in the internal cost–benefit computation that determines how energetically and quickly to act given perceived reward and urgency.5–7 In PD, dopaminergic dysfunction appears to blunt the assignment of urgency or incentive to fast movements, leading to slower movement even when mechanical capability exists, 8 a perspective that opens avenues to modify behavior by manipulating temporal or motivational context.
Cueing-based interventions, in which people with PD perform goal-directed movements in response to external sensory stimuli, have shown beneficial effects on gait parameters such as step length, speed, and cadence in PD.9–12 In addition, a recent work combining reactive (slips/trips) and volitional step training in PD improved laboratory balance recovery and shortened choice-stepping reaction times. 13 However, their impact on bradykinesia in upper limb tasks and stepping exercises has been inconsistent across studies. In a recent home-based training program using reactive visual cues, people with PD performed reaching and stepping movements triggered by randomly appearing lights on an interactive device. While this protocol improved gait parameters, it failed to produce significant changes in bradykinesia-related measures for arm reaching or stepping. 14 A key limitation of that program was that the visual cues remained active until the participant responded, removing any temporal constraint and, thus, any externally imposed sense of urgency.
The present study aimed to evaluate whether introducing a temporal limitation on cue availability—that is, requiring the participant to respond before the visual stimulus disappears—enhances movement vigor and improves bradykinesia in reaching and stepping tasks. We implemented a controlled comparison in which one group repeats the original reactive visual training protocol (with persistent cues) and a second group performs the identical exercise regimen except that each imperative visual cue is presented for a restricted time window, inducing an externally imposed urgency. We hypothesized that the time-constrained cueing will induce faster responses by increasing perceived urgency and engaging motivational-motor pathways, thereby yielding greater improvements in bradykinesia than the original protocol.
Methods
Study design and setting
This study was a randomized controlled trial. The intervention sessions were conducted in the participants’ own homes under the supervision of exercise professionals specialized in chronic conditions. All assessments were performed at the Motor Control Laboratory of the Universidad Rey Juan Carlos (Madrid, Spain) following standardized protocols. Outcome assessors were blinded to group allocation to minimize the risk of measurement bias. This study was registered as clinical trial (ClinicalTrials.gov Identifier: NCT05829915). This study was conducted in full compliance with the Declaration of Helsinki 1964 (updated in Fortaleza, 2013) and approved by the Local Ethics Committee.
Participants
Seventy-five individuals with idiopathic PD were recruited from several local Parkinson's associations in Madrid, Spain and were assessed for eligibility (Figure 1). A fundamental inclusion criterion was the ability to walk independently >30 m without the use of walking aids or external assistance. Participants were excluded if they had a history of neurological conditions other than PD, orthopedic or visual disturbance that affected walking ability or the interaction with the electronic devices required to perform the intervention. Sixty-five individuals with PD were selected, and 60 completed the study (28 in the experimental group and 32 in the control group).
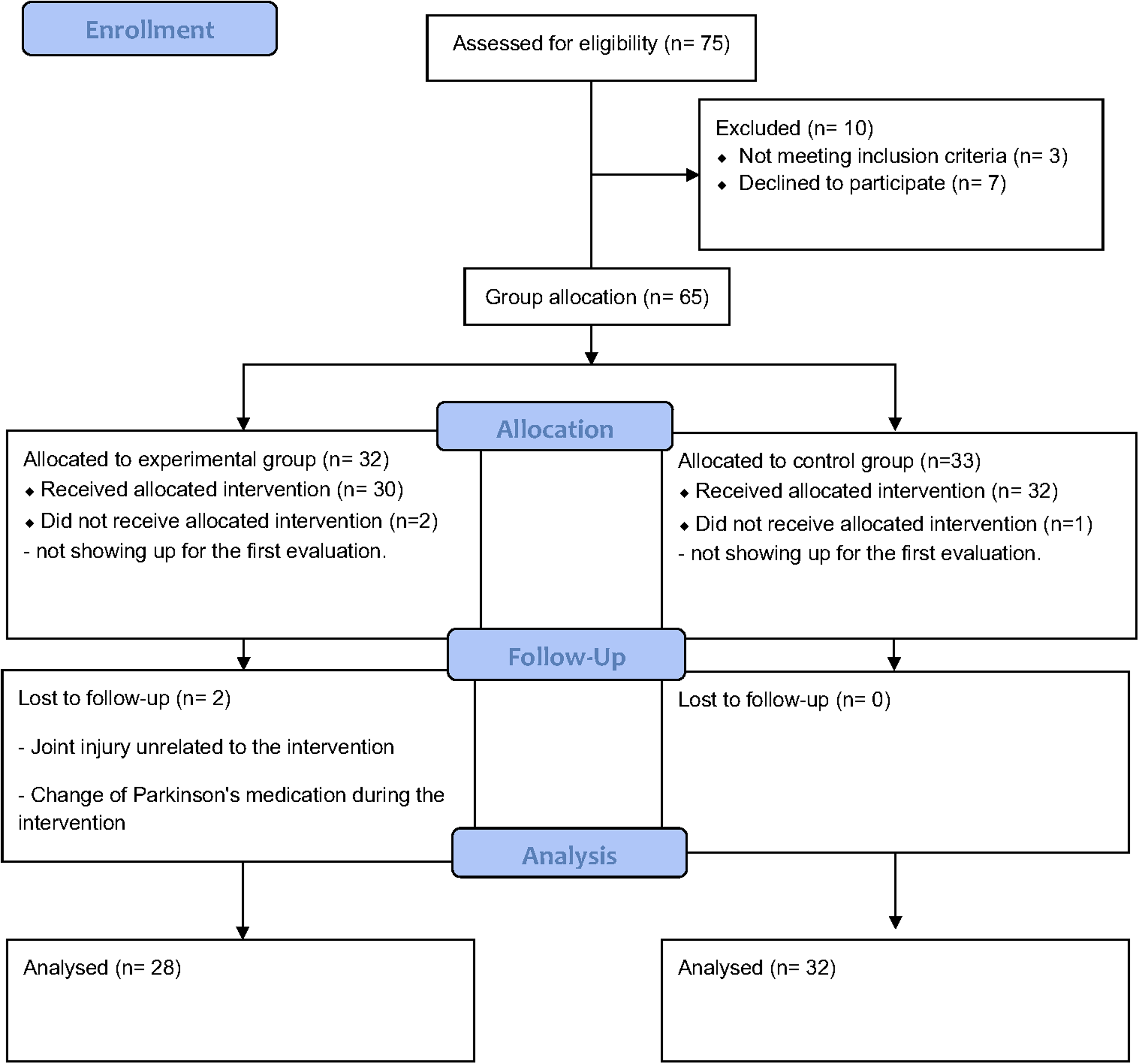
CONSORT flow diagram of participant recruitment, allocation, follow-up, and analysis. Reasons for exclusion and loss to follow-up are detailed for each group.
Motor severity was evaluated using the Movement Disorder Society–Unified Parkinson's Disease Rating Scale, Part III (MDS-UPDRS-III), and disease stages were classified according to the Hoehn and Yahr (H&Y) scale. Cognitive assessment was carried out by the Montreal Cognitive Assessment (MoCA). Eligibility required the capacity to provide informed consent and understand brief task instructions. Assessments were scheduled in the ON-medication state, 1.0–2.5 h after the participants’ usual antiparkinsonian medication (median 1.5 h), aligned with each participant's peak clinical effect as confirmed by the clinician and participant report. Table 1 shows the main characteristics of the participants in both groups. All participants provided written informed consent prior to enrollment.
Demographic data by group.

Participants were randomly assigned to an experimental group or control group, using a computer-generated randomization sequence with concealed allocation. All participants performed a reactive exercise program (intervention). Before (Pre) and after (Post) the program, subjects were evaluated on a battery of motor tests implemented by evaluators blind to subject allocation.
Intervention
The intervention consisted of 25 sessions (5/week) of reactive exercise program carried out in the subject's own home. The training program was developed by a multidisciplinary team of researchers and clinicians with extensive experience in PD and neurorehabilitation and implemented with mimo.fit system (https://mimo.fit). Mimo.fit is a set of small bluetooth-connected light/sensor “pods” that emit light cues and a mobile app that records the time of movement between the appearance of the stimulus and contact with the device. Through its mobile application, it allows the development of numerous sensory-cognitive-motor exercises adapted to the characteristics of the subjects. The training program included a variety of exercises in which the objective was always to touch a visual target using upper limb reaching movements (Figure 2(A)) or stepping (Figure 2(B)). A progression of increasing difficulty in the exercises was established by modifying parameters such as the distance from the targets, the number of targets, the presence of distracting stimuli, or the subject's orientation relative to the targets.

Examples of a reaching task (A) and a stepping task (B). In both, participants perform a simple reaction task: a single pod lights up and they respond by touching/stepping on it as quickly as possible.
All participants performed the same type of exercise, number of sets, and repetitions. The duration of the sessions varied between 25 and 45 min depending on the participant's functional capacity.
In the control group
The intervention conducted in the control group has previously demonstrated efficacy in improving gait parameters when applied independently by people with PD in unsupervised home-based settings. 14 In the present study, however, the intervention for both groups were delivered in person under the supervision of university-trained exercise specialists with a Master's degree in clinical exercise physiology (exercise for chronic conditions) and experience in neurorehabilitation. This supervised format was adopted for two main reasons: (1) to facilitate the inclusion of participants who may face technological barriers to autonomous device use, and (2) to allow for real-time individual adjustment of the response time window in the experimental group, ensuring that the time-limited stimuli remained appropriately challenging and personalized throughout the intervention.
Study outcome measures
The primary outcome was the change in response time in two tests–choice stepping reaction time and choice arm reaching reaction time–from pre- to post-intervention.
Choice stepping reaction time (CRTstepping)
This test was adapted from Lord and Fitzpatrick 2001. 16 In the original protocol subjects stood on a nonslip black platform (0.8 m x 0.8 m) that contained four rectangular panels (32 cm x 13 cm) one in front of each foot and one to the side of each foot. In our study we replaced the four rectangular panels for four electronic devices (mimo.fit). These devices are shaped as squares with rounded corners, measuring 10 cm in width and length, and 4 cm in height. Subjects stood with their feet 10 cm apart and in line with the two side devices. Subjects were instructed to touch their foot to the illuminated device as quickly as possible, using the left foot only for the two left devices (front and side) and the right foot only for the two right devices. The devices were connected to a mobile application and contained sensors to record in milliseconds the response time from the moment of illumination to the moment of foot contact. The subjects had a pair of familiarization trials with 10 stimuli each. Next, and after a brief rest period, the test was carried out, consisting of a single attempt of 20 stimuli presented in a semi-random manner to obtain 5 responses per device. Response time was measured as the time between illumination of a device and foot contact with it, and the average time of the 20 trials was used in the analysis
Choice arm reaching reaction time (CRTreaching)
This test consisted of an adaptation of the CSRT where the subject sitting in a chair had to touch the light devices placed on a table in front of the subject and distributed in the same way as in the CSRT. The subject had to position his two hands between and in line with the lateral devices. Like the CSRT, the subjects had to touch only the devices on the left side (front and side) with their left hand and the right hand only for the two right devices. After two practice blocks, a single block of 20 trials distributed equally among the 4 devices was recorded. Response time was measured as the time between illumination of a device and hand contact with it, and the average time of the 20 trials was used in the analysis.
Secondary measures included kinematic parameters of gait performed at preferred and maximum speed (speed /m/s), amplitude (m) and cadence (steps/minute) and the time to complete the up & go test.
Timed Up and Go (TUG)
The TUG test 17 was used to assess functional mobility involving sit-to-stand, walking, turning, and sit-down transitions. Participants were seated on a chair, and were instructed to stand up, walk at their own comfortable and safe walking speed for 3 meters, turn, return, and sit down. Time (in seconds) was measured using a manual stopwatch from the moment the back left the chair until it returned to contact.
Gait at preferred and maximal speed
The participants walked the length of a room at their preferred and maximal speed, covering 8 meters with round-trip trajectories until completing a total time of one minute. Spatiotemporal gait parameters (speed, step length, and cadence) were recorded using the OptoGait system (Microgate, Italy), which uses infrared sensors with validated accuracy and reliability. 18 The OptoGait system was displayed in parallel along the central 5 meters, so that the start and end of the gait at each lap were not recorded. Instructions were to start walking at will (preferred speed) or as fast as possible (maximal speed).
Statistical analysis
None of the data violated the normality assumption necessary to conduct parametric statistical tests according to the Shapiro-Wilk test.
To evaluate the effect of the exercise program in the variables recorded, Data were analyzed using three-way repeated-measures ANOVAs with TEST (pre, post) as the within-subject factor, GROUP (experimental, control) as the between-subject factor, and baseline MoCA as a covariate. Baseline MoCA was included as a continuous covariate to account for inter-individual differences in global cognition—and the modest baseline imbalance between groups (p = 0.064)—thereby yielding more precise and unbiased estimates of the GROUP × TEST effects. Post hoc t tests with Bonferroni corrections were computed when required. p-values reported were based on two-sided comparison. Effect sizes were reported using partial eta squared (η²p) or Cohen's d, as appropriate. A p-value lower than 0.05 was considered statistically significant. All statistical analyses were performed using JASP 0.95c (University of Amsterdam, https://jasp-stats.org/).
Results
Both groups were homogeneous in their main characteristics, as indicated by the absence of significant differences between groups (Table 1).
There were no significant differences between the number of training sessions completed by each group (21 and 24 for the control and experimental groups, respectively). All participants completed a minimum of 20 sessions.
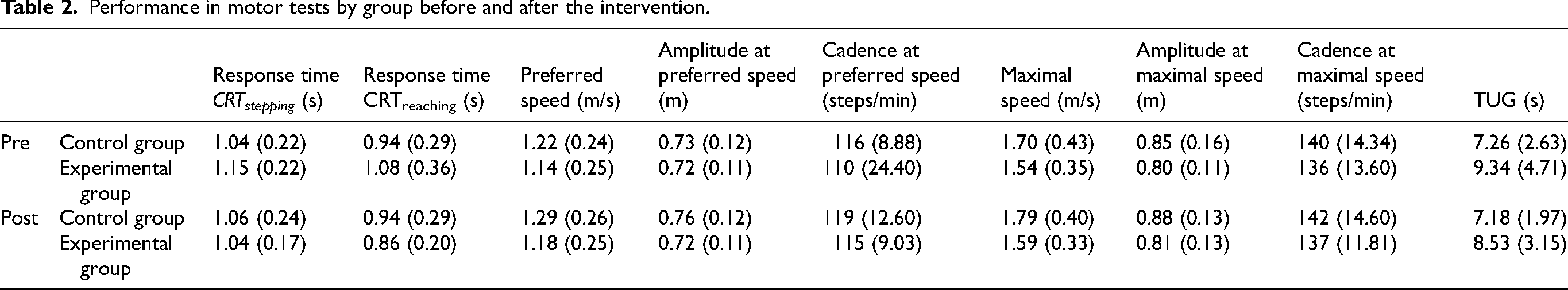
Table 2 shows the mean and standard deviation of performance in motor tests by group before and after the intervention.
Performance in motor tests by group before and after the intervention.
Primary outcome
The ANOVA revealed a significant effect of TEST (F = 4.18; p < 0.05; η²p = 0.07) and a significant TEST × GROUP interaction (F = 7.58, p < 0.01, η²p = 0.12) for response time in the CRT-stepping task, with no significant main effects of GROUP and MoCA. Post hoc analysis indicated that only the experimental group showed a reduction in response time following the intervention (p < 0.01, d = 0.51) (Figure 3(A)). Similarly, for the CRT-reaching task, there was a significant TEST × GROUP interaction (F = 12.45, p < 0.001, η²p = 0.18) and no significant main effects of GROUP and MoCA. Again, the experimental group demonstrated a post-intervention decrease in response time (p < 0.01, d = 0.69), whereas no change was observed in the control group (Figure 3B).
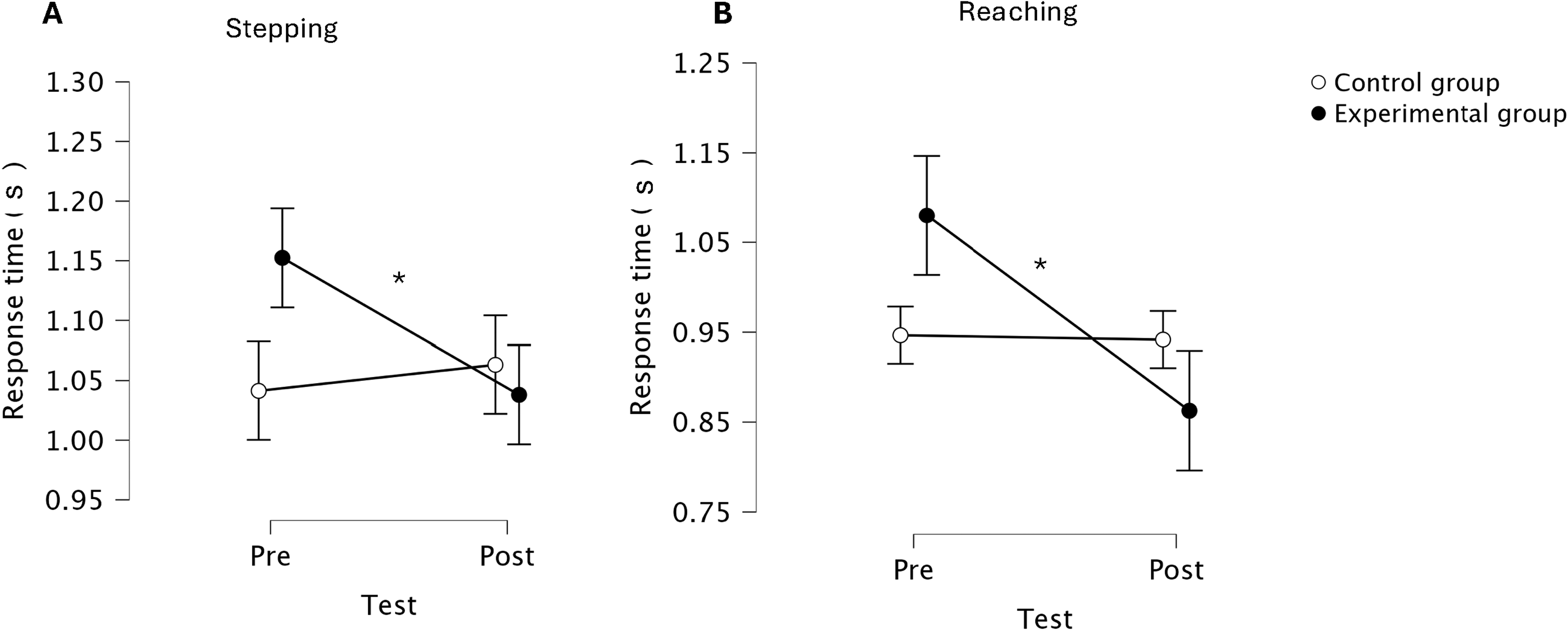
Mean (±sd) change in response time for the choice arm reaching reaction time test (A) and the choice stepping reaction time test (B) before and after the intervention in the experimental and control groups. * Indicate significant within-group changes (p < 0.01).
Figure 4 illustrates an example of performance during a reaching exercise, performed in all sessions by a subsample of participants. In the experimental group, there is a progressive reduction in response time across sessions, accompanied by reduced intra-subject variability (Figure 4(C)) and a logarithmic fit consistent with motor learning (Figure 4(D)). In contrast, the control group does not exhibit this pattern, showing greater intra-subject variability than the experimental group (Figure 4(A)) and a linear fit with no meaningful slope (Figure 4(B)).
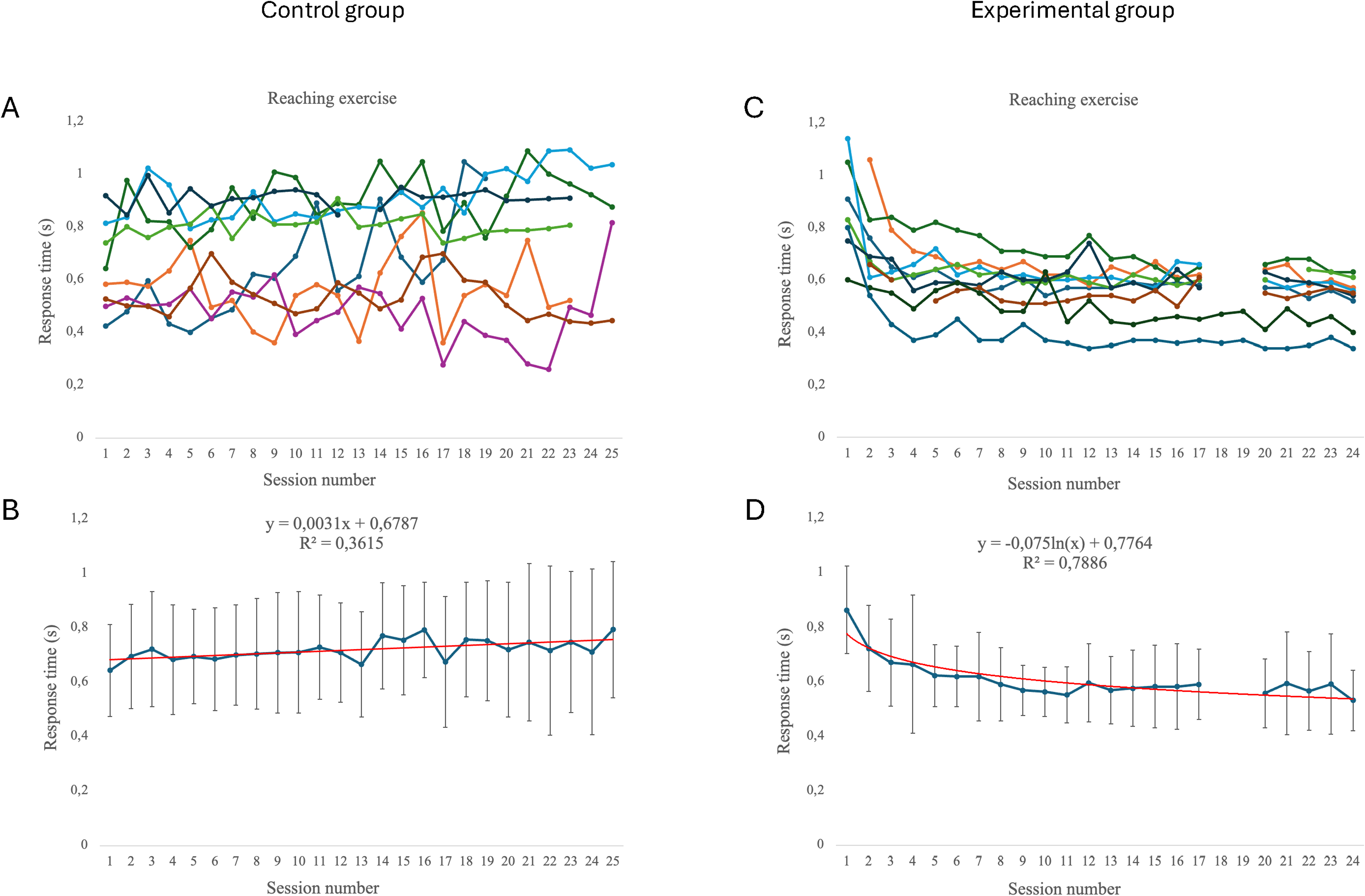
Example of individual performance during a reaching exercise in a subsample of participants across all training sessions. Panels A–B correspond to the control group and panels C–D to the experimental group. Panels A and C show trial-by-trial variability in response time, while panels B and D depict model fits (linear for control, logarithmic for experimental). Data are shown only for participants with app-recorded sessions; due to a storage malfunction, several sessions—including sessions 18–19—are missing for some individuals (Panel C), so we did not impute values and, to ensure comparability, the regression (Panel D) excludes sessions 18–19 for all subjects.
Secondary measures
The ANOVA conducted over the TUG test showed significant main effects for GROUP (F = 3.35, p < 0.05, η2p = 0.05) without other significant effects. Post-hoc analysis revealed lower values in the control than in the experimental group (p < 0.05, d = 0.53).
The ANOVA showed a significant main effect of TEST for speed in the preferred and maximal gait tests (F = 4.25, p < 0.05, η2p = 0.06 and F = 6.77, p < 0.01, η2p = 0.01, respectively). No significant effects of GROUP and MoCA, and no other significant interactions interaction were found. Post-hoc analysis revealed a significant increase in speed after the intervention for preferred and maximal gait tests (p < 0.01, d = 0.18 and p < 0.01, d = 0.19, respectively). No significant main effects or interactions were reported for amplitude and cadence for the preferred and maximal gait tests before and after the intervention.
Discussion
This randomized controlled trial investigated whether applying time constraints during visually guided reaching and stepping exercises could improve response time performance in individuals with PD. Our results demonstrate that progressively limiting the time available to complete motor responses significantly reduced both upper limb and lower limb response times in the experimental group, compared to controls who performed the same exercises without time pressure. These findings support the hypothesis that motor urgency, induced through constrained temporal windows, may enhance motor execution in PD.
The improvement in response time in reaching and stepping movements contrasts with our previous study using the same home-based mimo.fit platform, which showed improvements in gait parameters but no significant changes in reaching and stepping response time. 14 The key difference in the current intervention was the inclusion of time-limited stimuli and individualized feedback—two components that appear essential to engaging motivational-motor circuits in PD. 8 Therefore, the time constraint component itself is a critical factor in driving the observed improvements, rather than training volume or cues alone, which were identical in both groups. This aligns with theories of motor vigor and urgency, which posit that PD-related slowness may stem in part from altered cost-benefit computations that disfavor rapid responses in the absence of time constraints.19,20 Besides, limiting response time can act as a form of implicit motor learning, encouraging faster movement initiation and execution.21,22 The performance observed in the experimental group during the execution of the same reaching exercise throughout all the intervention sessions suggests that time-constrained training may promote more consistent performance gains and a learning curve characteristic of skill acquisition, whereas the absence of time pressure appears to limit such adaptation.23,24 In addition, the reduced intra-subject variability in the experimental group in the performance across sessions may reflect greater stability in motor output as participants adapted to the training demands. 25
The improvement in stepping response time could have clinical relevance for fall prevention in PD. As Caetano et al. noted, 26 stepping errors and slow response times—especially under cognitive load—are closely linked to increased fall risk. Beyond this behavioral study, fNIRS work has shown that when inhibitory demands rise during stepping, people with PD exhibit blunted prefrontal–premotor activation, pointing to limitations that can slow or destabilize reactive steps. 27 In line with this mechanism, our program was designed to minimize cognitive load and emphasize fast cue–action mapping; accordingly, including baseline MoCA as a covariate did not alter the training effects, and this low-load approach may serve as a pragmatic first stage before progressing to more cognitively demanding paradigms. By improving reactive stepping time, our intervention may translate into better real-world responses to balance disturbances or environmental hazards.
Notably, our results demonstrate that time-constrained training improves choice reaction time for both reaching and stepping, which may reflect a broader enhancement in movement planning and execution under temporal demand. Although upper-limb bradykinesia is a common and functionally relevant symptom in PD, the assessment of movement speed in reaching tasks has been used predominantly in experimental laboratory settings rather than as a primary outcome in clinical trials. Most existing studies have employed isolated pointing or reaching tasks to characterize motor slowness or explore the underlying mechanisms associated, often focusing on kinematic parameters such as reaction time, peak velocity, or movement time. 3 Systematic reviews and meta-analyses on exercise and rehabilitation in PD typically include very few interventions targeting upper-limb bradykinesia or reporting reaching speed as a primary endpoint, 28 limiting the ability to draw firm conclusions about its responsiveness to rehabilitation. In contrast to this limited evidence, the current study demonstrated significant improvements in reaching speed following the intervention, suggesting that targeted, ecologically valid reaching tasks embedded within a structured rehabilitation program can effectively enhance upper-limb motor performance in people with PD. Such improvements may translate into faster and more efficient execution of daily activities requiring upper-limb movements, thereby contributing to greater independence and quality of life.
It is also important to highlight that the present study replicates the previous improvements obtained in gait speed reported after using the same training system at home 14 which support the therapeutic potential of home-based technology in PD.29,30
Study limitations
Nevertheless, several limitations must be acknowledged. First, the study did not include long-term follow-up, so it remains unclear whether the observed improvements are retained over time. Second, although training was home-based, assessments were conducted in a laboratory setting, which may affect generalizability to daily function. Third, the intervention's impact on real-world mobility outcomes—such as falls, freezing of gait, or functional independence—was not assessed directly. While improvements in response time are promising, future trials should include functional outcomes to confirm clinical relevance. As a final limitation, we did not apply a MoCA cutoff for inclusion/exclusion. Although, the intervention imposed low cognitive demands and was delivered under one-to-one supervision, this choice may have increased cognitive heterogeneity and limits comparability with trials that exclude lower-MoCA participants. Sensitivity analyses adjusting for baseline MoCA yielded the same pattern of results, but residual confounding cannot be fully ruled out.
Conclusions
In conclusion, our results support the incorporation of temporally constrained cueing into home-based rehabilitation for individuals with PD. By leveraging the concept of urgency to modulate movement vigor, this approach addresses bradykinesia beyond conventional repetition-based paradigms. Future studies should explore its integration with pharmacological or cognitive strategies and investigate retention, transferability, and impact on quality of life.
Footnotes
Acknowledgments
Villamil-Cabello, Molinero-Martín, Fernandez-del-Olmo were supported by Ministry of Science and Innovation (grant reference PID2022-374 139930NB-I00). Luque-Casado was supported by Ministry of Science and Innovation (grant reference PID2023-152807NA-I00).
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Ministry of Science and Innovation, Spain, (grant number PID2022-374 139930NB-I00, PID2023-152807NA-I00).
Declaration of conflicting interests
Miguel Angel Fernández del Olmo is a cofounder of mimo.fit; however, this company provided no funding and had no role in the design, conduct, analysis, or reporting of this study.
Data availability statement
The data that supports the findings of this study are available and can be requested from M Fernandez-del-Olmo. The data are not publicly available due to privacy considerations.
Clinical trial registration number
ClinicalTrials.gov Identifier: NCT05829915.
