Abstract
Background
Impaired gait initiation is a debilitating motor symptom in people with Parkinson's disease (PD). During self-paced (uncued) gait initiation, anticipatory postural adjustments (APAs) are often absent or attenuated, and the first steps are abnormally short. External sensory cues can significantly improve APAs.
Objective
The effect of external cueing on lower limb muscle activation during gait initiation, compared to self-initiated steps, was examined in people with PD and healthy older adults (HOA).
Methods
Ground reaction forces, center of pressure excursions, and lower-limb surface electromyographic profiles (in seven bilateral muscles) were examined in 32 individuals with PD (off-medication) and 10 age-matched HOA during the APA and first step of self-paced or acoustically cued gait initiation.
Results
Anterior (tibialis anterior, vastus lateralis, rectus femoris) and gluteus medius muscles were primarily activated during the early phases of gait initiation, while later phases predominantly involved posterior (soleus, gastrocnemius, biceps femoris) and gluteus medius activations. Cueing facilitated anterior muscles and suppressed posterior muscle activity in both groups, however, activation patterns in PD were not restored to HOA levels. Instead, the PD group had lower early activity during the APA (compared to HOA) and higher late activity.
Conclusions
Cueing increased anterior muscle activation during gait initiation, rather than evoking a global gain across muscles and timings, demonstrating that cueing predominantly facilitates neural circuitry critical for early APA phases. People with PD showed enhanced late phase activity, probably to compensate for ineffective APAs, and thus have a stronger need to facilitate cue-evoked improvements.
Plain language summary
People with Parkinson's disease (PD) have difficulty with the weight shift pattern that is typically used to start walking (called an anticipatory postural adjustment, or APA). In people with PD, APAs are typically smaller and slower than in people without PD (controls). However, people with PD can improve their APAs when they are given an external cue (e.g., a “get ready” beep, followed ∼3 s later by a “go” beep). In this study, we measured muscle activity (surface electromyography) associated with APAs during cued and uncued (self-initiated) walking in people with PD and controls. For both cued and self-initiated walking, people with PD had less muscle activity during the APA, and more muscle activity around the time of the first foot lifting off the floor (toe-off), compared to controls. Cueing increased APAs and muscle activity along the fronts of the legs (anterior muscles), especially in the early phases of the APA, and decreased activity in muscles along the backs of the legs (posterior muscles), in both groups. Our results demonstrate that cues help people with PD initiate walking by improving muscle activation and force generation during the early phase of the weight shift pattern.
Introduction
Posture and locomotion coupling is impaired in people with Parkinson's disease (PD) and contributes to disturbances in self-initiated forward stepping.1–3 In healthy adults, the transition from standing to walking involves a sequence of weight shifts called anticipatory postural adjustments (APAs). APAs are characterized by an initial phase of step limb loading and stance limb unloading, during which the center of pressure (CoP) under the feet shifts posteriorly and laterally toward the initial stepping leg. This sequence acts to propel the body's center of mass forward and laterally toward the stance limb (APA Phase 1). 4 The initial phase is followed by concurrent step limb unloading and stance limb loading and a shift of the CoP forward (slightly) and toward the stance limb (APA Phase 2), followed by a transition to a period of increasing stance leg loading (above 50% body weight) and step leg unloading (below 50% body weight) prior to toe-off (Execution Phase). These later phases allow for step leg lift-off and subsequent push-off with the stance leg (Figure 1).4,5 APAs are generally preserved across the adult lifespan, 6 but in people with PD, the APAs generated during self-initiated (uncued) stepping are often absent or reduced in amplitude and prolonged in duration.1,2,7–11 Impaired APAs are associated with decreased initial gait velocity,12,13 a shortened first and second step,12,14–16 and an increased fall risk.17,18 In people with PD and freezing of gait (FOG), the initial two steps of uncued gait initiation are typically shorter and wider than those without FOG. 15 Others have provided evidence, using different gait initiation paradigms, that people with FOG may have more impaired CoP trajectories during APAs compared to those without FOG.16,19,20 Impaired gait initiation is especially problematic for people with PD and FOG as it may trigger a freezing episode, or the feeling that the feet are transiently glued to the floor while trying to take a step. 3
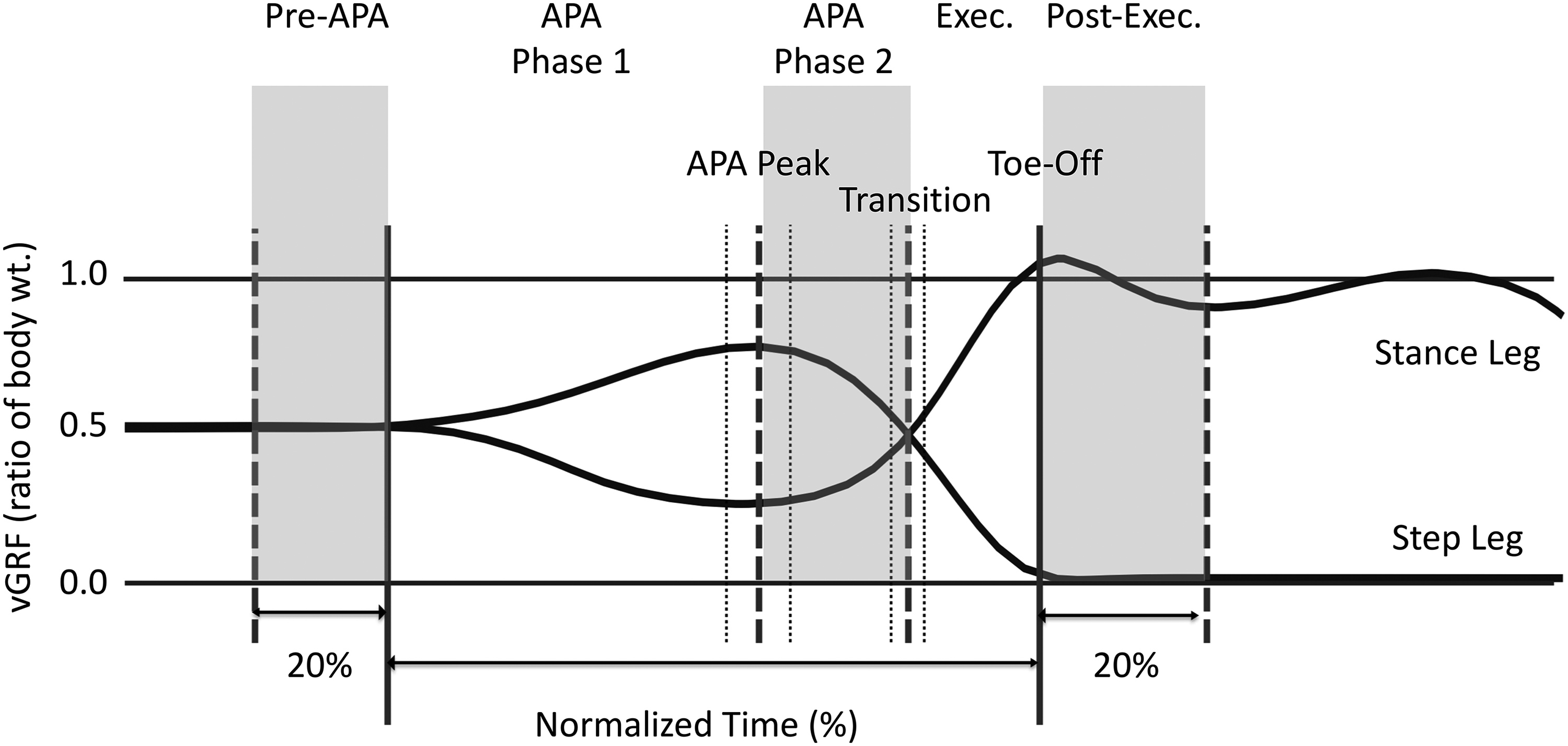
Time-normalized phases of the Anticipatory Postural Adjustment (APA), defined via the vertical ground reaction forces (vGRF) of the step and stance legs. Time is normalized with 0% set at the onset of the weight shift and 100% at initial step leg toe-off. The gait initiation cycle was divided into five phases: (1) a Pre-APA phase from -20% to 0%, (2) an APA phase 1 (APAI) from weight shift onset (0%) to the peak vGRF of the step leg (APA peak), (3) APA Phase 2 (APAII) from the APA peak to the transition point (defined as the percent time at which the step leg vGRF returns to the baseline value), (4) the Execution phase from the transition point to the step leg toe-off, and (5) the Post-Execution phase from toe off to an additional 20% of time.
External sensory cues (e.g., acoustic, visual, vibrotactile) can markedly improve gait initiation in people with PD (with or without FOG).1,11,21 External cueing significantly reduces the incidence of trials without an APA, increases the amplitude and reduces the duration of the CoP shifts compared to self-initiated stepping1,2,14,16 in people with PD, as well as healthy younger and older adults.1,22 The improvements in APAs evoked by cueing when people are in the off-medication state are comparable to performance improvements observed following the administration of levodopa.23,24 However, in both freezers and non-freezers, improvements in APA magnitude and timing may not translate to alterations in first step kinematics and timing,14,16,25,26 raising the possibility that imperative sensory cues may affect the early and later components of the gait initiation sequence differently.
Currently, little is known about the changes in lower limb muscle activity that mediate the facilitatory effects of external cueing in gait initiation. A variety of studies have used surface electromyographic (sEMG) to characterize the muscle activation patterns associated with impaired APAs in PD. For the most part, these studies have focused on the tibialis anterior (TA) and soleus (SOL) muscles.10,11,27–29 The start of the APA is typically characterized by activation of the TA and suppression of tonic standing activity in soleus. 27 TA activation moves the CoP posteriorly, which generates an anterior ground reaction force27–29 and a gravitational moment that accelerates the center of mass forward. 27 Reduced force generation and excursion of the CoP in PD during self-initiated stepping is due, in part, to fractionated muscle activity (multiple short duration bursts of low amplitude) in TA10,11 reduced and prolonged soleus activity, and reduced synergist activation of distal and proximal effectors on the step and stance legs.27,29,30 Cueing is associated with increased synchrony of step and stance TA activation, increased magnitude and duration of the initial TA burst and improved suppression of SOL activity.11,28,29,31 Currently, little is known about the effects of cueing on other primary mover muscles of the leg during gait initiation in people with PD.
The primary purpose of our study was to quantify the changes in the magnitude and timing of distal and proximal muscle activity associated with externally cued gait initiation compared to self-initiated stepping in people with PD (with and without freezing of gait, PD+FOG and PD-FOG) and matched healthy older adults (HOAs). Based on earlier work showing cue-evoked TA facilitation and soleus suppression,11,28,29,31 we hypothesized that, (1) cueing would preferentially facilitate muscle activity involved in the generation of the early APA phase of gait initiation (APA Phase 1), rather than a global increase in gain across all muscles and timings, (2) people with PD would have a larger cue-evoked increase in muscle activity, compared to HOA, and (3) there would be no significant difference in sEMG and the effects of cueing between PD+FOG and PD-FOG.
Methods
Participants
The data for this study was obtained from two previous experiments that examined the effects of cueing on gait initiation kinematics and kinetics.2,15,22 Two groups of participants were included: 32 people with PD (mean ± standard deviation: 65 ± 9 years; 9 female) and 10 age-matched healthy older adults (HOA, age 66 ± 10 years; 1 female). Participants were excluded if they had a diagnosis of a neurological disease other than PD, peripheral neuropathy in the lower limbs, clinically significant uncorrected reductions in vision or hearing, history of dementia or cognitive impairment, musculoskeletal disorders of the lower limbs that significantly impacted gait (overt changes in range of motion, strength, postural stability or symmetry due to disorder; e.g., knee replacement), or required an assistive device such as a cane or walker. Participants with PD were excluded if they had moderate-to-severe tremor (score >>2 on items 20 or 21 of UPDRS) in any limb or a history of neurosurgery to treat PD (e.g., deep brain stimulation). Healthy older adult controls were selected from volunteers who were age-matched to the individuals in the PD group in study the Petrucci et al. (2021) study, 22 and checked to be non-significantly different from the entire PD cohort when combined with the Lu et al. (2017) cohort. 2 Three of 25 people with PD from the Lu et al. study and one person with PD out of ten from the Petrucci et al. study were excluded from this analysis due to the absence of an APA in any trials. One control (out of 11) from Petrucci et al. was also excluded, due to technical issues. Participants with PD had a time since diagnosis of 6.5 ± 3.7 years, levodopa equivalent dose of 700 ± 460 mg (range: 0–1560 mg) (based on 32 ), off-medication score of 39.2 ± 12.9 on the Unified Parkinson's Disease Rating Scale Subsection III, Motor Scale (UPDRSIII), and a median Hoehn and Yahr score of 3 (Interquartile range: 2.4–3). Fifteen people with PD (PD+FOG) were classified as definite freezers (from self-report and visual confirmation in the lab), with 5 from the Petrucci et al. experiment, and 10 from Lu et al. (overall New Freezing of Gait Questionnaire mean score 15.8 ± 8.1). However, no participants froze during the trials included in this study. Participants with PD were tested in the morning after overnight withdrawal of PD medications (at least 12 h since taking their last dose). Written informed consent was obtained from the participants prior to inclusion in the study and all procedures were approved by the Institutional Review Board of the University of Minnesota (1302M27746 and 1407M52163).
Protocol
Data collections for the two primary studies were performed in Movement Disorders Laboratory at the University of Minnesota with the same group of investigators. Self-initiated and cued trials (blocks of trials utilizing acoustic, fixed delay cues were selected from each study) were performed with consistent protocols across studies, with small differences in cue timing and number of sEMG recorded, as highlighted below.2,22 Analysis code was also kept consistent. We have previously shown that exposure to multiple cued trials of different types does not affect performance of self-initiated gait 2 and thus, we were confident that selecting matching blocks of trials would not significantly affect our findings.
Participants performed blocks of 6 trials of cued and self-initiated (SI) gait initiation with their preferred stepping leg. Blocks were presented in randomized order for each participant within larger protocols including 11 (Lu et al. 2 ) or 5 (Petrucci et al. 22 ) blocks of trials. During the task, participants stood using a natural stance width, with each foot on one of two force plates embedded in the walkway (Kistler, Novi, MI). The outline of the feet was taped on the floor to ensure a consistent starting position across trials. For each trial, participants were instructed to look at an “X” placed 3 m in front of them and to “hold steady” until they started walking forward “as quickly as possible”. For cued trials, participants were given an acoustic warning cue (80 dB, 100 ms, 500 Hz) followed by an acoustic “go” cue (80 dB, 100 ms, 1000 Hz) after a fixed delay (2.5 or 3 s, depending on the primary study). For the self-initiated trials, participants were instructed and coached to wait at least 3–5 s after the researcher said “anytime”. Trials were repeated if the participant started walking in reaction to the “anytime” instruction.
During the gait initiation task, analog data from the force plates and surface electromyography (sEMG, Delsys, Natick, MA) were collected for 8 to 12 s at 1000 Hz and combined using SIMI Reality Motion Systems hardware and software (Munich, Germany). Force and moment data from the force platforms were used to determine the vertical ground reaction forces (vGRF) of the step and stance legs and the net center of pressure under the feet in the medial-lateral (ML-CoP) and anterior-posterior (AP-CoP) directions. Fourteen surface EMG electrodes were placed bilaterally on 7 leg and hip muscles including tibialis anterior (TA, dorsi flexor, important for initiating the APA 27 ), soleus (SOL, plantar flexor, typically inhibited initially and later activated 27 ), lateral gastrocnemius (GS, plantar flexor and knee flexor), vastus lateralis (VL, knee extender), rectus femoris (RF, hip flexor), biceps femoris (BF, hip extender and knee flexor), and gluteus medius (GM, hip abductor). Lateral gastrocnemius and rectus femoris sEMG were not included in one of the protocols, 2 so this data was only available for 9 people with PD (4 PD+FOG, and 5 PD without FOG, PD-FOG) and all controls.
Data analysis
The magnitudes and timings of key APA components were calculated from the force plate data (GRFz and CoP). These included the magnitude and time from APA onset to four primary peaks (local minima or maxima): peak step leg loading, stance leg unloading, ML shift of the CoP towards the stepping leg, and first posterior shift of the CoP. The second posterior shift of the CoP, which occurs around first toe-off, and time to toe-off, were also determined (see arrows in Figure 2 here and Figure 1 in Lu et al., 2017 2 ).
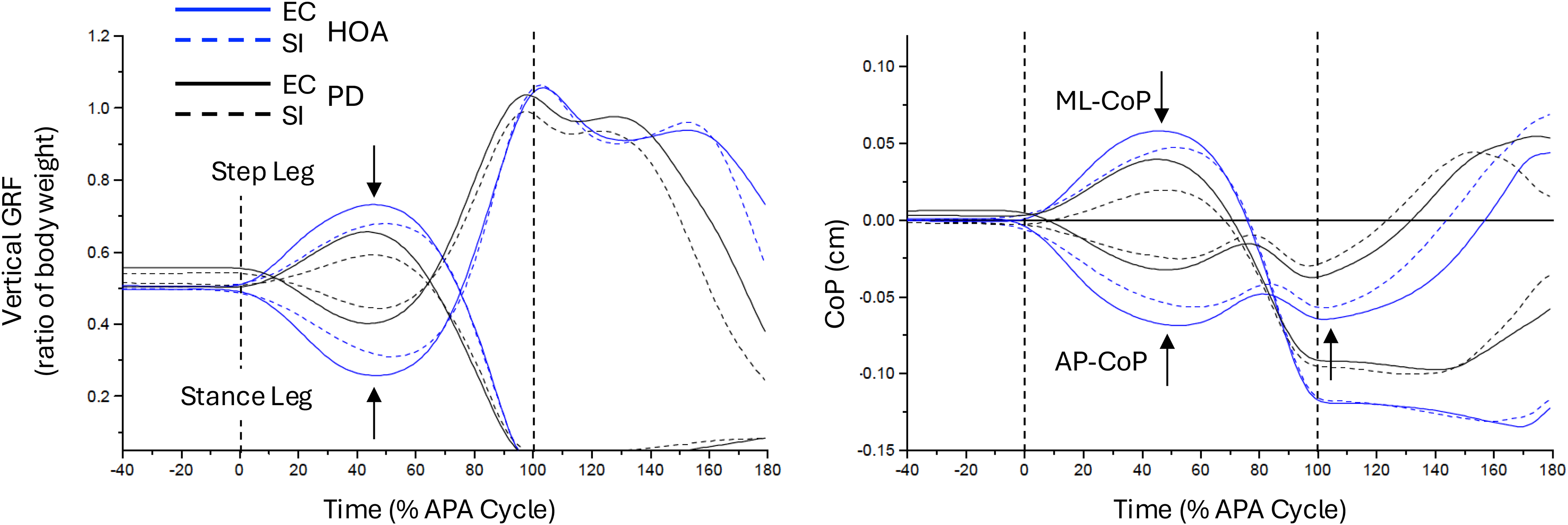
Cueing increased the magnitudes of the APAs. Subject- and group-averaged APA data (vertical ground reaction forces, vGRF, left plot; and center of pressure data, CoP, right plot) are shown for externally cued (EC, solid lines) and self-initiated trials (SI, dashed lines) for each group (Healthy older adults, HOA, in blue/gray; Parkinson’s disease, PD, in black). vGRFs are shown for the step and stance legs and the medio-lateral CoP (ML-CoP) and anterior-posterior CoP (AP-CoP), as labeled. Data is time-normalized as shown in Figure 1.
Only trials with all APA components (step leg loading, stance leg unloading, lateral and posterior shift in the CoP) were included in the data analyses (44 trials were excluded out of an available 462 trials, or 9.5%). Data from one person with PD was removed due to the lack of an APA in all six self-initiated (SI) trials. The sEMG data was post-processed in MATLAB (Mathworks, Natick, MA, USA). sEMG data were zero-meaned, full-wave rectified, and filtered with a 2nd order low-pass Butterworth filter at 20 Hz. The temporal profiles of sEMG were time normalized to the percentage of APA duration (Figures 1 and 2), from the time of APA onset (0%) to step leg toe-off (100%; based on the vertical ground reaction force). An additional 20% of time was added on each side of the window to capture sEMG activity preceding onset of the APA and during the initial phase of the first step. sEMG amplitudes were normalized to the peak amplitude observed in that participant's muscle trace across all SI and cued trials within the normalized time-period of −20% to 120%. The time- and amplitude-normalized profiles of sEMG activity were averaged across trials within each cueing condition, within each participant, and then across all participants (Figure 3).
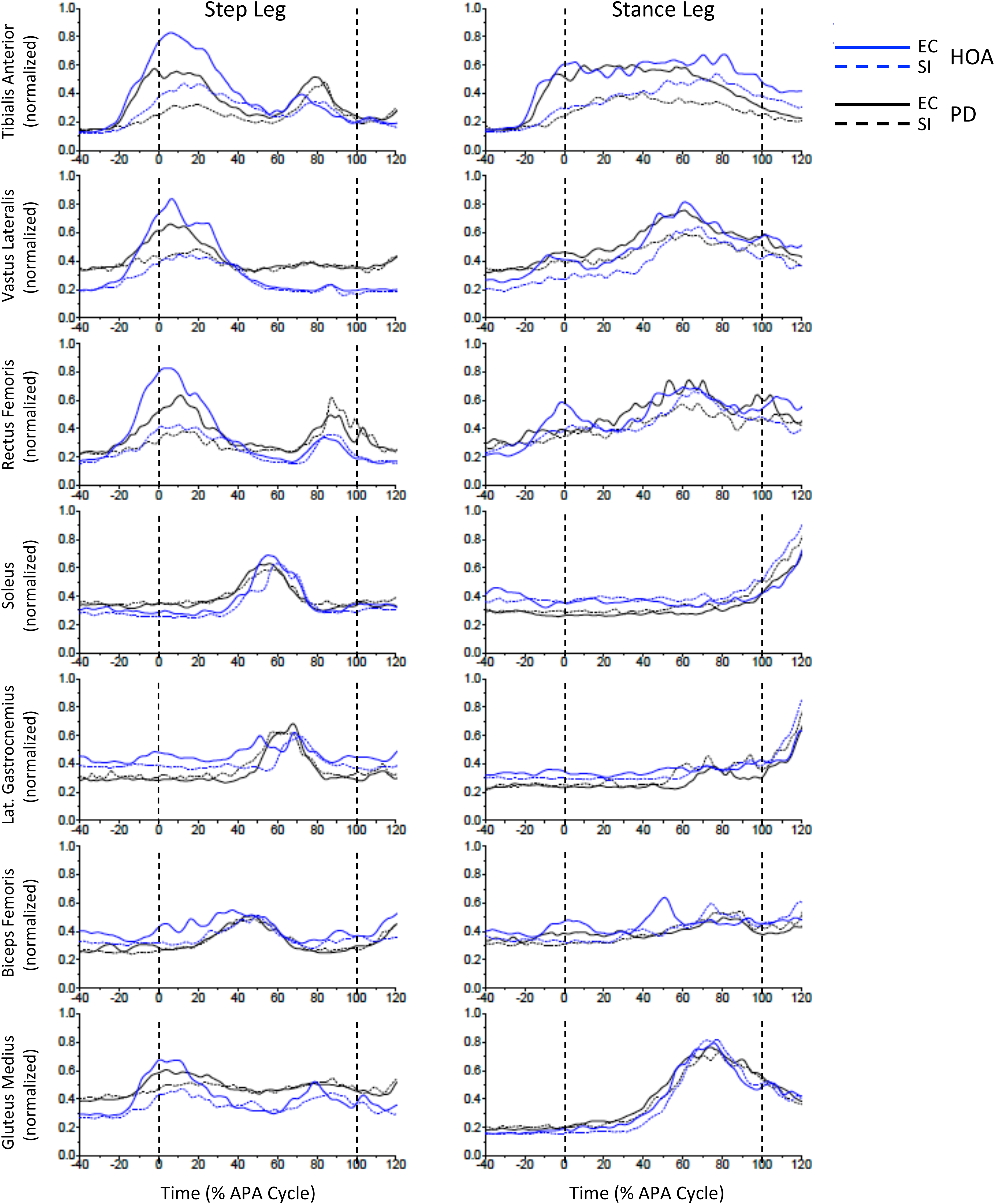
Cueing effects on subject- and group-averaged EMG. External cueing (EC, solid lines) evoked an increased rate of rise and magnitude of EMG activity compared with the self-initiated condition (SI, dashed lines) in all groups, particularly in anterior muscles during the early phases of gait initiation. Subject- and group-averaged EMG data are shown for each muscle for the step (left plots) and stance legs (right plots) for both groups (Healthy older adults, HOA, in blue/gray; Parkinson’s disease, PD, in black).
In order to investigate changes in the amplitudes and general timing of muscle activity at different times the gait initiation process, each trial of gait initiation was divided into five phases (Figure 1): 1) Pre-APA, 20% normalized time prior to APA onset; 2) APA Phase 1, from the onset of loading (increased vGRF) to the peak loading of the stepping leg; 3) APA Phase 2, from peak step leg loading to when the step leg vGRF returned to baseline values (transition point); 4) Execution: from the time of the transition point to step leg toe-off; 5) Post-Execution: from toe-off to 20% of normalized time. For each of the five phases, the (normalized) sEMG amplitude was integrated over normalized time (iEMG).
Statistical analysis
To address primary hypotheses 1 and 2, the iEMG data were analyzed using a linear mixed effects model (lmer, lme4 package 33 ) in R. 34 Effects included main effects of gait initiation phase (P1 – P5), group (HOA vs. PD), and cueing condition (SI vs. cued), along with their interactions, and with participant as a random effect. 35 Type III repeated-measures ANOVAs with Satterthwaite's method were used for each data set. The iEMG data was log10 transformed to improve the normality of the residuals’ distributions (tested before and after transformation via the Shapiro-Wilks test, and inspected via qq-plots and histograms of the residuals). 36 We repeated these models to compare PD+FOG vs. PD-FOG (without controls) to investigate the effects of freezing phenotype on the iEMG (Hypothesis 3).
For each significant main and interaction effect from the linear mixed effects model, the p-values were corrected with the false discovery rate (FDR) corrections for multiple comparisons in this exploratory study. 37 All significant ANOVA results were followed by post hoc tests (“emmeans” function in R) using Tukey-Kramer's method (to account for uneven sample sizes and correct for multiple testing).36,38 The significance threshold (including alpha 39 ) was set to p < 0.05. Effect sizes were estimated Cohen's d for each significant follow-up effect in the model (calculated from log10 transformed iEMG data, and non-transformed APA data).40–42 Although Cohen's d has limited interpretability when calculated from linear mixed effects models, 40 we calculated it for each significant post hoc t-test as a standardized metric to explore the effect size in our analysis. We also calculated the median difference and 95%% confidence intervals (CI) between the two compared variables (using non-transformed data, to maintain interpretability of the values) in each follow-up as a measure of unstandardized effect size (the larger of the two was used first for difference and CI calculations). 40 The two-sided 95% bootstrap CI of the difference between the medians of the datasets was based on 9999 bootstrap replications and the bias-corrected and accelerated (BCa) method (using the “ci_median_diff” function in R).
A secondary analysis explored any APA differences between groups (HOA vs. PD and PD+FOG vs. PD-FOG) and cueing condition in this cohort combined from multiple studies. For this, we investigated APA magnitudes and timings using linear mixed effects models with the main effects of condition (SI vs. cued) and group (both comparing HOA vs. PD and PD+FOG vs. PD-FOG), and their interactions, with participant as a random effect to account for repeated measures across multiple cueing conditions. Cohen's D were calculated as above. The APA measures were not log transformed for any analysis.
Results
External cueing facilitated APA kinetics
As shown in the primary experiments,2,22 external cueing significantly increased the magnitude and decreased the duration of the APAs (Supplemental Table 1). A summary of the time-normalized average profiles of the APA kinetics (vGRF beneath the stepping and stance legs, ML-CoP and AP-CoP) in each group is shown in Figure 2. The four primary APA magnitudes (peak step loading, stance unloading, and ML and first AP CoP shift) all showed significant main effects of condition (cue > self-initiated, p < 0.001) and group (HOA > PD, p < 0.01). There were no interaction effects on these magnitudes (p > 0.977). The time to peak GRFz and CoP shifts were significantly decreased with a cue (p < 0.01), but there were no main effects of group (p > 0.127) or an interaction effect (p > 0.977). Cueing effects on the APA were medium to large and impacted all the metrics (Cohen's d range: 0.48–0.89), with cues improving the amplitude and timing of the APA kinetics (Supplemental Tables 1 and 2).
Cues also affected performance of the execution phase. Cueing significantly increased the magnitude of the second peak posterior shift of the CoP (PPS2) (p < 0.001) and decreased the time from APA onset to PPS2 (p = 0.014). There was a main effect of group for the PPS2 magnitude (p = 0.020), with PD having a smaller magnitude than HOA, but not for the PPS2 timing (p = 0.133). There were no interaction effects on PPS2 magnitude or timing (p >> 0.977). Overall, the time from APA onset to toe-off decreased with a cue (p = 0.018), an effect of group (p < 0.009), and no interactions (p = 0.977), with post hoc assessments showing that PD had a longer time to toe-off than HOA.
Cueing differentially impacted anterior and posterior muscle activity
The average profiles of the sEMG activity across all fourteen muscles during self-initiated and cued gait initiation are shown for both groups in Figure 3. The three anterior muscles (TA, VL, and RF) on both legs had significant main effects of cueing condition (p < 0.001, Cohen's d range: 0.2–0.6), with greater activity in cued trials compared to self-initiated trials (Figure 4, Table 1, Supplemental Tables 3 and 4). However, there were interactions in phase x condition (p < 0.05) in bilateral TA activity, such that the cueing predominantly facilitated activity bilaterally in TA early in the APA cycle (Step TA in Pre-APA to APA phase 2, and stance leg TA in Pre-APA to Execution; Cohen's d range: 0.41–1.5).
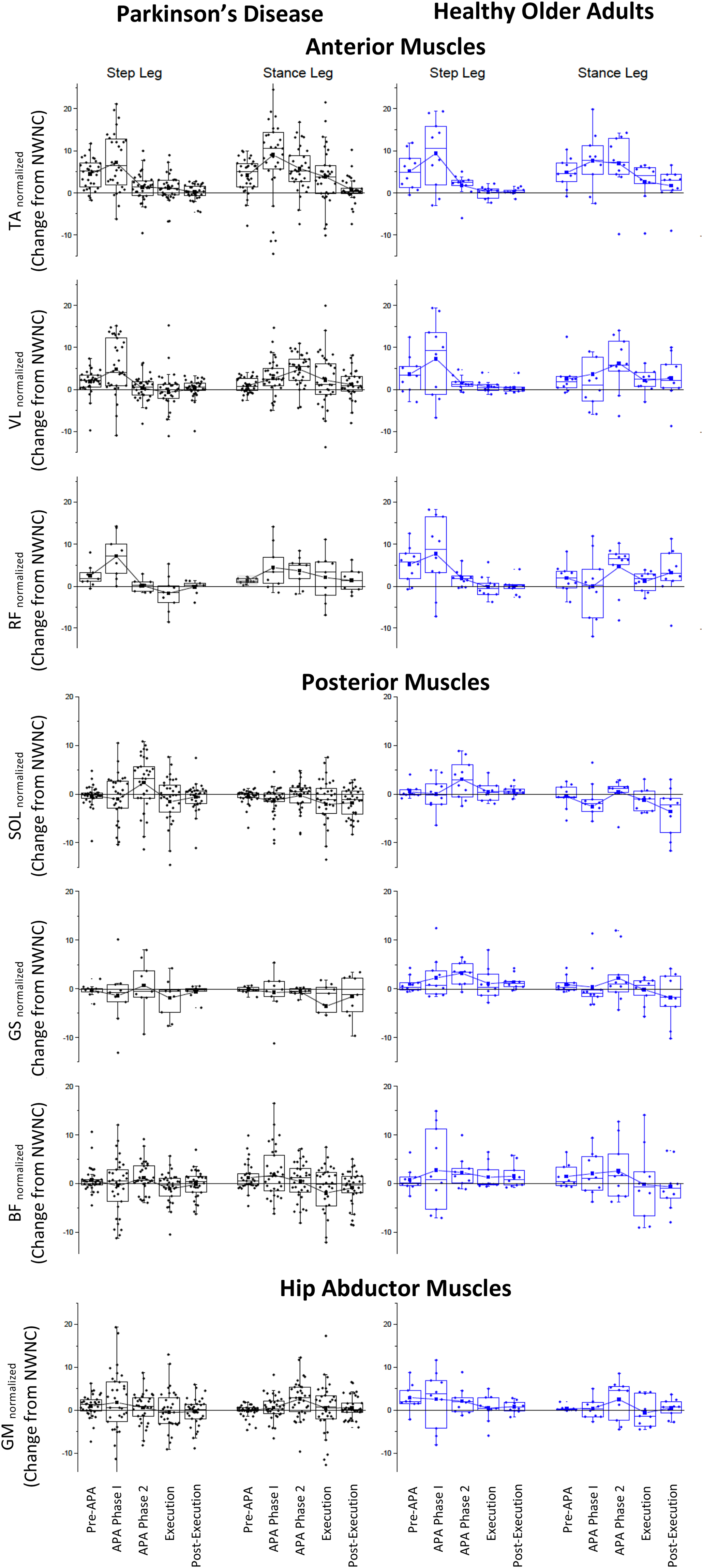
Box plots of cue-evoked changes in in integrated EMG (iEMG) magnitude relative to the self-initiated condition for each phase of gait initiation for each group and muscle. Amplitude-normalized EMG was integrated over normalized time for each of the five phases. The difference score (the average integral of cued trials minus uncued trials), for each phase and each subject is shown as an individual data point. The square symbol in the box plot denotes the within-group mean. Parkinson’s disease, PD, black plots on the left; Healthy older adults, HOA, blue/gray plots on the right. On each plot, the step and stance leg muscle data are shown on the respective left and right sets of connected bars. Muscle abbreviations: TA = tibialis anterior, VL = vastus lateralis, RF = rectus femoris, SOL = soleus, GS = lateral gastrocnemius, BF = biceps femoris, GM = gluteus medius. See Figure 1 for definitions of gait initiation phases. Each plot has a set of 5 phases for each leg.
iEMG results. Primary results of the linear mixed models analyses (Type III Repeated-measures Anova with Satterthwaites’ method) on measures of the changes in integrated surface EMG (iEMG) across gait initiation phase, group (PD vs. HOA), and cueing condition (cued vs. self-initiated). All data were log10 transformed for statistical analysis. F-values are given and post-hoc testing for significance used the false discovery rate (FDR) corrections for multiple comparisons. Please see Supplemental Tables 3 and 4 effect sizes. Table 1 Summary: Of note here, is the consistent effect of cue condition on anterior muscles (“Condition” column), the effect of cue condition in certain phases (“Condition x Phase” interactions) for anterior and posterior muscles, and group differences in certain phases (Group x Phase interaction terms) for TA muscles.
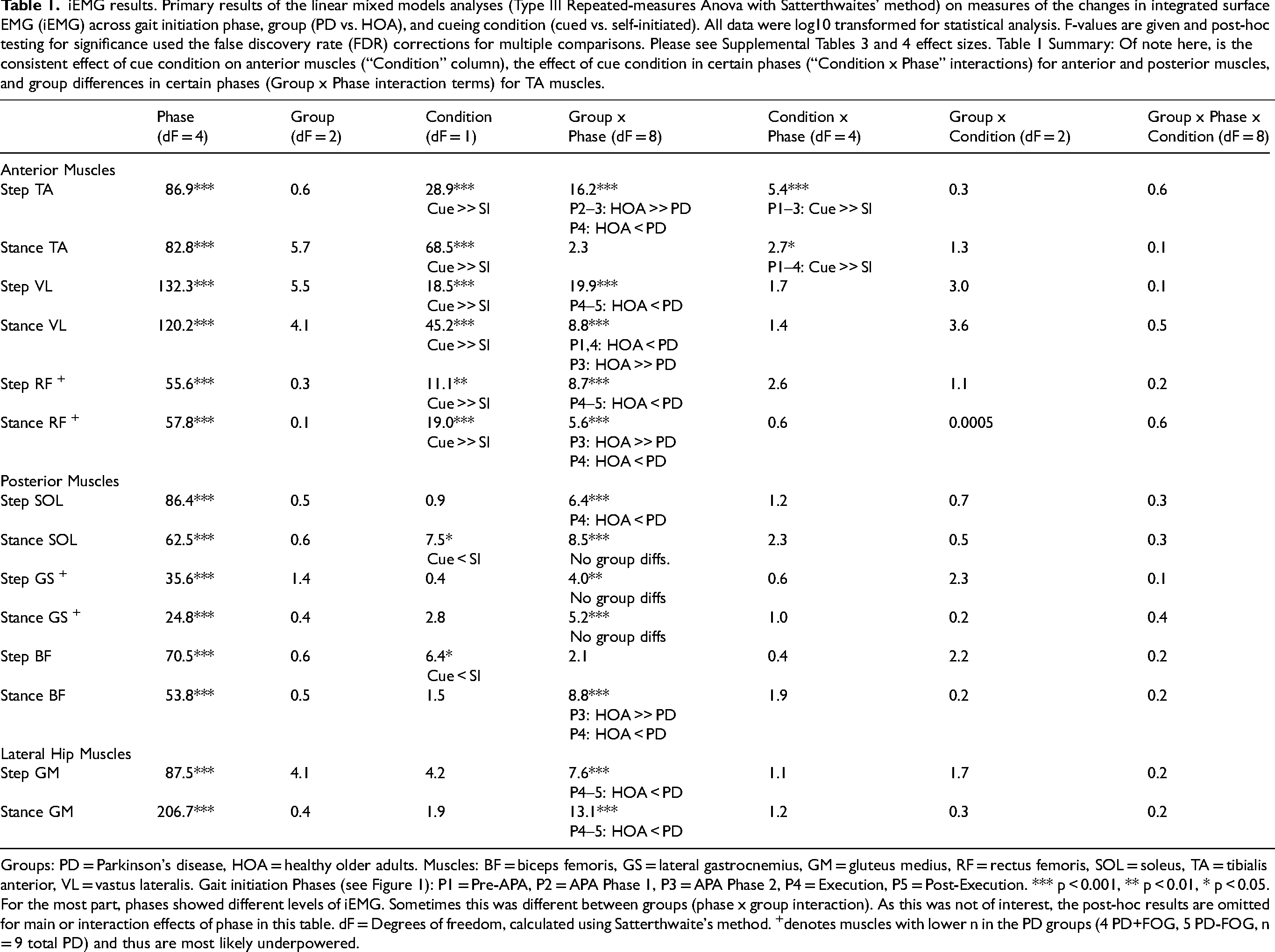
Groups: PD = Parkinson's disease, HOA = healthy older adults. Muscles: BF = biceps femoris, GS = lateral gastrocnemius, GM = gluteus medius, RF = rectus femoris, SOL = soleus, TA = tibialis anterior, VL = vastus lateralis. Gait initiation Phases (see Figure 1): P1 = Pre-APA, P2 = APA Phase 1, P3 = APA Phase 2, P4 = Execution, P5 = Post-Execution. *** p < 0.001, ** p < 0.01, * p < 0.05. For the most part, phases showed different levels of iEMG. Sometimes this was different between groups (phase x group interaction). As this was not of interest, the post-hoc results are omitted for main or interaction effects of phase in this table. dF = Degrees of freedom, calculated using Satterthwaite's method. +denotes muscles with lower n in the PD groups (4 PD+FOG, 5 PD-FOG, n = 9 total PD) and thus are most likely underpowered.
In the posterior muscles, there was a main effect of condition on the stance leg SOL (p = 0.012) and step leg BF (p = 0.021, Cohen's d range: 0.12–0.13), with cueing suppressing activity in those muscles. The lateral hip muscles (bilateral GM) showed no significant changes with cueing. Finally, there were no significant group x condition or group x phase x condition interactions in any muscles, suggesting that cues facilitated muscle activity similarly across groups.
Muscle activity was dynamically modulated across gait initiation phases
There was a significant main effect of phase for all muscles (p < 0.001, Table 1) reflecting the temporal dynamics of muscle activity throughout the gait initiation sequence. The patterns of sEMG were similar between groups. In the early phases of gait initiation, including the pre-APA and APA Phase 1, increased iEMG was predominantly observed in anterior muscles of the step and stance legs, which included the tibialis anterior (TA) and quadriceps (vastus lateralis, VL, and rectus femoris, RF), along with the step leg lateral hip muscle: the gluteus medius (GM). The anterior and GM muscles of the step leg reduced activity over the latter portion of APA Phase 1 and approached baseline levels near 60% of the APA cycle. In contrast, anterior muscles of the stance leg increased activity throughout the course of the APA with peak activity occurring near the transition point (∼60%) (halfway through what is sometimes called the unloading phase 23 ) and reducing throughout the first toe-off (100–120%). Activity increased again in the step leg TA and RF during the execution phase (transition point to toe-off, which would be the second half of the unloading phase 23 ).
Posterior muscles of the step leg, including the biceps femoris (BF), lateral gastrocnemius (GS) and soleus (SOL) muscles, increased in activity around 30–40% of the cycle (near initial weight shift and CoP movements), with activity in SOL preceding the GS. Posterior muscles peaked near 50–65% of the cycle (near the transition point). Activity in the stance leg plantar flexors increased near toe-off. In the hip muscles, stance leg GM sEMG increased in concert with the timing of the loading of the stance leg (APA Phase 2 and execution phases).
The dynamic activity was slightly different between groups and conditions, as there were phase x group interactions in 12 of 14 muscles (p < 0.01) and condition x phase interactions in 2 of 14 muscles (p < 0.05, see Table 1 and Supplemental Table 3). However, the temporal patterns of activation were sufficiently similar between cueing conditions and groups, such that post hoc assessments of the differences between phases were relatively small. For this reason, and because the change in muscle activity across gait initiation was not of interest, the primary tables presented here are simplified by omitting the follow-ups describing the iEMG differences between phases for any effects.
Groups differed in muscle activation in some phases of gait initiation
No muscles had a main effect of group, however, all anterior muscles, except for the stance leg TA, had significant group x phase interactions (p < 0.001), with group differences appearing in some phases. The PD group had lower step leg TA activity compared to the HOA group early in the APA sequence (APA phase 1 and 2, seen in Figure 3 around 0–20% APA cycle), but higher activation during Execution phase (∼80% APA cycle). The PD group also had significantly higher activity in the step leg VL and RF compared to the HOA group in late phases (Execution, Post-execution). The stance leg VL had significantly higher activity than HOAs during the Pre-APA and Execution phases, but lower activity in the middle of the cycle (APA Phase 2). Similarly, in the stance leg RF, the PD group had higher lower activity during APA Phase 2, but was higher during the Execution phase. In anterior muscles, Cohen's d range was 0.27–1.64.
Posterior muscles had fewer muscles with significant group x phase interactions (p < 0.01) compared to the anterior muscles. People with PD had larger activity in the step leg SOL during the Execution phase, compared to the HOAs. In the stance leg BF, people with PD had lower activity in the APA Phase 2, but higher activity in the Execution phase. Cohen's d range was 0.60–0.77.
The hip abductor muscles (Step and Stance GM) showed significant group x phase interaction effects (p < 0.001), with the PD group having significantly higher bilateral GM activity late in the cycle (Execution, Post-Execution), compared to the HOA group (Cohen's d range 0.08–1.4, see Supplemental Table 4).
Effect of FOG presence on gait initiation EMG
As secondary analyses, we compared APAs and iEMG between participants with PD with (PD+FOG) and without FOG (PD-FOG). We have previously shown that there were no significant group effects (p >> 0.6) or group x condition effects (p >> 0.1) on APA magnitude or timing measures in this cohort. 2 However, analyses of the iEMG data between only the PD groups (excluding controls) showed differences between groups in certain phases (group x phase interactions) for two of the step leg muscles: step leg BF (p = 0.008) and step leg GM (p = 0.048). Post hoc tests demonstrated that PD+FOG group had higher iEMG than the PD-FOG group in early phases (Pre-APA and APA Phase 1) for the step leg GM (P1: Cohen's d = 0.79, median difference = 2.99, 95% CI = 0.06–5.03; P2: d = 0.71, median difference = 6.19, 95% CI = 0.98–10.47), and the Execution phase for the step leg BF (P4: d = 0.58, Median difference = 1.63, 95% CI = −3.48–5.64).
Discussion
Summary
This study demonstrates that external cueing significantly increases activity in anterior muscles and decreases activity in selective posterior muscles during an in-laboratory gait initiation task. Cueing was associated with a marked facilitation of anterior muscles (VL, RF) in general, while the tibialis anterior muscles were specifically facilitated by the cue during earlier APA phases (partially supporting Hypothesis 1). In contrast, the effects of cueing on posterior muscle activation, which were generally active later in the gait initiation sequence, were relatively modest, and included a significant suppression of activity in the stance leg SOL and step leg BF. The effects of cueing were comparable between the PD and HOA groups such that, despite significant improvement in the muscle activation profiles in the PD cohort, the magnitude of muscle activation and corresponding APA kinetics remained proportionally different from controls during key phases (failing to support Hypothesis 2). PD activity was often lower than controls during the pre-APA and APA phases, and higher during execution and post-execution phases. Nonetheless, cueing produced significant improvements in the spatiotemporal profile of muscle activation in both PD and controls, which translated to improved APA kinetics. Consistent with our previous findings including a subgroup of this study's participants,2,22 there were no significant differences in APAs between the PD+FOG and PD-FOG subgroups. Additionally, for the most part, the EMG profiles were comparable between freezers and non-freezers, except for increased GM and BF activity in the PD+FOG group (partially supporting Hypothesis 3).
Cueing improved gait initiation through the facilitation of early anterior muscle activity
Previous studies that examined muscle activation during self-initiated or cued stepping have principally focused on the amplitude and timing of activity in the ankle plantar and dorsi-flexors.11,28,29,31 These experiments showed that external cueing is associated with an increased magnitude and duration of the initial TA burst in people with PD and matched controls.11,29,31 Cueing is also associated with a significant increase in the suppression of the soleus H-reflex during the initiation of the APA, possibly reflecting improved presynaptic inhibition during the early part of the gait initiation sequence. 31 Our work further expands upon this by demonstrating (in an extended set of muscles) that cues improve gait initiation by altering muscle activity in an effector- and phase-specific manner within and across legs. Cueing-evoked improvements in bilateral TA activity were accompanied by significant increases in activation of both the step and stance leg VL and RF. Anterior muscle activation is critical for the posterior excursion of the center of pressure and generation of an anterior acceleration of the center of mass. In contrast, significant effects of cueing on posterior muscle activation were limited to the stance leg SOL and step leg BF. While visual inspection of Figures 3 and 4 suggests that cueing may reduce plantar flexor activity during the execution phase of gait initiation for some people, these changes did not reach significance after FDR correction. This suppression possibly reflects a reduced need for propulsive forces from the plantar flexors due to the improved weight transfer and anterior acceleration generated during early phases of the APA.12,43 However, our findings should be interpreted with some caution as the multiple comparisons included in this exploratory study were corrected with moderately conservative adjustments using the False Discovery Rate.12,43 This could have resulted in some errant rejections of the null hypothesis (Type II errors). This is especially true for the RF and GS muscles, as recordings in these muscles were obtained in only a subset of participants (n = 19) and may be particularly underpowered.
Hip abductor (gluteus medius, GM) activity is critical for stabilization of the pelvis during loading/unloading of the legs and the generation of medial/lateral acceleration of the center of mass. 44 The temporal dynamics of hip abductor activity were similar between groups. The step leg GM activity was characterized by an initial burst during the Pre-APA and APA 1 Phases (Figure 3), which acts to stabilize the pelvis during unloading of the stance leg and assist with the generation of an acceleration of the center mass towards the stance leg. Figures 3 and 4 show that cueing was associated with a facilitation of step leg GM activity during the Pre-APA and APA 1 phases, but these changes did not reach statistical significance after FDR corrections. The stance leg GM showed a distinct burst of activity during the late APA and execution phases that counteracts the hip adductor moment generated by the pelvis/trunk during unloading the step leg in preparation for toe-off.
Importantly, the modulation of muscle activity produced by external cueing was not simply an increase in the magnitude of the sEMG bursts, which would reflect a generalized increase in the gain of muscle activity across the gait initiation sequence. Instead, effects of cueing were selective to specific muscles and phases of the APA cycle. Specifically, the early bursts of activity in the TA were significantly increased by cueing, but later bursts in TA during the transition (step leg unloading to toe-off) were not facilitated. Anterior muscles (VL and RF, more active earlier in the cycle) were generally facilitated, while selective posterior muscles were suppressed (Stance SOL and Step BF, which were more active in middle to late portions of the cycle). These results support the idea that cueing acts on neural circuity directly involved in the generation of the APA activation pattern, rather than evoking a generalized increase in the gain of muscle activation across the gait initiation sequence.
Cues improve gait initiation APA kinetics
Sensory cues have been shown to improve gait initiation in a variety of laboratory and clinical contexts and can be effective for overcoming freezing episodes in people with PD.45,46 Our results show that cueing improves the magnitude and timing of the early APA kinetics (forces and CoP) across groups (Figure 4).1,2,11,16,22 The effects of external cueing on later phases of the gait initiation sequence were relatively modest. While cueing had little effect on the forces and CoP beneath the stance leg during the step execution (what Palmisano et al. called the “unloading phase” 47 ) and post-execution phases of gait initiation, cues significantly increased the magnitude of the second posterior peak of the CoP excursion during the execution phase (Supplemental Table 1) and thereby facilitated the push-off kinetics of the first step. Enhanced posterior CoP shifts are associated with a larger first step length and velocity,12,14,16,26 especially for people with PD and FOG.15,48 However, different cueing methods may alter the cue's efficacy (e.g., whether or not a warning cue is given before the imperative go cue, or if the cue is internally or externally driven).22,26,49,50
Deficits in the transition from standing to walking in people with PD
Deficits in the transition from standing to stepping contribute to the slowness of gait initiation in PD. 3 Previous studies have described changes in the magnitude and timing of TA and SOL muscle activity that correlate with deficits in the APA and first step. 51 People with advanced PD can have abnormally prolonged soleus activity, which continues through initial TA burst, causing co-contraction, a delayed weight shift, and slower gait initiation. 29 Our dataset did not show a clear suppression of soleus activity prior to onset of the APA. In both groups, activity in the TA, VL, RF, and GM muscles during the initiation of the APA was lower during self-initiated compared to cued stepping (Figure 3). This corresponds well with the reduced forces generated during uncued gait initiation irrespective of age or the presence of PD pathology (p < 0.001, see also2,22). However, APAs remain smaller and slower in people with PD (in the off-medication state) relative to matched controls, even when cues are presented (p < 0.001, see also1,2,16,22). In line with these findings, Falaki et al. 30 found that people with PD had lower muscle synergy indices in preparation for gait initiation, reflecting impaired stability in activating groups of muscles (i.e., muscle synergies). Deficits in presynaptic inhibition (agonist-antagonist interactions) during the early phases of the APA, particularly in people with freezing of gait, 52 may also contribute to impaired activation and sequencing of muscle activity during the APA.
Contrary to our second hypothesis, the changes in sEMG modulation evoked by a cue were relatively consistent across groups, as there were no group x condition or group x phase x condition interactions. However, the analysis of group x phase interactions illuminated critical group differences in muscle activity throughout the APA cycle. Compared to controls, PD had lower muscle activity during the critical weight shift portions of the APA (APA Phase 1 and 2), and larger activity in Execution and Post-Execution phases in selective muscles. The enhanced muscle activity seen late in the gait initiation cycle in PD likely reflects a compensatory mechanism for the reduced forces generated in the early phases of the APA (most evident in anterior muscles). Increased late activity in the step leg SOL, VL and RF would act to generate the forward propulsive forces and flexion of the hip needed to achieve toe-off and a safe initial clearance of the foot during the first step.
Effect of FOG on parkinsonian impairment
We have previously shown (in a subgroup of this PD cohort) that cues have comparable effects on APA kinetics in people with and without FOG. 2 We were able to replicate this result in the larger combined PD cohort studied in this experiment.2,22 While some studies have reported a similar result, others have found significantly worse impairment in APAs in people with FOG.16,50,52–55 These discrepancies likely reflect differences in the experimental conditions used to test gait initiation, such as the type and/or presence of cues, medications status, and disease severity.16,50,52–55 While the data used in our analyses did not include trials during which a FOG episode occurred, significant differences in muscle activation were observed between the PD+FOG and PD-FOG groups during specific phases. Step leg GM activity was significantly higher in the PD+FOG group early in the gait initiation cycle (Pre-APA and APA Phase 1), and the step leg hamstrings (BF) activity was significantly higher during the execution phase. Given that the magnitude of stance leg unloading was similar between the PD+FOG and PD-FOG groups, it is unlikely that the increased GM activity in the PD+FOG group reflects an increased need for stabilization of the pelvis. Instead, it may indicate alterations in the muscle activation synergies required for coupling of postural control (trunk stabilization) with the gait initiation sequence. Lira et al. 52 showed that freezers had reduced presynaptic inhibition of the soleus muscle and lower magnitude APAs compared to people with PD without FOG, reflecting an impaired capacity to uncouple the postural activity present during standing from the dorsiflexor activity required to initiate the APA. Disturbances in this transition in people with PD and FOG are commonly associated with shorter first steps, 15 freezing episodes, 3 and an increased incidence of falls,17,18 compared to PD without FOG.
Limitations
The data presented in this paper was obtained from two separate experiments.2,22 Although the protocols for self-initiated and cued stepping were the same for both experiments, the protocols differed with respect to number and types of additional experimental conditions. Differences in the length of visit, and the type and number of total trials performed across experiments may have increased the between-subject variability. However, we previously showed that, in 23 of our PD participants, a block of self-initiated trials performed at the beginning of the data collection were not significantly different from trials collected at the end of the experiment (with ∼ 45–54 trials of other experimental conditions in between), suggesting that fatigue did not play a significant role in our study, and the cues did not have a carry-over effect on later self-initiated trials. 2 The combining of PD data from two studies resulted in differences in the sample sizes of the PD vs. HOA cohorts. While the Tukey-Kramer method used is robust to uneven sample sizes, the mismatch may have reduced the robustness of the findings. Similarly, recordings from the RF and GM muscles were only obtained in a subset of individuals. For this reason, the findings in these muscles should be interpreted with caution. The exclusion of individuals with PD and significant tremor, deep brain stimulation, and more severe stages of PD reduces the generalizability of our findings across the PD population. Similarly, we did not control or account for any influence of other heterogeneities within our PD group that may have affected our results (e.g., other underlying gait and balance impairments, subgroups such as by tremor vs. akinetic-rigid dominant). Due to the need to normalize the temporal profiles of sEMG activity across the APA cycle, trials that did not have an APA (9.5% of all trials) were excluded from the analyses. Trials without an APA may utilize different muscle synergies, which warrants future investigation. 55
Additionally, our results are applicable to the acute use of cues in a controlled laboratory setting in people with PD in the off-medication state. However, it remains unclear if these effects will persist in real-world situations. While cueing has been shown to provide consistent benefits on gait over time, 56 less in known about the long-term benefits of cueing on gait initiation. Our results are also specific to initiating forward stepping with only the preferred stepping leg. It is unclear if results would be different for non-preferred stepping legs. 48
Conclusion
Cueing evoked changes in muscle activity during gait initiation that were effector- and phase-specific in both the PD and HOA groups. The most marked effects of cueing were seen in anterior muscles (both proximal and distal) of the initial stepping leg during the early phases of gait initiation. In contrast, the effects of cueing on posterior muscle activity, which are utilized later in the gait initiation sequence, were relatively small. While cueing induced similar effects in both groups, anterior muscle activity and APAs in the PD group remained lower than the HOAs in early phases of the APA, but were comparable to, or larger than, HOA levels during later phases of gait initiation. These findings highlight the critical role of early anterior muscle activity in the generation of APAs and successful transition from standing to walking. Interventions designed to train and improve gait initiation in PD should be combined with strengthening of proximal and distal leg muscles57,58 and hip abductors 44 to ensure individuals retain the capacity produce the weight shift required for an APA. Cueing can thereby be used to facilitate muscle activation and force generation, improve the quality of the first and second steps, and potentially reduce the risk of falls in people with PD.
Supplemental Material
sj-docx-1-pkn-10.1177_1877718X251382575 - Supplemental material for Surface electromyographic profiles during gait initiation in people with Parkinson's disease: The effects of external sensory cueing
Supplemental material, sj-docx-1-pkn-10.1177_1877718X251382575 for Surface electromyographic profiles during gait initiation in people with Parkinson's disease: The effects of external sensory cueing by Lesley A Perg, Chiahao Lu, Matthew N Petrucci, Jae Woo Chung, Paul J Tuite, Colum D MacKinnon and Sommer L Amundsen-Huffmaster in Journal of Parkinson's Disease
Footnotes
Acknowledgements
We thank the volunteers for participation in this research. We thank Joshua De Kam and Jacqueline Vachon for coordinating the research, Hans Zander and Devin McCoy for help in data collection, and Abigail Kohut-Jackson, Danielle Berres, Daniel Bergevin-Smith, Hannah Bergeson, Sarah Lund, Rebecca George, and Kristin Seely for data post-processing.
ORCID iDs
Ethical considerations
All procedures were approved by the Institutional Review Board of the University of Minnesota (1302M27746 and 1407M52163).
Consent to participate
Written informed consent was obtained from the participants prior to inclusion in the study.
Consent for publication
Not applicable.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the following grants: NIH NINDS RO1 NS070264, NS088679 (CDM, SLAH, CL, PT, JC), R01 NS136323 (SLAH, CDM, JC, PT), R01 NS085188 (SLAH, CDM, PT), P50 NS09857 (CDM, SLAH, CL, PT) and MnDRIVE (SLAH, MP), the Engdahl Family Foundation (SLAH), the Clinical and Translational Science Institute at the University of Minnesota (NIH UL1TR002494), and the REDCap database (UL1TR000114).
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
Data will be shared upon reasonable request and approval of the University of Minnesota Institutional Review Board.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
