Abstract
Background
Levodopa-induced dyskinesia (LID) in Parkinson's disease (PD) is linked to exaggerated gamma oscillations. Buspirone, a 5-HT1A receptor agonist, is a potent medication for psychiatric conditions, predominantly prescribed for anxiety treatment.
Objective
This study aims to investigate whether buspirone alleviates dyskinesia in LID rat and its effects on pathological oscillatory activity and cortico-striatal functional connectivity.
Methods
We collected motor behavior and electrophysiological data of cortico-striatal from Sham rats, unilateral 6-hydroxydopamine (6-OHDA)-lesioned PD model rats, and rats with LID. We further examined the behavioral and electrophysiological changes in LID rats following buspirone intervention.
Results
PD rats showed increased beta activity and aperiodic components at 10–50 Hz, while LID rats exhibited excessive gamma oscillations and aperiodic activity at 50–150 Hz. Additionally, gamma-band functional connectivity within the cortico-striatal circuit was significantly enhanced during on-state dyskinesia, when rats exhibited abnormal involuntary movements. Administration of buspirone effectively reduced dyskinesia severity, suppressed gamma activity, decreased aperiodic components (50–150 Hz), and disrupted gamma-band functional connectivity without compromising the antiparkinsonian effects of levodopa.
Conclusions
Excessive gamma oscillations represent a key electrophysiological marker of dyskinesia. Altered gamma-band connectivity within the cortico-striatal network may contribute to its pathophysiology. Buspirone appears to be a promising candidate for the treatment of LID, potentially offering a novel therapeutic strategy.
Plain language summary
Parkinson's disease is a brain disorder that causes shaking, stiffness, and difficulty moving. It happens when certain brain cells that produce a chemical called dopamine are lost. Levodopa is the most effective medicine for improving movement in Parkinson's disease, but after taking it for a long time, many people develop uncontrolled, jerky movements called dyskinesia. These abnormal movements can greatly affect quality of life, and there are currently limited treatment options. In this study, we tested whether buspirone—a drug that affects the brain's serotonin system and is already approved for anxiety—could reduce dyskinesia in a rat model of Parkinson's disease. The rats were given a toxin to mimic Parkinson's disease and then treated with levodopa to cause dyskinesia. We measured both their movements and their brain activity. Rats with dyskinesia showed unusually strong fast brain rhythms, especially in areas controlling movement, and stronger-than-normal connections between these areas. When buspirone was given together with levodopa, the rats’ abnormal movements were reduced without losing the beneficial effects of levodopa. Their brain rhythms also returned closer to normal. Our findings suggest that buspirone could be a promising way to control dyskinesia in Parkinson's disease, and they point to specific brain activity patterns that could guide future treatments.
Keywords
Introduction
Parkinson's disease (PD) is a common neurodegenerative disorder, primarily characterized by the degeneration of dopaminergic neurons in the substantia nigra and striatum. 1 The standard treatment for PD is dopaminergic replacement therapy. 2 However, long-term administration of levodopa (L-dopa) can result in the development of dyskinesia, primarily manifesting as involuntary movements of the trunk, limbs, and oral-facial regions. 3 Currently, there is no effective medications for treating levodopa-Induced dyskinesia (LID) in clinical practice. 4
Local field potentials (LFP), which reflect aggregate neural activity across neuronal populations, can be analyzed through advanced electrophysiological methods to infer functional connectivity and directional flow between brain regions. 5 These features hold promise as clinical biomarkers for both disease diagnosis and long-term therapeutic prognosis. 5 Current researches suggest a strong relationship between beta oscillations and PD, as widely validated in both patients and animal models.6,7 In the PD state, the functional connectivity in beta-band activity within basal ganglia circuits is significantly increased, and studies have shown that the strength of beta oscillations is closely related to the severity of PD.8,9 Deep brain stimulation of the subthalamic nucleus (STN-DBS) treatment can reduce the activity of beta oscillations, accompanied by an improvement in PD motor symptoms. 8 Additionally, study has shown that L-dopa administration in PD patients suppresses beta oscillations while eliciting gamma oscillations. 10 This phenomenon has also been confirmed in animal models, where L-dopa treatment elicits gamma oscillations activity in LID rats.11–13
5-hydroxytryptamine (5-HT) neurons have been shown to convert exogenous L-dopa into dopamine (DA), store it in synaptic vesicles, and release it in an activity-dependent manner. 14 However, due to the absence of an intrinsic negative feedback mechanism to regulate synaptic DA levels, this release is poorly controlled. 15 As a result, 5-HT neurons contribute to sustained and dysregulated DA release, leading to abnormally elevated striatal DA concentrations and ultimately the development of LID . Drugs targeting 5-HT receptors, including 5-HT1A agonists, 5-HT1A/B agonists, 5-HT2A antagonists, and 5-HT transporter inhibitors, have been tested and found effective in alleviating LID in animal studies.16,17 The primary issue is that, despite promising preclinical results, these drugs have not demonstrated sufficient efficacy in clinical trials. 18 Buspirone is a partial agonist of the 5-HT1A receptor, which has been shown to effectively treat generalized anxiety disorder, with good tolerance in elderly populations, and may also be effective in treating anxiety in PD.19,20 Through its action on the 5-HT1A receptor, buspirone may offer additional benefits in reducing LID. The study aims to explore, from an electrophysiological perspective, whether buspirone can alleviate LID behaviors, thereby providing scientific evidence for the clinical treatment of LID.
Methods
Experimental procedures
Animals
To exclude potential interference from hormonal cycles (e.g., estrogen and progesterone fluctuations), only male rats were utilized in this study. Fifty-six healthy male Sprague-Dawley rats (12 weeks old, weighing 280–300 g) were obtained from Beijing Vital River Laboratory Animal Technology Co., Ltd (Beijing, China). Animals were housed under controlled conditions (25°C, 12 h light/dark cycle). The experimental protocol received approval from Zhengzhou University and followed the National Institutes of Health Guide for laboratory animal care and use.
Experiment 1
Eighteen rats (Sprague-Dawley rat, male, 280–300 g) were implanted with electrodes and sleeve tubes on day 1, followed by a 12-day recovery period (Figure 1A). Electrophysiological data recording commenced on day 14. On day 15, we injected 4 µl 6-hydroxydopamine (6-OHDA, Sigma, St Louis, MO, USA) solution (4 µg/µl) dissolved in 0.2 mg/mL L-ascorbic acid (Macklin, Shanghai, China) in 0.9% w/v saline unilaterally into the right medial forebrain bundle (MFB) through sleeve tube. 21 Electrophysiological recordings of the rats were conducted on day 28, followed by the apomorphine (APO, Sigma, USA) rotation test on the same day to confirm successful PD modeling.
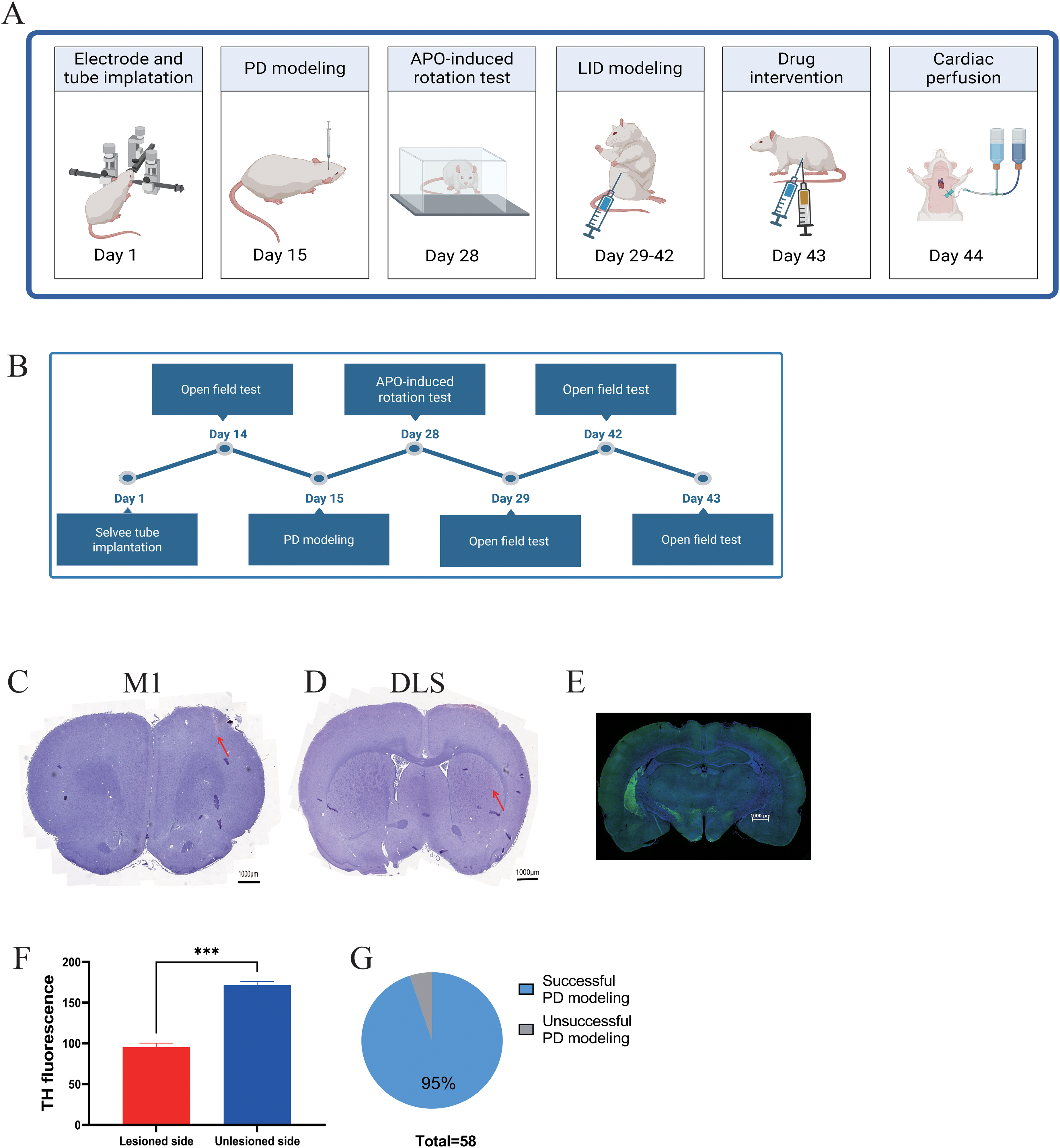
(A) Overview of the experimental design for Experiment 1. (B) Experimental workflow chart in experiment 3. (C) Electrode placement diagrams for the M1 brain regions. (D) Electrode placement diagrams for the DLS brain regions. Scale bars: 1000 μm. (E) Representative TH immunolabeling of dopaminergic neuron in the SNc of PD rats. Scale bars: 1000 μm. (F) Quantitative result of lesioned and unlesioned side (n = 54, t = 22.12, p < 0.001). (G) Success rate of the PD model (n = 58). Data are expressed as mean ± SEM. ***p < 0.001. Paired Student's t-test with two-tailed was used for Figure1F.
Subsequently, from day 29 to 42, the rats underwent LID modeling that received coadministration of L-dopa (8 mg/kg, Macklin, Shanghai, China) and benserazide (12 mg/kg, Macklin, Shanghai, China) via intraperitoneal injection (ip) for 14 days. On day 42, electrophysiological recordings were performed 80 min after drug injection, along with the recording of abnormal involuntary movements (AIMs). On day 43, 16 LID rats were divided into two groups: one group received L-dopa + benserazide + saline (n = 8, LID + L-dopa group, ip), while the other received L-dopa + benserazide + buspirone (n = 8, LID + L-dopa + Buspirone group, ip). Electrophysiological recordings were conducted 80 min after drug administration. All rats were euthanized on day 44 (Figure 1A).
Dose-response study of buspirone in LID
Experiment 2 assessed the dose-response relationship of buspirone to determine the optimal concentration for alleviating LID. Doses ranged from 0 to 4 mg/kg in 1 mg/kg increments, with 6 rats per group. Thirty rats were subjected to PD modeling on day 1, followed by a 14-day PD modeling period. From days 15 to 28, the rats were treated with L-dopa and benserazide. On day 29, five groups of rats received L-dopa and benserazide, along with buspirone at doses of 0 mg/kg, 1 mg/kg, 2 mg/kg, 3 mg/kg, or 4 mg/kg. Behavioral assessments of LID were subsequently recorded via video and evaluated using a blinded scoring system.
Experiment 3
Experiment 3 aimed to demonstrate that buspirone alleviates dyskinesia without antagonizing the antiparkinsonian effects of L-dopa (Figure 1B). Eight rats were implanted with sleeve tube on day 1 and allowed to recover for 13 days. Baseline motor function was assessed on day 14 using the open field. On day 15, 6-OHDA (4 µL, 4 µg/µL) was injected into the MFB via the sleeve tube to induce a unilateral Parkinsonian lesion. APO-induced rotation test was performed on day 28 to confirm successful PD modeling. On day 29, the open field was performed to assess motor deficits before LID modeling. From days 29 to 42, rats received daily intraperitoneal injections of L-dopa (8 mg/kg) and benserazide (12 mg/kg) to induce dyskinesia. The open field was performed on day 42, respectively, 80 min after L-dopa injection during on-state dyskinesia, characterized by the presence of abnormal involuntary movements. On days 43, rats received L-dopa, benserazide, and buspirone. The open field test was performed on day 43 at 80 min post-injection. All rats were euthanized on day 44 for histological analysis (Figure 1B).
Electrode and sleeve tube implantation
Rats were anesthetized using 1% pentobarbital (50 mg/kg, i.p.). Following anesthesia, they were secured in a stereotaxic frame. After skull exposure, bregma and lambda were leveled to establish the horizontal plane. Stereotaxic coordinates relative to bregma and skull surface were determined for the MFB, anterior-posterior (AP) −1.8 mm from bregma; medial- lateral (ML) 2.0 mm from bregma; and dorsal-ventral (DV), −8.35 mm from the skull surface, the motor cortex (M1, AP: −3.5 mm, ML: 2.5 mm, and DV: −8.2 mm), and the dorsolateral striatum (DLS, AP: −0.12 mm; ML: 3.5 mm; DV: −5.5 mm) were determined. Bone windows were created using a cranial drill. Electrode bundles, comprising eight Teflon-insulated stainless steel microwires (50 μm diameter; Plexon, Hong Kong, China) arranged in a 3 × 3 × 2 array (250 μm inter-wire spacing), were implanted alongside insulating sleeves (RWD, Shenzhen, China) at designated targets. Skull screws (RWD, Shenzhen, China) were affixed to the surface of the skull, and finally, dental cement (RWD, Shenzhen, China) was employed to secure the electrodes and tubes in place.
Behavioral test
APO-induced rotation test
APO-induced rotation (0.75 mg/kg, i.p.; Sigma, USA) was utilized to evaluate unilateral dopamine depletion. Rotation performance was assessed 10 min post-injection, with successful lesion creation indicated by the occurrence of 20 contralateral rotations within a 5-min period. 22 APO-induced rotation was conducted on day 28.
Open field test
The open field test assessed spontaneous locomotor activity. Individual rats were placed in the center of a square opaque plastic arena (100 × 100 × 40 cm; non-reflective surface), with video-tracking software (EthoVision XT, Noldus, Netherlands). Each 5-min session recorded total distance traveled. Between trials, the arena was cleaned with 75% ethanol to eliminate olfactory cues. All testing occurred during the light phase in a quiet, temperature-controlled room.
AIMs
AIMs scores were determined following a standardized protocol, 6 focusing on three categories: locomotive (axial torsion and rotation toward the contralateral side), limb (involuntary movements of the forelimb or digits on the lesioned side), and orolingual (including jaw activity, tongue protrusion, and contralateral licking). Each subtype was rated on a five-point scale (0–4): 0 = no movement; 1 = occasional (<50% of the time); 2 = frequent (>50%); 3 = continuous but interruptible with strong sensory input; 4 = continuous and resistant to interruption. 15 Scoring was conducted across ten time points at 20-min intervals (0–180 min), with each session lasting 2 min. On days 42 and 43, AIM was evaluated following pharmacological treatment. For each observation, the severity of all three subtypes was documented, yielding a maximum score of 12 per session (4 per subtype), and a potential total of 120 per day.
Electrophysiological data recording
A Plexon system with 64 channels (USA) was utilized for electrophysiological data recording, configured with a sampling rate of 1000 Hz. The acquired signals underwent bandpass filtering using a sixth-order zero-phase Butterworth filter, set within a frequency range of 0.1 to 1000 Hz. Awake and freely moving rats were situated in a cage for 5-min local field potential (LFP) recordings on days 13, commencing at 9:00 AM. Additionally, on days 42 and 43, 5-min LFP recordings were conducted 80 min post-drug administration. 23
Histological analysis
The rats were anesthetized via an intraperitoneal injection of pentobarbital sodium (50 mg/kg) and then subjected to a 30-s +20-μA to mark the recording electrode sites. The rats then underwent transcardial perfusion with 0.9% saline (400 mL) at 2 °C, followed by perfusion with 4% paraformaldehyde (400 mL) to preserve the tissue. Post-perfusion, brains were carefully extracted, post-fixed in 4% paraformaldehyde for 24 h, dehydrated, and embedded in paraffin. Using a paraffin microtome (Leica, Germany), coronal sections (8 μm thickness) were precisely cut through the M1 and DLS regions. Hematoxylin staining (10 min) preceded differentiation in 1% acid-alcohol, bluing reagent treatment for nuclear contrast enhancement, and distilled water washes. Cytoplasmic counterstaining with eosin (3 min) was succeeded by graded ethanol dehydration (80%:5 s; 95% and 100%:2 min each). Final clearing in xylene (2 × 4 min) and neutral resin mounting completed tissue processing. Subsequently, the stained sections were examined under an optical microscope (Magnification 1x, Leica, Wetzlar, Germany) for verification the position of electrode. 24
Immunofluorescence staining
Immunofluorescence staining was performed to assess tyrosine hydroxylase (TH) levels in the substantia nigra pars compacta (SNc). Tissue sections were heated at 65°C for 1 h, followed by paraffin removal and rehydration through sequential washes: (1) dimethylbenzene (3 × 10 min), (2) ethanol (100%, 95%, 85%, and 75%, 5 min each), and (3) ddH2O (3 × 5 min). Antigen retrieval was performed by immersing sections in preheated citrate buffer (Beyotime) and heating in a microwave until boiling, followed by continued boiling for 15 min. After cooling to room temperature (25°C), sections were washed three times with phosphate-buffered saline (PBS) for 5 min each. Next, sections were blocked with 5% normal donkey serum (Jackson ImmunoResearch, 017-000-121-E, Lancaster, PA, USA) in PBS containing 0.03% Triton X-100 at room temperature for 1 h. They were then incubated overnight at 4°C with a rabbit anti-TH (extracellular) polyclonal antibody (1:500, Abcam, Cambridge, UK; Cat# EP1532Y, RRID: AB_2801410). Following three PBS washes, sections were incubated for 2 h at room temperature with Alexa Fluor 594-conjugated donkey anti-rabbit secondary antibody (1:500, Jackson ImmunoResearch, Cat# 711-545-152, RRID: AB_2313584). After additional PBS washes, nuclei were counterstained with 4′,6-diamidino-2-phenylindole (DAPI) (C0065-10, Solarbio, Beijing, China) for 3 min. Fluorescence images were captured using an ECLIPSE Ti2-E inverted microscope (10x, Nikon Corporation, Tokyo, Japan). TH + cell quantification was performed using ImageJ software (1.53 K, Java 1.8.0_172, NIH, Bethesda, MD, USA), based on the optical fractionator principle.
Electrophysiological data analysis
Data processing
The electrophysiological data were analyzed for power spectral densities, periodic and aperiodic components, and functional connectivity. The data were pre-processed using the Fieldtrip toolbox. The LFP was divided into segments using the ft_definetrial function, each segment lasting 1 s. The ft_artifact_zvalue and ft_rejectartifact functions were used to identify and remove artifacts. 24
Power and time-frequency analyses
The power spectrum densities and time-frequency spectrum were computed using the fast Fourier transform algorithm with custom written code in Maltab (R2024a). A total of 180 processed data segments, each lasting 1 s, were used for calculating the power spectrum density. A Hanning window (1 ms) with a 0.9 overlap was applied during the analysis. The results of the time-frequency analysis were presented within the frequency range of 1 to 150 Hz with a step of 0.1 Hz. 25 The area under the curve of the power spectral density (PSD-AUC) within beta-band (25–35 Hz) and gamma-band (80–100 Hz) frequency were calculated. 14
Analysis of periodic and aperiodic components
Electrophysiological signals were analyzed using MATLAB to differentiate their periodic components (power, peak frequency, and bandwidth) and aperiodic components (exponent and offset). 26 The exponent indicates the slope of power spectra after adjusting periodic oscillations, while the offset reflects a consistent power shift across frequencies. 26 The aperiodic components within the 10–50 Hz and 50–150 Hz bands were estimated by fitting the power spectral density using the Fitting Oscillations & One-Over-F (FOOOF) algorithm, which separates periodic and aperiodic activity by modeling the background 1/f-like signal. This method was applied as previously described in our earlier work. 27
Functional connectivity
Coherence, measured as the similarity of LFPs across frequencies (ranging from 0 for no linear association to 1 for perfect similarity), was assessed to quantify functional connectivity between the M1 and DLS using the ft_connectivity analysis function in Fieldtrip. The parameters for spectral analysis were defined as cfg.foilim = 1:1:150, indicating that power was calculated across frequencies from 1 to 150 Hz in 1 Hz steps. The smoothing parameter cfg.tapsmofrq = 5 applied a ± 5 Hz frequency smoothing to each estimate to improve the stability of the spectral output. 14
PLV was computed using the filtfilt function (Matlab 9.3.0.713579, R2017b, MathWorks) on preprocessed M1 and DLS data within the 25–35 Hz (beta) and 80–110 Hz (high gamma) frequency bands, which were selected based on their known association with motor control and dyskinesia in rodent models. 27 Instantaneous phases were extracted via Hilbert transformation, and phase differences were defined as PLV. Low PLV values indicated unsynchronized signals, while high PLV values reflected higher synchronization. 14
Statistical analyses
The areas under the linear graph curves of the power spectrum density were calculated. Data are presented as means with standard errors. Prior to statistical analysis, all data were tested for normality. Data that followed a normal distribution underwent parametric tests, while non-normally distributed data were analyzed using the nonparametric Kolmogorov–Smirnov test. Comparisons between groups were made using paired or unpaired one-way ANOVA, paired or unpaired Student's t-test, as appropriate. A p-value of < 0.05 was considered statistically significant. Statistical analyses were performed using Graphpad Prism (version 9.0.0 for Windows, GraphPad Software, Boston, MA, USA, www. graphpad.com).
Results
Evaluation of PD-like disease induction and electrode placement accuracy
In the experiment, one rat was excluded due to failure in the APO-induced rotation test, which is used to confirm PD-like symptoms. One rat during LID modeling was excluded due to mortality, but no rats were excluded for incorrect electrode placement. In Experiment 2, two rats were excluded due to failure in the APO-induced rotation test. No rat was excluded in experiment 3. Figure 1C and 1D show the electrode placements in the M1 and DLS brain regions, illustrating their specific locations within the brain. Figure 1E presents representative images of TH immunostaining in the SNc of PD model rats, illustrating dopaminergic neuron loss. The unlesioned side exhibited a higher number of TH-positive cells in the SNc compared to the lesioned side (Figure 1F). Figure 1G illustrates the induction success rate of the PD model, assessed by APO-induced rotational behavior. The contralateral rotations of PD rats during the APO-induced rotational test was present in Supplemental Table 1. Out of all the animals, three failed to meet the APO-based criteria, yielding a 95% success rate.
Excessive gamma activity during on-state dyskinesia
LFP analysis was conducted before PD modeling (day 1), after PD modeling (day 29), and after LID modeling (80 min post-L-dopa injection on day 42). The spectrogram and linear graph showed significant beta activity (25–35 Hz) in the PD state and gamma oscillations (80–100 Hz) in the LID state across the three groups (Figure 2A, B, E, and F). Paired one-way ANOVA revealed a significant increase in beta activity (25–35 Hz) in PD rats compared to Sham and LID rats (Figure 2C and G). Conversely, higher gamma activity (80–100 Hz) was observed in the LID state compared to Sham and LID rats (Figure 2D and H), with a more pronounced increase in the M1 brain region compared to the DLS area.
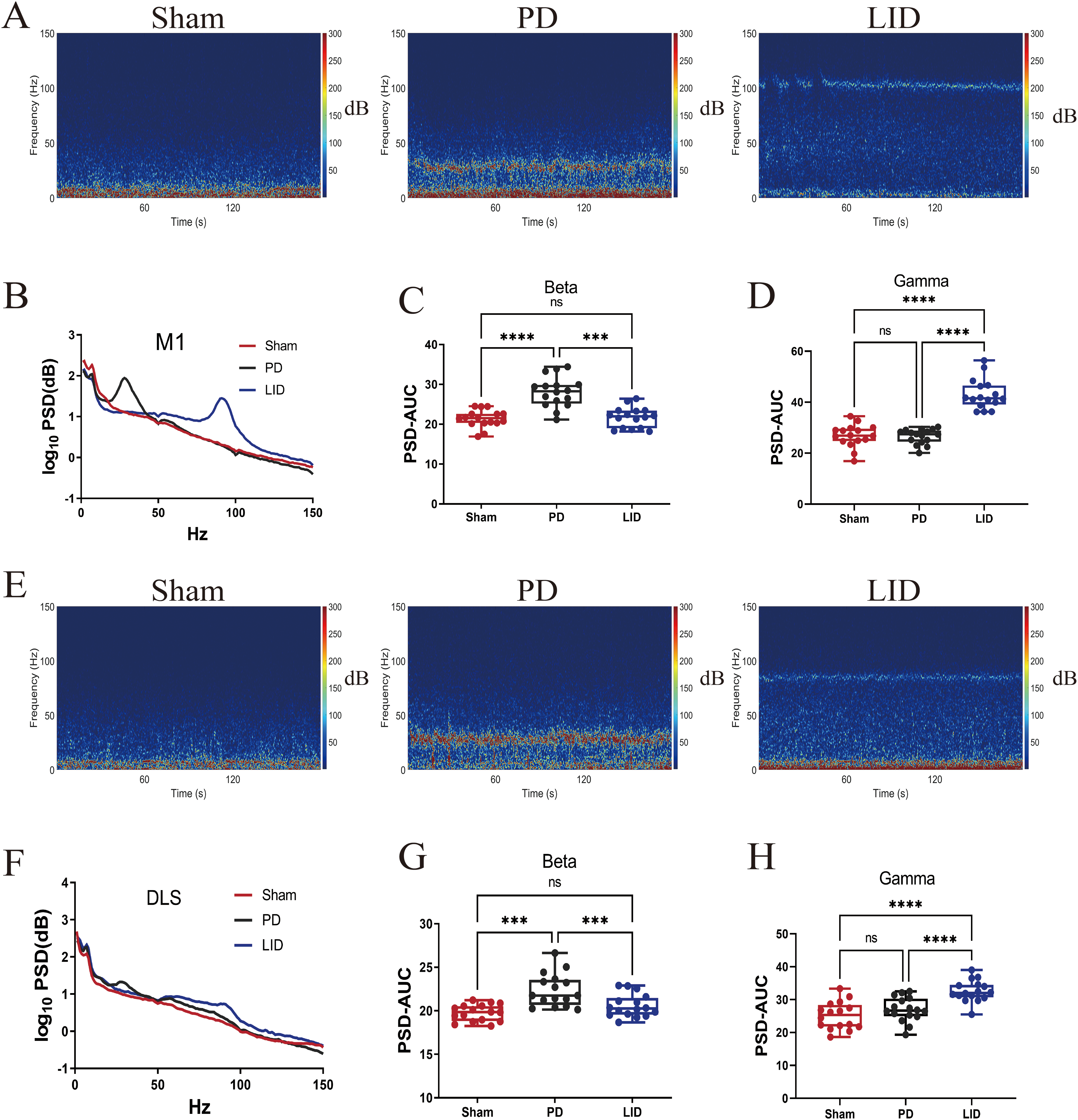
Exaggerated beta oscillations in PD and excessive gamma oscillations in LID. (A) Representative spectrograms of the M1 among groups. (B) Mean cortical power spectral density plots. (C) PD group showed higher beta power in M1 than Sham and LID groups (F = 30.70, PD vs. Sham, p < 0.0001; PD vs. LID, p = 0.0002). (D) LID group showed stronger gamma power than Sham and PD groups in M1 (F = 73.69, LID vs. Sham, p < 0.0001; LID vs. PD, p < 0.0001). (E) Representative spectrograms of the DLS. (F) Mean power spectral density plots of the DLS. (G) The PD group exhibited higher beta power in the DLS compared to Sham and LID groups (F = 19.39, PD vs. Sham, p = 0.0004; PD vs. LID, p = 0.0004). (H) The LID group demonstrated stronger gamma power in the DLS than Sham and PD groups (F = 24.75, LID vs. Sham, p < 0.0001; LID vs. PD, p < 0.0001). ns, p > 0.05, ***p < 0.001, ****p < 0.0001. Sham, PD, and LID groups; n = 16 per group. Paired one-way ANOVA followed by Tukey's HSD post hoc test was used for Figure 2C, D, G, and H.
Increased aperiodic components at 50–150 Hz during on-state dyskinesia
We then analyzed the changes in aperiodic components among the three groups of rats, as shown in Figure 3A. Similar to the power, aperiodic components (exponent and offset) at 10–50 Hz increased in the M1 region in the PD state (exponent, Figure 3C; offset, Figure 3D). During on-state dyskinesia, the aperiodic components at 50–150 Hz increased in the M1 region in the LID group compared to the other two groups (exponent, Figure 3E; offset, Figure 3F). Similarly to M1, increased aperiodic components at 10–50 Hz in PD (Figure 3H and I) and excessive aperiodic components at 50–150 Hz in LID (Figure 3J and K) were identified in the DLS region.
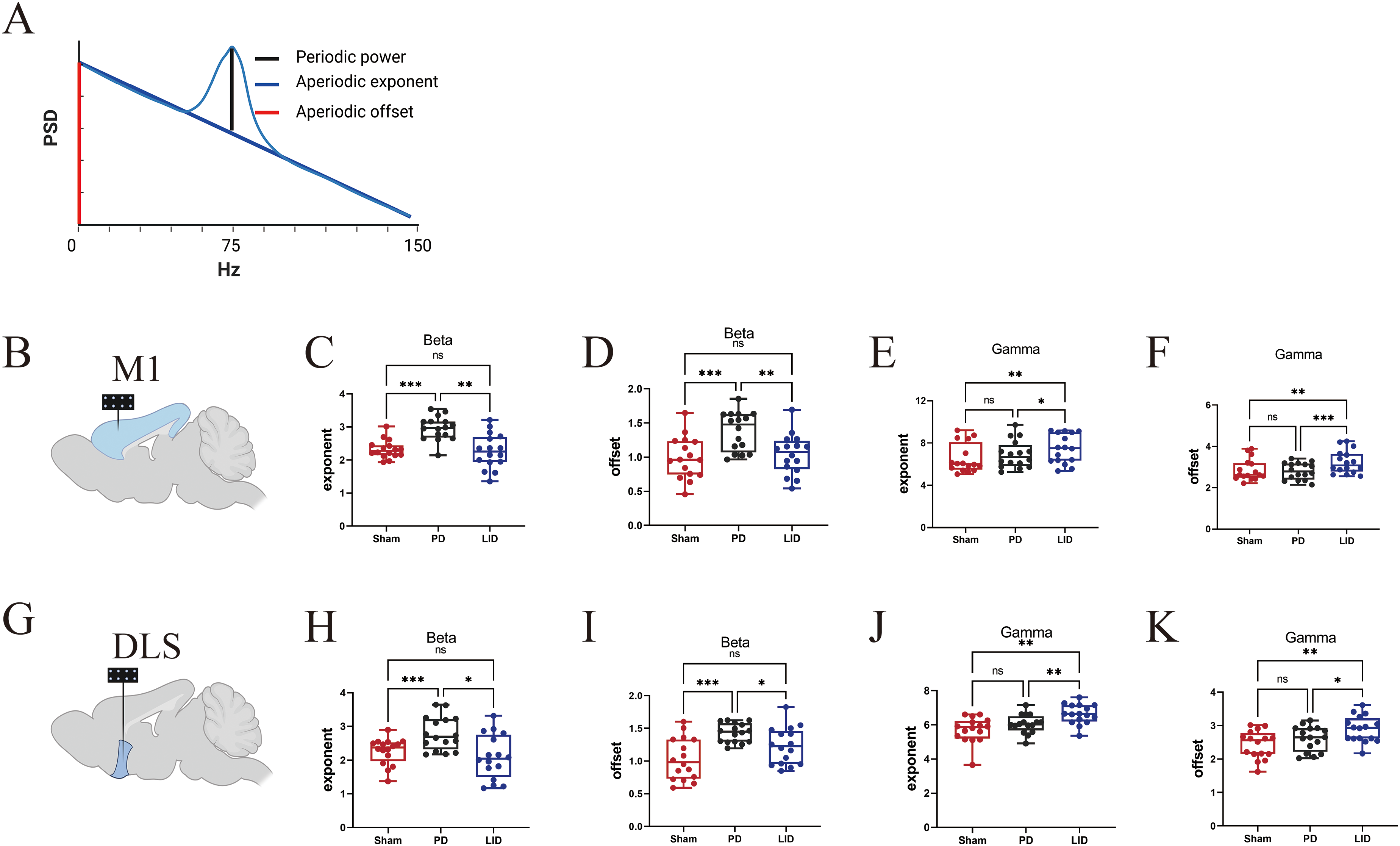
Exaggerated aperiodic components at 10–50 Hz in PD and excessive aperiodic components at 50–150 Hz during on-state dyskinesia. (A) Schematic of aperiodic and periodic components. (B) Schematic of electrode placement in the M1. (C) Exaggerated aperiodic exponent at 10–50 Hz in the PD group in the M1 (F = 14.91, PD vs. Sham, p = 0.0004; PD vs. LID, p = 0.0014). (D) Elevated aperiodic offset at 10–50 Hz in the PD group in the M1 (F = 11.05, PD vs. Sham, p = 0.0006; PD vs. LID, p = 0.0012). (E) Excessive aperiodic exponent at 50–150 Hz in the LID group in the M1 (F = 9.47, LID vs. Sham, p = 0.0025; LID vs. PD, p = 0.0133). (F) Excessive aperiodic offset at 50–150 Hz in the LID group in the M1 (F = 10.15, LID vs. Sham, p = 0.0079; LID vs. PD, p = 0.0007). (G) Schematic of electrode placement in the DLS. (H) Exaggerated aperiodic exponent at 10–50 Hz in the PD group in the DLS (F = 9.89, PD vs. Sham, p = 0.0004; PD vs. LID, p = 0.012). (I) Exaggerated aperiodic offset at 10–50 Hz in the PD group in the DLS (F = 12.09, PD vs. Sham, p = 0.0002; PD vs. LID, p = 0.0378). (J) Excessive aperiodic exponent at 50–150 Hz in the LID group in the DLS (F = 8.73, LID vs. Sham, p = 0.006; LID vs. PD, p = 0.0034). (K) Excessive aperiodic offset at 50–150 Hz in the LID group in the DLS (F = 8.04, LID vs. Sham, p = 0.0029; LID vs. PD, p = 0.024). ns, p > 0.05, *p < 0.05, **p < 0.01, ***p < 0.001, ****p < 0.0001. Sham, PD, and LID groups; n = 16 per group. Paired one-way ANOVA followed by Tukey's HSD post hoc test was used for Figure 3C-F and H-K.
Enhanced gamma-band functional connectivity between M1 and DLS during on-state dyskinesia
To explore the impact of L-dopa treatment on the cortical-striatal circuit under different conditions, we computed coherence between the M1 and DLS using FieldTrip's magnitude-squared coherence function, which reflects the frequency-specific consistency in phase and amplitude between signals (Figure 4A). Beta-band coherence (25–35 Hz) was significantly higher in the PD group compared to the Sham and LID groups (Figure 4B and C). In contrast, gamma-band coherence (85–105 Hz) was strongest in the LID group compared to Sham and PD groups (Figure 4B and D). Likewise, beta-band PLV (25–35 Hz) was increased in the PD group relative to the Sham and LID groups (Figure 4E and F), while gamma-band PLV (80–110 Hz) was significantly higher in the LID group compared to both the Sham and PD groups (Figure 4E and G).
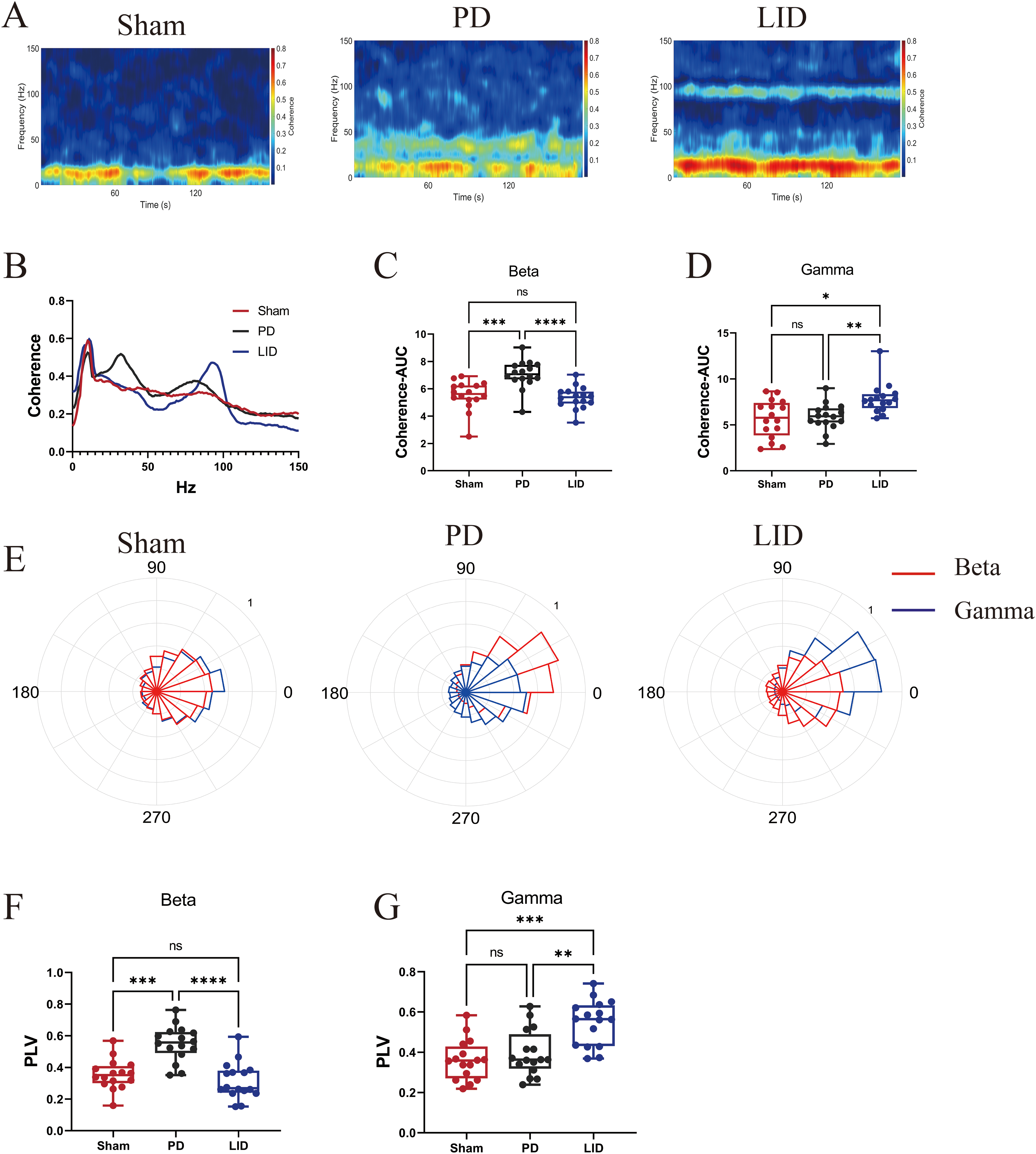
Increased functional connectivity between M1 and DLS. (A) Representative coherence spectrograms among groups. (B) Linear graph of M1-DLS coherence among groups. (C) PD group showed higher beta-band coherence than Sham and LID groups (F = 4.17, PD vs. Sham, p = 0.0001; PD vs. LID, p < 0.0001). (D) LID group showed higher gamma-band coherence than Sham and PD groups (F = 6.59, LID vs. Sham, p = 0.0261; LID vs. PD, p = 0.0054). (E) Representative polar histograms of phase locking between M1 and DLS; red lines indicate beta oscillations, blue lines indicate gamma oscillations. (F) Increased beta-band PLV in PD group (F = 21.05, PD vs. Sham, p = 0.0004; PD vs. LID, p < 0.0001). (G) Excessive gamma-band PLV in LID group (F = 12.18, LID vs. Sham, p = 0.0002; LID vs. PD, p = 0.0039). ns, p > 0.05, *p < 0.05, **p < 0.01, ***p < 0.001, ****p < 0.0001. Sham, PD, and LID groups; n = 16 per group. Paired one-way ANOVA followed by Tukey's HSD post hoc test was used for Figure 4C, D, F and G.
Buspirone alleviated dyskinesia without diminishing the antiparkinsonian effects of l-dopa
In Experiment 2, we investigated the effects of buspirone on LID in rats by evaluating AIMs. We identified that the optimal dose of buspirone for reducing hyperkinesia was 3 mg/kg (Figure 5A). This dose was then used in Experiment 1 and 3.
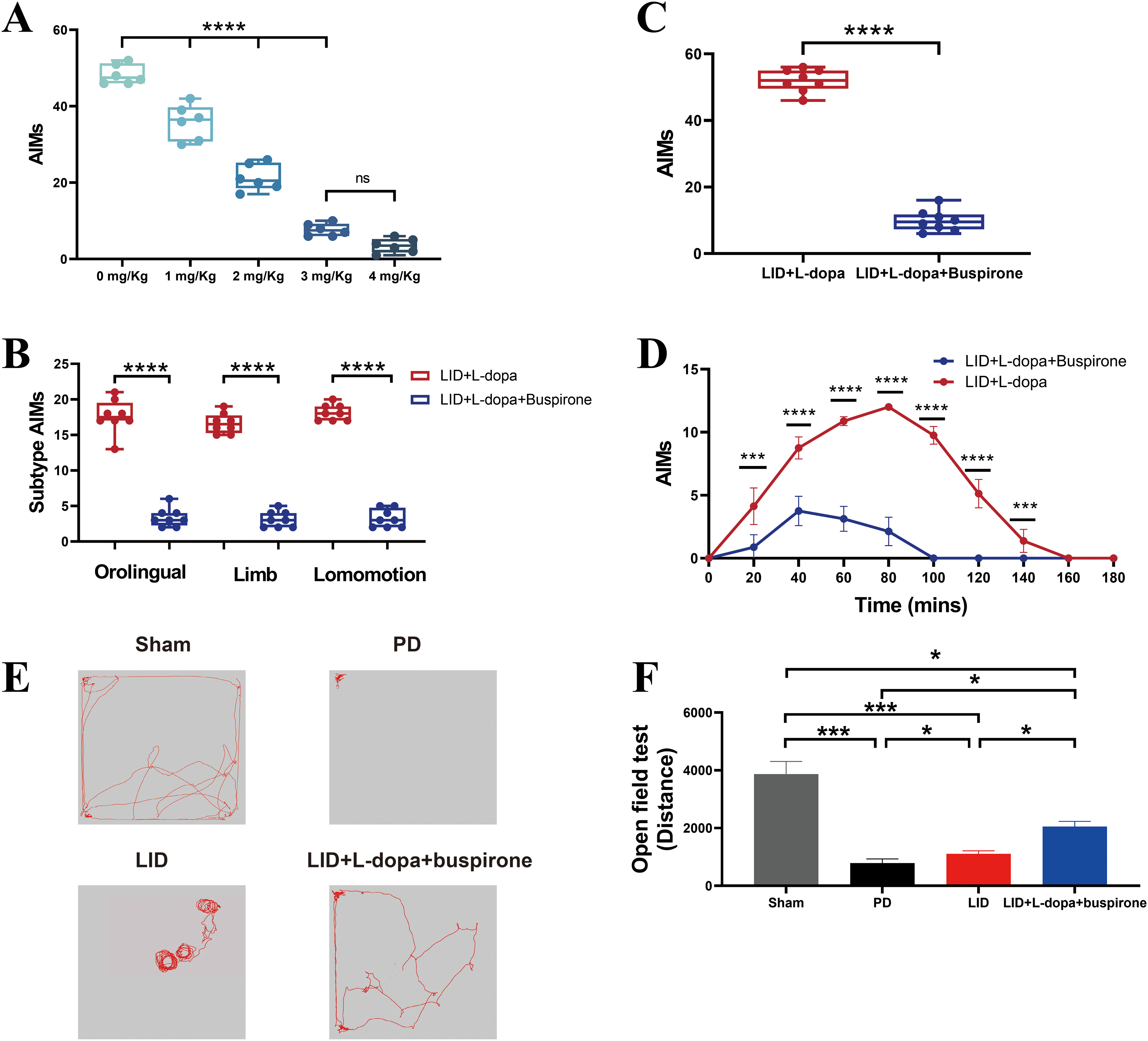
Buspirone administration alleviated LID. (A) Total AIMs of LID rats following different concentrations of buspirone administration among groups (n = 6 per group, F = 299.90, 0 mg/Kg vs. 1 mg/Kg, 0 mg/Kg vs. 2 mg/Kg, 0 mg/Kg vs. 3 mg/Kg, 0 mg/Kg vs. 4 mg/Kg, 1 mg/Kg vs. 2 mg/Kg, 1 mg/Kg vs. 3 mg/Kg, 1 mg/Kg vs. 4 mg/Kg, 2 mg/Kg vs. 3 mg/Kg, 2 mg/Kg vs. 4 mg/Kg, p < 0.0001). (B) LID + L-dopa group showed higher total AIMs than LID + L-dopa + buspirone group (t = 25.50, p < 0.0001). (C) LID + L-dopa group showed higher subtype AIMs than LID + L-dopa + buspirone group (Orolingual AIMs, t = 14.82, p < 0.0001; Limb AIMs, t = 16.63, p < 0.0001; Lomomotion AIMs, t = 24.66, p < 0.0001). (D) AIMs at each time points of LID + L-dopa and LID + L-dopa + buspirone groups (20 min, t = 5.22, p = 0.0001; 40 min, t = 9.66, p < 0.0001; 60 min, t = 20.83, p < 0.0001; 80 min, t = 24.81, p < 0.0001; 100 min, t = 39.00, p < 0.0001; 120 min, t = 12.87, p < 0.0001; 140 min, t = 4.25, p < 0.0001). ns, p > 0.05, ***p < 0.001, ****p < 0.0001. n = 8 per group for LID + L-DOPA and LID + L-DOPA + buspirone conditions. (E) Trajectory map of open field test among groups (n = 8 per group). (F) Quantitative analysis of total distance traveled revealed a significant increase in locomotor activity in the buspirone-treated group compared to the LID and PD groups (F = 28.08, Sham vs. PD, p = 0.0007; Sham vs. LID, p = 0.0010; Sham vs. LID + L-dopa + buspirone, p = 0.0244; PD vs. LID, p = 0.0244; PD vs. LID + L-dopa + buspirone, p = 0.0146; LID vs. LID + L-dopa + buspirone, p = 0.0244). Unpaired one-way ANOVA with Tukey's HSD post hoc test for Figure 5A, and unpaired Student's t test with two-tailed for Figure 5B-D. Paired one-way ANOVA followed by Tukey's HSD post hoc test was used for Figure 5F.
In Experiment 1, we compared motor behavior between day 43 (LID + L-dopa group) and day 44 (LID + L-dopa + buspirone group). Buspirone administration resulted in a significant reduction in total AIM scores (Figure 5B), with all AIM subtypes showing marked decreases (Figure 5C). Moreover, AIM scores at multiple time points (20, 40, 60, 80, 100, 120, and 140 min) were significantly lower on day 44 compared to day 43 (Figure 5D).
In Experiment 3, we used the open field test, a commonly employed behavioral test for evaluating motor function in both healthy and PD rats. In LID rats, co-administration of buspirone maintained L-DOPA–induced locomotor performance, resulting in higher motor activity compared with the PD group (Figure 5E and F).
Buspirone suppresses exaggerated gamma oscillations in LID
To investigate the potential effects of buspirone on gamma band activity in LID rats, buspirone (i.p.) was administered to LID rats on day 43, creating the LID + L-dopa + buepirone group. Simultaneously, eight LID rats were treated with L-dopa (8 mg/kg, i.p.) along with benserazide (12 mg/kg, i.p.) on day 43, forming the LID + L-dopa group.
As shown in Figure 5A-C, the buspirone increased beta power (25–35 Hz, Figure 6A-C) and decreased gamma power (80–100 Hz, Figure 6A, B, and D) in the M1 in LID rats. Similarly, buspirone increased beta power (25–35 Hz, Figure 6E-G) and decreased gamma power (80–100 Hz, Figure 6F-H) in the DLS of LID + L-dopa + buspirone group. These findings indicate that buspirone modulates aberrant oscillatory activity in both M1 and DLS by enhancing beta activity and suppressing pathological gamma oscillations in LID rats.
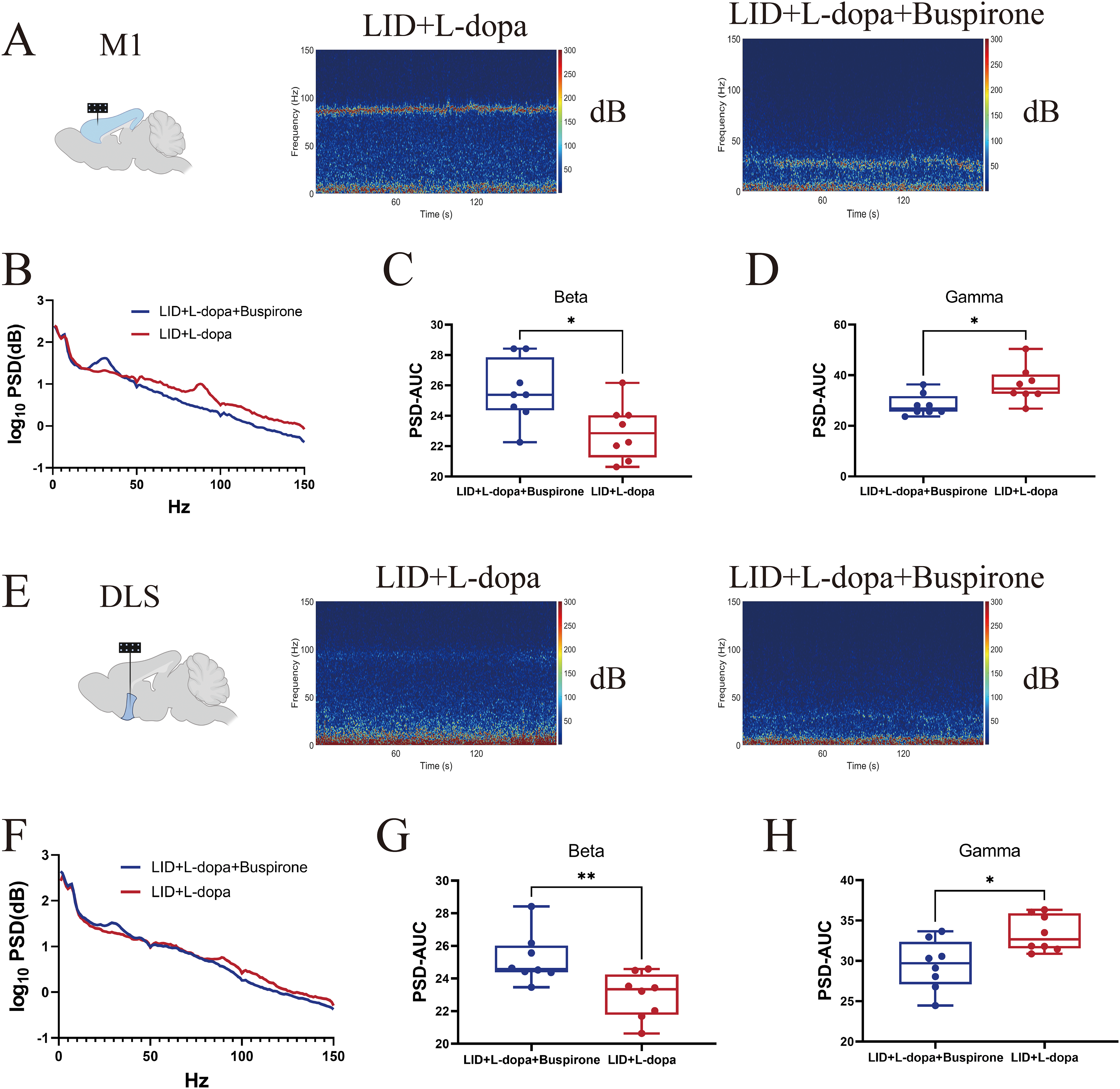
Buspirone administration decreased gamma oscillations in corticostriatal projections. (A) Representative spectrograms of the M1 among groups. (B) Mean cortical power spectral density plots. (C) Increased beta oscillations in M1 following buspirone administration (t = 2.72, p = 0.0167). (D) Decreased gamma oscillations (t = 2.77, p = 0.015) in M1 response to buspirone administration. (E) Representative spectrograms of the DLS. (F) Mean power spectral density plots of DLS. (G) Increased beta oscillations in DLS following buspirone administration (t = 3.07, p = 0.0084). (H) Decreased gamma oscillations (t = 2.92, p = 0.0112) in DLS following buspirone administration. n = 8 per group for LID + L-DOPA and LID + L-DOPA + buspirone conditions. *p < 0.05, **p < 0.01. Unpaired Student's t test with two-tailed for Figure 6C, D, G, and H.
Effect of Buspirone on the aperiodic components in rats with LID
Aperiodic components analysis revealed that aperiodic components at 10–50 Hz increased in the M1 following buspirone administration in the LID + L-dopa + buspirone group (exponent, Figure 7A; offset, Figure 7B). The augmented aperiodic components at 50–150 Hz were significantly reduced by buspirone (exponent, Figure 7C; offset, Figure 7D). Additionally, aperiodic components at 10–50 Hz were enhanced in the DLS following buspirone administration in the LID + L-dopa + buspirone group (exponent, Figure 7E; offset, Figure 7F). The elevated aperiodic components at 50–150 Hz observed in the dyskinetic state were also decreased in the DLS after buspirone treatment (exponent, Figure 7G; offset, Figure 7H).
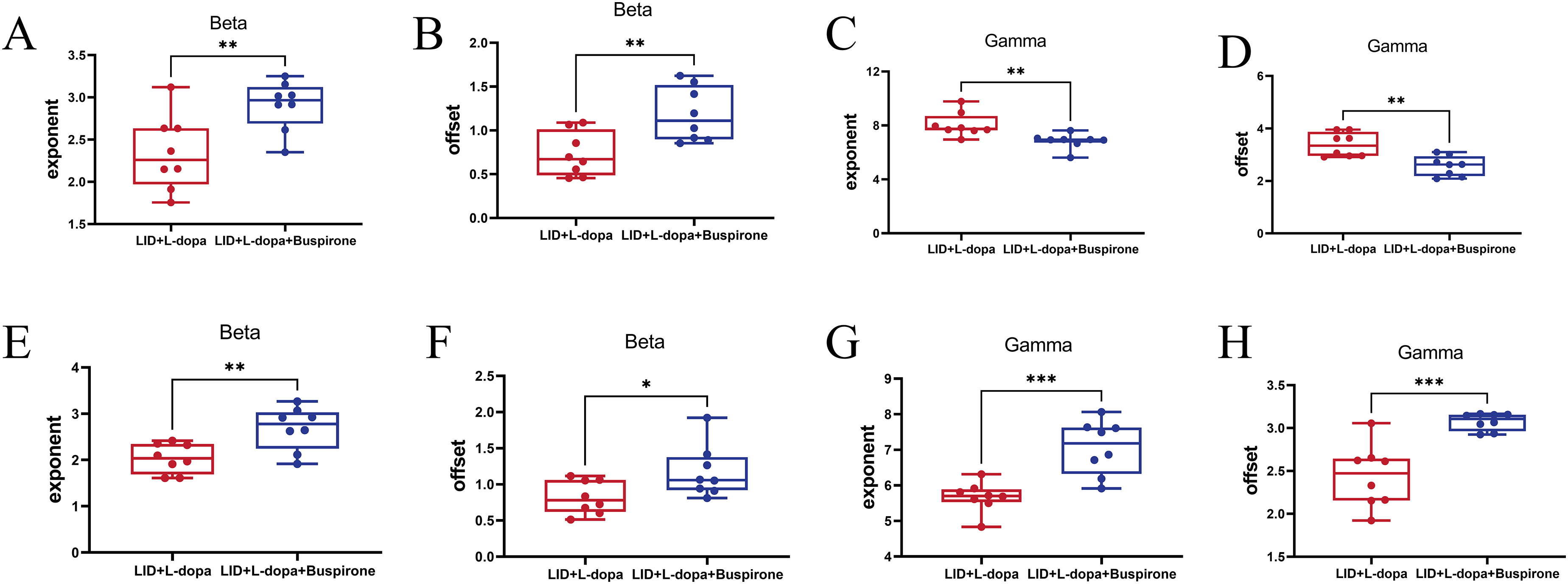
Increased aperiodic components at 10–50 Hz and decreased aperiodic components at 50–150 Hz following buspirone administration. (A) The LID + L-DOPA + buspirone group showed increased cortical aperiodic exponent at 10–50 Hz compared to the LID + L-DOPA group (t = 3.01, p = 0.0095). (B) Cortical aperiodic offset at 10–50 Hz was elevated in the LID + L-DOPA + buspirone group (t = 3.23, p = 0.0061). (C) The LID + L-DOPA group exhibited higher cortical aperiodic exponent at 50–150 Hz (t = 3.27, p = 0.0056). (D) Cortical aperiodic offset at 50–150 Hz was exaggerated in the LID + L-DOPA group (t = 3.86, p = 0.0017). (E) In the DLS, the aperiodic exponent at 10–50 Hz was higher in the LID + L-DOPA + buspirone group (t = 3.24, p = 0.0059). (F) The DLS aperiodic offset at 10–50 Hz was elevated in the LID + L-DOPA + buspirone group (t = 3.32, p = 0.036). (J) At 50–150 Hz, the LID + L-DOPA group showed increased aperiodic exponent in the DLS (t = 4.52, p = 0.0005). (K) Aperiodic offset at 50–150 Hz in the DLS was elevated in the LID + L-DOPA group (t = 4.76, p = 0.003). n = 8 per group for LID + L-DOPA and LID + L-DOPA + buspirone conditions. *p < 0.05, **p < 0.01, ***p < 0.001. Unpaired Student's t test with two-tailed for Figure 7A-H.
Buspirone normalized gamma-band functional connectivity between M1 and DLS
Coherence analysis revealed that beta-band coherence (25–35 Hz) increased following buspirone administration in the LID + L-dopa + buspirone group (Figure 8A-C). The augmented gamma-band coherence (80–100 Hz) was significantly reduced by buspirone (Figure 8A, B, and D). Additionally, the beta-band PLV (25–35 Hz) was enhanced in the LID + L-dopa + buspirone group following buspirone administration (Figure 8E and F). The elevated gamma-band PLV (80–110 Hz) between the M1 and DLS in the dyskinetic state was also decreased (Figure 8E and G).
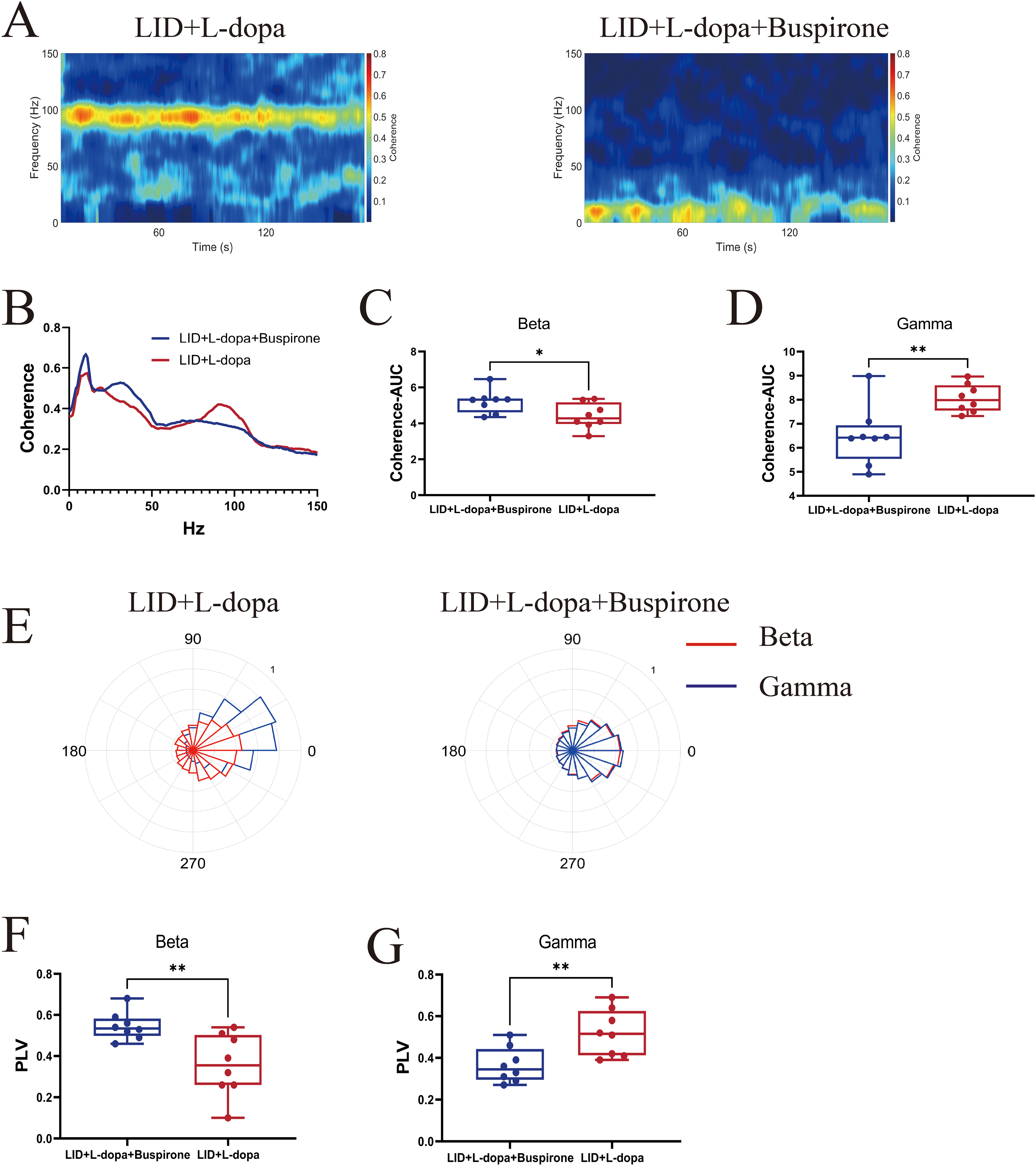
Buspirone administration suppressed function connectivity between M1 and DLS. (A) Representative coherence spectrograms among groups. (B) Linear graph of M1-DLS coherence among groups. (C) Increased beta-band coherence in LID + L-dopa + buspirone group (t = 2.36, p = 0.0336). (D) Decreased gamma-band coherence in LID + L-dopa + buspirone group (t = 3.26, p = 0.0057). (E) Representative polar histograms of phase locking between M1 and DLS; red lines indicate beta oscillations, blue lines indicate gamma oscillations. (F) Increased beta-band PLV in LID + L-dopa + buspirone group (t = 2.23, p = 0.006). (G) Elevated gamma-band PLV in LID + L-dopa group (t = 3.15, p = 0.0071). n = 8 per group for LID + L-DOPA and LID + L-DOPA + buspirone groups. *p < 0.05, **p < 0.01. Unpaired Student's t test with two-tailed for Figure 8C, D, F, and G.
Discussion
In this study, excessive gamma oscillations and increased aperiodic components at 50–150 Hz were observed during on-state dyskinesia. Functional connectivity analysis showed enhanced gamma-band coherence and PLV between M1 and DLS during on-state dyskinesia. Buspirone administration alleviated dyskinesia and suppressed exaggerated gamma oscillations, increasing beta power in M1 and DLS regions in LID rats. Buspirone also normalized gamma-band functional connectivity between M1 and DLS, increasing beta-band coherence and decreasing gamma-band coherence and PLV.
Exaggerated gamma-band activity and aperiodic components at 50–150 Hz during on-state dyskinesia
The M1 is a crucial component of the motor system, receiving feedback from the basal ganglia circuits to control voluntary body movements . Additionally, M1 is a common target for non-invasive neuromodulation techniques in clinical practice, such as transcranial direct current stimulation and transcranial magnetic stimulation.28,29 The DLS, a key component of the basal ganglia, plays a critical role in motor control. 30 Therefore, this experiment investigates the effects of Buspirone on LID by exploring the electrophysiological changes in cortico-striatal projections.
Consistent with previous research, chronic L-dopa administration led to a suppression of increased beta-band mixed power in the parkinsonian state, 31 while inducing exaggerated gamma-band power. 32 Additionally, exaggerated aperiodic components at 10–50 Hz frequency range have been identified as a sensitive biomarker for early diagnosis of PD. 33 Our study demonstrated that exaggerated aperiodic components at 10–50 Hz frequency range could also be suppressed by L-dopa administration. To avoid interference from the theta frequency band (4–8 Hz), which has been reported to be involved in LID, we selected this frequency range for our analysis.
Furthermore, our study revealed a novel finding: chronic L-dopa administration induced an increase in the aperiodic components at 50–150 Hz frequency range. Several lines of evidence suggest that the aperiodic components carry important physiological information, such as the excitation-inhibition balance, overall firing rate, and the sum of the pre- and postsynaptic currents. 34 Therefore, we speculated that the observed aperiodic changes in our study reflect important physiological information related to the effects of chronic L-dopa administration on the neural activity in the M1 and DLS of LID rats.
Overall, our study provides new insights into the potential use of aperiodic components as biomarkers for PD and LID, and highlights the importance of further investigation into the physiological significance of these components in the context of these neurological disorders.
Increased gamma-band functional connectivity between M1 and DLS
The results indicated enhanced gamma-band coherence and PLV in response to L-dopa administration, signifying abnormal neural connectivity in LID rats. Increased cortico-STN coherence has been detected in PD patients on and off L-dopa.35,36 Previous studies suggested strengthened cerebellar dentate nucleus and putamen connectivity reduces LID. 37 Additionally, structural and functional connectivity independently predict motor improvement in PD. 38 Our findings regarding altered functional connectivity patterns in LID) may provide neuroelectrophysiological insights into the pathophysiological mechanisms underlying this movement disorder.
Buspirone alleviates LID by regulating excessive gamma oscillations
In this study, we demonstrated that buspirone effectively attenuated dyskinesia, as evidenced by significantly reduced AIMs, while preserving the therapeutic motor benefits of L-dopa. Importantly, buspirone administration did not impair L-dopa's ability to improve parkinsonian motor deficits, as locomotor performance—measured in the open field test—remained intact or was even enhanced following co-administration. These findings suggest that buspirone can selectively suppress dyskinetic activity without compromising the antiparkinsonian efficacy of L-dopa. However, no study has examined how buspirone may affect the altered output basal ganglia activity in PD and LID. Here, we first show that the excessive gamma oscillations of M1 and DLS during on-state dyskinesia were reduced when buspirone was administered. Subsequently, the strengthened aperiodic components at 50–150 Hz were decreased in cortico-striatal projections. The gamma-band functional connectivity between M1 and DLS was suppressed by buspirone administration.
Medications aimed at the 5-HT1A and/or 5-HT1B receptors have been developed with the intent of relieving LID. 39 Selective 5-HT1A agonists have been shown to suppress the sensitization of cortico-basal ganglia-thalamic circuits and modulate striatal glutamate transmission without affecting striatal ectopic dopamine release, which may explain their anti-dyskinetic effects. 40 In addition, buspirone may improve LID symptoms by restoring the burst activity of the substantia nigra pars reticulata induced by dopamine loss, making it a promising target for testing new drugs for the treatment of PD and LID. 41 Previous study has shown that both gamma power and gamma bursts in the M1 correlate with motor behavior. 42 Hence, from an in vivo electrophysiology perspective, we infer that buspirone modulates exaggerated gamma activity, accompanied by the restoration of aberrant neural functional connectivity and flow in the M1-DLS projection, to alleviate LID symptoms. We elucidated, for the first time, the potential neural electrophysiological mechanism underlying buspirone's efficacy in dyskinetic states and provided supporting evidence for its clinical application.
This study is the first comprehensive investigation into the electrophysiological characteristics of experimental antidyskinetic treatment with buspirone. Through recording state changes induced in cortico-striatal projections in dyskinetic rats, we characterized these compounds electrophysiologically and identified their potential therapeutic relevance. Our findings suggest that the common antidyskinetic effects of the evaluated drugs may primarily be due to the reversal of a brain state characterized by excessive gamma oscillations. Moreover, differential effects on excessive gamma oscillations may suggest variations in pharmacological profiles that could be pertinent for motor symptoms. However, this study is limited to the investigation of neurophysiological mechanisms, and the molecular basis of buspirone's effects remains to be elucidated. Further research into its molecular mechanisms would help clarify the clinical relevance of these electrophysiological changes.
Supplemental Material
sj-docx-1-pkn-10.1177_1877718X251380196 - Supplemental material for Buspirone regulates cortico-striatal gamma oscillations to ameliorate dyskinesia
Supplemental material, sj-docx-1-pkn-10.1177_1877718X251380196 for Buspirone regulates cortico-striatal gamma oscillations to ameliorate dyskinesia by Han Liu, Ruizuo Wang, Yuming Xu, Weina Dai and Pengfei Wang in Journal of Parkinson's Disease
Footnotes
Ethical considerations
Healthy male rats (280–300 g) were obtained from the Animal Experiment Center of Zhengzhou University, China. All procedures complied with the university's animal care guidelines and were approved by its Animal Ethics Committee.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by funds from the Youth Natural Science Foundation of Henan (no. SBGJ202103046)
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The data supporting the findings of this study are available from the corresponding author, Yuming Xu, upon reasonable request.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
