Abstract
Background:
The gold-standard treatment for Parkinson’s disease is L-DOPA, which in the long term often leads to levodopa-induced dyskinesia. Serotonergic neurons are partially responsible for this, by converting L-DOPA into dopamine leading to its uncontrolled release as a “false neurotransmitter”. The stimulation of 5-HT1A receptors can reduce involuntary movements but this mechanism is poorly understood.
Objective:
This study aimed to investigate the functionality of 5-HT1A receptors using positron emission tomography in hemiparkinsonian rats with or without dyskinesia induced by 3-weeks daily treatment with L-DOPA. Imaging sessions were performed “off” L-DOPA.
Methods:
Each rat underwent a positron emission tomography scan with [18F]F13640, a 5-HT1AR agonist which labels receptors in a high affinity state for agonists, or with [18F]MPPF, a 5-HT1AR antagonist which labels all the receptors.
Results:
There were decreases of [18F]MPPF binding in hemiparkinsonian rats in cortical areas. In dyskinetic animals, changes were slighter but also found in other regions. In hemiparkinsonian rats, [18F]F13640 uptake was decreased bilaterally in the globus pallidus and thalamus. On the non-lesioned side, binding was increased in the insula, the hippocampus and the amygdala. In dyskinetic animals, [18F]F13640 binding was strongly increased in cortical and limbic areas, especially in the non-lesioned side.
Conclusion:
These data suggest that agonist and antagonist 5-HT1A receptor-binding sites are differently modified in Parkinson’s disease and levodopa-induced dyskinesia. In particular, these observations suggest a substantial involvement of the functional state of 5-HT1AR in levodopa-induced dyskinesia and emphasize the need to characterize this state using agonist radiotracers in physiological and pathological conditions.
INTRODUCTION
Parkinson’s disease is a neurodegenerative disorder characterized by a wide variety of motor and non-motor symptoms. This disease affects approximately 1% of individuals older than 60 years and represents a major public health problem in developed countries [1]. At the onset of the disease, the gold-standard treatment for motor symptoms remains L-3,4-dihydroxyphenylalanine (L-DOPA) [2]. This ‘dopamine replacement’ therapy is the most effective and the best tolerated in the short term [3–5]. However, long-term use of L-DOPA commonly leads to the development of debilitating dyskinesia known as levodopa-induced dyskinesia (LID) [6].
The pathological hallmark of Parkinson’s disease is the loss of dopaminergic neurons in the substantia nigra pars compacta (SNc) and subsequent loss of dopamine release in the projection areas, but it is known that other monoaminergic systems are involved in the motor and non-motor symptoms. Indeed, the serotonergic system plays an important role in the pathophysiology of the disease [7–9] and the appearance of LID.
Although the exact mechanisms are not completely elucidated, LID is associated with the conversion of L-DOPA into dopamine by serotonergic neurons and its aberrant striatal release as a ‘false neurotransmitter’ by lack of autoregulatory mechanisms [10, 11]. Importantly, several studies have shown that activation of 5-HT1A receptors decreases LID [12–16], which may be attributed to the inhibition of the 5-HT system via the stimulation of 5-HT1A autoreceptors (thereby reducing the release of dopamine by 5-HT terminals in the striatum). It should be noted that a direct activation of post-synaptic 5-HT1A receptors in the striatum is also able to decrease LID [17, 18]. 5-HT1A receptor agonists have therefore been proposed as promising candidates for the treatment of LID, and may also be useful to treat motor or non-motor symptoms of Parkinson’s disease [19].
In this context, a better understanding of the pathophysiological role of this receptor in Parkinson’s disease and LID seems critical to facilitate development of future treatments. To this end, several PET and autoradiography studies have been conducted using 5-HT1A receptor (5-HT1A R) radioligands [20]. However, results have been inconsistent: some studies suggested an upregulation [21–23], whereas others suggested a loss of 5-HT1AR [24–26]. In addition, 5-HT1AR PET studies used antagonist radiotracers, such as [11C]WAY-100635 or [18F]MPPF, and not agonist radiotracers. This is important because G protein-coupled receptors (GPCRs) such as 5-HT1AR can exist in a high or a low affinity states for agonists, depending on whether they are coupled or not to their G proteins [27]. Whereas 5-HT1A receptor antagonists bind with equal affinity to coupled and uncoupled 5-HT1AR, agonists bind preferentially to the coupled receptors [28] in other words, to the active state of the receptor [29]. Thus, previous studies using antagonist radiotracers did not evaluate the functional state of 5-HT1AR in Parkinson’s disease. Given that GPCR coupling is potentially altered in pathological conditions [30–32] it seems critical to investigate the active state of 5-HT1AR in order to better understand their role in Parkinson’s disease and LID [33].
We recently developed and validated in animal the first fluorinated agonist radiotracer of 5-HT1A receptors, [18F]F13640 [34], and we have transferred it to humans as a new PET radiopharmaceutical [35]. This radiotracer is a full 5-HT1AR agonist and displays a high affinity and selectivity for 5-HT1A receptors [36, 37]
Here, the aim was to investigate changes of 5-HT1AR coupling in the 6-OHDA-lesioned rat model of Parkinson’s disease using both a 5-HT1A antagonist radiotracer, [18F]MPPF, and the agonist radiotracer [18]F13640. We also studied the functional state of 5-HT1AR in LID, using HPK rats that were chronically treated with L-DOPA.
METHODS
Animals
Twenty-eight male Sprague-Dawley adult rats (Charles River laboratories, France) of 364.92±60.71 g were used. Animals were housed in standard temperature and humidity conditions with a 12 h/12 h light/dark cycle. Food and water were provided
Unilateral 6-OHDA lesions in the medial forebrain bundle
Rats were anesthetized using isoflurane. Prior to surgery, buprenorphine (0.05 mg/kg, s.c.) was administered for peri- and post-surgical analgesia. Lidocain was used for local analgesia.
Rats were lesioned in the right medial forebrain bundle (MFB) according to the Paxinos and Watson’s atlas [38] at coordinates AP = –4.0 mm,
After 3 weeks, the rats were challenged with apomorphine (0.05 mg/kg, s.c.; Sigma-Aldrich) and turning behavior assessed over 30 min. The rats making > 10 net full turns contralateral to the lesion were used in further experiments. This value was chosen in order to ensure a striatal dopamine depletion of at least 90% with no risk of false positives, as virtually no rotations are observed following apomorphine administration in the absence of such a lesion [39].
Development of L-DOPA-induced motor behaviors
For the study of LID, 6-OHDA lesioned rats received chronic daily treatment with L-DOPA (6 mg/kg) and benserazide (12 mg/kg) for 3 weeks in order to induce stable reproducible abnormal involuntary movements. After this induction phase, the rats were maintained with 2 injections of L-DOPA per week for the duration of the experiments (L-DOPA was not administered the days of imaging sessions, so that there was at least a 24-h interval between an injection and the PET scan). Induction of LID was assessed by observing for 1 min each rat at t: 20, 40, 60, 80, 100 120, 140, 160, and 180 min post L-DOPA administration, at day 1 (D1), day 7 (D7), day 14 (D14), and day 22 (D22) of treatment, using the following scale: Absence of dyskinesia (normal) Orolingual dyskinesia Orolingual+moderate limb and axial dyskinesia Orolingual+pronounced limb and axial dyskinesia.
PET/CT imaging
Animals were divided in three groups: 6-OHDA-lesioned rats (HPK;
Concerning the voxel-based analysis, data processing was carried out using the statistical parametric mapping software (SPM12, Wellcome Trust Center for Neuroimaging, London; UK). Parametric images of Binding Potential Non-displaceable (BPND) were generated for all rats using the Logan graphical method with reference region (cerebellum) calculated from 20 to 60 min. Individual PET images were realigned and spatially normalized on a custom PET template for each radiotracer which was coregistered on an anatomical MRI template. The quality of the registration was visually checked between all images to verify that they fitted well together and matched the MRI template in terms of cortical boundaries. Each PET volume was smoothed using an isotropic Gaussian filter [1.4×1.4×1.4 mm].
Statistical analyses
For the comparisons of DVR in the regions of interest, a two-way ANOVA with Tukey’s multiple comparisons test was performed (
The comparisons of parametric images between the different groups of animals were performed voxel by voxel using a
RESULTS
Development of L-DOPA-induced motor behaviors
Six rats chronically treated with L-DOPA were scored as dyskinetic during the 60 min post L-DOPA administration (peak effect): 2.3±0.2, 2.7±0.2, 2.5±0.2 and 2.8±0.2 (average dyskinetic score±SEM, at D1, D7, D14 and D22, respectively). The other rats from the group were also confirmed as dyskinetic but not systemically quantified.
PET imaging of total 5-HT1A receptors using [18F]MPPF
[18F]MPPF binding was compared between hemiparkinsonian rats with or without dyskinesia and control rats (Fig. 1). At the voxel level, [18F]MPPF BPND was significantly lower in hemiparkinsonian rats versus controls in various cortical areas, with few differences between the hemispheres (Fig. 1A). On the contrary, a local significant increase of [18F]MPPF BPND was found in the contralateral orbital cortex. In the ROIs (Fig. 1D), the most significant decreases of distribution volumes were found in the right and left hippocampus (–11%,
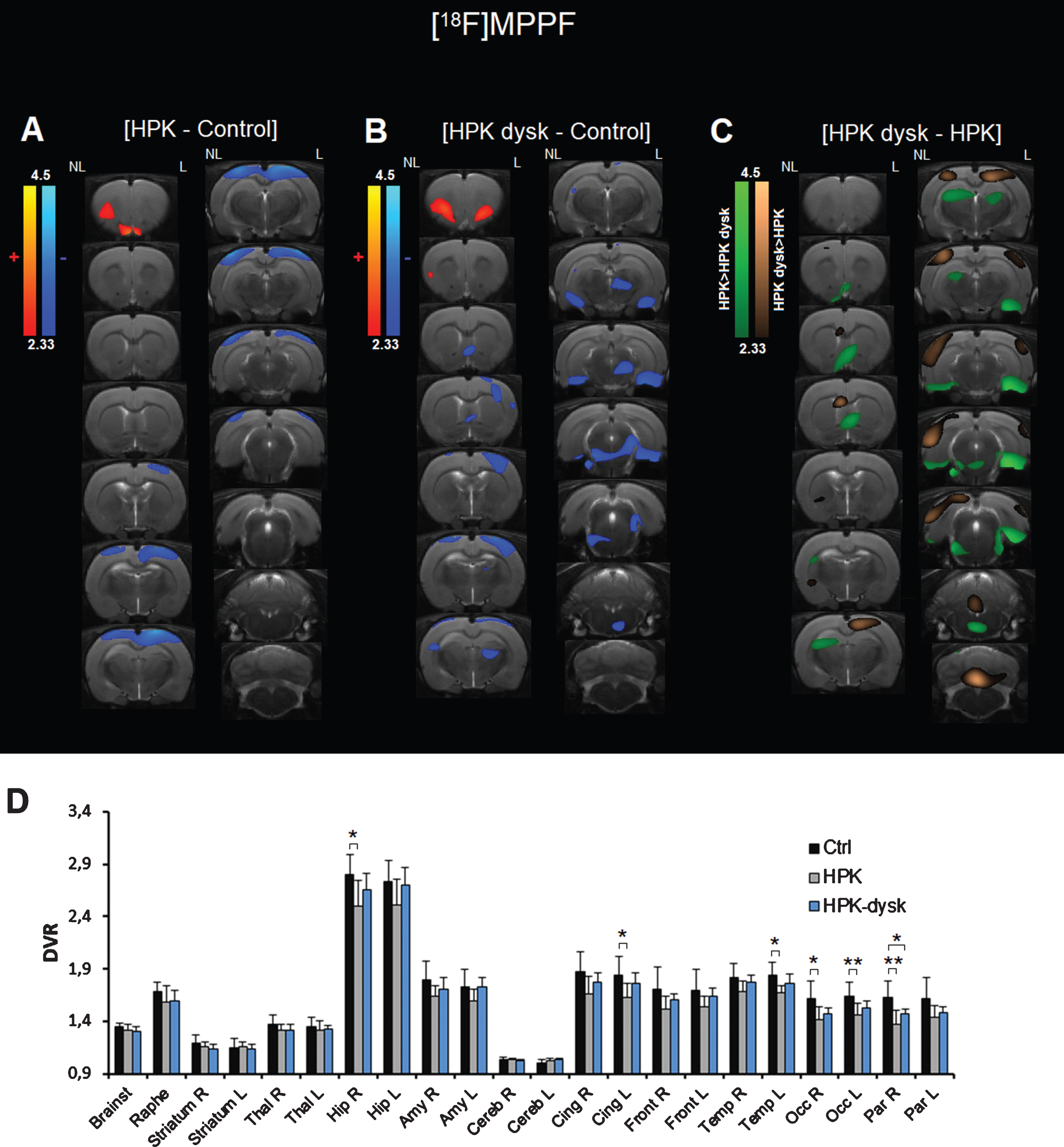
Effect of 6-OH-DA lesion and LID induction on [18F]MPPF PET imaging in rats. A) Voxel-to-voxel statistical comparisons of [18F]MPPF BPND between control rats and hemiparkinsonian rats. Z scores in color scales (significant increases in red, significant decreases in blue;
In the dyskinetic HPK rats, [18F]MPPF uptake was also modified in several areas (Fig. 1B). Fewer changes were found in the cortex, and were mainly located in the right somatosensory and motor cortices. Other decreases were found mainly in the limbic system: medial septum, lateral entorhinal cortex, and ventral hippocampus. [18F]MPPF uptake was also decreased in the right thalamus and the brainstem. Significant increases were found in the orbital cortex, similarly to HPK rats, but bilaterally. In the ROIs, significant decrease of DVR were found only in the ipsilateral side of the hippocampus, the parietal cortex and the occipital cortex and ranged from –5 to –9%.
We also compared [18F]MPPF binding between HPK and HPK-LID rats (Fig. 1C). [18F]MPPF BPND was significantly higher in non-dyskinetic animals in some areas of the limbic system (the medial septum and the entorhinal cortex), the raphe (median and magnus) and the dorsal thalamus (in green). On the contrary, [18F]MPPF BPND was higher in dyskinetic animals in parts of the parietal and occipital cortex, and the medial deepest part of the cerebellum (in brown). Results from the ROIs analysis also showed that [18F]MPPF uptake was higher in both sides of the hippocampus in the dyskinetic animals (Fig. 1D).
PET imaging of 5-HT1A receptors in high-affinity state using [18F]F13640
Binding potentials of [18F]F13640, reflecting changes in 5-HT1A receptors in the high-affinity state for agonists, were compared between hemiparkinsonian rats with or without dyskinesia and control rats (Fig. 2).
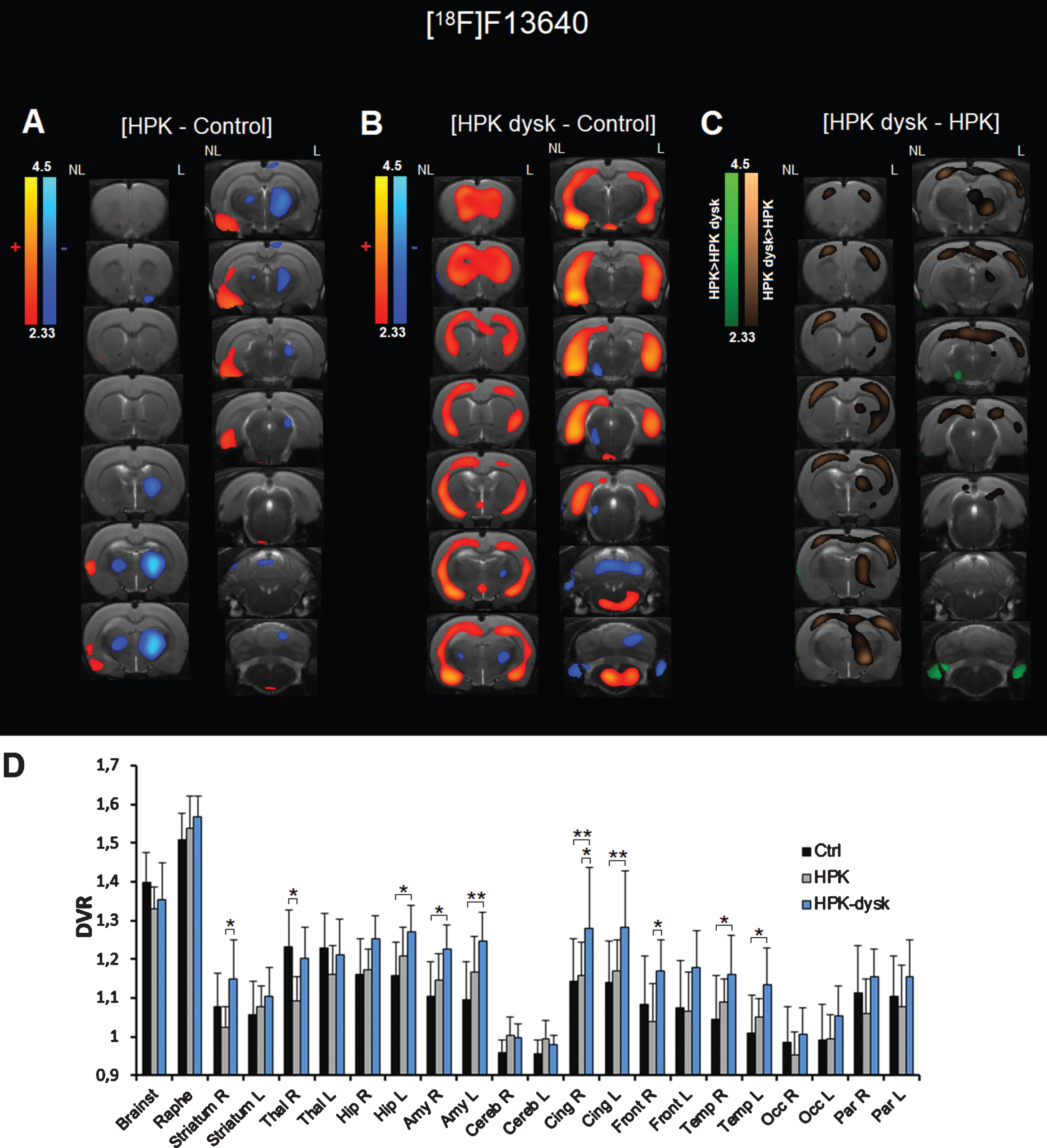
Effect of 6-OH-DA lesion and LID induction on [18F]F13640 PET imaging in rats. A) Voxel-to-voxel statistical comparisons of [18F]F13640 BPND between control rats and hemiparkinsonian rats. Z scores in color scales (significant increases in red, significant decreases in blue;
When comparing HPK and control rats (Fig. 2A), the voxel-based analysis revealed a significant decrease of [18F]F13640 binding potential in a large cluster of the ipsilateral hemisphere, spreading from the globus pallidus to the thalamus. A similar but smaller cluster was found on the contralateral side. HPK rats also displayed a higher [18F]F13640 binding in several limbic areas of the contralateral hemisphere, spreading from the insular cortex to the piriform cortex, the entorhinal cortex and parts of the hippocampus. When comparing the distribution volume ratios in the different regions of interest (Fig. 2D), we only found a significant decrease in the right thalamus in HPK rats versus control rats (–11.5%,
Significant decreases of [18F]F13640 binding in HPK-LID rats were fewer than HPK rats (Fig. 2B); small clusters were found rostrally in the ventral thalamus, and caudally in the left substantia nigra and parts of the cerebellum. On the contrary, many areas displayed a significant increase of binding potential. Similarly to HPK rats, the most significant increases were located in areas of the limbic system, such as the piriform and entorhinal cortices, the amygdala and the ventral hippocampus. Other cortical regions were also affected, such as the prefrontal cortex and widespread areas of the temporal and parietal cortices. Other small increases were found in the hypothalamus and the brainstem. In general, increases were observed in both sides of the brain but were more significant in the contralateral hemisphere. Such increases were also significant when comparing the DVR between dyskinetic and control animals in several regions of interest (Fig. 2D), namely the left hippocampus (+9.9%,
We also compared voxel by voxel [18F]F13640 binding between HPK and HPK-LID rats (Fig. 2C). [18F]F13640 binding was significantly higher in dyskinetic animals (in brown) in various cortical areas in both hemispheres and the right striatum and thalamus. On the contrary, [18F]F13640 binding was higher in non-dyskinetic animals (in green) in the left substantia nigra and the paraflocculus of the cerebellum. In the regions of interest (Fig. 2D), DVR were higher in dyskinetic compared to non-dyskinetic rats in the right striatum (+12.2%,
DISCUSSION
5-HT1AR as a promising therapeutic target for Parkinson’s disease
In addition to the dopaminergic system, it has been compellingly demonstrated that the serotonergic system is involved in the pathophysiology of Parkinson’s disease, whether in terms of motor symptoms [24, 42] or non-motor symptoms such as cognitive impairment [43] and mood disorders [44], but also in the etiology of LID [45, 46]. In this context, the 5-HT1AR appears to be a promising therapeutic target: in addition to having a potential therapeutic effect in treating motor [47–50] and cognitive [51] symptoms or mood disorders [52–55] associated with Parkinson’s disease, 5-HT1A agonists are highly effective for treating LID in preclinical models [13, 56–61]. The main proposed mechanism for their antidyskinetic effects is the stimulation of autoreceptors in the raphe nuclei. However, striatal 5-HT1AR may also play a role in control of LID [17, 62]. Nevertheless, there are also discrepancies regarding the effect of 5-HT1A agonists on motor symptoms of Parkinson’s disease [63–65]. Few clinical studies have been conducted with 5-HT1A agonists to treat LID, and they reported either promising results [66–68] or worsening of parkinsonian features in a high proportion of patients [69].
Therefore, more information is needed to understand the mechanism of action of 5-HT1A agonists in treating LID and how to diminish dyskinesia without worsening motor symptoms in patients. Indeed, little is currently known about the impact of Parkinson’s disease and LID on 5-HT1AR levels and function.
The contribution of PET imaging in deciphering the involvement of 5-HT1AR in Parkinson’s disease
We recently reported the radiosynthesis, preclinical validation [34] and first-in-man administration [35] of the first highly selective 5-HT1AR full agonist PET radiotracer [18F]F13640. Unlike antagonist radiotracers of GPCRs, agonists radiotracers are known to bind preferentially to receptors in a high-affinity state for agonists, that is, to receptors that are fully functional and accessible to the endogenous neurotransmitter [70–73]. We therefore investigated changes in 5-HT1AR in a rat model of Parkinson’s disease using [18F]F13640 in comparison with an antagonist radiotracer, [18F]MPPF. Furthermore, changes of 5-HT1AR levels and functionality were also studied in rats that were chronically treated with L-DOPA to elicit LID. Importantly, unlabeled F13640 (also known as befiradol or NLX-112) has shown promising antidyskinetic properties in rat, marmoset and macaque models of Parkinson’s disease [14, 59]. Therefore, the binding of [18F]F13640 points directly to the population of 5-HT1AR that are targeted by this antidyskinetic 5-HT1A receptor agonist and is likely to provide important information regarding the therapeutic mechanism of this class of drugs.
Previous studies using antagonist radiotracers have shown disparate results. In rats that were unilaterally injected with 6-OH-DA in the striatum, binding potential of [18F]Mefway was significantly lowered in the hippocampus [74, 75]. In cynomolgus monkeys treated with 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine (MPTP), [18F]MPPF binding was increased in various structures at early stages, whereas fewer changes were found in the fully symptomatic state [26]. An autoradiography study using [3H]WAY-100635 showed decreases of 5-HT1AR binding in the external layers and increases in the middle layers of the premotor and motor cortices of MPTP-lesioned macaques [21, 22]. 5-HT1AR levels were also investigated in human studies using 5-HT1A antagonist radiotracers. In 2003, Doder et al. reported a decrease of [11C]WAY-100635 binding potential in the raphe of parkinsonian patients, which was significantly correlated with the tremor score [24]. Another study using [18F]MPPF showed decreases of 5-HT1AR binding in patients, depending on the comorbidity with depression [26]. A postmortem autoradiography study reported an increased binding of [3H]WAY-100635 in the temporal cortex of patients [21]. These data show a complex involvement of 5-HT1AR in the pathophysiology of Parkinson’s disease. The variability of these findings may be partly due to different species and models, but also by factors such disease progression, the development of dyskinesia or the chronic effects of L-DOPA treatment by itself. Indeed, long-term treatment with L-DOPA produces marked changes in the 5-HT system [76], as shown by a previous study showing increased tritiated WAY-100635 binding in MPTP-lesioned macaques chronically treated with L-DOPA compared with untreated macaques [22].
Once again, it should be emphasized that all the above studies used antagonist radiotracers to label 5-HT1AR: such tracers cannot characterize underlying changes in receptor-G-protein coupling state. In contrast, the use of the innovative agonist radiotracer, [18F]F13640, allowed us for the first time to examine changes in active state 5-HT1AR in the present model of Parkinson’s disease.
PET imaging of high-affinity state 5-HT1A receptors allows revisiting the pathophysiology of LID
The main finding of this study is that the changes of the population of 5-HT1AR in the active (i.e., G-protein-coupled) state, as determined by [18F]F13640 binding, were completely distinct from the changes in the whole population of receptors, as visualized using [18F]MPPF.
Using [18F]MPPF, 5-HT1AR binding appeared to be decreased in cortical areas only in HPK animals. In dyskinetic animals, the lower cortical binding was more restricted to the somatosensory, motor and entorhinal areas, but [18F]MPPF uptake was also lowered in subcortical areas such as the thalamus and the raphe. For both pathological groups, few increases were found and restricted to the orbital cortex.
Such changes occurring with a 5-HT1AR antagonist radiotracer are unlikely to be related to the functional state of receptors as they are known to bind equally to the coupled or uncoupled receptors [28], or to the competition with endogenous serotonin because [18F]MPPF is poorly sensitive to supraphysiological changes in serotonin concentration [34]. Therefore, cortical decreases of [18F]MPPF binding potential could be related to an altered affinity for receptors or to a lower density of 5-HT1A receptors, which could be related to various non-motors symptoms of Parkinson’s disease such as cognitive impairment, depression, and anxiety [43, 44], as well as to motor and sensory deficits, since the somatosensory and motor cortices also displayed a decreased uptake. Another worthy observation is that in the raphe, [18F]MPPF binding was decreased in dyskinetic rats, which would suggest a decreased serotonergic autoinhibition that could be related to the dyskinetic behavior (as conversely, the stimulation of 5-HT1A autoreceptors is able to block LID). Interestingly, the local increase in the ventral orbital cortex is consistent with a previous autoradiography study using a 5-HT1A antagonist radiotracer that showed an increased binding in the most internal parts of the frontal cortex in parkinsonian primates that were also chronically treated with L-DOPA [21], this was suggested to be linked to visual hallucinations symptoms.
Numerous changes of [18F]F13640 uptake were also found in HPK and dyskinetic rats. These changes are more difficult to interpret but also more reflective of the pathophysiological involvement of 5-HT1AR, as contrary to antagonists, agonists bind preferentially to the active state, i.e., G-protein coupled state of receptors [28, 77]. This means that an increased proportion of receptors in the uncoupled state could lead to a decrease in [18F]F13640 binding. Another important consideration is that [18F]F13640 was shown to be very sensitive to competition with endogenous serotonin [34], therefore a local increase in serotonin concentration could also lead to a decrease in [18F]F13640 binding. As seen in the voxel analysis, [18F]F13640 binding modifications were spatially distinct from those of [18F]MPPF, therefore we propose that they were mainly due to one or both of these mechanisms (intracellular coupling or extracellular serotonin concentration) rather than changes in the density of receptors (which should also be observed using [18F]MPPF).
In HPK rats, [18F]F13640 uptake was decreased in the globus pallidus and the thalamus, especially in the lesioned side. There might be an increased serotonergic innervation in these areas, which would be consistent with a recent study in MPTP-lesioned cynomolgus monkeys showing a 2-fold increase of serotonergic axon varicosities in the globus pallidus [78]. Alternatively, 5-HT1AR coupling state might be altered in this region, which is supported by very recent findings in 6-OHDA lesioned rats showing that 5-HT1A agonists effects are decreased in the entopeduncular nucleus (which is analogous to the internal part of the globus pallidus) compared to control rats [79]. In the contralateral side, [18F]F13640 uptake was increased in limbic areas or limbic-related areas such as the insula, the entorhinal cortex, and the amygdala and the ventral hippocampus. Such increases could be due to the loss of serotonergic neurons in Parkinson’s disease [80] as they were observed in areas richly innervated by 5-HT, although it is unclear why they were more pronounced in the non-lesioned side. This deserves more investigation to understand if these increases are the signature of a compensatory mechanism and how they are involved in non-motor symptoms of Parkinson’s disease; given that they occur in limbic regions, they may be linked to the cognitive and mood symptoms.
Interestingly, dyskinetic animals displayed more pronounced increases of [18F]F13640 binding in the same regions as non-dyskinetic HPK animals but that also spread to cortical areas, and to the ipsilateral side of the brain. These observations are consistent with the known involvement of the serotonergic system in LID and could be due to a possible decrease of 5-HT basal levels in many regions in dyskinetic animals. The decreased 5-HT concentration would acutely result in a decreased competition towards 5-HT1AR and higher binding of [18F]F13640, but it can also be expected that a chronic depletion of 5-HT release would result in an overexpression of 5-HT1AR in the high-affinity state as a compensatory mechanism due to the chronic lack of stimulation by 5-HT. Indeed, it has been shown in 6-OH-DA-lesioned rats that L-DOPA decreases extracellular concentration of serotonin in the prefrontal cortex and hippocampus after acute administration [81] and decreases basal extracellular 5-HT levels in the PFC, striatum, hippocampus and substantia nigra after chronic administration [82]. Deficits in 5-HT release can arise from several mechanisms [83]: first, after decarboxylation of L-DOPA in 5-HT neurons, dopamine competes with 5-HT for storage into vesicles and exocytotic release [10, 84]; secondly, L-DOPA treatment can induce oxidative stress that is known to induce 5-HT neuron loss
Another important result of our study is the increase of [18F]F13640 uptake in the lesioned striatum of dyskinetic animals compared to untreated animals. This could be explained by a lower serotonergic innervation of the lesioned striatum in dyskinetic animals, resulting in higher binding of the radiotracer, but this hypothesis is contradicted by a previous study which showed that L-DOPA chronic treatment in HPK rats results in a higher SERT binding density in the lesioned striatum [89]. Therefore, the present observations more likely reflect a switching of a striatal population of 5-HT1AR to a high-affinity state, suggesting a hypersensitivity of 5-HT1AR in the lesioned side of the striatum.
As previously discussed, changes occurring at the level of presynaptic serotonergic transmission could explain some of our results with [18F]F13640. In the future, it would be valuable to evaluate the changes of [18F]F13640 along with the extracellular 5-HT concentration and the level of serotonin transporter. This is also important as the injection of 6-OHDA could induce serotonergic lesions, although we observed marked changes in the contralateral side of the brain, which would not be directly impacted by the toxin.
Finally, our results demonstrate that the coupling state of 5-HT1AR can be strongly disrupted in Parkinson’s disease and LID, supporting the use of agonist radiotracers in future clinical PET studies to revisit the pathophysiological involvement of these receptors. Such studies would contribute to a better understanding of the role of 5-HT1AR in the psychiatric and motor symptoms in Parkinson’s disease. Focusing on the active state of 5-HT1AR may also constitute a biomarker for monitoring disease progression, or for measuring occupancy changes after administration of clinical doses of 5-HT1A agonists (5-HT1AR antagonist radiotracers being poorly sensitive to competition with agonists—see [90–94]). Overall, the use of agonist PET tracers opens multiple avenues of investigation for brain imaging of Parkinson’s disease and other disorders involving serotonergic neurotransmission.
Footnotes
ACKNOWLEDGMENTS
We thank Ronan Depoortere for his contribution in the animal behavioural analyses, Jérôme Redouté and Nicolas Costes for their help in the data analysis and Véronique Gualda for the zootechnical assistance.
CONFLICT OF INTEREST
Dr. Newman-Tancredi is an employee and stockholder of Neurolixis. The other authors have no conflict of interest to report.
