Abstract
Exercise is an essential part of the treatment of Parkinson's disease. However, certain symptoms can make exercise difficult. In this viewpoint, we describe an under recognized symptom called running-induced dystonia (RID), which can prevent people with Parkinson's from exercising in their preferred way. We summarize the existing literature and use three case studies to outline potential mechanisms, aggravating and relieving factors and both pharmacological and non-pharmacological therapies to get rid of RID.
Plain Language Summary
Exercise is an important treatment for Parkinson's disease to help manage symptoms. Dystonia is a symptom of Parkinson's disease that can make exercise difficult. People with dystonia experience muscles spasms or contractions that lead to twisting or jerky movements of the body. In some people with Parkinson's disease, dystonia can occur in the foot when running, making this kind of exercise difficult. This paper summarizes what is known about running and foot dystonia. It describes three people with Parkinson's experiences with foot dystonia when running, and provides suggestions for management, including both medicines and rehabilitation strategies.
Introduction
Exercise is an essential treatment modality for people living with Parkinson's disease (PD). 1 We recently published a commentary paper entitled “Advice to People with Parkinson's in My Clinic: Exercise.” 2 This paper outlined the evidence for the benefit of exercise on motor and non-motor symptoms, the four components of the exercise prescription (aerobic, resistance, flexibility, and neuromotor), practical tips for implementation, and PD-specific considerations for exercise.
It is apparent that certain features of PD can make implementing the exercise prescription complicated. In our second paper we provided suggestions for addressing complications arising from autonomic dysfunction, 3 and our third paper outlined the importance of cardiopulmonary exercise testing for safety, precise exercise prescription, and determination of peak heart rate. 4 This fourth paper in the series focuses on an underrecognized phenomenon, called running-induced dystonia (RID), which is a disabling symptom that can stop people living with PD from exercising in their preferred way.
Running is a time and cost-efficient way to meet the aerobic component of PD exercise prescription. Running can also provide joy from the release of endorphins, seeing different places, and a feeling of freedom and flow, resulting in benefits to mood, sleep and confidence. Therefore, with the known benefits of exercise on motor 1 and non-motor symptoms, 5 early recognition and management of RID in PD is vital. In this commentary, we provide a background on what is known in the literature about RID, including its mechanisms and treatment. We enrich this with the experiences of three people living with PD and RID (MC, DG, ES), who describe their history, aggravating and relieving factors, medical management, and other tips and tricks they use to manage RID.
What is dystonia?
Dystonia is defined by expert consensus as: “a movement disorder characterized by sustained or intermittent abnormal movements, postures, or both. Dystonic movements and postures are typically patterned and repetitive and may be tremulous or jerky. They are often initiated or worsened by voluntary action and frequently associated with overflow movements.” 6
Dystonia is likely mediated by central processing abnormalities in areas such as the basal ganglia, cerebellum, supplementary motor areas and sensorimotor cortex, leading to an abnormal balance between motor activation and inhibition, abnormal plasticity and sensorimotor dysfunction. 7 Features can include task-specificity, relieving sensory tricks and a worsening with stress, anxiety or fatigue.8,9 Task-specificity is when the dystonia is triggered by a specific activity such as focal hand dystonia during writing, but the person can easily button a shirt without any dystonia. “Sensory tricks”, also known as ‘geste antagoniste’ temporarily relieve the dystonia and include actions such as touching a specific spot on the affected body part, or another remote body part. 10 Psychiatric disorders are now recognized as part of some dystonia's endophenotype, and disruption of corticostriatal connectivity to underlie not just motor control, but also anxiety, sensory and sleep disturbances. 9
Primary dystonia is usually due to a genetic cause, or idiopathic, and is not discussed here. Secondary dystonia occurs due to another cause such as drugs or a neurodegenerative disease. In this paper we focus on secondary dystonia caused by PD and specifically on RID.
How common is dystonia in Parkinson's disease?
Dystonia is found in approximately 30% of people living with PD but is more prevalent in early-onset PD (EOPD), where it can occur in approximately 60% of people. 11 In PD, dystonia can be the presenting sign in up to 14% of cases,11,12 and the foot is most commonly involved. 13 Certain monogenic causes of PD are more likely to be associated with dystonia, and those with autosomal recessive forms more likely to have dystonia as a presenting feature [See Table 1].14-16
In PD, dystonia commonly occurs when dopaminergic stimulation is low – i.e., before medications have started to have an effect in the morning, or when medications are wearing off. However, dystonia may also be seen as a peak-dose effect of levodopa. 11
Running-induced Dystonia (RID) in PD
RID has been reported as an initial symptom of PD, 13 occurring in the lower leg and foot after prolonged running, progressing to occurring earlier in a session, but disappearing after a few minutes of complete rest. 17 Over time dystonia may emerge during walking (Supplementary Video 1 and Figure 1). RID can feel like a severe cramp, with flexion of the toes and plantar flexion and inversion of the foot, sometimes with accompanying first toe extension. There are few published reports of RID in people living with PD,12,17,18 and therefore little to guide management. Therefore, we summarize the published literature, our clinical experience and importantly enrich this with the lived experience of three people with PD and RID.
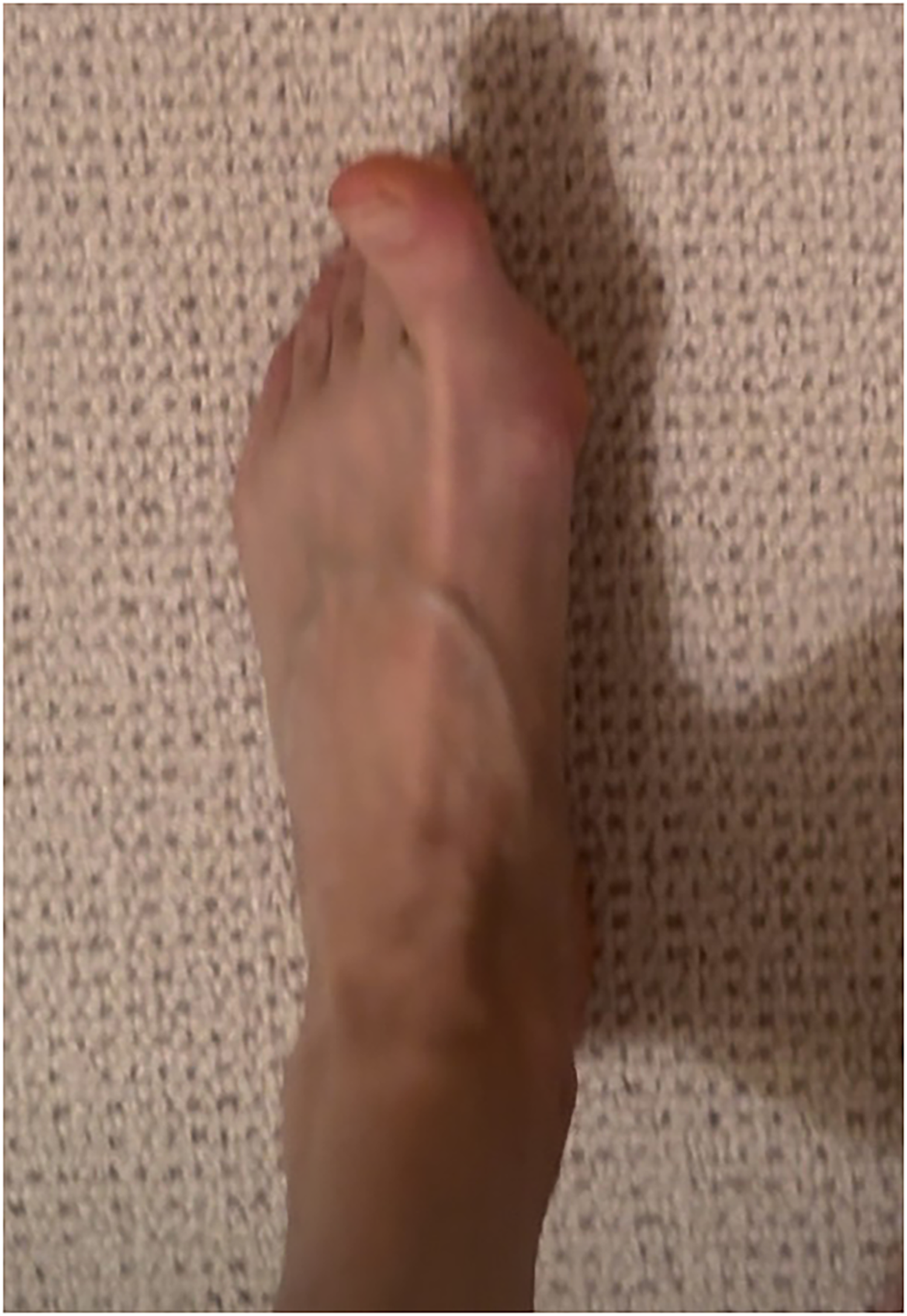
Dystonia of the left foot (Case 1). Supplementary Material: Video showing dystonia of the left foot walking with and without toe separators (Case 1).
Case 1 – female
I was 48-years old, when 2 h into a trail run on a downhill section I noticed tightness in my left calf and 2nd-5th toes. The symptoms resolved immediately on rest, and I was able to walk, but not run, the final 20 min home. I was healthy apart from a bilateral oophorectomy a few months prior. Over the next 6 months curling of the 2nd-5th toes progressed to inversion of my left foot accompanied by extension of the great toe (RID). At first this occurred mostly when running downhill but then it progressed to occurring on a flat terrain and after only 20 min of running. Rest and stretching my calf and toes alleviated the symptoms, but they would occur again within a few minutes of resumption of running. One year after first symptoms, I was only able to run for 5–10 min before debilitating symptom onset, and I stopped running altogether. Over the course of the following year, I developed rigidity and slowness in my left upper and lower limbs, reduced facial expression and left arm swing. The cramping and curling in the toes would come on even when walking, usually after approximately an hour.
I was diagnosed with PD 2 years after that first occasion of RID. Initiation of Madopar (benserazide-levopdopa) 25 mg/100 mg 4 times per day improved all symptoms, and I could recommence running, building up to 40 min. About 1.5 years after diagnosis, I developed early morning off-dystonia, with the same left foot posturing, mostly after a few minutes of walking and before my first morning medications. A Rotigotine (dopamine agonist) 4 mg transdermal patch was added, helping with the morning dystonia, and I was able to run for a duration of up to 1 h without RID.
Approximately 4.5 years after the initial RID symptom (2.5 years after diagnosis) I developed wearing-off symptoms while running, and occasionally walking, manifesting as dystonia of the left foot and left hand, as well as left sided stiffness and slowness. These wearing-off episodes were proceeded by ‘warning signs’ of ‘twitching’ in the toes, which provoked anxiety and hastened onset. I developed a fear of the dystonia, especially being stuck out on the trail unable to walk. I discovered that if the dystonia occurred, I was able to run backwards and sideways without dystonia, but not forward. Prevention of these episodes was treated by adding Opicapone (catechol-O-methyltransferase (COMT) inhibitor), sometimes taking an extra ½ tablet of Madopar ∼30 min into a longer run and carrying additional Madopar Rapid on runs as a back-up. An apomorphine injection was suggested by my neurologist, but I declined.
I found the following helpful in managing the dystonia: toe separators (Supplementary Material: Video 1), toe socks, running on softer surfaces and undulating terrain, a slower running speed and cadence, and practicing mindfulness. I noted the following worsened the dystonia: recovery socks where the spiky texture on the inside felt like a noxious stimulus, hard flat surfaces, running fast, anxiety regarding the dystonia, and poor sleep. Currently I can run for 1–1.5 h on undulating soft surface terrain. I run twice per week, play pickleball once per week and weight train at the gym twice per week – none of which provoke dystonia.
Case 2 - male
I was diagnosed with PD at 51-years-old, based on left-sided bradykinesia and rigidity. I did not take any medication initially, and I was otherwise healthy. I’d run prior to being diagnosed on the beach, without problems. At age 52, I followed evidence that suggests that 30 min of high-intensity treadmill training three times per week might slow down PD. 19 However, I noted that after 4–5 min of running, it became difficult to land properly on my left foot, with RID manifesting as left foot plantar flexion and inversion causing me to land on my toes. Stepping off the treadmill, resting and stretching the calf muscles for 30 s alleviated symptoms and then I could continue running. However, RID would occur again after another 4–5 min of running.
My neurologist first prescribed Sinemet (carbidopa-levodopa) 12.5 mg/50 mg taken 15–20 min prior to my morning run, which I tried for two weeks. Sinemet reduced the severity of dystonia, and it took longer to occur, but did not completely solve the problem. So, after this period, I was advised to try Madopar 25 mg/100 mg dissolving in water 15–20 min prior to my morning run, which solved the problem completely. Currently I run 6 km every second morning on the treadmill with only a quick stretch after 1 km and then I can complete the remaining 5 km without stopping or stretching. I also do resistance training on alternate days. I have never experienced left foot dystonia while rowing, walking, hiking, or weight training. About 4 years after been diagnosed I started Sinemet 12.5 mg/50 mg three times per day. Rarely I have experienced mild calf tightness on a long walk. My current medications are Sinemet 12.5 mg/50 mg three times per day, where the morning dose is replaced with Madopar 25 mg/100 mg on the days I run.
Case 3 - female
At 48 years of age, I experienced tremors in my left hand and was diagnosed with PD approximately 6 months later. I was otherwise healthy. My mother had PD and genetic testing revealed a monoallelic PRKN mutation. I had been running since age 34, typically between 45–60 min once per week, plus a variable long run in preparation for half-marathons or marathons. I continued to run after my diagnosis. At age 49, I began to experience a painless plantar flexion and inversion (with no toes involvement) of my left foot after 10–15 min of running. The abnormal posturing would occur and release repetitively. Initially, I was able to continue running, and the twisting motion would spontaneously self-resolve after pushing through for another 10–15 min. After a few months, however, the dystonic movement persisted for the duration of my run, causing instability. Even after a short rest, the dystonia would return in 1–2 min of running. Rasagiline 1 mg and amantadine 200 mg daily provided minor benefit. A neurologic physical therapist suggested taking large steps backwards or sideways to “break” the dystonia. These physical maneuvers resulted in abatement of symptoms, but the dystonia typically recurred within 2–15 min. This led to addition of carbidopa/levodopa 25 mg/100 mg taken at least 30 min before exercise, which resolved the dystonia. I don’t take levodopa unless I’m running, as my other symptoms don’t warrant it, and it also causes a transient, low-level nausea. On this regimen, I have been able to run 3–6 miles without severe dystonia, although my running gait is still not entirely normal, with a more “flat-footed” left mid-foot strike and decreased left leg drive. Additionally, I have not been able to increase weekly running mileages as I had done in the past when training for marathons.
In all the above cases dystonia was initially specific to running and relieved by increased dopaminergic stimulation. Rest, stretching, changing the motor pattern, the terrain or the sensory input led to temporary relief.
Pathophysiology of RID
The pathophysiology of RID in PD is not well understood, although there are several hypotheses as to why dystonia occurs first in running, rather than walking. One hypothesis is that dopamine release during prolonged exercise could lead to a state of relative dopamine deficiency due to low reserves, leading to reversible dystonia.12,18 This could explain why it is an initial symptom in some cases – akin to a cardiac exercise stress test exposing issues not present at rest. In one case study of a marathon runner 17 and the three cases presented here, individuals were able to resume running after a rest period of a few minutes to half an hour suggesting that the body may have replenished dopamine in this time. Others hypothesize that the increased release of dopamine during exercise could overstimulate hypersensitive or upregulated dopamine receptors. 18 However, this appears less likely in the above cases.
RID is seen in endurance runners without PD and may be caused by repetitive use, like the ‘task specific’ overuse dystonia seen in highly skilled actions of professional writers or violin players. 20 Case study 1 had a 30-year history of distance trail running and Case study 3 a 14-year history of half or full marathons. More developed motor pathways and larger cortical representation areas with a reduction in inhibition may have been responsible. 21 In the case studies presented, using a less well learned motor pattern such as running sideways or backwards helped temporarily. Alternatively, involvement mainly of the foot in RID could reflect altered somatotopic organization within the basal ganglia or patterns of dopamine depletion. 12
Case studies in the literature report that running over pebbles and massage temporarily relieved symptoms, 17 supporting abnormal sensorimotor integration as a cause of RID. In Case 1, toe-separators and running over varied terrain helped increase the latency before RID occurred. Running is typically faster than walking, and the vertical ground reaction forces are 2–3 times higher, requiring greater muscle activation to absorb shock. 22 Case 1 found faster speeds, downhill running and harder surfaces brought on the dystonia earlier, whereas slowing the cadence and softer surfaces allowed longer duration runs. This suggests that the greater stress on sensorimotor systems could be an additional factor.
Recommendations for evaluation
Our preliminary diagnostic criteria of RID in the context of PD are as follows:
A diagnosis of PD Dystonia that is task specific to running. Predominately affect the foot and toes with involuntary sustained posturing into plantarflexion inversion and flexion of the toes. The first toe may posture into extension. Relieved almost immediately by stopping running, although later may be associated with other similar tasks such as walking.
A thorough patient history and examination are needed to establish the clinical diagnosis of RID, and to guide management. The history queries body distribution, temporal pattern (including onset, course, time of day, frequency, variability and relationship to antiparkinsonian medications), along with specific activities, terrain and surfaces that trigger the symptoms, the presence of a sensory trick, and any other alleviating and aggravating factors. Using a diary to record motor and non-motor (i.e., anxiety and sleep) symptoms throughout the day, along with timing of medications can be helpful to identify potential triggers or exacerbating factors. 23 A gait examination, including assessment of walking and running is essential. However, due to its paroxysmal nature, it may be difficult to reproduce RID during the visit. If possible, videos taken during an episode in the field, or on a treadmill, would help to better characterize dystonia and evaluate response to treatment. There is ongoing research focusing on wearable devices and artificial intelligence to help with the diagnosis, monitoring, and treatment of patients with movement disorders.24,25
What we tell people with PD in our clinic
We tell our patients that high-intensity aerobic exercise is the treatment with the most evidence to suggest it might be disease modifying. We also tell them that the best form of exercise is the one they will actually do. So, if they love running, it's our job to work with them to keep them running.
Triggers of RID are likely to be multifactorial, so we tell our patients that a holistic rehabilitation approach is recommended. Education, tailoring medications, strengthening, stretching, taping, changing the motor pattern, sensory tricks and managing stress, anxiety and sleep may all be helpful (Figure 2).
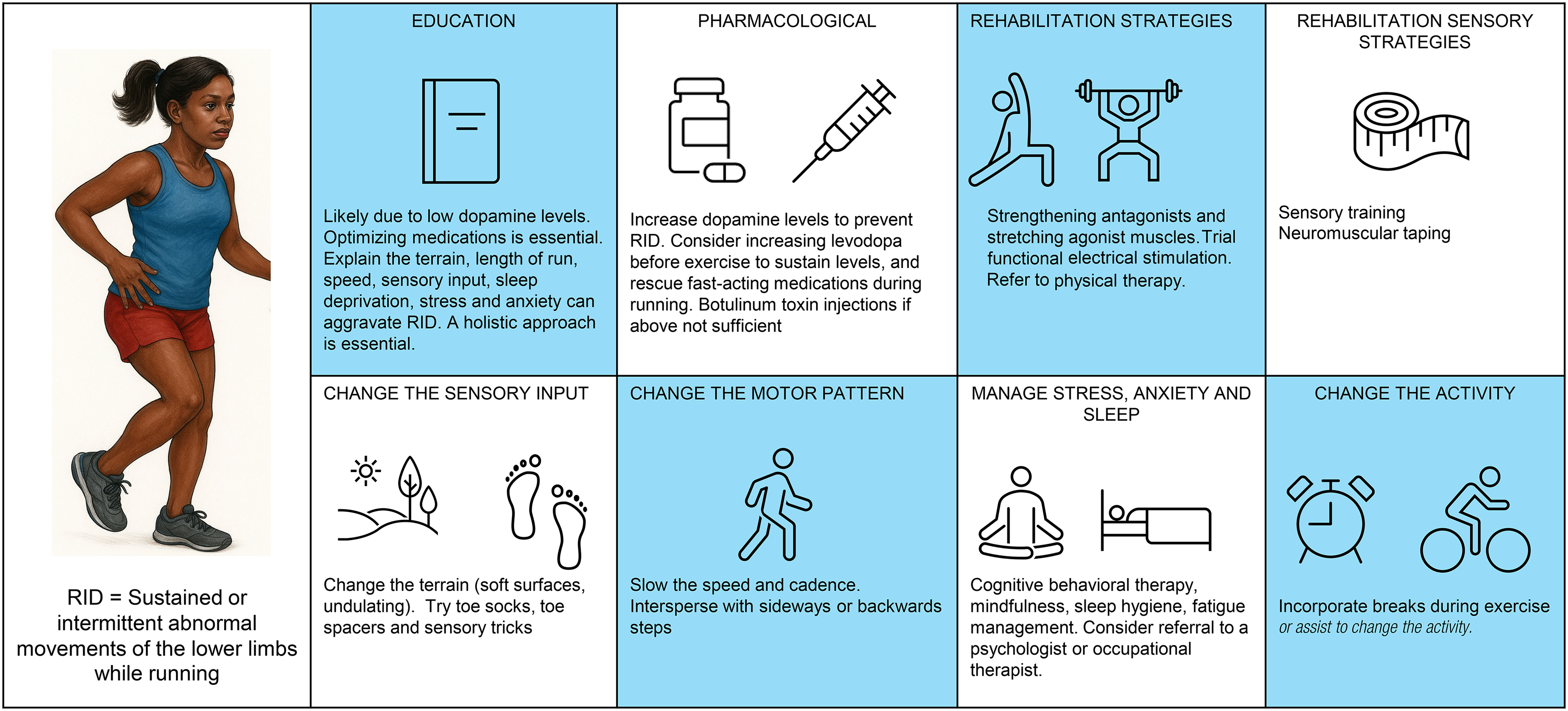
Management of RID. (Image created with AI based on the JAMA image drawn by Dr. Michael Okun and Dr. Melissa Armstrong 26 ).
Increasing the dose of levodopa right before, or during, a run may be beneficial in sustaining adequate dopamine levels. Others have found that dissolving the levodopa with vitamin C and sipping it may be helpful to provide more continuous dopaminergic stimulation. Rescue options may also be considered as soon as the dystonia starts (i. e. inhalation powder levodopa, apomorphine sublingual or apomorphine injections), but their use has not been investigated to treat RID in PD. In cases where RID persists despite medication adjustments, botulinum toxin injected into the muscles involved in the dystonia can be very helpful. 28 Although deep brain stimulation – specifically into the globus pallidus internus – is used to treat dystonia in people with and without PD, the effect of deep brain stimulation specifically on exercise-induced dystonia in people living with PD is unknown.
Epidemiological context and potential reporting bias
The three case reports were written by lived experience authors, two of whom have a health professional background (physician-scientist and physiotherapist). While we acknowledge that these personal accounts may add elements of selection or reporting bias, there is not to our knowledge an objective measure for RID that can easily be performed by a clinician, such as those that are performed as part of the MDS-UPDRS Part 3. In the absence of this, we believe that these narratives are a strength.
There is a need for systematic prospective studies to better understand RID and its risk factors. This includes the number of cases of running induced foot dystonia that convert to PD, the time period for conversion, the prevalence of RID in different age groups, and whether there are other factors, such as a history of long-distance running, that increase the risk of developing RID.
Summary
RID is a disabling symptom in PD that can impact an individual's preferred method of exercise. Medications should be tailored and optimized for those that wish to run. Although there is not a lot of research into non-pharmacological options for RID, we recommend a multidisciplinary approach that includes trial of sensory tricks, changing the terrain and motor pattern, altering sensory input, taping, stretching, strengthening and managing stress, anxiety, fatigue and sleep.
Supplemental Material
Footnotes
Ethical considerations
Not required. The three case studies are also authors of the paper.
Consent to participate
Confirmed consent for publication was received from each case who are also authors of this work.
Consent for publication
Confirmed consent for publication was received from each case who are also authors of this work. All authors have confirmed consent to publish this work.
Funding
DC and KEM - The research reported in this publication was supported by a generous philanthropic gift from the JCS Family Foundation and Robert and Mitra Ginsburg. It was also supported by the National Institute of Neurological Disorders and Stroke of the National Institutes of Health under Award Numbers U01NS113851. Research is also supported by the National Institutes of Health's National Center for Advancing Translational Sciences, Grant Number UL1TR001422. It is also supported by a generous philanthropic gift in honor of Howard Gilbert. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health.
Declaration of conflicting interests
The author(s) declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: MC – member National Parkinsons Alliance, past board member Parkinsons Tasmania. JA – member National Parkinsons Alliance, board member Parkinsons Tasmania. MC has received funds from Fight Parkinsons and Parkinsons Tasmania to attend or present at meetings. JA received funds from Abbvie and Interpharma for a Parkinsons symposium and has received funds from Abbvie, GP2, NovoNordisk and Parkinson's Tasmania to attend medical events. The other authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability
Not applicable.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.

