Abstract
Sleep problems are among the most frequently reported non-motor symptoms of Parkinson's disease (PD), with a broad range of disorders: insomnia, REM sleep behavior disorder, restless legs syndrome, excessive daytime sleepiness, and sleep-related breathing disorders. These disturbances evolve in complexity across PD severity stages, significantly impact the patients’ quality of life and may exacerbate motor and other non-motor symptoms. Neurodegenerative processes, impaired function of neurotransmitters, medication side effects, circadian rhythm dysfunction are among the most proposed mechanisms that may explain the frequent occurrence of sleep disorders in PD. However, there are still many unanswered questions related to the pathophysiological mechanisms of sleep disorders in PD that may offer the clue to better therapeutical options. Although the prevalence of sleep disturbances is very high, the treatment options are still limited. The current review focuses on main sleep disturbances encountered in PD, pathophysiological insights, current therapeutical options and future perspectives for a better and more personalized management of these disorders in PD.
Plain language summary
Sleep problems are very common in people diagnosed with Parkinson's disease and can take many forms. Some patients have difficulties falling or staying asleep (insomnia), act out their dreams while sleeping (REM sleep behavior disorder), feel the urge to move their legs at rest or night (restless legs syndrome), feel very sleepy during the day, or have breathing issues during sleep. These sleep problems can impair other symptoms of Parkinson's disease and reduce overall quality of life. Sleep disturbances tend to become more complex and more severe as the disease progresses. Researchers have found that several factors may cause sleep problems in Parkinson's disease, including the damage of specific brain areas that control sleep and wakefulness, loss of neurotransmitters (such as dopamine, serotonin, etc.) that normally help maintaining a normal and healthy sleep cycle, disruptions to the body's internal clock (the circadian rhythm), and side effects of medications used to treat other symptoms of Parkinson's disease. Even though these factors were found to play an important role in the occurrence of sleep problems, the exact causes of sleep problems in Parkinson's disease are still not fully understood. Gaining a better understanding of these underlying mechanisms could help researchers discover more effective and personalized treatments. Currently, treatment options for sleep problems in Parkinson's disease are limited. This review focuses on describing the main types of sleep problems found in Parkinson's disease, what is known so far about their causes, available therapies, and how future research could lead to more personalized care.
Keywords
Introduction
Sleep problems are a common non-motor symptom of Parkinson's disease (PD), affecting patients across all stages of the disease. A broad spectrum of sleep disturbances may be identified in PD patients: insomnia, parasomnia (particularly REM sleep behavior disorder, RBD), restless legs syndrome (RLS) and other sleep related movement disorders, circadian rhythm sleep-wake disorders, excessive daytime sleepiness (EDS), and sleep-related breathing disorders. 1 PD patients with late disease (over 6 years of evolution) ranked sleep problems (mainly insomnia and EDS) as the fourth most bothersome symptom. 2 Considering the high prevalence of sleep disorders in PD and their significant impact on patients’ quality of life, a non-motor subtype for PD patients with sleep disturbances was proposed—the Park Sleep Subtype. 3 In the prodromal stages, sleep disorders often serve as biomarkers of the disease. RBD is one of the most distinctive sleep-related symptoms during this phase. Studies suggest that up to 80% of individuals with isolated RBD eventually develop PD or other synucleinopathies. 4 EDS may also be a prodromal feature of PD, although the evidence is limited. 5 As PD progresses into its early motor stages, sleep disturbances tend to become more pronounced. 6 Longitudinal studies have noted an increased prevalence of insomnia 7 and EDS 8 with longer disease duration. There are several proposed pathophysiological mechanisms that may explain the high occurrence of sleep disorders in PD and their progression along PD stages.
Effective management of sleep disorders in PD requires a multidisciplinary approach tailored to patients’ complaints and associated motor and non-motor symptoms. Current management strategies include education, correction of other medications’ side effects, and specific pharmacological interventions. However, these strategies have several limitations, and further studies are mandatory for a better understanding of pathophysiological mechanisms of sleep disorders in PD, which may offer the clue to developing new pharmacological approaches or disease-modifying therapies (Figure 1).
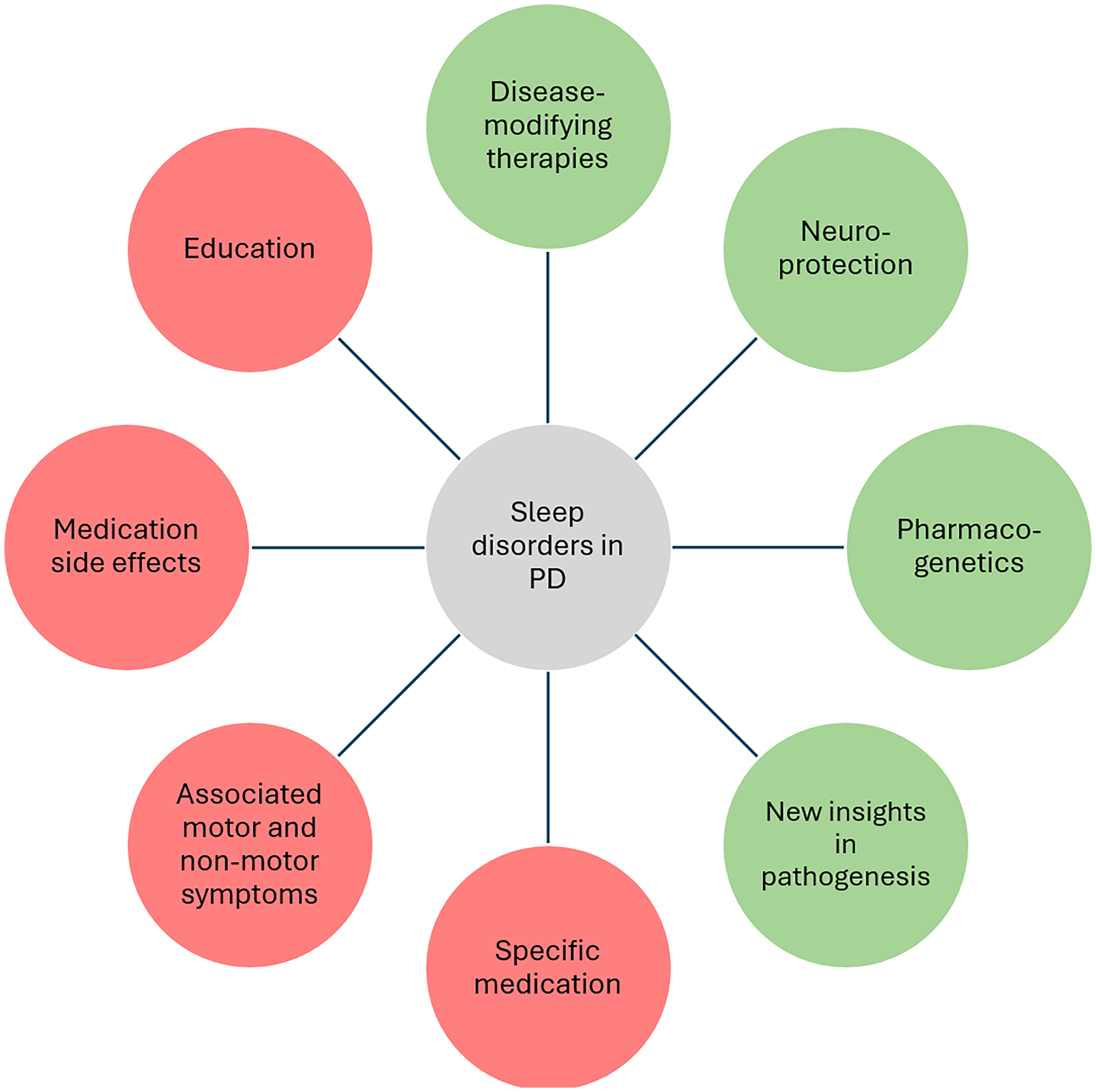
Management of sleep disorders in Parkinson's disease. Current management strategies (represented in red) include education, correction of medication side effects, treatment of associated motor and non-motor symptoms and specific medication. New therapeutic perspectives (represented in green) should focus on new insights in pathogenesis, pharmacogenetics, neuroprotection and disease-modifying therapies.
This current review aims to emphasize the main pathophysiological insights, the current state of the art and future directions for the management of sleep disorders in PD.
Insomnia
Insomnia is characterized by a difficulty in initiating or maintaining sleep and it is an integral component of the non-motor symptoms spectrum in PD. It is the most frequent sleep disturbance, with a reported prevalence of 30–80% across studies. 9 Furthermore, insomnia is associated with impairment in health-related quality of life, 10 increased disease duration, 11 cognitive decline 12 and mood disorders. 13
Pathophysiology of insomnia
Currently, there is no consensus related to a pathophysiological model of insomnia. 14 The multifaceted etiology of insomnia in PD arises from a variety of causes involving the neurodegenerative process (affecting key brain areas that modulate sleep), circadian rhythm dysfunction, the presence of nocturnal motor and non-motor symptoms, medication side effects and poor sleep habits. 15
Impaired activity in the suprachiasmatic nucleus (SCN) leads to lower levels of melatonin that have been found in patients with neurodegenerative disorders. 16 Beta activity in the basal ganglia was correlated with increased wakefulness and frequency of awakenings and decreased non-REM sleep in PD patients 17 as well as in animal models. 18
The presence of nocturnal motor symptoms (akinesia, tremor, dyskinesia, dystonia) as well as other non-motor symptoms (depression, anxiety, nocturia, pain) including other sleep disturbances (RBD, RLS) can lead to insomnia by sleep fragmentation, difficulty falling asleep and increased number of awakenings. Furthermore, dopaminergic medications such as amantadine, COMT inhibitors and dopamine agonists are essential for the management of motor symptoms, however the disruption of sleep architecture can be a potential side-effect of their use. This is also the case for medications used to treat PD comorbidities, such as acetylcholinesterase inhibitors and selective serotonin reuptake inhibitors (SSRIs). 19
Treatment
Careful investigation of the various etiological factors and their proper management represents the first step for a tailored management of insomnia. Non-pharmacological options, including Cognitive Behavioral Therapy for Insomnia (CBT-I), 20 which features structured programs incorporating education, behavioral interventions, and cognitive therapy, as well as bright light therapy 21 and exercise, 22 should be considered prior to starting other pharmacological agents. Optimization of current antiparkinsonian treatments should also be taking into consideration. As nocturnal motor symptoms are commonly associated with sleep fragmentation, optimization of dopaminergic treatment during nighttime is recommended. Controlled-released carbidopa-levodopa administered prior to bedtime may be helpful in improving akinesia, but with no significant effects on sleep parameters. 23 Long-acting ropinirole 24 and sustained-release pramipexole 25 showed improvements of nocturnal motor symptoms that might disturb sleep. A meta-analysis of 16 randomized controlled clinical trials supported the evidence that rotigotine transdermal patch improves motor symptoms and sleep quality, with no increased risk of adverse effects compared to placebo. 26 Therefore, rotigotine was considered “likely efficacious” in managing insomnia, according to the recommendations of the International Parkinson and Movement Disorder Society Evidence-Based Medicine Committee (MDS EBM). 27 In advanced PD stages, when deep brain stimulation is recommended to control motor fluctuations, improvements of subjective sleep parameters may also be noticed, even though objective assessments have shown that sleep architecture may be disturbed. 28 Continuous drug-delivery therapies may be considered with favorable outcomes in the sleep domain. Levodopa-carbidopa intestinal gel (LCIG) resulted in long-term improvements in insomnia and total sleep scores 29 while the novel molecule foslevodopa/foscarbidopa subcutaneous infusion also seems promising in improving sleep-related quality of life.30,31 Longer-acting levodopa formulations have the advantage to provide continuous 24-h drug delivery. There is an increasing body of scientific evidence that subcutaneous foslevodopa/foscarbidopa improves sleep problems.32,33 This treatment was shown to improve nocturnal akinesia, early morning off periods, sleep quality, and nocturia. 30 The improvements of sleep in patients undergoing foslevodopa/foscarbidopa were also associated with positive changes in quality of life and reductions in “Off” time. 34 Similar positive effects on sleep parameters, sleep quality, and reduced morning akinesia were observed in advanced PD patients treated with foslevodopa/foscarbidopa, with only mild side effects reported.35,36
In order to test the safety and efficacy of subcutaneous apomorphine during night-time for the treatment of insomnia in 46 PD patients, it was shown that night-time subcutaneous apomorphine improved the sleep disturbances, measured by the reduction of PDSS scores. 31 However, subcutaneous apomorphine was administered only during the night-time in this study and a 24-h regimen should be avoided, in order to prevent side effects.
When considering pharmacological treatment of insomnia, melatonin 37 and eszopiclone 38 were found to improve insomnia and sleep in small trials; there is insufficient data to support the effectiveness of melatonin and eszopiclone on insomnia in PD. MDS EBM recommends these drugs as “possibly useful” in clinical practice. 27 Although benzodiazepine or benzodiazepine receptor agonists are widely recommended in the general population for treatment of insomnia, there is insufficient data regarding their use in PD population. As these drugs might induce cognitive decline in non-PD population or might increase the risk of pneumonia in elderly PD patients, 39 it is advisable to avoid their use. Low doses of doxepin (10 mg) demonstrated positive effects on insomnia in PD patients. 40 Novel drug therapies for insomnia in PD include the drug suvorexant, an orexin receptor antagonist as high orexin plasma levels were correlated with the severity of non-motor symptoms, mainly REM sleep disorders. 41 A randomized, double-blind, placebo-controlled pilot study of suvorexant for insomnia in PD that aimed to assess the safety, tolerability and efficacy of suvorexant vs placebo in a cohort of PD patients with insomnia was conducted with results expected in the near future. 42
Current challenges and future directions
Firstly, the complex pathological pathways of insomnia in PD involve the neurodegenerative process, the abnormal functioning of the circadian rhythms, and factors related to PD treatment and symptoms. However, their molecular mechanisms are not yet fully understood. Furthermore, other factors such as race, gender and the environment may also play a role. Thus, further research is warranted in order to gain a better understanding of the complexity of the disease.
Secondly, there are no specific consensus-based recommendations for the treatment of insomnia in PD. Currently, the pharmacological management of insomnia is based on the insomnia guidelines in the non-PD and elderly populations as well as on the observed effect of PD medications on motor and non-motor symptoms. Future research, with randomized controlled trials that evaluate the efficacity and safety of medications for insomnia, specifically for PD patients, is needed.
A better understanding of the mechanisms of insomnia as well as the proper identification of the risk factors and tailored treatment could lead to an effective management of the disease as well as an improvement in the well-being of PD patients and their caregivers.
Parasomnia in PD: REM sleep behavior disorder
RBD is a parasomnia characterized by the loss of physiological muscle atonia found during REM sleep stage, with excessive muscle tone, abnormal behaviors—mostly dream enacting (varying from simple movements or vocalization to complex or violent behaviors), together with nightmares and vivid dreaming.43–45 RBD can be classified into two main forms, isolated or primary RBD (iRBD) and symptomatic or secondary RBD, the latter one occurring in neurodegenerative disorders (such as PD, dementia with Lewy bodies and multiple system atrophy) or other neurological disorders, such as narcolepsy type 1.
According to a meta-analysis, the estimated prevalence of RBD in PD is 23.6%, compared to 3.4% in the general population. RBD is an early marker in PD.46,47
Longitudinal studies have shown that a large proportion of patients with iRBD will eventually convert to a neurodegenerative disorder, this phenoconversion being observed within a mean interval of 10 years.48,49 The pedunculopontine nucleus may be considered a potential link between RBD and synucleinopathies, considering its role in REM-atonia circuitry and the bidirectional connectivity between this nucleus and substantia nigra, which is the main site of neurodegeneration in PD. 50
Pathophysiology of RBD
Dysfunction of the regulatory processes of the physiological REM sleep atonia is a key factor leading to RBD. There are several brain structures involved in maintaining REM sleep atonia, such as locus coeruleus/subcoeruleus, pons, and ventromedial medulla.51,52 Inhibitory projections from these regions modulate the motoneurons of the spinal cord via glutamatergic, GABAergic, and glycinergic systems and reduce the muscle tone during sleep. 53 The underlying pathological mechanisms for occurrence of RBD in PD can be explained by the neurodegenerative processes, mainly caused by accumulation of α-synuclein, that impair the function of the aforementioned structures. 52 PD patients with RBD exhibit abnormal glutamatergic signalling, 54 but also reduced glycine and GABA inhibition. 55 Abnormal activity within basal ganglia (increased beta power during REM sleep without atonia) may also contribute to occurrence of RBD in PD. 56 Latest research emphasizes the role of a pontine-medullary loop in REM sleep circuitry, which is dysfunctional in PD, as the neurons expressing corticotropin-releasing hormone-binding protein at this level are significantly numerically reduced and contain abnormal α-synuclein. 57
Management
Regarding specific pharmacological treatment, there are currently few interventions for the management of RBD. No randomized control trials were included in the International MDS-Evidence Based Medicine recommendations to prove the clinical benefit of pharmacotherapy on RBD, up to the present moment. 27 Data from clinical practice and small trials favor the use of clonazepam and melatonin. Low doses of clonazepam (0.25–1.0 mg) have been efficient to reduce dream enactment. 58 However, the efficacy of clonazepam was proved in retrospective studies/case series, and the mechanisms of action are insufficiently understood. Side effects of clonazepam are common and include dizziness, enuresis, gait difficulties and cognitive impairment. 59 Melatonin may be considered a better choice for elderly patients, for those with cognitive decline or increased risk of falls, 60 as it has fewer side effects (the most reported ones being headache and somnolence 58 ). Small clinical trials have shown improvements of the RBD symptoms and reduction of the muscle tone during REM sleep under melatonin treatment. 61 Melatonin can be used in monotherapy or in conjunction with clonazepam. For isolated RBD, the recommended dose of melatonin is 3 mg prior bedtime. This dose may be progressively increased up to 15 mg. 58 Other melatoninergic agonists have also been studied in small trials. Agomelatine was efficient in improving RBD symptoms in one case series of iRBD patients. 62 Ramelteon, a melatonin receptor agonist, was efficient in improving the RBD symptoms and also reduced the percentage of REM sleep without atonia, according to a case-report series, in two patients, one with multiple system atrophy and one with PD. 63 Beneficial effects of ramelteon in PD patients with RBD were also shown in a multicenter open trial. 64
Current medication should be revised, as RBD can be triggered by several antidepressant drugs – selective serotonin reuptake inhibitors, serotonin and noradrenaline reuptake inhibitors and tricyclic antidepressants.65–67 Comorbid obstructive sleep apnea should be identified and treated with continuous positive airway pressure. 68
There is data with potential some benefits, summarized below.
Dopamine agonists may also be tried in resistant cases. In one prospective open-label study on eleven PD patients, rotigotine partially improved the RBD clinical symptoms and sleep parameters, as evaluated by video polysomnography. 69 The effects of pramipexole are controversial. Improvement of the RBD symptoms and PSG parameters under pramipexole in iRBD patients was observed in some case series, 70 while in PD patients no significant effects were noted. 71 Significant improvements of RBD symptoms were noted during treatment with safinamide in PD patients, in a randomized, longitudinal, cross-over pilot study. 72 In one double-blind, crossover pilot trial, the cholinesterase inhibitor rivastigmine (4.6 mg patch/day) diminished the frequency of the RBD episodes in PD patients. 73 Donepezil, another cholinesterase inhibitor, in doses of 10 mg at bedtime showed positive effects on RBD symptoms in one case-series. 74 According to a pilot study, cannabidiol in doses up to 300 mg may improve the symptoms of RBD in PD. 75
Current challenges and future directions
To date, no large double-blind placebo-controlled trials have been conducted specifically for PD patients with RBD. Some of the current trials have not used polysomnography to certify the diagnosis of RBD, therefore the results, although promising (such as in the case of memantine 76 ) cannot fully support the recommendation for use in daily practice. Low sample sizes, inconclusive data and heterogeneity of the populations included in the studies conducted to date also limit the evidence basis for current pharmacological therapies.
As RBD is considered a prodromal symptom of PD and also a biomarker for all α-synucleopathies, efforts should be made in order to develop potential neuroprotective interventions. The importance of early rehabilitation should be recognized and recommended in the future. Early rehabilitation started in RBD may have a potential positive influence on functional independence and quality of life. 77 According to recent studies, melatonin may be considered as a chronobiotic/cytoprotective agent in RBD, 78 as it may be effective in reducing the phenoconversion to synucleinopathies. 79 However, further prospective clinical trials are necessary to determine the evidence base for current available pharmacological interventions in the PD population and the possible neuroprotective effects. New insights regarding pathophysiology of RBD in PD can contribute to the development of future possible targets for tailored pharmacological agents that can have better effectiveness and safety.
Excessive daytime sleepiness
EDS is a common sleep disorder among PD patients, with a described prevalence varying greatly between 20–70%, due to demographical and methodological differences among studies.80,81 Suffering from EDS results in lower quality of life, with a great impact on the activities of daily living, as well as on the psychological well-being. 82
Pathophysiology of EDS
Several risk factors for developing EDS have been described. These include older age, male gender, longer disease duration and increased disease severity, as some studies have shown positive correlations between EDS and the H&Y score, as well as the MDS-UPDRS score.81,83 EDS can be a consequence of decreased quality of nighttime rest due to the impact of other sleep disorders like insomnia, RBD, RLS or sleep apnea.84,85 Other non-motor symptoms like nocturia, depression, anxiety and pain can also lead to sleep dysfunction and subsequently to increased daytime sleepiness. 86 Furthermore, the antiparkinsonian medication itself, in particular dopamine agonists, have been found to possibly aggravate excessive daytime sleepiness. 87 In several studies, pramipexol was found to have the most significant effect, and should therefore be cautiously used in PD patients with concurrent sleep dysfunction.88,89
The pathophysiology of EDS is complex and involves dysfunction of multiple circuits of various neurotransmitters such as the dopaminergic, cholinergic and serotoninergic pathways. 86 Similarly to narcolepsy, the progressive loss of orexin-containing neurons with PD advancement, could also be involved in the development of EDS. 90
Treatment
Before considering any specific therapeutic option, modifiable risk factors described beforehand should be optimized. Screening for other comorbidities should be carried out and treatment of other non-motor symptoms revised. The need for dopamine agonists, in particular pramipexol and ropinirole should be re-assessed, keeping the dosage to a minimum. 3
As a good foundation for any pharmacological treatment, sleep hygiene should always be practiced. This includes, among others, going to bed and waking up at regular hours, engaging in physical activity during the day and avoiding high intake of stimulants like coffee, black tea and energy drinks. 91
When it comes to pharmacological therapy, options with sufficient evidence are so far scarce. The most used, “possibly useful” agent, as considered by the Movement Disorders Society Evidence-Based Medicine Committee (MDS-EBM), is modafinil. 27 Although the exact mechanism by which modafinil promotes wakefulness in patients with EDS is not entirely known, a meta-analysis of three trials showed its beneficial effects in significantly reducing the ESS score. 92 Another study found a reduction of the ESS score by methylphenidate. However, more research is warranted. 93
Current challenges and future directions
Tackling the treatment of excessive daytime somnolence is a challenging task, as EDS is both an individual sleep disorder, as well as a consequence of other non-motor symptoms, prevalent in PD. As mood, psychological state and emotional well-being are known to have a significant impact on sleep quality, cognitive-behavioural therapy can help improve symptoms related to insomnia and consequently help achieve a better rest during nighttime, leading to less somnolence during the day, but no specific studies on its effects on EDS have been yet conducted. 94 Several wake-promoting agents have shown promise in treating EDS in individual studies, however, more research needs to be conducted to confirm these findings and to support their use in everyday practice. These include sodium oxybate (which has been so far used for the treatment of narcolepsy-type 1), 95 pitolisant (a histamine-3 receptor inverse agonist), 96 solriamfetol (a dopamine/norepinephrine reuptake inhibitor), 97 and istradefylline (a selective adenosine A2A receptor antagonist). 98
Restless legs syndrome and other sleep related movement disorders
Restless legs syndrome, also known as Willis-Ekbom syndrome, is described as unpleasant and distressing sensations in the lower limbs, occurring during nighttime or rest, that provoke the urge to move the limbs, with partial or full relief of the symptoms after movement. 99 Mimics of RLS such as nocturnal cramps, venous stasis, myalgia, 99 neuropathy, akathisia, leg motor restlessness,100,101 should be carefully checked for and excluded before establishing the diagnosis. The estimated prevalence of RLS in the PD population varies widely (0–50%), 102 with the tendency to increase as the disease progresses. 103 There are higher odds ratios for RLS in PD patients compared to controls. 104 As both RLS and the motor symptoms of PD respond to dopaminergic therapy, the prevalence of RLS may be even higher, but masked by chronic dopaminergic treatment. 100 RLS in PD is associated with poor sleep quality, nocturnal pain, fatigue, 105 and mood disorders. 106 The link between RLS and PD is controversial. According to some studies, RLS may be a clinical early feature of PD, 107 while other research failed to find any conclusive link between RLS and PD. 104 According to a pilot study, idiopathic RLS syndrome may even delay the onset of PD and decrease PD progression. 108
Pathophysiology of RLS
The current knowledge regarding pathological mechanisms of RLS in PD is scarce. Dysfunction of the dopaminergic, adenosine and glutaminergic systems, low iron levels and genetic susceptibility are some of the most known factors leading to RLS in the general population.109,110 However, in PD, these mechanisms do not entirely explain the pathophysiology of RLS and several differences between idiopathic RLS and RLS in PD have been identified. Iron deposition in the substantia nigra in PD is increased, while in idiopathic RLS is decreased.111,112 The nigrostriatal dopaminergic function, substantially reduced in PD, may be normal 113 or even increased in idiopathic RLS. 114 Functional imaging studies showed reduced homogeneity and connectivity within the precentral and postcentral gyri in PD patients with RLS, regions that are related to abnormal functionality of the sensorimotor pathways. 115
Treatment
Certain drugs, such as antidepressants and neuroleptics 116 can induce RLS symptoms as side effects, therefore careful revision of the current medication regimen is advised before starting a management plan. Non-pharmacological measures, such as exercise, application of cooling sprays, hot or cold baths, reduction of alcohol, caffeine and nicotine intake, 117 the use of pneumatic compression devices should be tried. 118 Peroneal nerve stimulation, 119 cognitive behavior therapy120,121 or near-infrared light therapy 122 are some of the non-pharmacological options that showed reduction of the RLS symptoms in the general population, but these results were not confirmed in the PD population.
There are no randomized controlled trials conducted specifically for PD patients with RLS. Current therapeutic recommendations are based on data in idiopathic RLS.
According to latest guidelines for management of RLS in general population, first-line pharmacological agents should be α-2-δ ligands such as gabapentin, gabapentin enacarbil and pregabalin, as the risk of augmentation for these medications is the lowest. 123 Moreover, it was shown that long-term treatment with dopamine agonists may reduce the efficacy of other therapeutic choices, such as gabapentin enacarbil. 124 However, the evidence for the effectiveness of α-2-δ ligands in PD is limited. Common side effects of the α-2-δ ligands include sleepiness, fatigue, dizziness, and headache. These medications could be considered as first therapeutic options in PD patients with RLS and other associated non-motor symptoms, such as anxiety, insomnia or pain. 125
Levodopa and dopamine agonists (DA) were reported to be effective on RLS in PD patients. 126 However, considering that many PD patients are already treated with levodopa and/or DA, the use of higher doses is limited by side effects. Albeit very rare in PD, the augmentation syndrome (characterized by worsening of the RLS symptoms, extension of the symptoms to upper limbs or occurrence also during activity/daytime 127 ) is a side effect that should be considered. Rotigotine has shown improvements of the RLS symptoms in several open studies.128–131 Apomorphine may also be effective in PD patients with RLS, as it may reduce the number of spasms, awakenings, 132 and the index of periodic limb movements. 133
Current evidence regarding the efficacy of iron therapy in PD is scarce; although iron supplementation should be recommended in idiopathic RLS (in cases where serum ferritin is lower than 300 mg/ml 134 ), this intervention has not been validated in PD patients.
Other therapies that have been successful in the management of RLS in PD in some case-report studies (such as istradefylline, a selective adenosine A2A receptor antagonist 135 and safinamide, a monoaminoxidase-B inhibitor 136 ) need further validation in larger randomized controlled trials.
According to a retrospective study which involved twenty-two PD patients, DBS of the subthalamic nucleus (STN) significantly improved RLS symptoms, allowing reduction of the dosage of the dopaminergic medication, results that were constant over a 2-year period. 137 This study provided Class IV evidence for DBS-STN in PD patients with moderate to severe symptoms of RLS. 137
Periodic limb movements during sleep (PLMS) involve repetitive, stereotyped movements of the lower limbs occurring during sleep. 138 Periodic limb movement disorder (PLMD) is defined by PLMS that disrupt sleep or lead to daytime dysfunction, in the absence of other sleep disturbances such as RLS, RBD, untreated obstructive sleep apnea or narcolepsy.45,139
Although PLMS is common in PD, especially as PD worsens, and leads to more sleep disturbances and lower quality of life, 140 there are no clear associations between PLMS and PD 141 or between RLS and PLMS in PD. 142 The pathophysiology of PLMS is linked to dysfunctions of the dopaminergic system, 143 or side effects of antidepressant medications, such as selective serotonin reuptake inhibitors. 144 Overnight PSG remains the gold standard for the diagnosis of PLMS. 145 There are no evidence-based treatment recommendations for PLMD, 146 but in patients with RLS, dopamine agonists (particularly D3 selective agonists), levodopa and α-2-δ ligands may be effective. 147 In the latest guideline regarding the management of RLS and PLMD, the American Academy of Sleep Medicine (AASM) suggests against the use of triazolam and valproic acid on PLMD in the general population. 139
Bruxism is characterized by repetitive clenching or grinding of teeth and may often co-occur with obstructive sleep apnea. 148 Treatment generally involves a dental appliance worn during sleep. Currently, evidence on the effectiveness of pharmacotherapy for treating sleep bruxism is insufficient. 149 For cases where dental appliances are insufficient, botulinum toxin may be effective. 150
Current challenges and future directions
To date, no consensus regarding optimal management of RLS in PD is available. As mentioned before, no randomized controlled trials were conducted for PD patients with RLS to date. Validation of the current diagnostic criteria of RLS in the PD population is needed. Although there are available guidelines for the management of idiopathic RLS and augmentation, their application in PD is questionable, considering the possible different pathological mechanisms of the two entities and the particularities of the PD patients (for instance the need for higher doses of dopaminergic treatment in PD which in the general population may lead to augmentation). Moreover, α-2-δ ligands are recommended as the first-line therapy for RLS in the general population, but in PD, DA therapies may be considered as the first line choice in cases where RLS occur as an off symptom. Evidence from the clinic suggests that DA can be more effective than levodopa in some patients and immediate-release DA at night can be particularly helpful in patients not tolerating higher doses of controlled-release DA. Although continuous drug delivery for advanced PD patients (through formulations such as levodopa-carbidopa intestinal gel 151 ) was reported to improve sleep parameters, further larger studies are necessary to confirm the efficacy on RLS. While foslevodopa/foscarbidopa has been shown to improve sleep in several studies,35,36 its specific impact on RLS remains yet to be determined. Moreover, further research is needed to better understand the pathophysiology of RLS in PD and to develop more targeted treatments. Longitudinal studies are also mandatory to evaluate the progression of RLS in PD and its impact on the overall disease trajectory. Due to the complexities involved in diagnosing PLMD, it is essential to establish specific criteria for its evaluation and diagnosis to facilitate high-level research on this disorder, including in the PD population.
Efforts should be made to assess the efficacy of the current therapeutic options in large randomized controlled trials in PD patients and to investigate new pharmacological agents.
Other sleep problems in PD
Circadian rhythm sleep-wake disorders
An impaired circadian rhythm can lead to sleep disturbances, as well as daily fluctuations of both motor and non-motor symptoms in PD patients and is tightly correlated with the neurodegenerative process. The SCN is the main structure involved in the synthesis of melatonin on a circadian basis through integration of information received from the retina and adjustment of the internal circadian rhythms. 152 Therefore, pathological α-synuclein accumulations in the retinal dopaminergic cells 153 (leading to impaired signal transmission to SCN) and in the SCN contribute to clock gene dysregulation and circadian dysfunction. 154
Circadian rhythm disorders involve chronic sleep disruptions due to changes in the circadian system or misalignment with socially imposed sleep-wake schedules. 45 Research is ongoing into chronotherapeutics for PD, including bright light exposure, physical exercise, and melatonin. 9
Sleep-disordered breathing in PD
Obstructive sleep apnea (OSA) is the most common sleep-disordered breathing in PD, affecting 20–70% of patients. 155 Sleep-disordered breathing could be encountered more frequently in patients with PD that also have RBD. 47 OSA can cause cardiovascular and neurological issues, particularly cognitive decline, impacting the quality of life in PD patients.156,157 Continuous positive airway pressure (CPAP) is the gold standard treatment.
Main known pathophysiological mechanisms, current state of the art and weaknesses of current management strategies of sleep disorders in PD are summarized in Table 1.
Main known pathophysiological mechanisms, current state of the art and weaknesses of current management strategies of sleep disorders in Parkinson's disease.
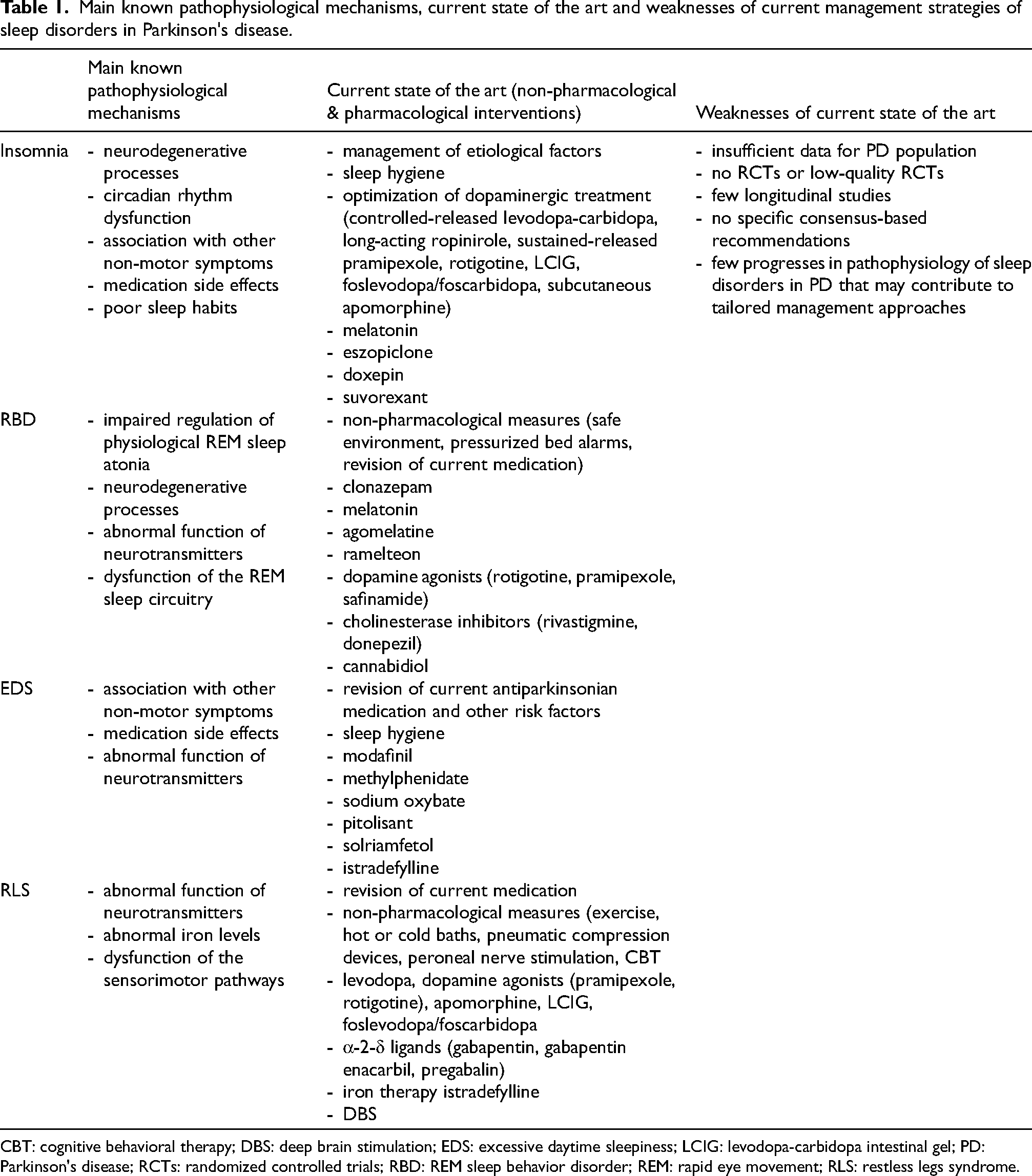
CBT: cognitive behavioral therapy; DBS: deep brain stimulation; EDS: excessive daytime sleepiness; LCIG: levodopa-carbidopa intestinal gel; PD: Parkinson's disease; RCTs: randomized controlled trials; RBD: REM sleep behavior disorder; REM: rapid eye movement; RLS: restless legs syndrome.
Footnotes
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
