Abstract
Background
Peripheral neuropathy in Parkinson's disease (PD) may not be an isolated pathological phenomenon but may interact with other non-motor symptoms.
Objective
To conduct a comprehensive investigation of the non-motor symptoms observed in PD patients, with a comparative analysis between patients with and without peripheral neuropathy, to explore the relationships between peripheral neuropathy and non-motor symptoms.
Methods
100 PD patients were recruited from the Neurology Department of Guangdong Provincial People's Hospital. Extensive clinical data were collected, and assessments were performed to evaluate both motor and non-motor symptoms in patients with and without peripheral neuropathy. Spearman's rank correlation analysis was employed to evaluate the association between peripheral neuropathy and non-motor symptoms. Multiple linear regression analysis was conducted to adjust for confounding variables to clarify the impact of peripheral neuropathy on non-motor symptoms.
Results
57 individuals (57%) were identified as having peripheral neuropathy. Of these, 38 individuals were diagnosed with small fiber neuropathy. After adjusting for confounding variables, higher scores on the 13-item Small Fiber Neuropathy Symptom Inventory Questionnaire were significantly correlated with heightened symptoms of depression (B = 0.498, 95% CI = 0.080–0.917, p = 0.020), anxiety (B = 0.504, 95% CI = 0.099–0.908, p = 0.015), autonomic dysfunction (B = 1.118, 95% CI = 0.828–1.409, p < 0.001), and sleep disturbances (B = 0.694, 95% CI = 0.309–1.080, p = 0.001).
Conclusions
PD patients with neuropathy, particularly those with small fiber neuropathy, demonstrate more pronounced non-motor symptoms. The severity of small fiber neuropathy is correlated with the extent of depression, anxiety, autonomic dysfunction, and sleep disturbances. These findings underscore the necessity for routine screening and early detection of peripheral neuropathy in PD patients.
Plain language summary
Parkinson's disease (PD) is a neurological disorder characterized by symptoms such as slowed movements and tremors in the hands or other body parts, commonly referred to as motor symptoms. Additionally, patients may experience discomfort primarily manifested as limb numbness, anxiety, loss of interest, poor sleep, and dysregulation of bowel and bladder function, which we call non-motor symptoms. These non-motor symptoms often occur together, suggesting that there may be certain connections between them. Our research explored the relationship between peripheral neuropathy and other non-motor symptoms like anxiety, loss of interest, and sleep disorders. We recruited 100 PD patients, collected their motor and non-motor symptoms, and assessed their peripheral nerve conditions. Among the 100 PD patients, as many as 57 patients had peripheral neuropathy. Then they were divided into groups based on the presence or absence of peripheral neuropathy. By comparing the non-motor symptoms of patients with and without peripheral neuropathy, we found that PD patients with peripheral neuropathy were more likely to experience anxiety, loss of interest, abnormal bowel and bladder function, and sleep disorders. Furthermore, the severity of symptoms related to peripheral neuropathy, such as numbness, was correlated with the severity of non-motor symptoms, meaning that patients with hand and foot numbness or pain were also more likely to experience related symptoms like sleep disorders. This highlights the importance of regularly checking for peripheral neuropathy in PD patients to address these additional challenges early on. Early detection can help manage and potentially improve non-motor symptoms, enhancing the overall quality of life for these patients.
Introduction
Parkinson's disease (PD) is a neurodegenerative disorder characterized by the presence of motor symptoms as well as a broad spectrum of non-motor symptoms. 1 Previous studies have demonstrated that the prevalence of large fiber neuropathy in PD patients ranges from 6% to 58%, while the incidence of small fiber neuropathy is reported to be between 37% and 91%. 2 These figures indicate that the occurrence of peripheral neuropathy in PD patients is higher compared to that in the general elderly population. Pathological studies have also confirmed that the abnormal accumulation of alpha-synuclein occurs not only in the central nervous system but also in the peripheral nervous system. 3
The diagnosis of peripheral neuropathy in PD is complicated by the absence of standardized methodologies. Large fiber neuropathy can be identified through nerve conduction studies, whereas the detection of small fiber neuropathy necessitates clinical evaluations and specialized testing procedures. 4 Non-invasive assessments, such as sympathetic skin responses (SSR) and cutaneous silent periods (CSP), have proven effective in identifying small fiber neuropathy and are practical for clinical application.5,6 Furthermore, the 13-item Small Fiber Neuropathy Symptoms Inventory Questionnaire (SFN-SIQ) is a reliable instrument for assessing small fiber neuropathy. 7
Mood disorders, cognitive impairment, autonomic nervous system dysfunction, and sleep disturbances are common non-motor symptoms observed in PD patients. Currently, there are no effective treatments available to ameliorate these non-motor symptoms.1,8 Research suggests that individuals with peripheral neuropathy, resulting from various causes such as chronic inflammatory demyelinating polyneuropathy, chemotherapy-induced peripheral neuropathy, and diabetic peripheral neuropathy, may also experience cognitive impairments and mood disorders.9–11 This suggests a potential relationship between peripheral neuropathy and these symptoms. Furthermore, previous studies have identified an association between pain and non-motor symptoms in PD patients, and the severity of peripheral neuropathy has been linked to disease severity and progression in PD.12–14 These findings imply that peripheral neuropathy in PD may not be an isolated pathological phenomenon but may interact in complex ways with other non-motor symptoms.
Our study aims to integrate clinical evaluations that include assessments of both motor and non-motor symptoms, as well as peripheral nerve function evaluations through clinical history and electrophysiological testing in PD patients. The objective is to conduct a thorough analysis of the interrelationships between peripheral neuropathy and non-motor symptoms in PD patients, with the aim of elucidating the influence of peripheral neuropathy on non-motor symptoms and overall quality of life within this population.
Methods
Study approval and patient consents
The study adhered to the ethical principles established in the 1964 Declaration of Helsinki and its subsequent amendments. Ethical approval was granted by the Institutional Ethical Committee of Guangdong Provincial People's Hospital (KY-Q-2021-105-01). Informed written consent was obtained from all participants after a after thorough explanation of the study procedures.
Participants
Participants were recruited from the Neurology Department of Guangdong Provincial People's Hospital from May 2021 to October 2023. The eligibility criteria for inclusion were as follows: (1) Participants had to meet the 2015 International Movement Disorder Society (MDS) diagnostic criteria for confirmed PD. 15 (2) Participants were required to be aged between 50 and 80 years. (3) Participants had to provide informed consent to complete the necessary evaluations. Furthermore, the diagnosis of confirmed PD was independently verified by two experienced neurologists specializing in movement disorders. The exclusion criteria were as follows: (1) Participants who had undergone deep brain stimulation therapy or possessed a heart pacemaker, making them unsuitable for electrophysiological assessment. (2) Participants with comorbid peripheral neuropathy-related disorders, including but not limited to diabetes mellitus, Vitamin B12 deficiency, hepatic or renal failure, and HIV.
Data collection
All data were collected during the patients’ hospitalization.
The collection of general clinical data includes variables such as age, gender, height, disease duration and the levodopa equivalent daily dose (LEDD).
After a fasting period of no less than 12 h, all participants underwent a panel of blood tests designed to screen for additional disorders associated with peripheral neuropathy. These assessments included measurement of serum vitamin B12, homocysteine, folate, blood glucose levels, hemoglobin A1C, among others.
Motor and non-motor symptoms were evaluated by clinicians who were blinded to the results of the peripheral nerve evaluation. Motor function was assessed using the Movement Disorder Society Unified Parkinson's Disease Rating Scale (MDS-UPDRS) Part III and the Hoehn and Yahr (H&Y) stage. Participants were evaluated in an “off” state, as those undergoing anti-parkinsonian therapy were instructed to discontinue all dopaminergic medications at least 12 h prior to the evaluation. Non-motor symptoms were evaluated as follows: depression was measured using the Hamilton Depression Scale (HAMD), anxiety was evaluated with the Hamilton Anxiety Scale (HAMA). Cognitive function and autonomic dysfunction were evaluated using the Mini-Mental State Examination (MMSE) and the SCOPA-AUT, respectively. Sleep disorder was evaluated using the Parkinson's Disease Sleep Scale-2 (PDSS-2) scale. Furthermore, the quality of life was evaluated using the Activities of Daily Living (ADL) scale.
Peripheral nerve assessment
The evaluation of peripheral nerve was performed by clinicians who were blinded to the non-motor outcomes. This evaluation consists of two main parts. The first part involves the acquisition of peripheral neuropathy symptoms and signs, which were obtained through medical history, physical examination and 13-item SFN-SIQ. The second part involves electrophysiological testing of the peripheral nerve. The evaluation of large fiber nerves is conducted through nerve conduction studies, encompassing motor conduction and F-wave analysis of the median, ulnar, and tibial nerves bilaterally, in addition to sensory conduction assessments of the median, ulnar, and sural nerves on both sides. The evaluation of small fiber nerves is assessed through SSR of four limbs and CSP of both hands.
Diagnostic criteria for peripheral neuropathy
Participants were diagnosed large fiber neuropathy if any nerve conduction results were abnormal.
Participants were diagnosed small fiber neuropathy if they met at least two of the following three criteria: (1) 13-items SFN-SIQ score ≥ 6.5; (2) Abnormal SSR result of any limbs; (3) Abnormal CSP result of any hands.
Statistical analysis
Statistical analyses were performed using SPSS 25.0. The distribution of continuous data was evaluated through histograms, probability-probability (P-P) plots, and quantile-quantile (Q-Q) plots. Continuous variables exhibiting a normal distribution were reported as mean ± SD, whereas those not following a normal distribution were reported as median and interquartile range (IQR). Categorical variables were presented as frequencies and percentages. T-test was employed to compare normally distributed continuous variables between two groups, whereas the Mann-Whitney U test was used for variables that did not follow normal distribution. Chi-square tests or Fisher's exact tests were utilized to compare categorical variables across groups, depending on the sample size. Spearman's rank correlation analysis was used to assess the correlation between SFN-SIQ scores and non-motor symptoms. Multiple linear regression analysis was conducted to adjust for confounding factors while investigating the impact of peripheral neuropathy on non-motor symptoms in PD. The models incorporate variables that account for potential confounding factors, as well as those variables that demonstrated differences between patients with and without peripheral neuropathy. These variables include age, gender, disease duration, UPDRSIII scores and H&Y stage. The distribution of residuals within the models was assessed graphically to evaluate the assumption of normality. Both residuals and collinearity were considered in evaluating the models’ fit to the data. The missing data were distributed as follows: there was one missing case for HAMD, four missing cases for both MMSE and PDSS-2, and five missing cases for both UPDRS-IV and educational background. To optimize data utilization, the pairwise deletion method was employed to address the missing data. The significance level was established at p ≤ 0.05.
Results
Demographic and clinical characteristics
Initially, the study enrolled 134 patients. However, 11 patients were excluded due to incomplete peripheral nerve assessments, 6 were excluded because of abnormal blood test results, and 17 patients were excluded due to a probable diagnosis of PD. Consequently, the final cohort comprised 100 patients, with 49% being male. The mean age of the participants was 62.63 ± 7.70 years, and their mean height was 160.98 ± 9.15 cm. The median disease duration was 3.75 years, with an interquartile range of 1.5 to 6 years. A total of 70 (70%) participants were undergoing anti-Parkinson treatment. The mean UPDRS III score was 37.07 ± 15.48, and the median H&Y stage was 2, with an interquartile range of 2 to 2.5. 51 (51%) were classified as tremor-dominant, 41 (41%) as postural instability gait difficulty (PIGD) type, and 8 (8%) as intermediate type.
In our study, 57 (57%) individuals exhibited peripheral neuropathy (PD-PN). Specifically, 30 individuals presented with large fiber neuropathy (PD-LPN), 38 individuals presented with small fiber neuropathy (PD-SPN). No statistically significant differences were identified in terms of gender ratio, height, education level, drug-naive proportion, motor subtype distribution, and LEDD between the PD-PN group and the PD-NPN group (Table 1). Patients in PD-PN group were older (64.18 ± 7.88 vs. 60.58 ± 7.02, p = 0.020) and had a longer disease duration (4 (2.5–7) vs. 2 (1.08–5.25), p = 0.018) than those in PD-NPN group.
The non-motor characteristics of Parkinson's disease accompanied by peripheral neuropathy.

*p < 0.05; n: number; PD-NPN: Parkinson's disease patients without peripheral neuropathy; PD-PN: Parkinson's disease patients with peripheral neuropathy; LEED: Levodopa equivalent dose; UPDRS: Unified Parkinson's Disease Rating Scale; H&Y: Hoehn & Yahr stage; TD: tremor-dominant; PIGD: Postural instability/gait difficulty; HAMD: Hamilton Depression Scale; HAMA: Hamilton Depression Scale; MMSE: Mini-Mental State Examination; PDSS-2: Parkinson's Disease Sleep Scale-2; ADL: activities of daily living.
No statistically significant differences were identified in terms of gender ratio, height, education level, drug-naive proportion, motor subtype distribution, and LEDD between the PD-LPN and PD-NPN groups. Patients in PD-LPN group were slightly older (66.23 ± 7.15 vs. 60.58 ± 7.02, p = 0.001), and had a longer disease duration (4 (3–7) vs. 2 (1.08–5.25), p = 0.020) compared to those in PD-NPN group (Table 2).
The non-motor characteristics of Parkinson's disease accompanied by large fiber neuropathy.

*p < 0.05; n: number; PD-NPN: Parkinson's disease patients without peripheral neuropathy; PD-LPN: Parkinson's disease patients with large fiber neuropathy; LEED: Levodopa equivalent dose; UPDRS: Unified Parkinson's Disease Rating Scale; H&Y: Hoehn & Yahr stage; TD: tremor-dominant; PIGD: Postural instability/gait difficulty; HAMD: Hamilton Depression Scale; HAMA: Hamilton Depression Scale; MMSE: Mini-Mental State Examination; PDSS-2: Parkinson's Disease Sleep Scale-2; ADL: activities of daily living.
No statistically significant differences were identified in terms of gender ratio, age, height, disease duration, education level, drug-naive proportion, education level, motor subtype distribution and LEDD between the PD-SPN group and the PD-NPN group (Table 3).
The non-motor characteristics of Parkinson's disease accompanied by small fiber neuropathy.

*p < 0.05; n: number; PD-NPN: Parkinson's disease patients without peripheral neuropathy; PD-SPN: Parkinson's disease patients with small fiber neuropathy; LEED: Levodopa equivalent dose; UPDRS: Unified Parkinson's Disease Rating Scale; H&Y: Hoehn & Yahr stage; TD: tremor-dominant; PIGD: Postural instability/gait difficulty; HAMD: Hamilton Depression Scale; HAMA: Hamilton Depression Scale; MMSE: Mini-Mental State Examination; PDSS-2: Parkinson's Disease Sleep Scale-2; ADL: activities of daily living.
Relationship between peripheral neuropathy and depression, as well as the relationship between peripheral neuropathy and anxiety
PD-PN group demonstrated higher HAMD scores than PD-NPN group (11 (5.5–17) vs. 8 (3.5–13), p = 0.046). Subgroup analysis revealed PD-SPN group had higher HAMD scores than the PD-NPN group (12 (5–17) vs. 8 (3.5–13), p = 0.034). These findings suggest that PD patients with small fiber neuropathy might experience more pronounced depressive symptoms. To further investigate the relationship between small fiber neuropathy and depression symptoms, a Spearman correlation analysis was conducted to assess the association between HAMD scores and the 13-item SFN-SIQ scores, which served as indicators of small fiber neuropathy symptoms. The analysis revealed a positive correlation between the 13-item SFN-SIQ scores and HAMD scores (r = 0.313, p = 0.002, Figure 1(a)). Furthermore, multiple linear regression analysis indicated that HAMD scores increased with 13-item SFN-SIQ scores in PD patients (B = 0.498, 95% CI = 0.080–0.917, p = 0.020), after adjusting for age, gender, disease duration, UPDRS-III scores and H&Y stage (Supplemental Table 1).
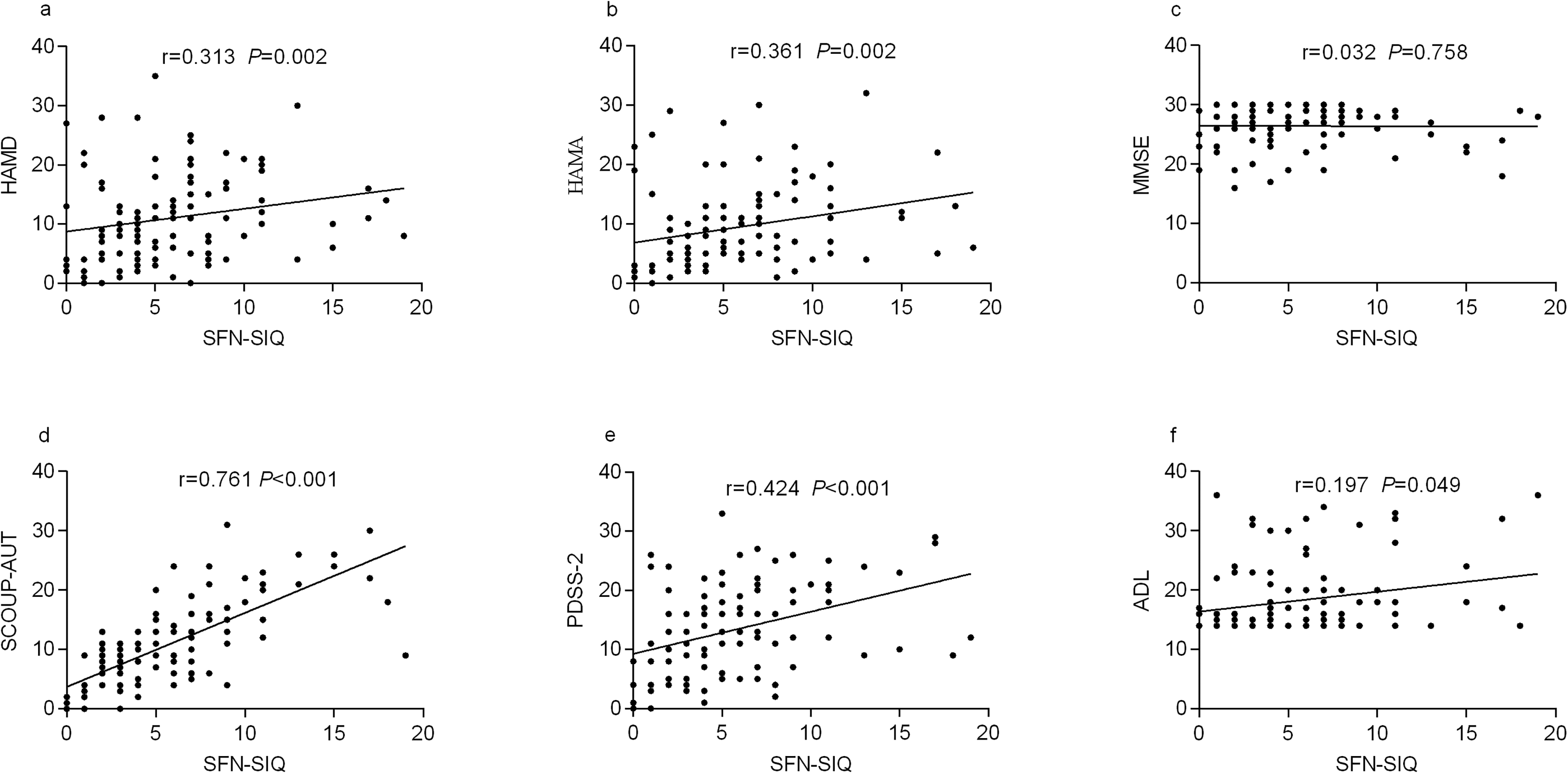
Relationship between small fiber neuropathy symptoms, non-motor symptoms, and quality of life. (a) Relationship between 13-item SFN-SIQ scores and depression symptoms. (b) Relationship between 13-item SFN-SIQ scores and anxiety symptoms. (c) Relationship between 13-item SFN-SIQ scores and cognition symptoms. (d) Relationship between 13-item SFN-SIQ scores and autonomic dysfunction symptoms. (e) Relationship between 13-item SFN-SIQ scores and sleep disturbances symptoms. (f) Relationship between 13-item SFN-SIQ scores and activities of daily living.
PD-PN group demonstrated higher HAMA scores than PD-NPN group (9 (5–15) vs. 6 (2.5–11), p = 0.020). Subsequent subgroup analyses revealed PD-SPN group had higher HAMA scores than the PD-NPN group (11 (5–15) vs. 6 (2.5–11), p = 0.010). These findings suggest that PD patients with small fiber neuropathy might experience more pronounced anxiety symptoms. To further investigate the relationship between small fiber neuropathy and anxiety symptoms, a Spearman correlation analysis was conducted to evaluate the association between HAMA scores and the 13-item SFN-SIQ scores. The analysis revealed a positive correlation between the 13-item SFN-SIQ scores and HAMA scores (r = 0.361, p = 0.002, Figure 1(b)). Furthermore, multiple linear regression analysis indicated that HAMA scores increased with 13-item SFN-SIQ scores in PD patients (B = 0.504, 95% CI = 0.099–0.908, p = 0.015), after adjusting for age, gender, disease duration, UPDRS-III scores and H&Y stage (Supplemental Table 2).
Relationship between peripheral neuropathy and cognition
PD-PN group demonstrated lower MMSE scores than PD-NPN group (26.5 (23–29) vs. 28 (27–29), p = 0.021). Subsequent subgroup analyses revealed PD-SPN group exhibited lower MMSE scores than the PD-NPN group (27 (23–28.5) vs. 28 (27–29), p = 0.012). These findings suggest that PD patients with small fiber neuropathy might experience more pronounced cognition symptoms. To clarify the relationship between small fiber neuropathy and cognition, a Spearman correlation analysis was conducted to evaluate the association between MMSE scores and the 13-item SFN-SIQ scores. The analysis revealed no correlation between the 13-item SFN-SIQ scores and MMSE scores (r = 0.032, p = 0.758, Figure 1(c)).
Relationship between peripheral neuropathy and autonomic dysfunction
PD-PN group demonstrated higher SCOUPA-AUT scores than PD-NPN group (12.40 ± 7.72 vs. 9.12 ± 6.07, p = 0.023). Subsequent subgroup analyses revealed PD-SPN group exhibited higher SCOUPA-AUT scores than the PD-NPN group (14.61 ± 7.86 vs. 9.12 ± 6.07, p = 0.001). The results suggest that PD patients with small fiber neuropathy might experience more pronounced autonomic dysfunction symptoms. To clarify the relationship between small fiber neuropathy and autonomic dysfunction, a Spearman correlation analysis was conducted to evaluate the association between SCOUPA-AUT scores and the 13-item SFN-SIQ scores. The analysis revealed a positive correlation between the 13-item SFN-SIQ scores and SCOUPA-AUT scores (r = 0.761, p < 0.001, Figure 1(d)). Furthermore, multiple linear regression analysis indicated that SCOUPA-AUT scores increased with 13-item SFN-SIQ scores in PD patients (B = 1.118, 95% CI = 0.828–1.409, p < 0.001), after adjusting for age, gender, disease duration, UPDRS-III scores and H&Y stage (Supplemental Table 3).
Relationship between peripheral neuropathy and sleep
No statistically significant differences were observed in PDSS-2 scores between PD-PN and PD-NPN group. Subsequent subgroup analyses revealed PD-SPN group exhibited higher PDSS-2 scores than the PD-NPN group (13 (9–21) vs. 11 (5–16), p = 0.037). The result suggested that PD patients with small fiber neuropathy might experience more pronounced sleep disturbance. To clarify the relationship between small fiber neuropathy and sleep disturbance, a Spearman correlation analysis was conducted to evaluate the association between PDSS-2 scores and the 13-item SFN-SIQ scores. The analysis revealed a positive correlation between the 13-item SFN-SIQ scores and PDSS-2 scores (r = 0.424, p < 0.001, Figure 1(e)). Furthermore, multiple linear regression analysis indicated that PDSS-2 scores increased with 13-item SFN-SIQ scores in PD patients (B = 0.694, 95% CI = 0.309–1.080, p = 0.001), after adjusting for age, gender, disease duration, UPDRS-III scores and H&Y stage (Supplemental Table 4).
Relationship between peripheral neuropathy and life quality
PD-PN group demonstrated higher ADL scores than PD-NPN group (16 (14–22) versus 14 (14–19), p = 0.024). Subsequent subgroup analyses revealed there were no difference between PD-NPN and PD-LPN or PD-NPN and PD-SPN group in ADL scores. Despite the negative findings from the subgroup analysis, the ADL scores for the PD-PN group were higher than those for the PD-NPN group. It is plausible that the limited sample size in the subgroup analysis contributed to a false negative outcome. Consequently, we conducted a correlation analysis between the 13-item SFN-SIQ scores and ADL scores, which revealed a positive correlation between the 13-item SFN-SIQ scores and ADL scores (r = 0.197, p = 0.049, Figure 1(f)). Furthermore, multiple linear regression analysis indicated that ADL scores are not significantly influenced by SFN-SIQ score (B = 0.260, 95% CI = −0.057–0.578, p = 0.107), after adjusting for age, gender, disease duration, UPDRS-III scores and H&Y stage (Supplemental Table 5).
Discussion
Our study investigated the relationship between peripheral neuropathy and non-motor symptoms in PD. The findings suggest that PD patients with peripheral neuropathy, especially small fiber neuropathy, exhibited more pronounced non-motor symptoms. After adjusting for variables such as gender, age, disease duration, and motor symptoms, we observed that a higher 13-item SFN-SIQ score was correlated with heightened symptoms of depression, anxiety, autonomic dysfunction, and sleep disturbances.
Previous studies have shown that PD patients with peripheral neuropathy exhibit heightened motor and non-motor symptoms, consistent with the results of our study.16–18 However, our study concurrently explored the relationship between large fiber neuropathy and non-motor symptoms, as well as the relationship between small fiber neuropathy and non-motor symptoms, thereby serving as a supplement to previous research. Notably, the differences in non-motor symptoms between PD-LPN and PD-NPN were not statistically significant, which stands in contrast to previous research findings. Merola et al. reported that MoCA scores for PD patients with large fiber neuropathy is 26.69 ± 2.73, in contrast to 24.05 ± 4.24 for those without peripheral neuropathy. 16 This statistically significant difference indicates that PD patients with peripheral neuropathy exhibit reduced cognitive function compared to those without peripheral neuropathy. Furthermore, the alterations in sural nerve amplitude was correlated with lower PDQ-39 scores (r = −0.336, p = 0.037) and MMSE scores (r = 0.331, p = 0.042). 19 These findings suggest that large fiber neuropathy may also be associated with more non-motor symptoms. The negative results observed in our study may be attributed to two primary factors. Firstly, the patients in our study had a relatively short disease duration, with a median of 3.75 years, and exhibited relatively mild symptoms, as indicated by a median H&Y stage of 2. Previous research has established a correlation between disease duration, motor symptoms, and peripheral neuropathy in PD patients. 20 Therefore, patients with longer disease durations and more severe motor symptoms may produce positive results. Secondly, the sample size was relatively small, consisting of 30 patients with large fiber in PD-LPN group and 43 patients in PD-NPN group, which could have contributed to the false-negative outcomes.
In cases of diabetic polyneuropathy, the prevalence rates of depression and anxiety have been reported as 46.7% and 60.7%, respectively. 21 A recent large-scale clinical investigation into mood disorders among patients with small fiber neuropathy revealed that 36.3% of these individuals may exhibit symptoms of anxiety and depression. Furthermore, the study established a positive correlation between the severity of small fiber neuropathy symptoms and the scores for anxiety and depression. 22 Previous research has identified a correlation between pain and depression in individuals with PD. Given that pain is a common symptom of small fiber neuropathy, it is plausible to hypothesize that small fiber neuropathy in PD may influence symptoms of depression and anxiety. 23 This aligns with findings from our research. The locus coeruleus plays a crucial role in the transmission of sensory information, including nociceptive signals, and is a key component of the central stress response network. Consequently, it may serve as a critical hub for modulating sensory processing and stress responses. 24
Patients diagnosed with hereditary transthyretin amyloidosis exhibited both autonomic dysfunction and small fiber neuropathy, with a moderate correlation identified between these two conditions. 25 This finding aligns with our observations in PD patients, highlighting a significant association between autonomic dysfunction and small fiber neuropathy. Peripheral small fibers, which include αδ fibers and unmyelinated C fibers, are responsible for providing sensory and autonomic innervation to the skin and viscera. Small fiber neuropathy can result in both autonomic and sensory dysfunctions. Autonomic dysfunctions symptoms may encompass disturbances in thermoregulation, sexual dysfunction, orthostatic hypotension, and gastrointestinal dysmotility, among others. 26 Therefore, it is not surprising that PD patients with small fiber neuropathy exhibit elevated levels of autonomic dysfunction symptoms.
Patients diagnosed with small peripheral neuropathy demonstrate an increased likelihood of developing sleep disorders. Moreover, reduced sleep quality appears to exacerbate the symptoms associated with small fiber neuropathy.27–30 Small fiber neuropathy often co-occurs with chronic pain, which would further compromise overall sleep quality. 31 Experiences of pain can disrupt patients’ ability to maintain stable nocturnal sleep, subsequently impairing daytime attention, and perpetuating a detrimental cycle. 32 As previously mentioned, small fiber neuropathy can also result in dysregulation of the autonomic nervous system. Hyperactivity of the sympathetic nervous system may induce anxiety, potentially exacerbating insomnia, while hypoactivity of the parasympathetic nervous system impairs the body's capacity to relax, thus affecting sleep initiation. 33 Collectively, these findings support the hypothesis of a bidirectional relationship between small fiber neuropathy and sleep disturbances.
PD patients with peripheral neuropathy exhibited higher ADL scores than those without peripheral neuropathy. Previous research has demonstrated that PD patients with peripheral neuropathy tend to experience a reduced quality of life. Moreover, an inverse correlation exists between the alterations of the sural nerve amplitude and the PDQ-39 score. 19 Non-motor symptoms of PD, such as cognitive impairment, autonomic dysfunction, mood disorders, and sleep disturbances, are critical factors influencing patients’ quality of life. 34 Small fiber neuropathy may independently or synergistically exacerbate the quality of life, as it is frequently concomitant with more pronounced no-motor symptoms in PD patients.
This study is subject to several limitations. Firstly, the single-center design and relatively small sample size warrant caution when generalizing the findings to broader populations. Specifically, during subgroup analysis, there were 30 cases in the PD-LPN group and 43 cases in the PD-NPN group. No significant differences were observed in non-motor symptoms between these groups, suggesting the potential for false negatives due to the limited sample size. This underscores the need for further research involving larger cohorts. Secondly, the diagnosis of small fiber neuropathy relies on questionnaire surveys and electrophysiological examinations, without additional validation through skin biopsy. Thirdly, although no restrictions were placed on disease severity during patient recruitment, patients in H&Y stages 4 and 5, were less inclined to participate. Consequently, the study predominantly included patients in H&Y stages 1–3, only eight in stage 4, and none in stage 5. Therefore, the applicability of the study's conclusions to patients in H&Y stages 4 and 5 requires further validation. Furthermore, the cross-sectional nature of the analysis limits the ability to establish causal relationships between small fiber neuropathy and non-motor symptoms. Consequently, further multi-center longitudinal studies are required to substantiate these findings.
Conclusion
In conclusion, this study investigated the relationship between peripheral neuropathy and non-motor symptoms in individuals with PD. The results demonstrate that patients with small fiber neuropathy exhibit more pronounced non-motor symptoms. Moreover, the severity of small fiber neuropathy is correlated with the extent of depression, anxiety, autonomic dysfunction, sleep disturbances. These results underscore the necessity for routine screening and early detection of peripheral neuropathy, particularly small fiber neuropathy, in PD patients.
Supplemental Material
sj-docx-1-pkn-10.1177_1877718X251371538 - Supplemental material for Patients with small fiber neuropathy exhibit more pronounced non-motor symptoms in Parkinson's disease
Supplemental material, sj-docx-1-pkn-10.1177_1877718X251371538 for Patients with small fiber neuropathy exhibit more pronounced non-motor symptoms in Parkinson's disease by Chunge Xie, Jieling Chen, Rong Gan, Yuyuan Gao, Limin Wang, Guixian Ma, Yuhu Zhang, Kun Nie and Lijuan Wang in Journal of Parkinson's Disease
Footnotes
Acknowledgements
We would like to thank all the participants.
ORCID iDs
Ethical considerations
The study was approved by the Institutional Ethical Committee of Guangdong Provincial People's Hospital (KY-Q-2021-105-01) before the study initiation.
Consent to participate
Informed written consent was secured from all participants following a comprehensive explanation of the study procedures.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the National Natural Science Foundation of China (No. 82471257, 82371249, 82101377, 82071419), the Science and Technology Planning Project of Guangzhou (No.202201000005).
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
Data that support the results of this study are available on request from the corresponding authors.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
