Abstract
Background:
Assessment of affective-behavioral states in patients with Parkinson’s disease (PD) undergoing deep brain stimulation (DBS) is essential.
Objective:
To analyze well-established questionnaires as a pilot-study with the long term aim to develop a screening tool evaluating affective-behavioral dysfunction, including depression, anxiety, apathy, mania, and impulse control disorders, in PD patients screened for DBS.
Methods:
Two hundred ninety-seven inpatients with PD underwent standardized neuropsychiatric testing including German versions of Beck Depression Inventory-II, Hospital Anxiety and Depression Scale, Apathy Evaluation Scale, Self-Report Manic Inventory, and Questionnaire for Impulsive-Compulsive Disorders in PD-Rating Scale, to assess appropriateness for DBS. Statistical item reduction was based on exploratory factor analysis, Cronbach’s alpha, item-total correlations, item difficulty, and inter-item correlations. Confirmatory factor analysis was conducted to assess factorial validity. An expert rating was performed to identify clinically relevant items in the context of PD and DBS, to maintain content validity. We compared the shortened subscales with the original questionnaires using correlations. To determine cutoff points, receiver operating characteristics analysis was performed.
Results:
The items of the initial questionnaires were reduced from 129 to 38 items. Results of confirmatory factor analyses supported the validity of the shortened pool. It demonstrated high internal consistency (Cronbach’s alpha = 0.72–0.83 across subscales), and the individual subscales were correlated with the corresponding original scales (r s = 0.84–0.95). Sensitivities and specificities exceeded 0.7.
Conclusion:
The shortened item pool, including 38 items, provides a good basis for the development of a screening tool, capturing affective-behavioral symptoms in PD patients before DBS implantation. Confirmation of the validity of such a screening tool in an independent sample of PD patients is warranted.
Keywords
INTRODUCTION
Deep brain stimulation (DBS) of the subthalamic nucleus (STN) is a well-established treatment to improve motor symptoms, activities of daily living, and quality of life in patients with Parkinson’s disease (PD) carefully selected for this procedure [1]. Multiple factors need to be considered in deciding whether a patient with PD is a suitable candidate for DBS. Clinically relevant psychiatric disorders such as depression, anxiety, apathy, mania, impulse control disorders (ICD), and hallucinations may have a large impact on the overall outcome of DBS and may constitute a relative contraindication for DBS [2, 3]. At the same time, recent studies suggest that DBS may improve psychiatric disorders in PD, especially in the context of ICD and anxiety [4–6]. For this reason, preoperative assessment of affective-behavioral dysfunctions is essential to determine an individual’s psychiatric benefit-risk profile, and for evaluating both, contraindication and indication, for DBS surgery.
Indication testing for DBS at the University Hos-pital Cologne is based on multidisciplinary assessments by movement disorders specialists, stereotactic neurosurgeons, neuropsychologists, and psychiatrists. The neuropsychiatric assessment of affective-behavioral symptoms includes five questionnaires (Beck Depression Inventory-II [7, 8], Hospital Anxiety and Depression Scale [9, 10], Apathy Evaluation Scale [11, 12], Self-Report Manic Inventory [13, 14], and Questionnaire for Impulsive-Compulsive Disorders in PD-Rating Scale [15]) which have proven their usefulness in the clinical context over the years. Using the five separate scales for all relevant psychiatric symptoms of concerns was found to be time-consuming, sometimes exhausting for the patient then resulting in incomplete answering. Furthermore, general measures may not address areas salient to specific diseases and may contain items that are not suitable for PD, leading to incorrect results.
There are different scales to assess non-motor symptoms in PD, like the Non-Motor Symptom Scale (NMSS) [16], the Movement Disorder Society-Unified Parkinson’s Disease Rating Scale (MDS-UPDRS) part one [17], the Movement Disorder Society-Non-Motor Rating Scale (MDS-NMS) [18, 19], and the Non-Motor Symptom Questionnaire (NMSQ) [20]. However, the NMSS, MDS-UPDRS part one, and MDS-NMS are rater-administered versions and not for self-rating purposes. Only the NMSQ is a patient-based questionnaire, but it does not cover mania and ICD and each symptom is covered by a single dichotomous question.
Therefore, this pilot study aimed to analyze well established questionnaires in the context of PD with the long term aim to develop a non-commer-cial self-rating screening tool assessing the broad affective-behavioral state including depression, anxiety, apathy, mania, and ICD in PD patients under-going DBS, to reduce the burden of the patients and increase the practicability and informative value of the assessment.
METHODS
Participants and ethical approval
Three hundred thirty-nine in-patients with PD who underwent evaluation testing for DBS implantation at the University Hospital of Cologne from March 2014 to April 2019 were retrospectively analyzed. Patients records were examined for age, sex, disease duration, PD motor-subtype, levodopa equivalent daily dose (LEDD) [21], Hoehn and Yahr stage [22] and Unified Parkinson’s Disease Rating Scale-III (UPDRS-III) in Medication-ON and -OFF [23]. The retrospective pilot study was carried out following the Declaration of Helsinki and approved by the local ethics committee (University of Cologne, Germany).
Measures
The neuropsychiatric examination was carried out using the following scales:
The
The
Apathy was evaluated using the self-report version of the
The
The
Statistical item reduction
The item reduction was conducted with IBM SPSS version 25, separately for each questionnaire. Kaiser-Meyer-Olkin Test was used to analyze how suitable the data is for factor analysis, with values≥0.6 indicating adequate sampling [30]. An exploratory factor analysis, informed by eigenvalues and scree plots, was used to determine the number of factors that best represent the data. Factor structures in the given sample were compared with the ones described in the literature. Exclusion criteria were defined as follows: a factor loading of > 0.5 was used to determine those items to be retained in a factor; items with lower factor loadings (< 0.5) were excluded [31]. Furthermore, items reducing internal consistency (indicated by Cronbach’s alpha) [32] were excluded. An item-total correlation < 0.4 [30] and itemdifficulty
Factorial validity
To assess factorial validity, confirmatory factor analyses (CFA) were conducted for the reduced item pool’s subscales and the overall model. We implemented robust maximum-likelihood estimation methods and goodness-of-fit statistics because distributions of some model variables deviated from normality [34]. For each analysis, models were identified by setting the variances of each factor to 1.0. Model fit was evaluated with the Satorra-Bentler robust chi-square statistic, while chi-square/ degrees of freedom ratio of 3 or less was taken as an indicator of acceptable model fit [35]. Based on the recommendations of Hu and Bentler we used additional fit indices, including the standardized root mean square residual (SRMR), the root mean square error of approximation (RMSEA), the comparative fit index (CFI), and the Tucker-Lewis index (TLI). Values of CFI greater than 0.95 [36] and TLI greater than 0.90 are indicative of well-fitting models [37], while SRMR values below 0.08 and RMSEA values ≤ 0.06 (N > 250)/ ≤ 0.08 (N < 250) indicate a reasonable error [36].
Expert rating
As recommended in previous research, we combined the statistically driven strategy with a judgmental approach [38, 39]. This process is usually based on the Delphi method [40]. Here, a modified Delphi method was used, i.e., we presented the results of the statistical test shortening process to three renowned experts of psychiatric comorbidities in PD (KW, PK, and DW). The experts had to decide independently whether they agree or disagree with each excluded and included item in the clinical context of indication testing for DBS. Therefore, we presented the experts a table with the statistical results. They had to check every single item for clinical relevance. They also had the opportunity to comment each decision. The first-round results were presented anonymously to all three experts in a second round, in which they had the opportunity to reconsider their decision based on the other experts’ decision and comments. Afterwards, they were able to adjust their decision about the item selection. Based on the results of the second round, it was decided whether a further round was needed, since Delphi procedures include at least two rounds. Criteria for this decision were the achievement of consensus or the occurrence of ‘diminishing returns’ [41].
Internal consistency and comparison of the long and short form
As a measure of the reduced item pool’s reliability (internal consistency), we calculated Cronbach’s alpha for each subscale [32]. A value of≥0.7 was considered acceptable, ≥0.8 good, and≥0.9 excellent [42]. To assess the relationship between the initial questionnaire and the reduced item pool, we computed Spearman correlations between the original questionnaires’ total scores and the subscale scores of the shortened pool. The degree of association of≥0.7 was considered strong [43].
Correlation between the short form subscales
To investigate potential overlaps between the different subscales, we computed Spearman correlations between the five subscales of the reduced item pool (depression, anxiety, apathy, mania, and ICD). The degree of association of < 0.4 was considered weak, 0.4 –0.6 moderate, and > 0.6 strong [43, 44].
Cutoff points
Receiver operating characteristics (ROC) analysis was performed to determine cutoff points for each shortened subscale of the reduced item pool. The area under the curve (AUC), as well as sensitivity and specificity, were calculated. Final cutoffs were defined using the Youden index [J = Sensitivity +Specificity –1] [45]. As a criterion for positive test results in the initial questionnaires, the following cutoffs were used: BDI-II≥14 [7], HADS-D≥8 [9], HADS-A≥8 [9], AES≥16 [11], SRMI≥14 [27], QUIP-ICD≥10, QUIP-Hobbyism≥4, QUIP-Punding≥3, QUIP-DDS≥3 [15, 29].
RESULTS
Participants
Patient records of 339 PD patients with neuropsychiatric examination were retrospectively screened. Forty-two patients were excluded because they were diagnosed with atypical PD. The final study sample consisted of 297 patients (200 male, 97 female) with a mean age of 65.2 years (standard deviation±9.4) at the time of indication testing with 9.4 years (±5.5) disease duration, 906.1 mg (±495.2) dopaminergic medication requirement (LEDD), and a median Hoehn and Yahr score of 3.0 (interquartile range: 2.0-3.0). All PD types were represented, with the majority of patients having akinetic-rigid PD (n = 123), followed by mixed PD (n = 103) and tremor-dominant PD (n = 71). Mean UPDRS-III-ON was 23.49 (±11.95) and UPDRS-III-OFF 37.4 (±13.86).
Statistical item reduction
Descriptive statistics of the five questionnaires are reported in Table 1. According to the Kaiser-Meyer-Olkin test, the sample was adequate for factor analysis (all≥0.6). Exploratory factor analyses reproduced the factor structures of the five questionnaires (two-factor-structures for depression, apathy, mania, and ICD, one-factor structure for anxiety). Of 129, 99 items were excluded, resulting in a reduced item pool containing 30 items, including 7 items for depression, 5 items for anxiety, 6 items for apathy, 7 items for mania, and 5 items for ICD. Exploratory factor analyses (EFA) of the shortened subscales reproduced the long versions’ factor structures, except for the depression and ICD subscales. Depression and ICD subscales resulted in one factor, as most items of the second factor were excluded in the item reduction process. The items of the statistically evaluated reduced item selection and the excluded items, including mean, standard deviation, item-total correlation, and item difficulty, can be found in the Supplementary Material (Supplementary Table 1). Results of the exploratory factor analyses are displayed in the Supplementary Table 2.
Descriptive statistics of the five questionnaires in the sample
Descriptive statistics of the five questionnaires (BDI-II [8], HADS [10], AES [12], SRMI [14], QUIP-RS [15]) in the sample. AES, Apathy Evaluation Scale; BDI-II, Beck Depression Inventory-II; DDS, Dopamine dysregulation syndrome; HADS-D/-A, Hospital Anxiety and Depression Scale-Depression/-Anxiety; ICD, Impulse control disorders; QUIP-RS, Questionnaire for Impulsive-Compulsive Disorders in Parkinson’s Disease-Rating Scale; SD, standard deviation; SRMI, Self-Report Manic Inventory;
Factorial validity
Structural equation modeling was used to model relationships between directly observed variables (items of the reduced item pool) and indirectly observed latent variables (factors). Loadings on each factor were defined by scale memberships of the items and the preceding exploratory factor analyses. The resulting seven-factor model, including standardized factor loadings, is shown in Fig. 1. Depression, anxiety, and ICD each load for one factor, and mania and apathy each load for two factors. Confirmatory factor analyses for the subscales and the overall model yielded adequate-to-excellent fit across all fit indices (Table 2).
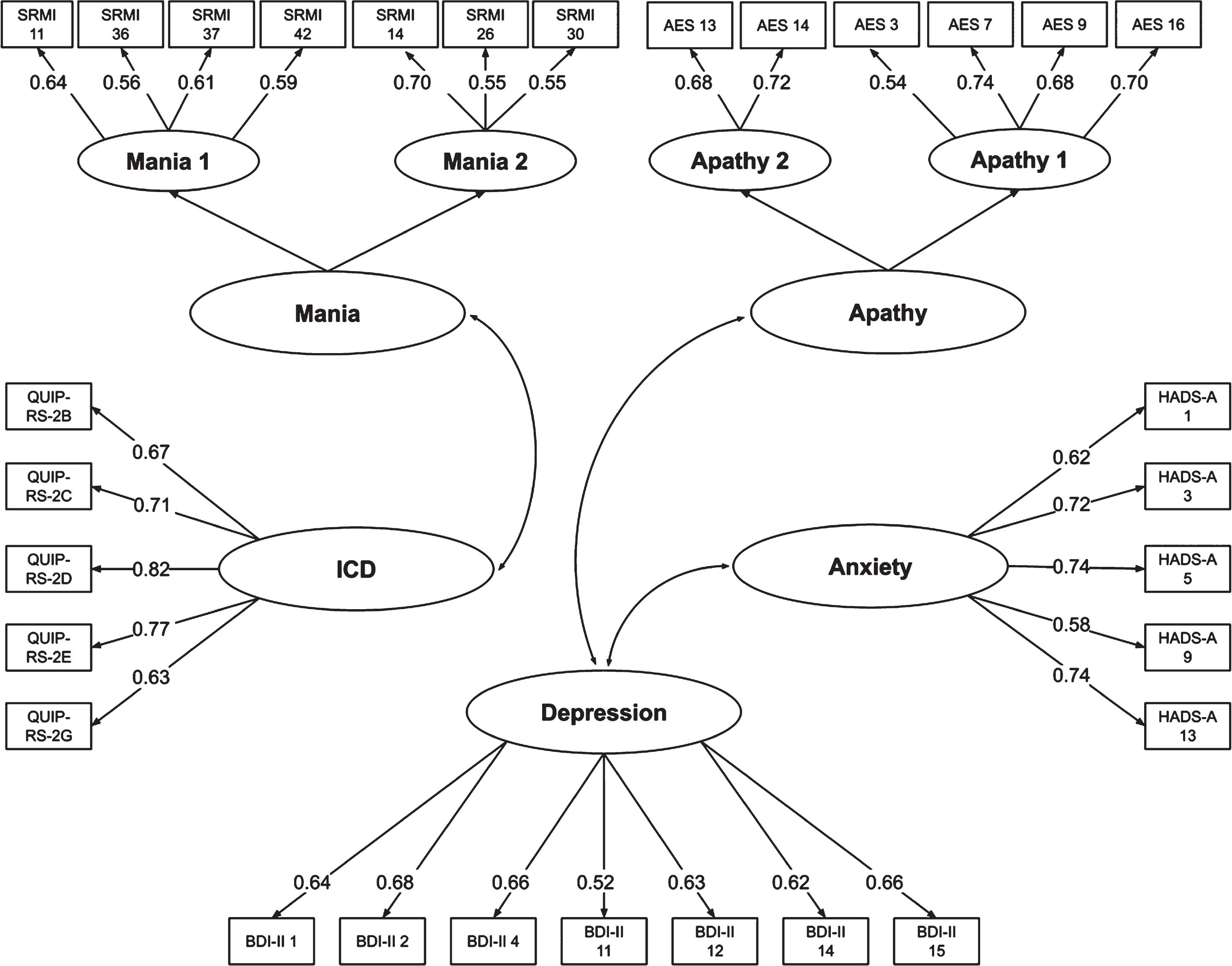
Structural equation model of the reduced item pool after the statistical item reduction. Structural equation model of the 30-item selection after the statistical item reduction showing standardized coefficients for the model fitted to the total sample. All coefficients are statistically significant (p < 0.001). Observed variables are displayed in rectangles (number of the items of the original questionnaires), the names of latent variables in ovals. Straight lines with arrow-heads on one end are paths that link a predicting latent and a predicted observed variable. Curves with arrow-heads on both sides stand for correlations between latent variables. The items are from the questionnaires BDI-II [8], HADS [10], AES [12], SRMI [14] and QUIP-RS [15]. AES, Apathy Evaluation Scale; BDI-II, Beck Depression Inventory-II; HADS-A, Hospital Anxiety and Depression Scale-Anxiety; ICD, Impulse control disorders; QUIP-RS, Questionnaire for Impulsive-Compulsive Disorders in Parkinson’s Disease-Rating Scale; SRMI, Self-Report Manic Inventory.
Fit Indices for tested models
Results of the confirmatory factor analysis of each individual subscale and the complete model with all categories. CFI, Comparative fit index (≥0.95); df, degrees of freedom; ICD, Impulse control disorders; p-value (>0.1); RMSEA, Root mean square error of approximation (≤0.06 (N > 250)/≤0.08 (N < 250); SRMR, Standardized root mean square residual (≤0.08); TLI, Tucker-Lewis index (≥0.9); χ2, Chi-square; χ2 / df (< 3).
Expert rating
The expert rating included two-rounds and resulted in a final item pool with 38 items, excluding three items and adding eleven items to the statistically-generated reduced item selection. After the second round, the exclusion of all three items was decided consensually by the experts. Out of the eleven included items, eight items were included consensually, three items were included based on majority opinion (SRMI 10, 16, and 21). We decided not to perform a third round, as this would not have added further results, since neither of the experts changed their mind within the two rounds regarding the three items. The criterion of the occurrence of ‘diminishing returns’ was fulfilled. We presented the final results of the two rounds to the experts, with which all three experts agreed. Results of the expert rating are displayed in Fig. 2. Notably, the experts unanimously suggested to include an item about visual hallucinations, since it is a common and highly relevant psychiatric symptom in PD [46]. In fact, no item from the original questionnaires addresses hallucinations in general, or visual hallucinations specifically. Therefore, the experts decided to include a suitable item about hallucinations and psychosis based on the UPDRS (part one, item 2): “Have you seen or heard things that were not really there?” [23]. The item was added to the reduced item pool in a separate category. Corresponding characteristic values of the final item selection (without the additional hallucination item) are displayed in Table 3. The final item pool includes eight items for depression, five items for anxiety, eight items for apathy, ten items for mania, six items for ICD and related disorders, and one item for hallucination. This reduced item pool as a potential basis for a new screening tool was developed as a scientific project, not for commercial purposes.
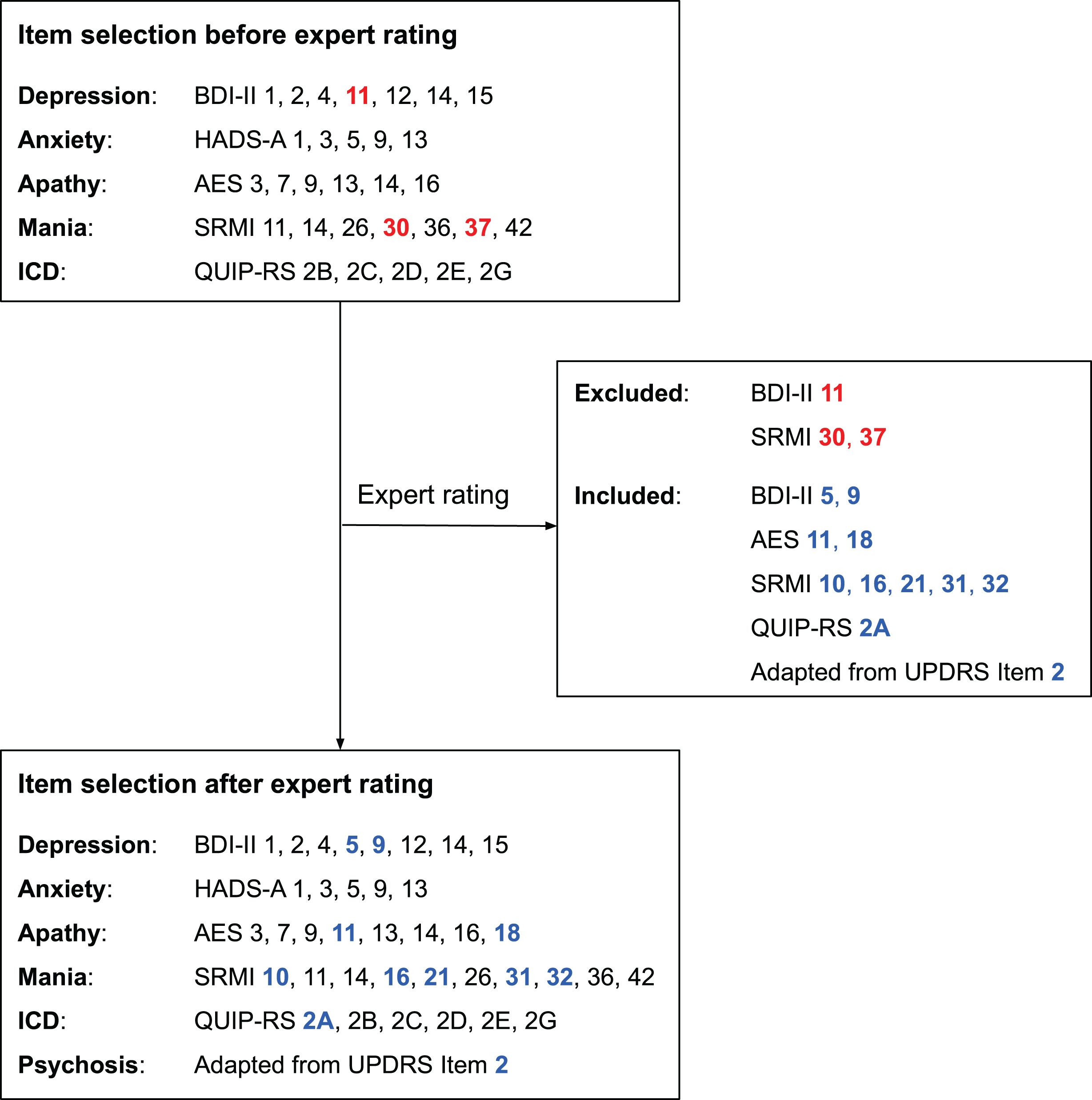
Flowchart with the item selection before and after the expert rating. Excluded items during the expert rating are highlighted in red and included items in blue (both boldface). The items are from the questionnaires BDI-II [8], HADS [10], AES [12], SRMI [14], QUIP-RS [15] and UPDRS [23]. AES, Apathy Evaluation Scale; BDI-II, Beck Depression Inventory-II; HADS-A, Hospital Anxiety and Depression Scale-Anxiety; ICD, Impulse control disorders; QUIP-RS, Questionnaire for Impulsive-Compulsive Disorders in Parkinson’s Disease-Rating Scale; SRMI, Self-Report Manic Inventory; UPDRS, Unified Parkinson’s Disease Rating Scale.
Final item selection with corresponding characteristic values
In the brackets behind the items, the number of the items in the original questionnaires (Beck Depression Inventory-II [8], Hospital Anxiety and Depression Scale-Anxiety [10], Apathy Evaluation Scale [12], Self-Report Manic Inventory [14], Questionnaire for Impulsive-Compulsive Disorders in Parkinson’s Disease- Rating Scale [15]) are displayed. ITC, item-total correlation; P, item difficulty; PD, Parkinson’s disease; SD, standard deviation;
Internal consistency and comparison of the original questionnaires and the shortened subscales
All subscales of the short version demonstrated acceptable-to-good internal consistency, with Cronbach’s alpha coefficients of 0.83 for the depression subscale; 0.80 for anxiety; 0.79 for apathy; 0.72 for mania, and 0.79 for ICD. Summary scores of the original questionnaires were highly correlated with the corresponding shortened subscales (depression, r s = 0.87; anxiety, r s = 0.90; apathy, r s = 0.95; mania, r s = 0.85; and ICD, r s = 0.84, all p < 0.001).
Correlation between the short form subscales
The correlation between the short version’s subscales were as follows: Correlation of depression with anxiety, r s = 0.57; depression with apathy, r s =0.42; depression with mania, r s = 0.30; depression with ICD, r s = 0.23; anxiety with apathy, r s = 0.29; anxiety with mania, r s = 0.37; anxiety with ICD, r s = 0.29; apathy with mania, r s = 0.23; apathy with ICD, r s = 0.24; and mania with ICD, r s = 0.30 (all p < 0.001).
Cutoff points
Optimal cutoffs for the reduced item pool’s subscales are: ≥4 for depression,≥5 for anxiety,≥8 for apathy,≥3 for mania,≥3 for ICD,≥1 for hobbyism, and≥1 for DDS. The cutoffs with corresponding sensitivities, specificities, and areas under the curve (AUC) are displayed in Table 4, ROC-curves are displayed in Fig. 3.
Cutoffs with corresponding sensitivities and specificities
*p < 0.001; **defined by Youden-index [J = Sensitivity + Specificity –1]. AUC, Area under the curve; DDS, Dopamine dysregulation syndrome; ICD, Impulse control disorders.
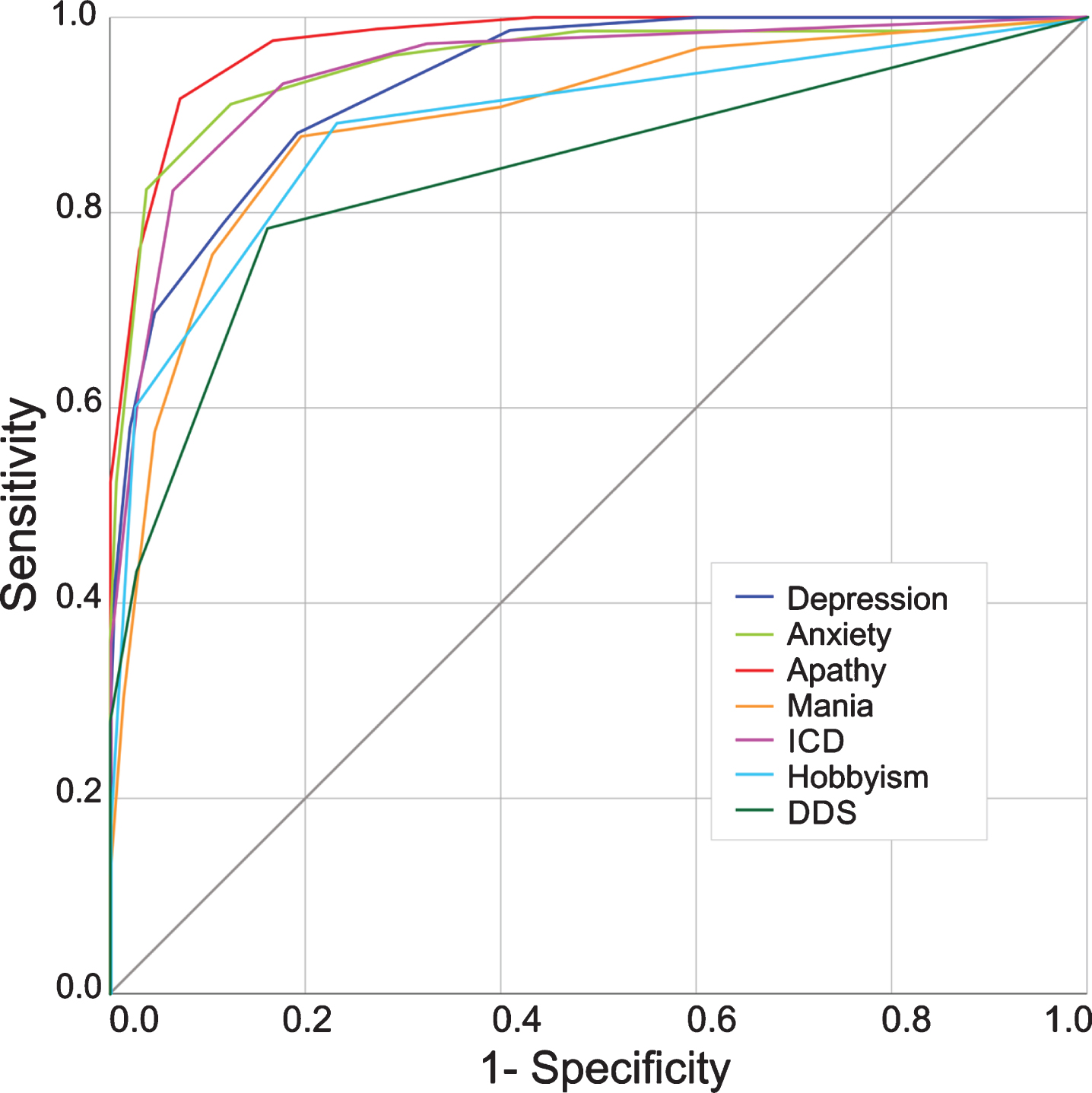
Receiver operating characteristic curves. Receiver operating characteristic curves are displayed for the short forms of depression, anxiety, apathy, mania, ICD (including gambling, sex, eating and buying), hobbyism, and DDS. All areas under the curve are statistically significant (p < 0.001). DDS, Dopamine dysregulation syndrome; ICD, Impulse control disorders.
DISCUSSION
In the present pilot study, we performed an item reduction of existing questionnaires with the long term aim to develop a screening tool to assess PD patients’ affective-behavioral states before DBS. The initial item pool was reduced from 129 to 38 items. Results of confirmatory factor analyses supported the validity of the shortened item pool. An expert rating subsequent to the statistical procedure confirmed the clinical relevance of the reduced item pool.
The need for adequate affective-behavioral assessment is essential. Clinically relevant psychiatric disorders may constitute relative contraindication for DBS and may have a large impact on DBS’s overall outcome [2, 3]. Further, more recent studies suggest that DBS may also improve psychiatric symptoms in PD, especially in the context of ICD and anxiety [4, 5]. Therefore, the identification of affective-behavioral dysfunctions pre-DBS surgery is mandatory, not only for evaluating contraindications for DBS but also for indication. A careful assessment is essential to determine a patient’s individual benefit-risk for DBS. Covering all aspects of affective-behavioral changes with the original separate questionnaires is burdensome, time-consuming, and might not be feasible at all DBS centers.
The major advantages of a novel screening tool would include reduced patient burden, increased practicability and meaningfulness in research and clinical practice, and minimization of administration time. Since 70.5%of the items were excluded in the reduced item pool, a potential screening tool with comparable number of items could be completed much faster than the full battery of questionnaires, while information loss due to incomplete answering would be minimized. Compared to other screening instruments of non-motor symptoms in PD, the reduced item pool covers depression, anxiety, apathy, mania, ICD and hallucinations. Especially in the context of DBS it is important to assess those symptoms carefully. The potential screening tool’s benefit is to enable the psychiatrist to focus on important clinical symptoms without missing relevantinformation.
The reduced item pool was developed by removal of items from well-established, international self-assessment scales, measuring depression (BDI-II [8], HADS-D [10]), anxiety (HADS-A [10]), apathy (AES [12]), mania (SRMI [14]), and ICD (QUIP-RS [15]) in PD patients. To develop the reduced item pool we applied a two-step procedure, with the first step including a statistical item reduction based on classical test theory [30]. In the second step, we aimed to increase content validity of the reduced item pool by including an expert rating, which is a common and recommended method in the context of questionnaire development [38, 47]. This is advantageous, as several items of the original questionnaires were not applicable in PD. For example, restlessness (HADS-A item 11) [10] may not be indicative of anxiety in PD, and insomnia (BDI-II item 16) [8] can occur for several reasons irrespective of the presence of depression in PD patients. Including those items may result in misclassification [48]. Furthermore, by following a statistical approach only, items like, e.g., BDI-II item 9 “suicidal thoughts or wishes” [8] would have been excluded through statistical exclusion criteria, since selectivity and item difficulty were low. However, a careful preoperative assessment of suicidal tendencies is still essential, even if it rarely occurs, as suicidal tendencies might increase after DBS [49] and therefore constitute contraindication. Moreover, the QUIP-RS item about pathological gambling [15] would have been excluded, as only 2.7%reported to suffer from pathological gambling in our sample (item difficulty > 90%). Also, selectivity of the items was low (item-total correlations < 0.4), suggesting that pathological gambling might not correlate highly with the presence of other symptoms of ICD. Therefore, gambling might be interpreted as an independent symptom of ICD. This finding is consistent with DSM-V changes, as the gambling diagnosis was reclassified from ICD to substance-related and addictive disorders [50]. This modification was based on previous studies, which observed correlations between pathological gambling and substance-related disorders [51]. However, although pathological gambling in PD does not occur very often, consequences can be tremendous [52]. This underlines the judgmental approach’s importance, as a solely statistically procedure may miss rare but highly relevant aspects of a careful clinical assessment.
During the expert rating, there was an extensive discussion on the item “I heard voices when people weren’t there” (SRMI 46) [14]. The experts consensually decided that it is important to include one item about psychotic symptoms in the reduced item pool, as it is a common phenomenon in advanced PD. However, the item from the SRMI does not suit PD patients, as these mostly suffer from visual, but not auditory hallucinations [46]. The original questionnaires used to develop the reduced item pool do not include an item about visual hallucinations. The experts suggested to include one question about hallucinations and psychosis based on the original UPDRS part one, item two (“Have you seen or heard things that were not really there?”) [23]. This item was added to the reduced item pool in a separate category, as we were interested in a comprehensive assessment of the affective-behavioral state in PD patients.
Considering depression, only items from the BDI-II were included. Items from HADS-D were excluded as they were very similar to items of the BDI-II but had lower selectivity. This is consistent with a study by Schrag et al., who compared depression rating scales in PD and recommended the BDI-II for screening purposes [24].
The correlations between the reduced item pool subscales revealed moderate correlations between depression and anxiety (r s = 0.57), and depression and apathy (r s = 0.42). This is reasonable, as symptoms of depression overlap with symptoms related to anxiety and apathy [53]. Degrees of association between the other subscales were weak, which indicates minimal overlap between those subscales.
The scoring of the original questionnaires was kept within the reduced item pool, to enable the calculation of cutoff points. In a potential future screening tool this should be more unified. Further, the time frame to which patients shall refer their symptoms to while answering the questions is different between the original questionnaires. For a potential future screening tool we would suggest four weeks, although BDI-II originally covers two weeks [8], and HADS covers one week [10]. AES, SRMI and QUIP-RS cover four weeks [12, 15], and since the potential future screening tool favors a high sensitivity, a longer period of time seemed more appropriate.
The reduced item pool’s subscales exhibit high correlations with the corresponding original questionnaires (all r > 0.8). Also, although the number of items was reduced substantially, there was no fundamental loss of internal consistency for subscale scores. The complete reduced item pool’s internal consistency was found to be good (Cronbach’s alpha = 0.88). This suggests that the reduced item pool is a promising foundation for an alternative to the full battery of questionnaires to assess affective-behavioral symptoms in PD for screening purposes.
Limitations
A few limitations apply to this study. There are several other self-rating scales to assess affective-behavioral symptoms than the ones’ we used to develop the reduced item pool for the future screening tool. However, the short version was developed retrospectively, the BDI-II [8], HADS [10], AES [12], SRMI [14], and QUIP-RS [15] were chosen by an interdisciplinary team at the University Hospital Cologne, to assess affective-behavioral symptoms in PD patients in 2014. Clinical experience supported that those questionnaires cover relevant affective-behavioral symptoms in PD undergoing DBS. Therefore, we decided to choose existing data of those questionnaires for the development of the reduced item pool. Responses to the short form’s items may deviate from answers retrieved for these items in the original long form, as responses to these questions might be impacted by the other questions (e.g., sequence effects). However, the item reduction approach is common practice, as it is not functional to include the original and the shortened pool in the same assessment [54]. The reduced item pool was developed in the context of indication testing for DBS in PD patients. At this point of time, the patients had an advanced disease stage (median Hoehn and Yahr score of 3.0; interquartile range: 2.0-3.0) with a mean disease duration of 9.4 (±5.5) years. Thus, the given results cannot be readily transferred to a more general PD cohort without further validation. Moreover, although the present study relates to the time point of indication testing, it is reasonable that the future screening tool might be advantageous for follow-up examinations, too, as the original questionnaires are used at all time points, and the correlation of the reduced item pool with the initial questionnaires was high (all r > 0.8).
The statistical results of the analysis of the reduced item pool suggest that such a screening tool is a reliable alternative to assess affective-behavioral states in PD before DBS. Construct validity explored by the CFA revealed good to excellent model fit, indicating that the subscales represent the allocated states. By means of increasing content validity, those results were analyzed by movement disorders specialists in a second step, to improve the informative value of the reduced item pool in the clinical context. Although, future studies are needed to validate such a reduced item pool as a screening tool against an external criterion, such as DSM diagnoses of behavioral syndromes in a separate sample and to define its floor- and ceiling-effects, its variation, sensitivity to change and test-retest reliability. Further validation should also analyze such a screening tool in relation to quality of life, since this is a highly relevant predictor for DBS outcome in PD [55].
As it will only be a screening tool, it cannot replace a more detailed neuropsychiatric assessment before DBS, if one or more of the subscales have a positive test result. Naturally, it cannot replace a comprehensive psychiatric in-person interview, which is also required when assessing DBS eligibility and which should occur in parallel to psychometric neuropsychiatric assessments. The potential screening tool’s benefit will be to enable the physician to focus on certain symptoms in an interview. Ensuing investigations must evaluate for false positives, as these may lead to incorrect rejection from DBS and increase potential stigma. Moreover, non-motor fluctuations should be considered, as these often occur in PD patients with motor fluctuations [56]. Therefore, non-motor fluctuations should be addressed in the psychiatric interview, as these may also lead to misclassification.
For the test evaluation, each subscale of such a screening tool (depression, anxiety, apathy, mania, ICD, hobbyism, DDS, and hallucinations) would have to be analyzed separately with an individual cutoff. There is no total score, as each psychiatric dysfunction may occur separately or in combination. One total score, including all categories, could lead to misinterpretation by not being able to differentiate between a high score in one category or combined low scores in several categories. We used the cutoffs described in the literature for the original questionnaires to define if the patient has a positive or negative test value. As for some of the original questionnaires, different cutoffs had been recommended. By choosing the cutoff for the original questionnaire, we focused on recommendations in the context of PD as well as the most frequently recommended cutoffs. As the new cutoffs were chosen based on this criterion, it supposes that the original questionnaires’ classification of positive and negative test results is correct. Future studies should include psychiatric expert ratings as an external criterion to confirm the cutoffs. As we had no data about the hallucination item, sensitivity and specificity for this modified single dichotomous item could not be established. As in the other categories, if there is a positive result (in this case, the answer is “yes”), further information is needed to verify if this should be considered an exclusion criterion for DBS.
CONCLUSION
The reduced item pool is an efficient alternative to assess the affective-behavioral state in PD patients in the context of DBS. The present results suggest that the reduced item pool is a reliable base for a future screening tool that improves the original questionnaires’ diagnostic utility in the context of PD and DBS. Further validation of such screening tool in a different sample and against an external criterion is needed.
