Abstract
Background
The association between body mass index (BMI), metabolic conditions, and incident Parkinson's disease (PD) is quite complex.
Objective
To investigate the relationship between these variables, particularly the impact of metabolically healthy overweight/obese on the risk of PD, in the general population.
Methods
A total of 402,059 participants from the UK Biobank were categorized into four phenotypes according to the presence of overweight/obesity and/or metabolically abnormal status: overweight/obesity was defined as BMI ≥25 kg/m2; metabolically abnormal status was defined as having one or more metabolic risk factors including elevated blood pressure, fasting glucose, or triglyceride level, or reduced high-density lipoprotein cholesterol level. Cox proportional hazard regression analyses using four different models were performed to compare the risk of developing PD among the four BMI-metabolic status phenotypes.
Results
During the median follow-up of 13.5 years, 2283 (0.6%) patients were newly diagnosed with PD. Cox regression models demonstrated that individuals with overweight/obesity and those with metabolic abnormalities were at a higher risk of developing PD than their counterparts. Compared with the metabolically healthy non-overweight group (reference group), the two metabolically abnormal groups (either overweight/obese or non-overweight) showed a higher incidence of PD. The metabolically healthy overweight/obese group exhibited a comparable risk of developing PD to the metabolically healthy non-overweight group.
Conclusions
This study demonstrated that metabolically abnormal conditions are more relevant to incident PD than overweight/obesity. In particular, a metabolically healthy overweight/obese status does not increase the risk of developing PD compared with a metabolically healthy non-overweight status.
Plain Language Summary
Researchers have long studied whether body weight and metabolic health affect the risk of developing Parkinson's disease, but results have been mixed. In particular, it was unclear whether people who are overweight but have good metabolic health—known as “metabolically healthy overweight or obesity"—face a higher risk of Parkinson's disease. To explore this, we analyzed data from over 400,000 people in the UK Biobank who did not have Parkinson's disease at the start of the study. We grouped participants into four categories based on whether they were overweight and whether they had metabolic problems such as high blood pressure, high blood sugar, or abnormal cholesterol levels. After following them for an average of 13.5 years, about 2300 people developed Parkinson's disease. We found that being overweight per se did not increase the risk of Parkinson's disease if metabolic health was normal. However, people with metabolic problems had a higher risk of developing Parkinson's disease regardless of their weight. These results suggest that poor metabolic health is more strongly linked to Parkinson's disease than body weight alone. Addressing metabolic issues early may help prevent or delay the onset of Parkinson's disease.
Introduction
Parkinson's disease (PD), the second most common neurodegenerative disorder, is characterized by disabling cardinal motor signs, including bradykinesia, rigidity, tremor, and postural instability. This neurodegenerative condition affects body weight during the disease course, even in the premotor prodromal phase. 1 However, the relationship between body weight or body mass index (BMI) and the risk of developing PD remains inconclusive. Weight loss is often observed several years before the diagnosis of PD, and an underweight status is associated with an increased risk of PD.2–5 Other studies have reported that individuals with overweight or wider waist circumferences have a higher risk of developing PD.6–8 Meanwhile, some studies have found no association between the risk of PD and BMI or weight variability.8–12
Several epidemiological studies have explored the association between the incidence of PD and metabolic conditions, such as metabolic syndrome, besides overweight/obesity. In particular, ample evidence suggests that compared with individuals without type 2 diabetes mellitus (T2DM), those with T2DM have an approximately 30% increased relative risk of developing PD. 13 Hypertension might increase the risk of PD, 14 although the evidence is still inconclusive. Recent studies have provided relatively more support for high cholesterol levels as a protective factor against the pathogenesis of PD,15,16 which can be confounded by statin use17,18 and the presence of comorbidities. Inconsistent results from previous studies suggest that the relationship between metabolic conditions and the risk of PD is complex.
Recently, the concept of metabolically healthy overweight/obesity (MHO) was introduced to explain the improvement in clinical outcomes related to cardiovascular disease and all-cause mortality in a subgroup of subjects with overweight/obesity.19–21 These subjects exhibit normal metabolic profiles and relatively lower insulin resistance and visceral adiposity despite being overweight or obese. Regarding the relationship between MHO and neurodegenerative disorders, some studies have suggested that individuals with MHO have a reduced risk of incident Alzheimer's disease (AD) compared to those without metabolic abnormalities and obesity.22–24 However, few studies have reported the association between BMI-metabolic status phenotypes and the development of PD. 25 Therefore, in the present study, we aimed to investigate the association of BMI and metabolic conditions with incident PD, particularly the impact of MHO on the risk of PD, in the general population using the UK Biobank database.
Methods
Participants
The UK Biobank, a prospective observational cohort study, has data and biospecimens compiled from a cohort of over half a million participants aged 40–69 years throughout the United Kingdom between 2006 and 2010. Baseline information, encompassing detailed questionnaires and biospecimens, was systematically collected from 22 centers across England, Scotland, and Wales. A comprehensive account of the study rationale, design, methodologies, and protocols can be found in a previous publication. 26 Ethical clearance for data utilization in this study was obtained from the UK Biobank Access Committee (application 73873), ensuring strict adherence to ethical guidelines, regulatory standards, and the acquisition of informed consent from all participants.
Of the initial 502,419 participants, 933 with a history of PD, progressive supranuclear palsy, or multiple system atrophy were excluded. We further excluded 97,863 participants with missing metabolic indicators, such as blood pressure (n = 45,458), fasting glucose (n = 72,774), triglycerides (n = 33,215), and high-density lipoprotein cholesterol (HDL-C) (n = 72,471). We also excluded 1566 participants without BMI data. The final analysis included 402,059 participants, as illustrated in Figure 1.
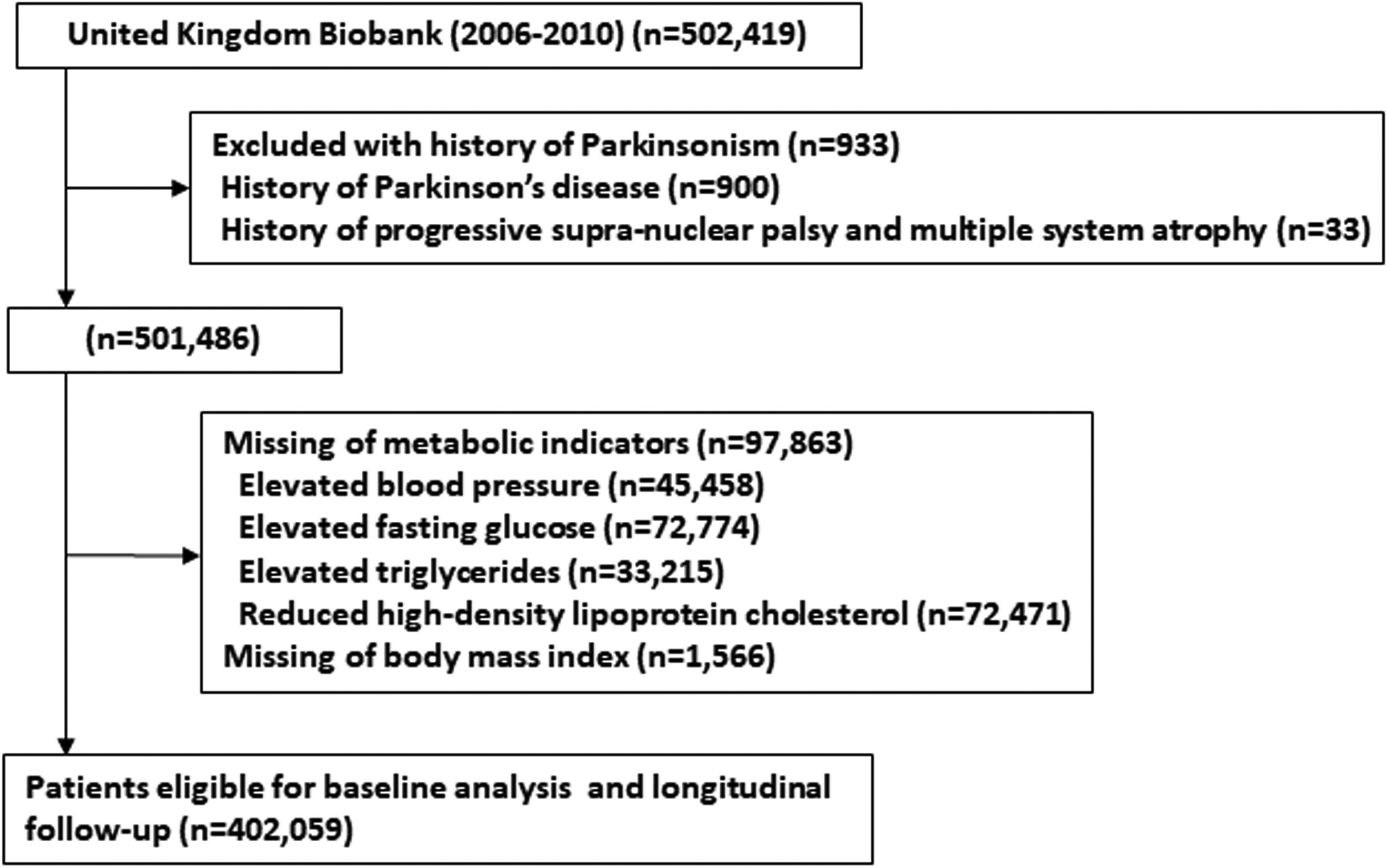
Flow diagram of the study participants.
Data collection and measurements
During the participant recruitment, medical histories, lifestyle habits, and socio-demographic characteristics were collected using a detailed touchscreen questionnaire. Trained staff conducted physical measurements, including height, weight, and blood pressure, using a Seca 202 height measure, Tanita BC-418 MA body composition analyzer, and an electronic sphygmomanometer (Omron 705 IT), respectively. BMI was calculated as body weight (kg) divided by height squared (m2). Medical histories of hypertension (I10–13 and I15), DM (E10–14), and PD were assessed using self-reports, ICD-10 codes, and medication records from the UK Biobank dataset. Detailed definitions, corresponding ICD-10 codes, and UK Biobank Field IDs used to define each condition are summarized in Supplemental Table 1. Self-reported smoking and alcohol consumption habits were categorized as never or ever. Annual household income was divided into four groups: 1) less than €18,000; 2) €18,000 to €30,999; 3) €31,000 to €51,999; and 4) over €52,000. Physical activity, measured in the metabolic equivalent task (MET) minutes per week, was grouped into tertiles (<600, 600–3000, and >3000 min/week).
Venous blood samples were collected after overnight fasting and analyzed for relevant metabolic parameters, including fasting glucose, triglycerides, HDL-C, and low-density lipoprotein cholesterol (LDL-C). These laboratory measurements were obtained using standardized enzymatic and immunoassay methods, as detailed in the UK Biobank protocol (https://biobank.ctsu.ox.ac.uk/showcase).
Assessment of metabolic abnormality and overweight/obesity
Currently, a universally accepted standard for defining metabolically healthy or abnormal states remains elusive. Lavie et al. 26 suggested a unified framework for delineating MHO consisting of seven fundamental components. According to these recommendations, metabolically healthy is defined by the absence of metabolic indicators: 1) elevated blood pressure, denoted by systolic blood pressure ≥130/85 mmHg, a history of hypertension, or the use of antihypertensive medications; 2) increased fasting glucose, specified as fasting plasma glucose ≥125 mg/dL, a history of T2DM, or the use of anti-diabetic medications; 3) elevated triglycerides, indicated by triglyceride levels ≥150 mg/dL or the use of fibrates; 4) decreased HDL-C, described as HDL-C ≤ 40 mg/dL in men and ≤50 mg/dL in women, or the use of lipid-lowering drugs. In contrast, metabolic abnormality (MA) was defined as the presence of one or more metabolic indicators, as described above. The validity of these definitions was further supported by our findings, which observed a significant increase in the risk of PD development among individuals have one or more metabolic indicators (Figure 2).
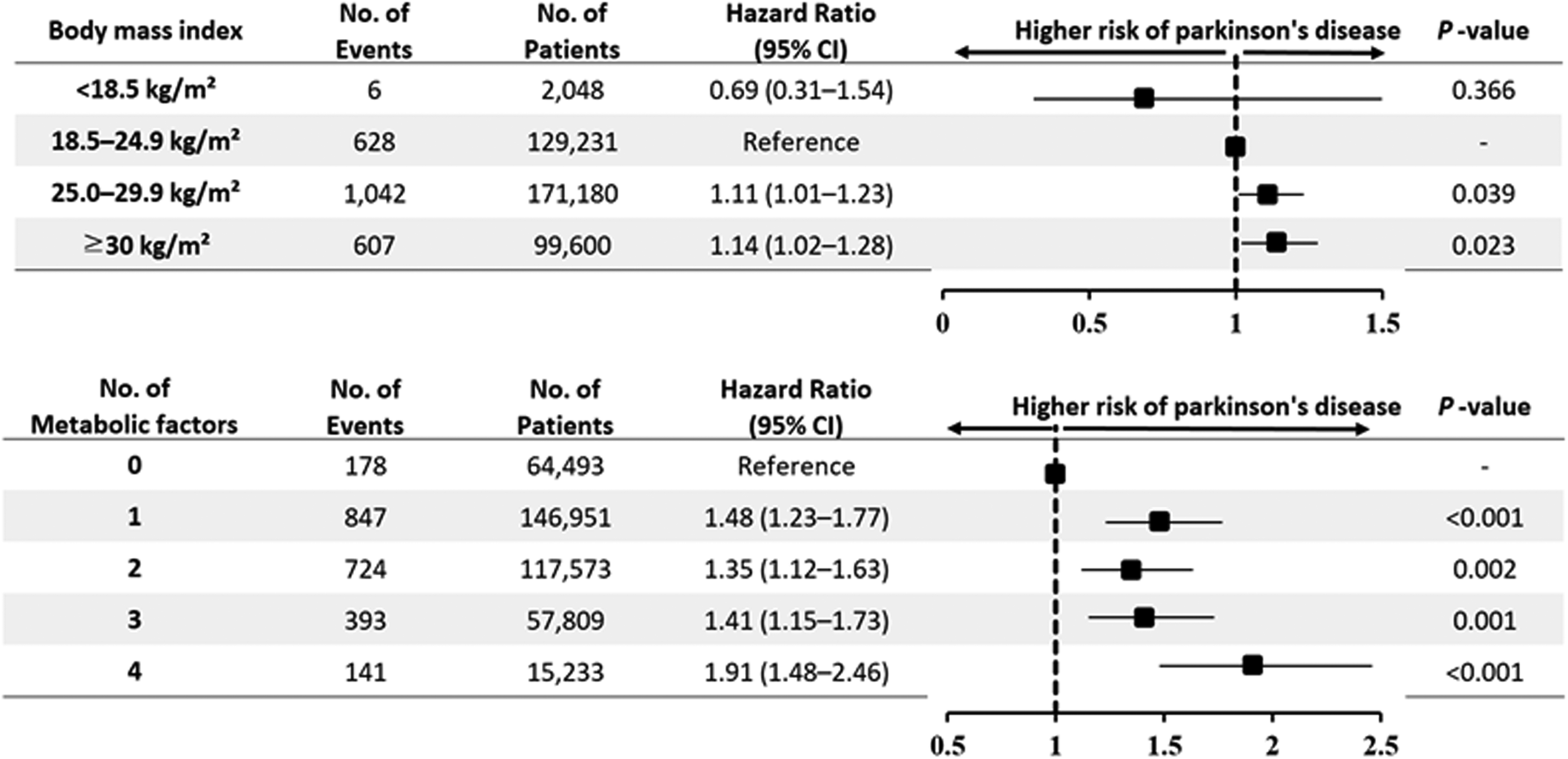
Hazard ratios for the occurrence of Parkinson's disease according to body mass index and the number of metabolic factors. Age, sex, race, smoking, alcohol habits, income, and metabolic equivalent task were included as covariates in adjusted model. No.: number; CI: confidence interval.
Similarly, the cut-off value of overweight/obesity was defined as BMI ≥25.0 kg/m² in accordance with the standard definition provided by the U.S. National Institute of Diabetes and Digestive and Kidney Diseases and the World Health Organization, which classify BMI ≥25.0 kg/m² as overweight. Notably, a significant increase in PD risk was also observed among individuals with BMI ≥25.0 kg/m² in our cohort (Figure 2). Consequently, the participants were categorized into four different groups: 1) BMI <25.0 kg/m2 without MA (group 1); 2) BMI <25.0 kg/m2 with MA (group 2); 3) BMI ≥25.0 kg/m2 without MA (group 3); and 4) BMI ≥25.0 kg/m2 with MA (group 4).
Assessment of outcome
The primary endpoint was defined as the first onset of PD within the primary care or hospital inpatient setting. The diagnosis of PD was based on ICD-10 code G20. PD was identified using the algorithmically defined outcome provided by UK Biobank (Data-Field 42032), which is constructed from linked data sources including hospital records, primary care data, death registries, and self-reports. We additionally compared this variable with the self-reported PD diagnosis (Field ID 20002, code: 1262) to confirm consistency. Outcomes were assessed from the initial assessment to the final follow-up, concluding on February 28, 2021, for participants in England and Scotland and on February 28, 2018, for participants in Wales.
Statistical analysis
The baseline characteristics of the participants are presented as mean (standard deviation) for continuous variables, count (%) for categorical variables, and median with interquartile range for skewed distributions. To assess the differences between continuous and categorical variables according to metabolic disorders and BMI, one-way analysis of variance and chi-square test were used, respectively. The Kruskal–Wallis test was used for variables with a skewed distribution. Using Kaplan–Meier estimates, we calculated the cumulative incidence rate of PD according to metabolic status and BMI and compared the results using the log-rank test. The period between the initial assessment and development of PD was referred to as the survival time. The scaled Schoenfeld residual approach was used to confirm the proportionality assumption. 27
Multivariate Cox proportional hazards regression models were used to assess the risk of developing PD. Model 1 represented the crude risk without adjustment. Model 2 was adjusted for demographics including age, sex, race, smoking habits, and alcohol consumption habits. We also created Model 3 after adjusting for household income and METs with the factors included in Model 2. We performed multiple comparisons among the four groups after statistical adjustment using the false discovery rate (FDR) correction method. In addition, we performed an analysis using a logistic regression model with inverse probability of treatment weighting (IPTW) to minimize selection bias and other possible confounding effects. Up to 10,000 iterations were conducted to minimize the absolute standardized mean difference in the effect size. The iteration stopped when the absolute standardized difference fell below 0.1 in baseline covariates between the study groups. After the IPTW adjustment, the maximum pairwise standardized mean difference showed that the variables used in the Cox model were well-balanced (Supplemental Table 2). Finally, we performed a sensitivity analysis with an additional BMI threshold of ≥30.0 kg/m2 for the robustness of primary results. Multivariate Cox proportional hazard regression results and IPTW models are presented as hazard ratios (HRs) with 95% confidence intervals (CIs). All statistical analyses were performed using Stata version 14.2 (StataCorp, College Station, TX, USA). The level of significance was set at p < 0.05.
Results
Demographic and clinical characteristics
Table 1 presents the baseline characteristics of the four metabolic phenotypes, based on MI and/or metabolic status. Group 1 corresponds to metabolically healthy non-overweight individuals, group 2 to metabolically abnormal non-overweight, group 3 to MHO, and group 4 to metabolically abnormal overweight/obesity. Among a total of 402,059 participants, 39,248 (9.8%), 91,945 (22.8%), 25,245 (6.2%), and 245,621 (61.0%) belonged to groups 1, 2, 3, and 4, respectively. The mean age of study participants was 56.6 years, and 215,586 (53.6%) participants were male. The prevalence of hypertension and T2DM were 33.5% and 5.6%, respectively. Regardless of the BMI cut-off value (BMI ≥25 kg/m2), individuals with MA were older, had a higher prevalence of hypertension and T2DM, and were more likely to be ever-smokers. These participants also had higher blood pressure, low METs, unfavorable lipid profiles, and higher levels of inflammation.
Baseline characteristics of study participants in four phenotypes classified by the BMI and/or metabolic status.
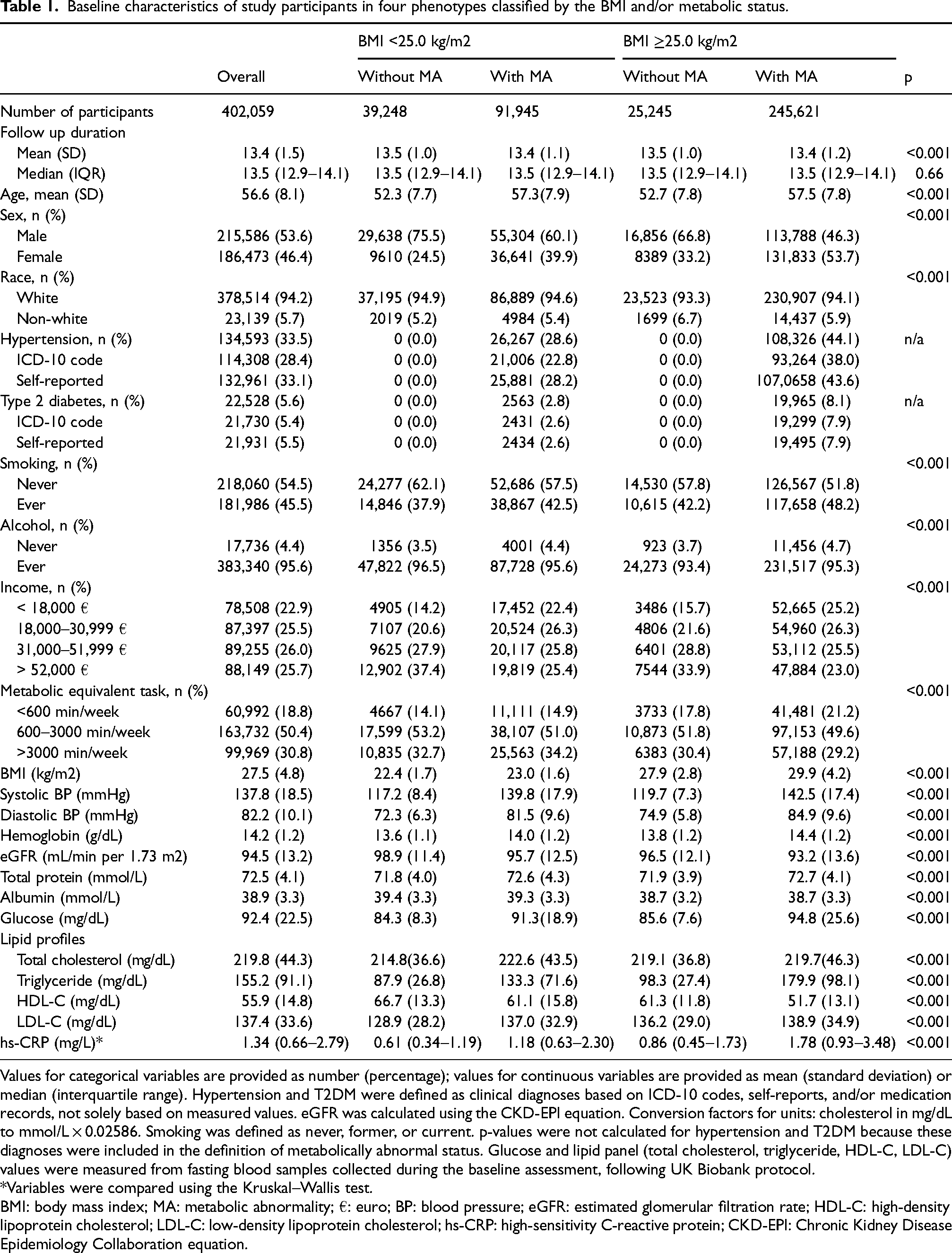
Values for categorical variables are provided as number (percentage); values for continuous variables are provided as mean (standard deviation) or median (interquartile range). Hypertension and T2DM were defined as clinical diagnoses based on ICD-10 codes, self-reports, and/or medication records, not solely based on measured values. eGFR was calculated using the CKD-EPI equation. Conversion factors for units: cholesterol in mg/dL to mmol/L × 0.02586. Smoking was defined as never, former, or current. p-values were not calculated for hypertension and T2DM because these diagnoses were included in the definition of metabolically abnormal status. Glucose and lipid panel (total cholesterol, triglyceride, HDL-C, LDL-C) values were measured from fasting blood samples collected during the baseline assessment, following UK Biobank protocol.
*Variables were compared using the Kruskal–Wallis test.
BMI: body mass index; MA: metabolic abnormality; €: euro; BP: blood pressure; eGFR: estimated glomerular filtration rate; HDL-C: high-density lipoprotein cholesterol; LDL-C: low-density lipoprotein cholesterol; hs-CRP: high-sensitivity C-reactive protein; CKD-EPI: Chronic Kidney Disease Epidemiology Collaboration equation.
BMI, MA, and the risk of PD
First, we analyzed the individual associations of BMI and metabolic status with the risk of PD. Over a follow-up period of 5,393,752 person-years (median 13.5 years), the primary outcome occurred in 2283 (0.5%) participants, resulting in an overall incident rate of PD at 4.2 per 10,000 person-years (Table 2). The incidence rate of PD was significantly higher in individuals with BMI ≥25.0 kg/m2 and those with MA compared to their counterparts. In multivariable Cox proportional analyses adjusting for confounding factors, compared with non-overweight (BMI <25.0 kg/m2), overweight/obesity (BMI ≥25.0 kg/m2) was associated with a 1.15-fold higher risk of PD development (HR, 1.15; 95% CI, 1.03–1.28; p = 0.01). Similarly, participants with MA had a 1.44-fold increased risk of PD (HR, 1.44; 95% CI, 1.21–1.72; p < 0.001) compared with those without MA. These results were consistent with those from the IPTW analysis, suggesting that both higher BMI and the presence of MA independently contribute to an increased risk of PD.
Hazard ratios for the incidence of Parkinson's disease according to the BMI and metabolic status.
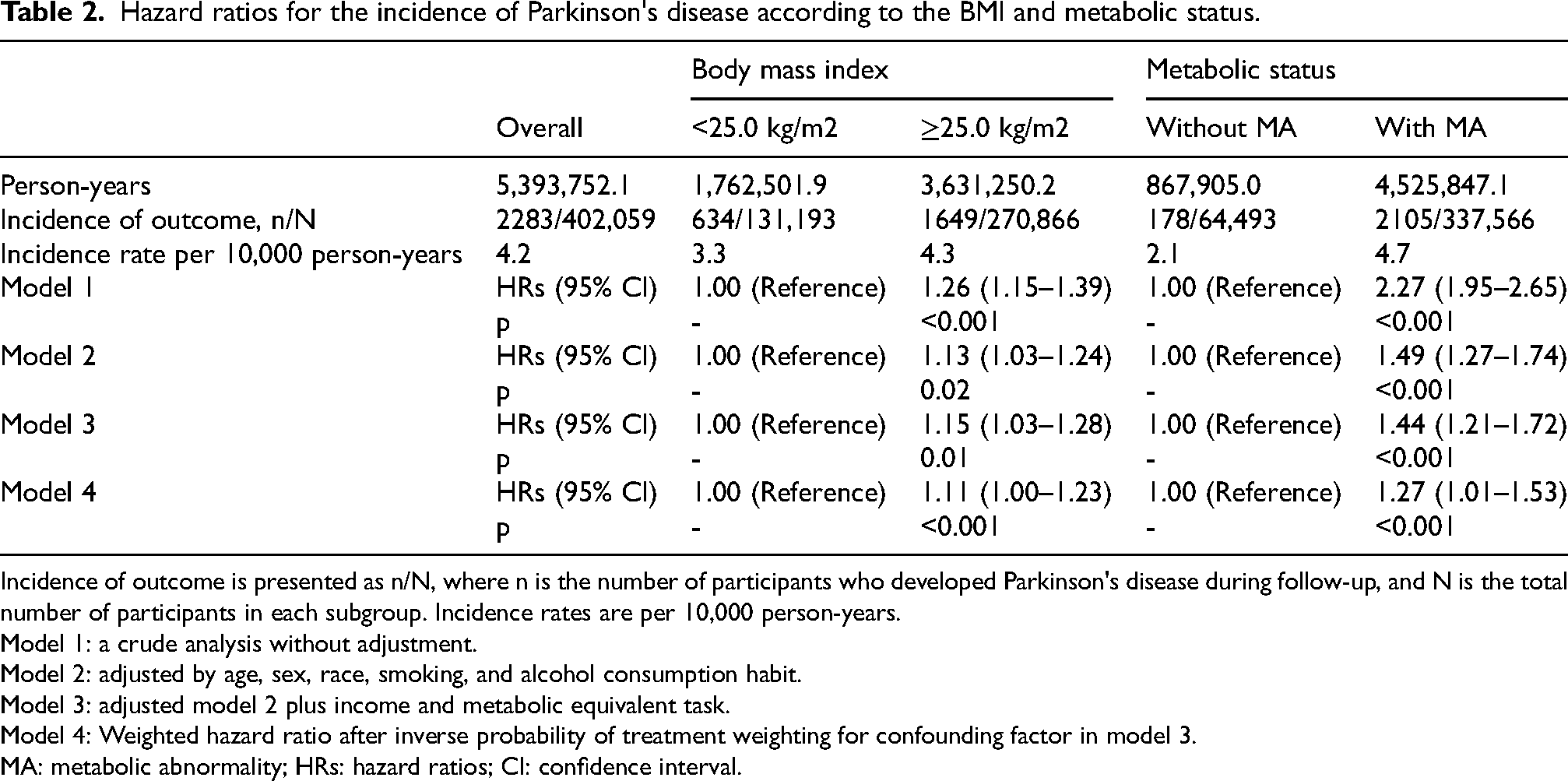
Incidence of outcome is presented as n/N, where n is the number of participants who developed Parkinson's disease during follow-up, and N is the total number of participants in each subgroup. Incidence rates are per 10,000 person-years.
Model 1: a crude analysis without adjustment.
Model 2: adjusted by age, sex, race, smoking, and alcohol consumption habit.
Model 3: adjusted model 2 plus income and metabolic equivalent task.
Model 4: Weighted hazard ratio after inverse probability of treatment weighting for confounding factor in model 3.
MA: metabolic abnormality; HRs: hazard ratios; CI: confidence interval.
Metabolic phenotype-based risk of PD
We further assessed the risk of incident PD based on the metabolic phenotypes. The incidence rates of PD were 1.9 (BMI <25.0 kg/m2 without MA), 4.3 (BMI <25.0 kg/m2 with MA), 2.2 (BMI ≥25.0 kg/m2 without MA), and 4.8 (BMI ≥25.0 kg/m2 with MA) per 10,000 person-years across the four groups (Table 3). Regardless of the BMI status, the incidence rate was significantly higher in participants with MA than in those without MA. The cumulative incidence rate of PD was also significantly higher in participants with MA, with the highest incidence observed in participants with BMI ≥25.0 kg/m2 and MA, followed by those with BMI <25.0 kg/m2 and MA (Figure 3). Consistent with the cumulative incidence rate, the risk of developing PD was 2.50-fold higher in participants with BMI ≥25.0 kg/m2 and MA (HR, 2.50; 95% CI, 2.04–3.06; p < 0.001) and 2.26-fold higher in participants with BMI <25.0 kg/m2 and MA (HR, 2.26; 95% CI, 1.83–2.80; p < 0.001) compared to those with BMI <25.0 kg/m2 without MA (Table 3). After adjusting for confounding factors, both BMI ≥25.0 kg/m2 participants with MA (HR, 1.58; 95% CI, 1.25–1.99; p < 0.001) and BMI <25.0 kg/m2 participants with MA (HR, 1.55; 95% CI, 1.22–1.98; p < 0.001) exhibited a higher risk of PD development than BMI <25.0 kg/m2 participants without MA (Model 3). These associations were consistent in the weighted HRs after IPTW analysis for confounding factors (Model 4) and in inter-group comparisons after statistical adjustment with FDR correction (Supplemental Table 3). Additionally, participants with BMI ≥25.0 kg/m2 without MA tend to have a lower risk for developing PD than those with BMI <25.0 kg/m2 with MA (HR, 0.79; 95% CI, 0.61–1.03; FDR-corrected p = 0.14) and those with BMI ≥25.0 kg/m2 with MA (HR, 0.76; 95% CI, 0.59–0.97; FDR-corrected p = 0.06). Meanwhile, participants with BMI ≥25.0 kg/m2 with MA exhibited a risk of PD development similar to that in participants with BMI <25.0 kg/m2 with MA (HR, 1.02; 95% CI, 0.91–1.15; FDR-corrected p = 0.69). Participants with BMI ≥25.0 kg/m2 without MA also exhibited a PD risk comparable to that in participants with BMI <25.0 kg/m2 without MA (HR, 1.17; 95% CI, 0.84–1.63; FDR-corrected p = 0.41). These findings suggest that the risk of PD is associated with the presence of MA, irrespective of BMI status.
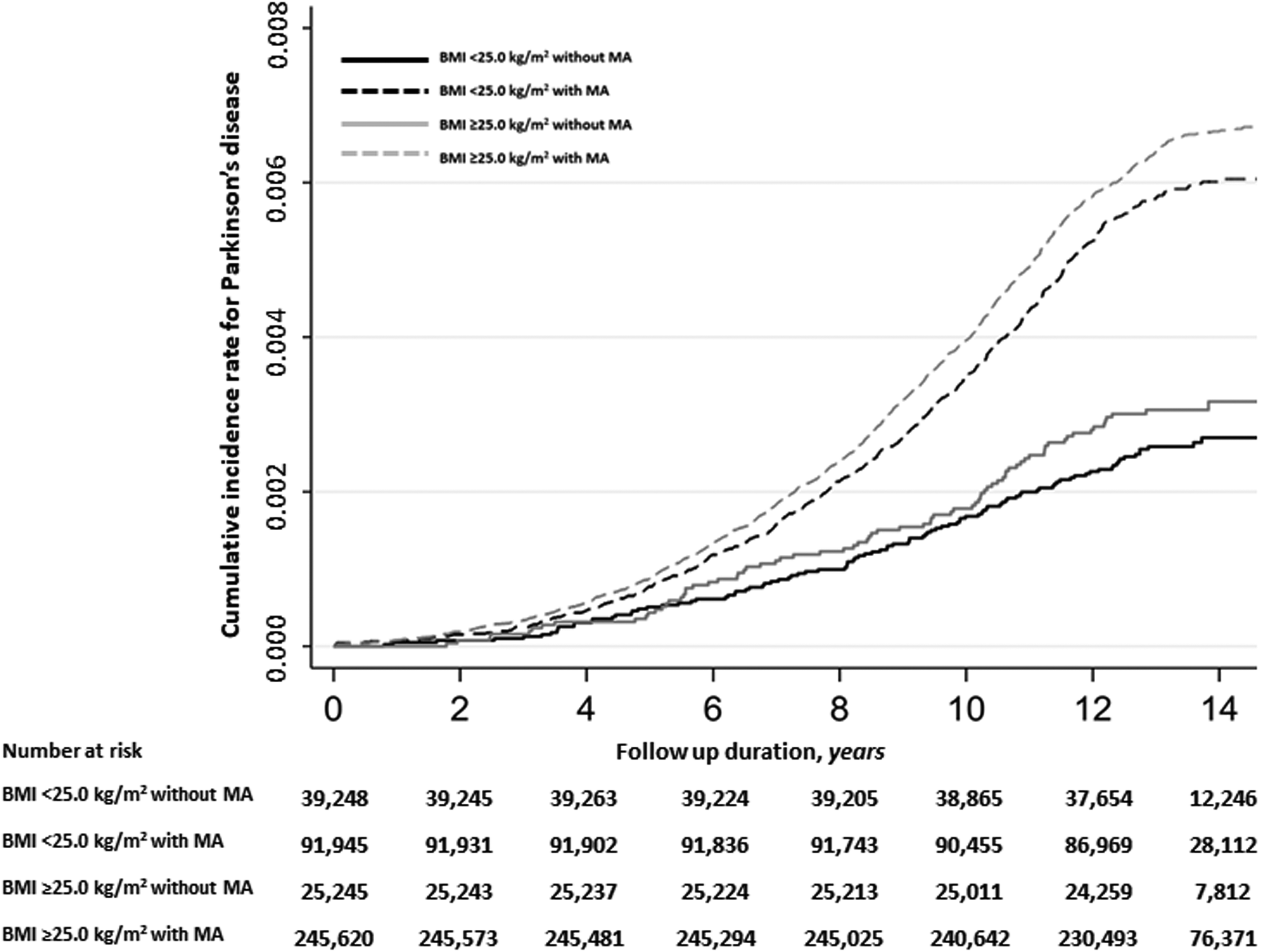
Cumulative incidence rate for Parkinson's disease according to metabolic subtypes. Subjects with metabolically abnormal status had a higher risk for developing PD than those with metabolically healthy status, regardless of overweight or obese status (either body mass index ≥25 kg/m2 or <25 kg/m2). The crosses in the graphs indicate censored data. BMI: body mass index; MA: metabolically abnormal.
Hazard ratios for the incidence of Parkinson's disease among 4 BMI-metabolic status phenotypes classified by the BMI and/or metabolic abnormality.
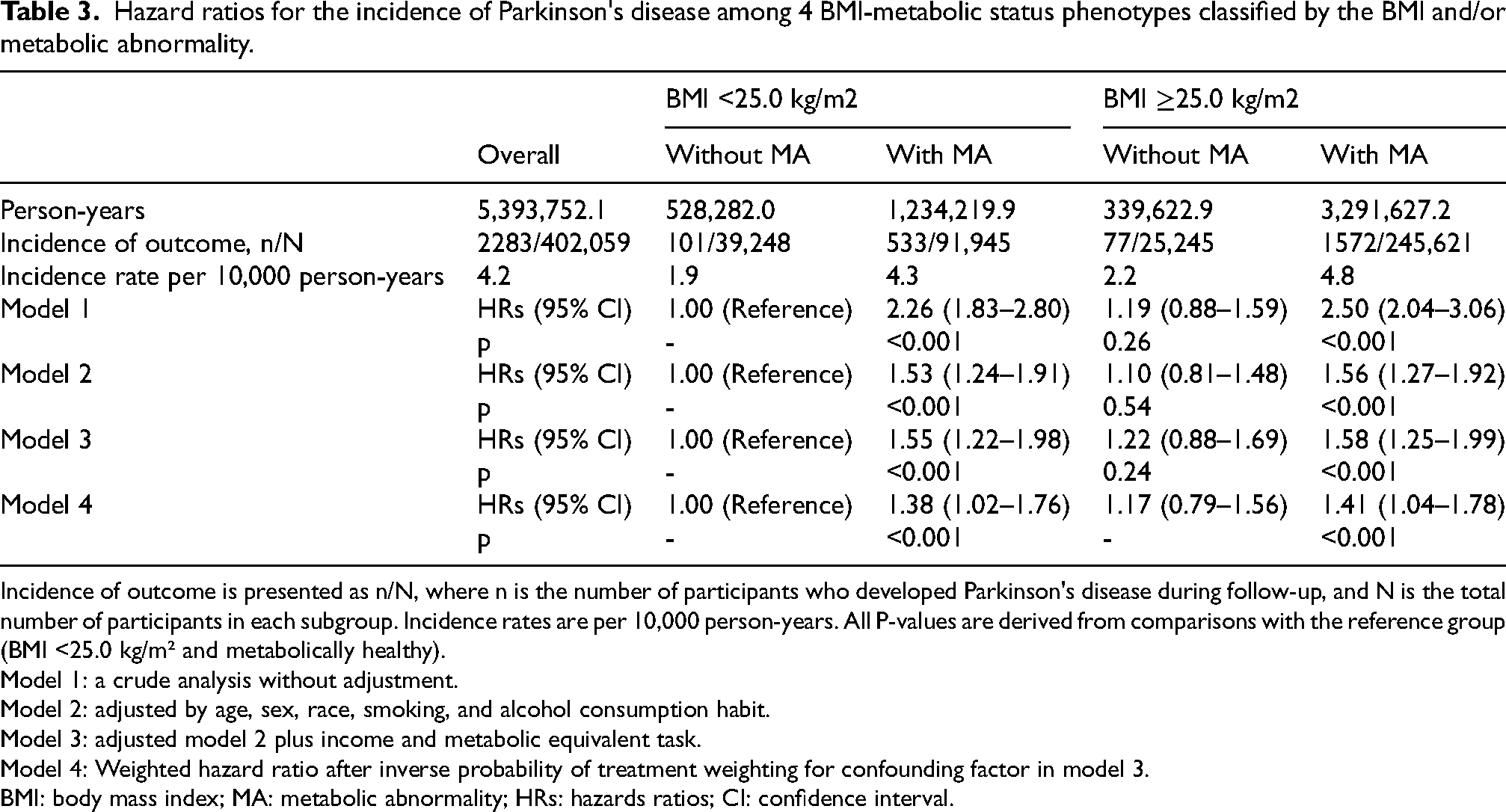
Incidence of outcome is presented as n/N, where n is the number of participants who developed Parkinson's disease during follow-up, and N is the total number of participants in each subgroup. Incidence rates are per 10,000 person-years. All P-values are derived from comparisons with the reference group (BMI <25.0 kg/m² and metabolically healthy).
Model 1: a crude analysis without adjustment.
Model 2: adjusted by age, sex, race, smoking, and alcohol consumption habit.
Model 3: adjusted model 2 plus income and metabolic equivalent task.
Model 4: Weighted hazard ratio after inverse probability of treatment weighting for confounding factor in model 3.
BMI: body mass index; MA: metabolic abnormality; HRs: hazards ratios; CI: confidence interval.
To further explore the time-dependent effect of metabolic status on PD risk, we conducted a stratified Cox regression analysis across three follow-up periods (0–5, 5–10, and 10–15 years; Supplemental Table 4). The risk differences between groups were minimal during the first 5 years. However, during the 5–10 and 10–15 years of follow-up periods, individuals with MA exhibited significantly increased risks of developing PD, particularly among those with BMI ≥25 kg/m².
Sensitivity analysis
To assess the robustness of our primary results, we conducted a sensitivity analysis using BMI >30.0 kg/m2 as an alternative cut-off value. Regardless of the BMI status, the cumulative incidence of PD was significantly higher Early intervention for metabolic abnormalities would be in participants with MA than those without MA (Supplemental Figure 1). The risk of developing PD remained significantly higher in both participants with obesity and MA (HR, 1.49; 95% CI, 1.22–1.81; p < 0.001) and participants without obesity with MA (HR, 1.45; 95% CI, 1.21–1.74; p < 0.001), compared to participants without obesity and MA. Meanwhile, participants with obesity without MA exhibited a comparable risk for developing PD to participants without obesity and MA (HR, 1.19; 95% CI, 0.64–2.20; p = 0.58), in the Cox proportional analysis (Model 3 in Supplemental Table 5). These findings are consistent with the IPTW analysis (Model 4 in Supplemental Table 5).
Metabolic component-specific and interaction effects on PD risk
We evaluated the effects of individual MA components—elevated blood pressure, glucose, triglycerides, and reduced HDL-C—on PD risk. In multivariable Cox regression models, elevated blood pressure (HR 2.07; 95% CI 1.85–2.33) and glucose (HR 1.46; 95% CI 1.33–1.61) were significantly associated with increased PD risk. Although the level of triglycerides (HR 1.07; 95% CI 0.98–1.16) and HDL-C (HR 1.09; 95% CI 0.99–1.20) did not reach statistical significance, both showed a trend toward an increased risk of PD. We also assessed the cumulative effect using the total number of abnormal components. Each additional component increased the PD risk by 24% (HR 1.24; 95% CI 1.19–1.28), supporting an additive relationship (Figure 2, bottom panel). Finally, interaction terms between all component pairs were tested in Cox regression models. None were statistically significant, although a borderline trend was observed for blood pressure × triglycerides (HR 0.80; p = 0.068), further supporting an additive rather than multiplicative effect.
Discussion
The present study investigated the association of overweight/obesity and metabolically abnormal status with the risk of PD using the UK Biobank database. The major findings were as follows: (1) Study participants were categorized into four BMI-metabolic status phenotypes, including the metabolically healthy non-overweight (9.8%), MHO (6.3%), metabolically abnormal non-overweight (22.9%), and metabolically abnormal overweight/obesity (61.1%) groups. (2) During the follow-up period, 2283 (0.6%) of 402,059 participants were newly diagnosed with PD. (3) Individuals with overweight/obesity and individuals with MA had a higher risk of developing PD than their counterparts. (4) Compared with the metabolically healthy non-overweight group (reference group), the MHO group had a comparable risk of developing PD, whereas the other two metabolically abnormal groups exhibited an increased incidence of PD. These findings suggest that a metabolically abnormal status is more relevant to incident PD than overweight/obesity; in particular, MHO status does not increase the risk of developing PD. Indeed, a sensitivity analysis investigated whether individual metabolic risk factors such as T2DM or hypertension are associated with the risk of PD, our study extended this approach by evaluating combined metabolic phenotypes, which integrated both BMI and metabolic health status. This allowed us to identify distinct subgroups, such as the MHO phenotype, that may not carry an increased PD risk. By doing so, our findings offer a more nuanced understanding of how metabolic health interacts with body weight in relation to PD development.
Overweight6–8 and underweight2–5 have both been reported as risk factors for PD; however, the relationship between BMI and risk of PD remains controversial. Determining BMI as a risk factor for PD is challenging, based on the results of epidemiological studies, since BMI is a variable that may change over time. Moreover, body weight change may represent a prodromal feature of PD rather than an independent risk factor. However, evidence suggests that BMI is determined by several factors that are closely linked to neurodegenerative processes in the premotor stages, including energy imbalance, hypothalamic dysregulation, dopaminergic signaling alterations (e.g., lower striatal D2 receptor availability in subjects with obesity 28 ), and the non-motor nature of PD (e.g., gastrointestinal dysfunction, impaired olfaction, and reduced motivation). 1 Furthermore, both high and low BMI can involve a series of pathological cellular responses leading to neurodegeneration, for example, via adiposity-related inflammation 29 and low central insulin levels, 30 respectively. In this study, using the UK Biobank database, individuals with overweight or obesity exhibited a higher risk of developing PD than those with normal weight, whereas individuals with underweight had a comparable risk of PD to those with normal weight. One possibility explaining these inconsistent results across studies is that the association between BMI and PD risk is likely attributable to two separate processes in the development of PD, depending on when BMI was measured.31,32 Higher BMI in middle-aged individuals may be linked to an increased risk of PD, whereas lower BMI associated with preclinical metabolic changes may be observed before PD diagnosis. Further, well-designed studies are required to unravel the effect of BMI on PD risk, considering that PD-related pathologies commence several years before the appearance of Parkinsonian motor symptoms and BMI may change dynamically during this period. 1
Concerning the contribution of metabolic status in determining the risk of PD, this study found that even a single metabolic risk factor was associated with an increased incidence of PD. Metabolic syndrome is a cluster of common pathologies, and we defined metabolically abnormal status as the presence of one or more metabolic risk factors. To date, epidemiological evidence linking T2DM and PD is well established, 33 and Mendelian randomization also suggests a causal effect of T2DM on PD risk. 34 T2DM and PD might share common pathogenic mechanisms (e.g., insulin receptor signaling 35 ) leading to protein aggregation, 36 mitochondrial dysfunction, 37 defective autophagy, 38 and inflammatory response. 39 Moreover, based on preclinical evidence for using glucagon-like peptide 1 receptor agonists and dipeptidyl peptide 4 inhibitors in PD models, several clinical trials of antidiabetic medications are currently ongoing to investigate their protective effects against PD pathology. 33 Meanwhile, the association between hypertension and the risk of PD remains controversial. A recent meta-analysis demonstrated that hypertension might increase the incidence of PD in population-based cohort studies, whereas the opposite results were obtained in case-control studies. 14 With regard to the relationship between lipid profile and PD risk, earlier studies report mixed results.40,41 However, recent studies provide more evidence that higher cholesterol levels are associated with a reduced incidence of PD.15,16 The relationship between statins and PD risk is complex. Preclinical studies have demonstrated both protective 42 and harmful effects of statins on PD models,43,44 and epidemiological studies have also reported inconsistent results that may be affected by statin type and duration of statin use.45–48 The situation for statin initiation (such as metabolic syndrome, cardiovascular disease, and physical inactivity) is itself a risk factor for developing PD, and the therapeutic effect of statins (i.e., lowering the cholesterol level) may negatively impact the incidence of PD via alterations in lysosomal membrane stability. 49 Additional research is required to elucidate the complex relationship between metabolic status and the risk of PD.
Although overweight/obesity and metabolic risk factors are often interrelated, they can also exist independently in some individuals, and their clinical implications may vary depending on the BMI-metabolic status phenotypes. In particular, one unique subtype of overweight/obesity, MHO, has received much attention as having relatively favorable clinical outcomes for obesity-related diseases such as cardiovascular disease. 50 Moreover, all lean individuals do not present a healthy metabolic profile, and the concept of “metabolically obese, but normal weight (MONW)” is also introduced to explain the heterogeneous nature of obesity. 50 Individuals with MHO and MONW exhibit insulin sensitivity and lipid and inflammatory profiles that are contrary to those predicted for an obese status. Since these profiles are also closely related to the biological mechanisms of neurodegenerative disorders, attempts have been made to investigate an association between BMI-metabolic status phenotypes and AD.22–24 However, this is much less studied in populations with PD. 25 The present study demonstrated that the MHO group had a comparable risk of developing PD to the metabolically healthy non-overweight group. Additionally, the metabolically abnormal non-overweight group had a similar PD risk as the metabolically abnormal overweight group. These findings are in line with the clinical relevance reported in previous studies for distinct obesity subtypes, including MHO and MONW,19,50 suggesting that metabolic conditions are more strongly associated with the risk of developing PD than BMI. Furthermore, when evaluating individual MA components, only elevated blood pressure and elevated glucose were independently associated with an increased PD risk, whereas elevated triglycerides and reduced HDL-C were not. In addition, no statistically significant interactions were observed between any pair of components, supporting the interpretation that MA contribute to PD risk in an additive rather than multiplicative manner. Additionally, our time-stratified analysis revealed that the effect of MA on PD risk became more pronounced over time, with significant increases in PD risk during the 5–10 and 10–15 years of follow-up periods. These findings support the hypothesis that the influence of metabolic dysregulation on neurodegeneration may accumulate gradually, highlighting the importance of early intervention. The significant association between BMI and PD risk observed in this study may be attributed to the relationship between BMI and metabolic status. However, because the metabolically abnormal group was older than the metabolically healthy group (age difference of approximately 5 years) in this study, caution should be exercised when interpreting the results in the context of the impact of metabolic syndrome on the development of PD. Indeed, a sensitivity analysis including non-linear age terms showed attenuation of the observed associations, suggesting that the relationship between metabolic status and PD risk may be partially influenced by complex age-related effects (Supplemental Table 6). This is in line with previous literature that aging is the primary risk factor for PD: some aging-related changes, such as impaired proteasome/lysosome function, oxidative/nitrative damage, and inflammation, can produce a vulnerable pre-parkinsonian state. 51
Our study had some limitations. First, the primary outcomes in our study are based on ICD codes. Thus, issues regarding diagnostic accuracy and the overestimation or underestimation of PD incidence may rise in such large-scale population-based studies. In particular, the diagnosis of PD based on Hospital Episode Statistics (HES) records may be delayed compared to the actual clinical diagnosis. To address this concern, we conducted a sensitivity analysis comparing baseline metabolic profiles between individuals diagnosed with PD within 5 years of baseline and those diagnosed 5 years later. No significant differences in baseline metabolic profiles were found between the groups (Supplemental Table 7), suggesting that the potential delay in the diagnosis of PD based on HES records is unlikely to have substantially biased our findings. However, the incidence rate of PD (4.2 per 10,000 person-years) reported in this study does not contradict prior estimates of PD incidence (an average estimate of 13 per 100,000 person-years in the general population and 180 in populations aged 65 years and older). Second, BMI and metabolic conditions may change over time, influencing the risk of developing PD in different directions. The present study did not reflect possible changes in BMI-metabolic status phenotypes during the follow-up period because of the unavailability of relevant data. Moreover, previous longitudinal studies have shown that metabolic health status is dynamic and may deteriorate with aging,52,53 suggesting that some degree of misclassification over time should be considered when interpreting the results. Third, MA is a complex condition comprising diverse clinical manifestations such as increased blood pressure, insulin resistance, dyslipidemia, and systemic inflammatory conditions. However, we could not independently assess the specific contributions of these factors to PD development. Thus, these factors may collaborate to induce inflammatory conditions during the development of PD. Fourth, our study findings may have limited generalizability to other populations, since the UK Biobank primarily comprises healthy volunteer participants and is predominantly confined to the UK. Finally, while this study used a literature-based definition of MA, future work may benefit from data-driven scoring systems that assign variable weights to individual metabolic components, or metabolomics-based risk stratification once such data becomes widely available in large cohorts. Additionally, identifying mediating biological pathways such as inflammation or insulin resistance may further clarify the mechanisms linking MA to PD risk.
In conclusion, our findings suggest that metabolically abnormal conditions are more relevant to incident PD than overweight/obesity. MHO status does not increase the risk of developing PD compared with metabolically healthy non-overweight status, whereas metabolically abnormal status is associated with an increased incidence of PD, regardless of overweight/obesity status. These results highlight the potential importance of addressing MA in future efforts to delay the onset or progression of PD.
Supplemental Material
sj-docx-1-pkn-10.1177_1877718X251367305 - Supplemental material for Body mass index, metabolically abnormal status, and incident Parkinson's disease: Data from the UK Biobank
Supplemental material, sj-docx-1-pkn-10.1177_1877718X251367305 for Body mass index, metabolically abnormal status, and incident Parkinson's disease: Data from the UK Biobank by Hae-Ryong Yun, Nak-Hoon Son, Hee Byung Koh and Seok Jong Chung in Journal of Parkinson's Disease
Footnotes
Ethical considerations
Ethical clearance for data utilization in this study was obtained from the UK Biobank Access Committee (application 73873), ensuring strict adherence to ethical guidelines and regulatory standards.
Consent to participate
Written informed consent was obtained from all individuals for their participation in UK Biobank
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by a faculty research grant of Yonsei University College of Medicine (6-2022-0059) and a research grant of Yongin Severance Hospital, Yonsei University College of Medicine (Z-2023-0013). This research was also supported by a grant from the Korea Health Technology R&D Project through the Korea Health Industry Development Institute (KHIDI), funded by the Ministry of Health & Welfare, Republic of Korea (RS-2023-KH134527).
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
For purposes of replicating procedures and results, any qualified investigator can request anonymized data after ethics clearance and approval by all authors.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
