Abstract
Mild cognitive impairment and dementia are common symptoms in people with Parkinson's disease (PwPD) that impair the quality of life of those affected. However, PD-specific concepts for the prevention of cognitive decline are rarely incorporated into routine care. Here, we provide key data on cognitive impairment in PwPD and a framework for primary, secondary, and tertiary prevention in this context. The importance of cognitive reserve as a protective buffer for cognitive decline in PwPD is highlighted. Relevant lifestyle and health-related factors, including cognitive aspects, physical and social activity, diet, hearing loss, and cardiovascular factors, are discussed. Evidence on the efficacy of possible cognition-enhancing interventions in PwPD—pharmacological, cognitive, physical, nutritional, and multidomain interventions—is summarized. On this basis, and the recommendations of the European Task Force for Brain Health Services, a proposal is developed outlining options for preventing cognitive impairment in PwPD that could be implemented in routine care, as well as further developments needed to achieve a best-case scenario. The main pillars of a strategic agenda for this purpose include: (i) regular assessment of cognitive state, overall risk, and risk factors for cognitive decline; (ii) risk communication and education concerning modifiable risk factors with standardized procedures; (iii) risk reduction with multi-domain interventions for secondary prevention; and (iv) cognitive enhancement with cognitive and physical training for tertiary prevention. As the proposal makes clear, the prevention of cognitive impairment in PwPD requires interdisciplinary collaboration organized throughout PD care networks.
Plain language summary
Many people with Parkinson's disease (PwPD) experience cognitive problems, relating to memory and thinking. This can impair their quality of life. However, there are few strategies in regular medical care to help prevent these issues. This text explains the main facts about cognitive problems in PwPD and suggests ways to prevent them. It describes how “cognitive reserve” can help protect cognitive skills. Lifestyle factors that are important for cognitive health, like staying mentally, physically, and socially active, and a healthy diet, are discussed. Managing hearing loss, heart disease, and other health issues is also important. Different interventions to support brain health in PwPD are reviewed. These include medication, mental exercises, physical activity, diet changes, and combined approaches. Based on scientific recommendations, a plan is proposed to include prevention strategies in routine care.
The key steps in this plan are:
Regularly checking cognitive skills, along with possible risk factors. Educating PwPD about ways to lower their risks. Using combined intervention approaches to help slow cognitive loss. Offering cognitive and physical training to support cognitive skills.
Preventing cognitive decline in PwPD requires teamwork between different medical experts. These efforts should be organized in special PD care networks to provide the best support.
Cognitive impairment and dementia in people with Parkinson's disease: why prevention is important
Cognitive impairment and dementia are common and severely debilitating symptoms of Parkinson's disease (PD). Together with affective disorders, such as depression, anxiety, and apathy, which also occur frequently, these non-motor symptoms have the greatest impact on the quality of life of people with PD (PwPD). They are also associated with a greater physical and emotional burden on relatives. The high direct and indirect care costs caused by cognitive impairment also place a considerable burden on healthcare systems. Dementia is associated with disease prognosis and life expectancy and is a common reason for admission to an inpatient nursing facility. As an exclusion criterion for deep brain stimulation, dementia also limits the treatment of motor symptoms. 1
The “cognitive continuum” in PD (see Figure 1) ranges from healthy, age-appropriate cognitive performance to mild cognitive impairment (PD-MCI) and Parkinson's disease dementia (PDD); PD-MCI represents a risk factor for the development of PDD. 1 While PD-MCI is characterized by psychometrically objective cognitive impairment without notable effects on activities of daily living (ADL), cognitive impairment must significantly interfere with ADL for a diagnosis of PDD. The cognitive profiles of PwPD can be very heterogeneous. Memory and executive functions are typically affected early, but attention, visuo-cognitive dysfunction, and language problems can also occur. 2 The early presence of deficits in memory, semantic verbal fluency, and visuo-cognitive tasks, reflecting more “temporal dysfunction” (and resembling more the Alzheimeŕs disease (AD) profile) have been discussed in particular as predictive of the later development of dementia. Accordingly, coexisting tau and Aβ pathology is common in PwPD with cognitive impairment and relates to a faster onset of dementia. 3 In contrast, deficits in executive function—considered more “frontostriatal” or dopaminergic in nature—are less strongly associated with cognitive decline.4,5 These different risk profiles for cognitive decline in PD and their underlying pathophysiology have also been conceptualized in the “dual syndrome hypothesis”. 6 With regard to the motor profile, the akinetic-rigid/ postural instability gait disturbance subtype in particular is considered a risk factor for the development of PD-MCI 7 and PDD 8 compared to the tremor-dominant subtype. 9
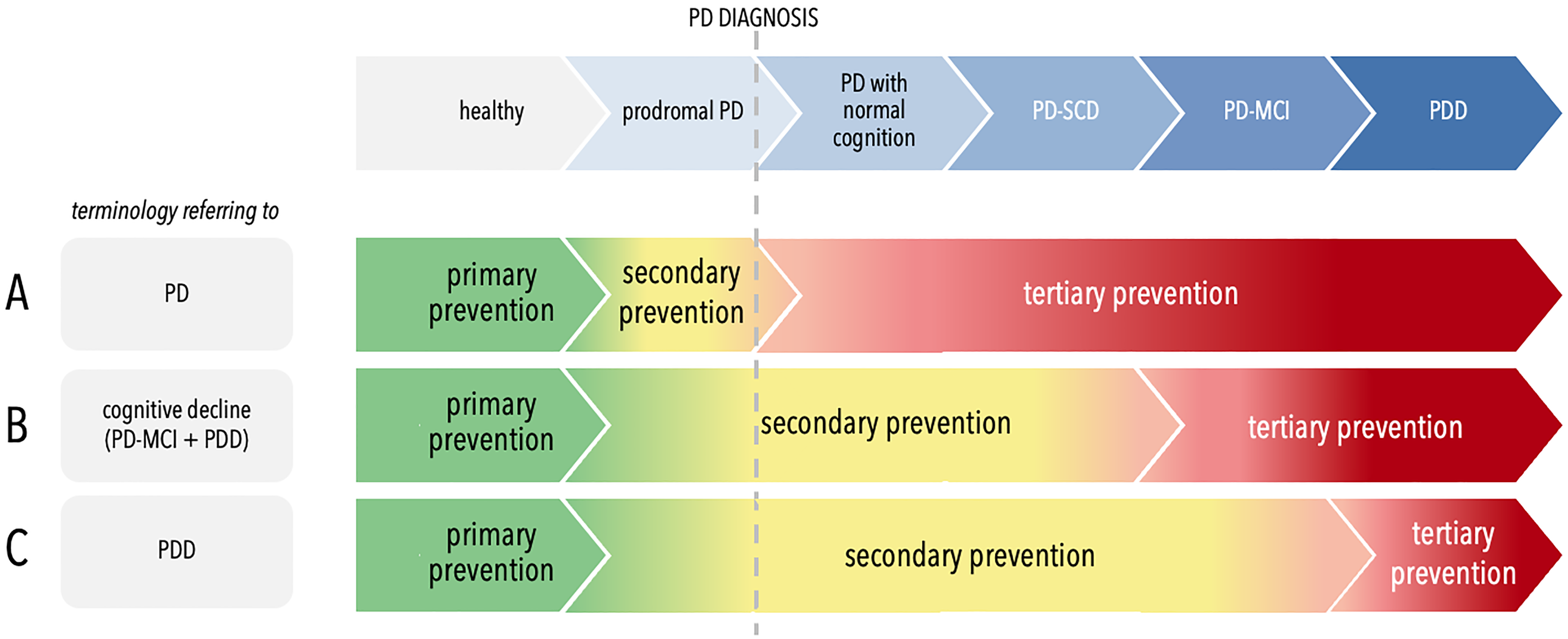
Primary, secondary, and tertiary prevention of Parkinson's disease and cognitive impairment in people with Parkinson's disease: different nomenclatures. Abbreviations. PD: Parkinson's disease, SCD: subjective cognitive decline; PD-MCI: mild cognitive impairment in PD; PDD: Parkinson's disease dementia. Primary prevention approaches are aimed at the healthy general population and should be implemented before disease processes begin.
Other risk factors for early progression to dementia include older age and older age at onset, male sex, low education, visual hallucinations, REM sleep behavior disorder, severity of motor symptoms, and depression; changes in specific biomarkers have also been identified, including cortical atrophy and AD-like changes in cerebrospinal fluid and functional imaging. 1
The point prevalence of PD-MCI in PwPD is around 40%. 11 In up to one-third (19%–36%) of PwPD, PD-MCI is already detectable at the time of clinical diagnosis. 12 The estimated point prevalence of PDD is 24% to 31%. 13 According to the analysis of two recent longitudinal cohort studies, the risk of dementia increases with disease duration, from 9% to 27% after 10 years, up to 50% after 15 years, and to 74% after 20 years. 14
The concept of subjective cognitive decline (SCD) in PwPD is also attracting increasing interest. SCD was first conceptualized in the context of AD. 15 SCD describes cognitive decline that is subjectively perceived by the patient, but which is not clinically detectable using cognitive tests. SCD in people with an AD-typical biomarker profile is often a precursor to clinical cognitive impairment and progression to AD dementia. 16 There is also increasing evidence in PwPD that SCD precedes PD-MCI and PDD. The relative risk of cognitive decline in PwPD with SCD but without objective cognitive impairment is 2.71 within an average follow-up period of 3.16 years. 17 The point prevalence of PD-SCD is currently estimated at 36%, 17 although the definitions and assessments used in the context of PD are very heterogeneous.
Cognitive changes can also occur in the prodromal stage of PD. 18 For example, people with isolated REM sleep behavior disorder (iRBD), which is indicative of early alpha-synucleinopathy, show poorer performance across different cognitive domains than healthy controls. 19 Up to 90% of people with iRBD develop PD or dementia with Lewy bodies (DLB) within a decade. 20 For example, in a multicenter cohort study of 1280 people with iRBD, 73.5% progressed to clinically advanced alpha-synucleinopathies within 12 years 21 - 52% of whom developed PD, 43% developed DLB, and 4.5% developed multiple system atrophy.
In summary, it is evident that PwPD—and in particular individuals who are already affected by PD-SCD or PD-MCI as possible PDD precursors—are important target groups for the prevention of cognitive impairment and dementia. It is also relevant in this context that no pharmacological therapies are currently recommended for the treatment of PD-MCI, and no disease-modifying pharmacotherapies are available for cognitive impairment in PwPD. 22
With regard to terminology, the terms primary, secondary, and tertiary prevention, which are used for PD in the context of motor symptoms, 10 are not necessarily applicable to the prevention of cognitive impairment in PD (Figure 1). Primary prevention refers to disease prevention measures in the healthy population. We propose that in the context of cognitive decline in PD, secondary prevention includes all measures aimed at preventing cognitive impairment (PD-MCI and PDD) in the following target groups: people with an increased risk of PD and prodromal PD, people with clinically manifest PD without cognitive impairment, and those with SCD. Tertiary prevention would then include interventions when PD-MCI or PDD is already present.
Cognitive reserve, lifestyle factors, and dementia prevention - and what we know about them in people with Parkinson's disease
The term “cognitive reserve” refers to the resistance of the brain to damage and related negative effects on cognitive performance. 23 The concept of cognitive reserve, which originates from the context of AD, is related to various influencing variables throughout the lifespan that impact the risk of dementia in old age. According to a recent report by the Lancet Standing Commission, around 45% of all-cause dementia cases worldwide could be prevented or delayed if all modifiable influencing factors were optimized. 24 The fourteen risk factors that met the defined evidence criteria include low education in childhood and adolescence, hearing loss, traumatic brain injury, hypertension, high LDL cholesterol levels, excessive alcohol consumption and obesity in middle age, smoking, depression, visual impairment, social isolation, physical inactivity, air pollution, and diabetes in old age. 24 Another relevant factor is engagement in cognitively stimulating activities over the lifespan, including both occupational and leisure-related activities.25,26 The latter can refer to a wide range of activities such as educational courses, board and card games, reading, and artistic and craft activities.
The concept of cognitive reserve is increasingly being applied to PwPD. A recent review and meta-analysis indicated associations of cognitive reserve with higher cognitive performance and fewer motor symptoms in PwPD and people with DLB. 27 Regarding the determinants of cognitive reserve, both higher levels of education and more frequent participation in cognitively stimulating activities were associated with better cognitive performance in PwPD; conversely, low education increased the risk of dementia in PwPD.28,29 Two large studies including more than 500 PwPD each demonstrated that cognition was associated with hypertension, diabetes, dyslipidemia, physical and cognitive activity, 30 education, socioeconomic status, and social engagement. 31 A recent paper indicated that activities in middle and older (but not younger) adulthood were particularly relevant for cognitive reserve in PwPD. 32 It should be mentioned that there is also increasing evidence of the influence of lifestyle factors on functional and structural brain health in the sense of a “brain reserve” in PwPD. 33 Finally, for some risk factors for dementia indicated by the Lancet Standing Commission, such as hearing and vision impairment, 24 the association with cognition in PwPD has been widely neglected so far.
Although the data available is significantly sparser than for people with AD, they indicate overall that the mechanisms of cognitive reserve described also apply to PwPD. Accordingly, a healthy lifestyle with cognitively stimulating activities, physical activity, and social engagement as well as the management of general health factors (including hearing, vision, and cardiovascular) play an important role in cognitive functioning and the risk of cognitive decline in PwPD.
Pharmacological and non-pharmacological interventions
Alongside lifestyle and general health factors, targeted interventions can also strengthen cognitive functioning in PwPD.
Pharmacological therapy for cognitive impairment and PDD—following the recommendations of the International Parkinson and Movement Disorders Society (MDS) 22 or the guidelines of the German Neurological Society (DGN) 34 —aims to improve cognitive function and alleviate neuropsychiatric symptoms. The preferred treatment options include acetylcholinesterase inhibitors (AChEIs) which stabilize cognitive functions by inhibiting the breakdown of the neurotransmitter acetylcholine. Within the class of AChEIs, rivastigmine and donepezil are recommended in particular for PDD, as several studies have shown a moderate improvement in cognitive performance and a delay in disease progression. 35 Galantamine, while used in AD, is not recommended for the treatment of PDD, and there are also no recommended pharmacological therapies for PD-MCI. While AChEIs are well tolerated in PDD, their effects on cognitive function are limited, meaning an individual risk-benefit assessment is required for each patient. A discontinuation trial is also recommended, to check whether an effect on cognitive performance can be detected after a 6-month treatment period. Concurrent treatment of neuropsychiatric symptoms, such as depression and hallucinations, should be considered, as these often occur in conjunction with cognitive impairment. Among others, selective serotonin reuptake inhibitors and atypical antipsychotics such as quetiapine are recommended for this purpose. 36
There is extensive evidence that cognitive training can induce plasticity processes in the brain; 37 functional 38 and structural 39 brain changes have been described specifically in PwPD after engaging in such training. Cognitive training can be applied in many different settings—digitally or real-world, individually or group-based, supervised or unsupervised. Also, single-domain (i.e., working memory) and multiple-domain trainings exist, with the latter showing stronger effects in PD. 40 A recent Cochrane review classified the effects of cognitive training on cognitive performance in people with PD-MCI and PDD as uncertain, due to methodological heterogeneity and other limitations of the studies; 41 however, other meta-analyses have found positive effects with small to medium effect sizes on both global cognition and specific cognitive domains in PwPD with and without PD-MCI.40–43 On the basis of this evidence, the recently published German PD guidelines recommended cognitive training for the treatment of PD-MCI. 34 However, longitudinal studies with several years of follow-up to provide information on the potential for the secondary prevention of PD-MCI or PDD are still lacking. An initial hint is provided by a randomized controlled trial 44 in which PwPD without PDD participated in a six-week cognitive training group, leading to lower rates of progression from PD to PD-MCI after one year. It should be noted that the effectiveness of cognitive training on cognitive performance has also been shown for neurologically healthy older people, people with non-PD MCI, and those with other brain diseases, with small effect sizes. 45 However, evidence for long-term effects on the development of cognitive impairment and dementia is also largely lacking in these wider populations. 46 The large “Advanced Cognitive Training for Independent and Vital Elderly (ACTIVE)” study conducted in the USA, in which the effects of different forms of cognitive training were compared with a passive control group in over 2800 older adults, was able to demonstrate long-lasting effects over up to 10 years 47 on cognition and ADL. However, conversion rates to MCI or dementia were not analyzed, so a possible preventive effect can only be assumed indirectly.
There is a vast body of evidence indicating the positive effects of exercise on brain health, including benefits to neuroplasticity as the physiological basis of cognitive reserve. 48 Physical inactivity is regarded as a modifiable risk factor for the development of dementia. 24 Positive effects of targeted physical interventions on cognition have also been demonstrated for neurologically healthy older people 49 and people with non-PD MCI 50 and dementia. 51 Positive effects on brain health and neuroplasticity processes induced by physical interventions have also been described in PwPD. 52 Furthermore, while cognition has not often been measured as an outcome in physical intervention studies on PD-MCI, 53 there is sufficient evidence to support the inclusion of endurance exercise as a recommendation in the current German PD guidelines for the treatment of PD-MCI. 34 However, due to a lack of evidence, it was not possible to make any recommendation for the use of physical interventions for secondary or tertiary prevention of cognitive decline in PwPD.
Regarding nutritional interventions, increasing evidence indicates that a Mediterranean diet leads to an overall reduced risk of developing age-related cognitive disorders, including cognitive impairment, dementia, and AD. 54 Strict adherence to a Mediterranean diet has also been reported to lead to improved cognition in PwPD. 43 However, there is no reliable evidence that dietary supplements provide a preventive benefit or are effective in treating cognitive deficits in PD-MCI or PDD.
Knowledge of the effects of lifestyle and non-pharmacological interventions on cognition has led to the development of so-called multidomain lifestyle interventions. Here, cognitive training is typically combined with physical and nutritional interventions alongside the management of other health factors, such as cardiovascular issues. These broad interventions have been applied to people with an increased dementia risk profile, for example in the largescale “Multidomain Alzheimer Preventive Trial (MAPT)”, “Finnish Geriatric Intervention Study to Prevent Cognitive Impairment and Disability (FINGER)”, and “Prevention of Dementia by Intensive Vascular care (preDIVA)“ studies. 55 Although the individual study results are heterogeneous with regard to the effects on cognitive performance, they have been evaluated positively overall, and multidomain interventions are considered an effective strategy for the prevention of cognitive decline in people at risk. 55 This approach has so far received little attention in PwPD. However, the “Programme for Dementia Prevention” nationwide integrated care concept in Luxembourg—which offers “personalized multidomain intervention” tailored to an individual's dementia risk profile 56 —is currently also being adapted specifically to PwPD (R. Krüger, personal communication, March 27, 2024). Furthermore, an ongoing study of people with iRBD is investigating the effects of digital cognitive training accompanied by a module promoting a healthy, active lifestyle on cognition, physical activity, and structural and functional brain parameters. 57
Overall, although there is robust evidence for the short-term effects of targeted interventions, especially cognitive training and physical activity, on cognition in PwPD, their potential for the prevention of cognitive decline in the long-term requires further research.
Prevention of cognitive decline in people with Parkinson's disease: a possible roadmap
How the prevention of cognitive decline can be implemented in the clinical care of PwPD remains a critical question. In 2023 58 and in an update in 2025, 59 the European Task Force for Brain Health Services published recommendations for evidence-based and ethical dementia prevention strategies for people at risk for dementia (not specific to PD), which are to be implemented via “second-generation memory clinics” providing “brain health services”. According to this initiative, the core elements of these strategies for “proactive, prospective risk reduction” in people at risk for dementia (rather than care for those already suffering from the impairment) are (i) the assessment of risk of cognitive decline, (ii) risk communication with ad-hoc protocols, (iii) risk reduction with multi-domain interventions for secondary prevention, and (iv) cognitive enhancement with cognitive and physical training for tertiary prevention. Notably, for PwPD who are routinely treated by neurologists, specific implementation of these measures may be possible, but are currently not available.
Based on these recommendations,
58
,
59
we propose the following care elements and specifications for PwPD. The current status of care, possible further developments, and a best-case scenario are presented in Table 1. Meanwhile, Figure 2 illustrates prevention care components and relevant actors of a possible best-case solution for PwPD at different cognitive stages.
The implementation of a standardized assessment of the individual risk for cognitive decline in PwPD from the time of diagnosis should be discussed. Frequently used instruments—such as the “Cardiovascular Risk Factors, Aging, and Incidence of Dementia (CAIDE)” or “LIfestyle for BRAin health (LIBRA)” risk scores (overview in
63
), represent possible options, but are not established in the context of PD. Dementia risk scores specifically for PwPD have also been developed: “Montreal Parkinson Risk of Dementia Scale (MoPaRDS)” and “Parkinson's disease Risk Estimator for Decline In Cognition Tool (pPREDICT)”.
64
It is striking, however, that the composition of risk and protective factors assessed in these four scores is very heterogeneous and includes different sociodemographic, neuropsychological, neuropsychiatric, clinical, lifestyle, and – for PwPD – PD-specific factors (Table 2). They also miss some of the risk factors proposed by the Lancet Standing Commission
24
(Table 2). Genetic risk factors for cognitive decline in PD, i.e., mutations of the glucocerebrosidase (GBA), have also not been considered yet. Additionally, it should be noted that these risk scores serve to predict the risk of dementia, not MCI. Finally, they are not suited for the systematic assessment of individual modifiable risk factors. There is a need for the development of easy-to-use questionnaires that give comprehensive information on individual risk profiles, and indicate suitable lifestyle changes and possible targeted interventions. Due to the generally increased risk of cognitive decline in PD, all of those affected—independent of their individual risk—should be informed about the possibility of strengthening cognition through an active lifestyle. As indicated above (section ib), the development of an easy-to-use questionnaire on modifiable risk factors is needed (for a first draft see the German KogniPark-Checklist
71
). It should identify suitable recommendations for a “brain-healthy lifestyle” with regard to aspects that can be optimized for the individual (such as cognitive activity, physical activity, and nutrition). Until such tools are available, medical history in routine care visits should touch upon these aspects, and appropriate counseling and motivation for a healthy lifestyle should be provided. Prospectively, a module on “cognition and the prevention of cognitive decline” could also be integrated into standardized educational programs for PwPD, which are currently being developed in several countries.72–74
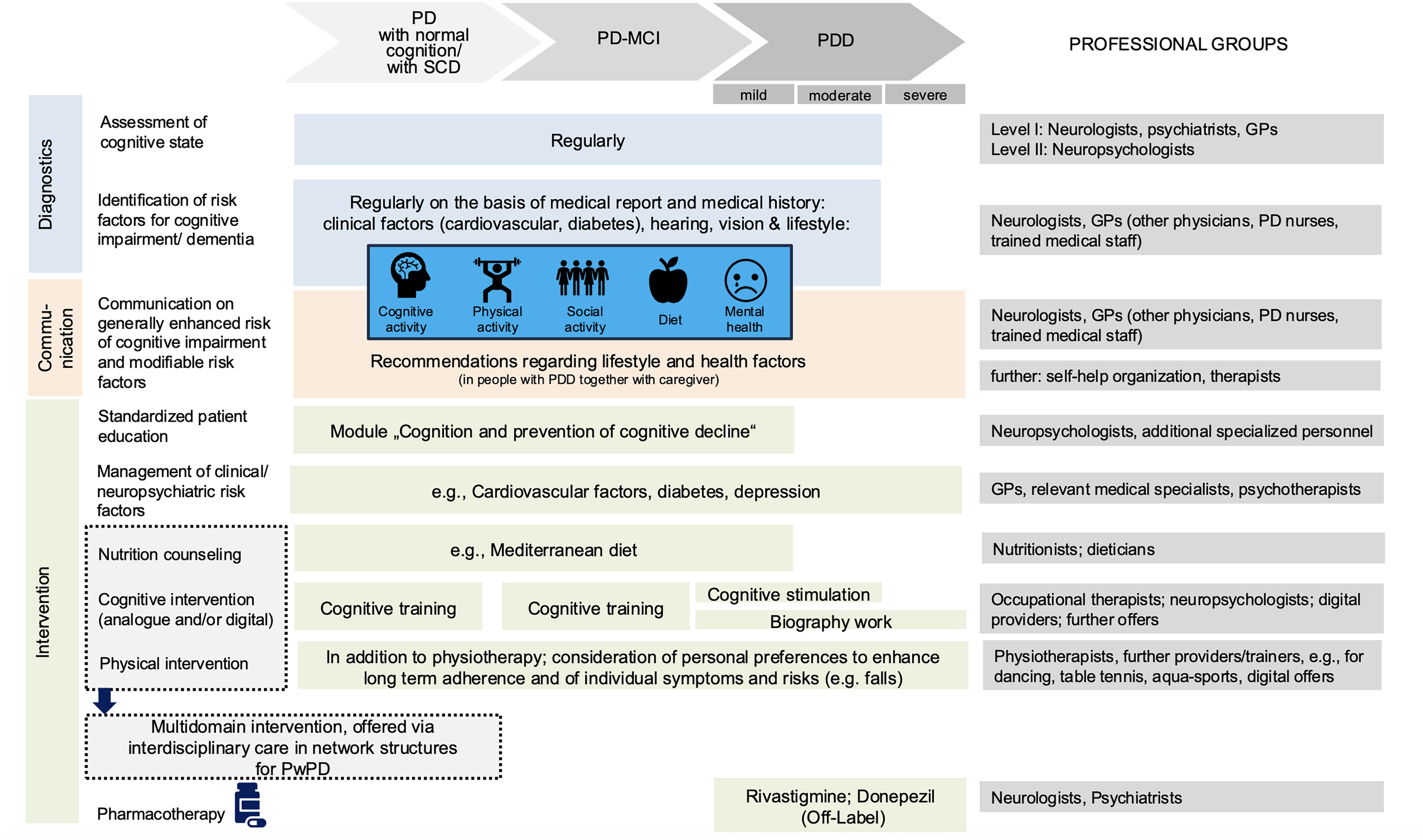
Proposed best-case care scenario for the prevention and treatment of cognitive impairment in people with clinical Parkinson's disease. Abbreviations. GP: general practitioner; PwPD: people with Parkinson's disease; PD-MCI: mild cognitive impairment in PD; PPD: Parkinson's disease dementia; SCD: subjective cognitive decline.
Prevention of cognitive impairment in people with Parkinson's disease: current options, with Germany as an example, possible developments, and best-case scenarios.
Dementia risk scores from Alzheimer's disease research and Parkinson's disease-specific research (modified according to Carlisle and colleagues 64 ).

Factors indicated by Lancet Standing Commission; 24 definition of specific factor or operationalization might differ;
Only considered in PD-specific LIBRA publication; 64
Only considered in original publication of LIBRA score, 67 but not in the PD-specific one.
Abbreviations. BMI: body mass index; CAIDE: cardiovascular risk factors, aging, and incidence of dementia; GDS-15: Geriatric Depression Scale (15 Items); HLD: hyperlipidemia; LIBRA: LIfestyle for BRAin health Index; MDS-UPDRS: Movement Disorders Society-Unified Parkinson Disease Rating Scale; MoCA: Montreal Cognitive Assessment; MoPaRDS: Montreal Parkinson Risk of Dementia Scale; PASE: Physical Activity Scale for the Elderly; pPREDICT: Parkinson's disease Risk Estimator for Decline In Cognition Tool; RSBDQ: REM Sleep Behavior Disorder Questionnaire; SBP: systolic blood pressure; STAI-S: State-Trait Anxiety Inventory; TIA: transient ischemic attack.
Conclusion
The aim of this paper was to summarize the current state of knowledge on cognitive impairment in PwPD and possibilities for the prevention of cognitive decline, and to present a best-case care scenario for implementation. This summary was based on currently available evidence on cognitive reserve and factors influencing the risk of cognitive decline, taken from the contexts of both AD and PD context; evidence for the effectiveness of interventions to strengthen cognition in PwPD; the recommendations of the European Task Force for Brain Health Services from 2023 58 and 2025 59 ; and recently proposed recommendations for therapy for cognitive impairment in PD from the German PD guidelines. 34
This paper proposes that all measures aimed at preventing progression to PD-MCI and PDD – in people at increased risk of PD, those with SCD, and those with clinically manifest PD without cognitive impairment – should be understood in terms of secondary prevention. Measures to strengthen cognition when PD-MCI or PDD is already present are conceptualized as tertiary prevention. Current therapy guidelines, such as those in Germany, 34 commonly only consider the latter, with cognitive and physical interventions being recommended. However, the reality of care currently shows insufficient implementation, related to a lack of service structures and failures of prescription and reimbursement practices. In terms of secondary prevention, there is increasing evidence for the influence of lifestyle-related factors on cognition in PwPD. However, a translation of this evidence into the intervention landscape for PwPD – as is being implemented for people at risk for AD in projects such as the worldwide “FINGERS network” (https://fbhi.se) – has not yet been initiated. Based on the biological definition of PD80,81 the knowledge about risk genes for cognitive decline, the progress in identifying prodromal PD, and increasing possibilities to predict the individual risk for cognitive decline in prodromal and clinical PD, the potential for personalized secondary prevention is enlarging. Finally, no standards for communication about the risk of cognitive impairment in PD and the possibilities of positively influencing cognition through a (brain-)healthy lifestyle have been established.
Given the increasing prevalence of PD and the prevalence and relevance of cognitive impairment in PwPD, there is an urgent need for action. Further developments and standardization in research and practice are required to achieve a possible best-case scenario, with the main pillars of (i) regular assessment of cognitive state, overall risk, and risk factors for cognitive decline, (ii) risk communication and education about modifiable risk factors with standardized procedures, (iii) risk reduction with (digitally-supported) multi-domain interventions for secondary prevention of cognitive decline, and (iv) cognitive enhancement with cognitive and physical training for tertiary prevention. This would be best realized in an interdisciplinary care landscape, organized by PD networks, as have been or are being established in the Netherlands 82 and Germany. 83 This roadmap requires the participation of healthcare providers for PwPD, stakeholders in the healthcare system, scientists, and those affected and their relatives.
Footnotes
Acknowledgments
We thank Jördis Dienst for her support with preparing the manuscript.
Author contributions
EK: concept, writing of manuscript; TW: concept, revision of manuscript; CE: concept, revision of manuscript; AO: concept, revision of manuscript; AKF: concept, writing of manuscript.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
EK has received grants from the German Federal Ministry of Education, RheinEnergie Stiftung Germany, and STADAPHARM GmbH as well as honoraria from EISAI GmbH, BIAL GmbH, Memodio GmbH, Kohlhammer Verlag, Hogrefe Testzentrale, ProLog Wissen GmbH. EK is one of the authors of the cognitive intervention series “NEUROvitalis” but receives no corresponding honoraria.
TW has received honoraria from BIAL, Abbvie, Desitin, Pftzer, Britannia, Esteve and Licher, consultancies from Stadapharm and Phagenesis, and funding from Abbvie and EVER Pharma.
CE received in the last 12 months payments as a consultant for Abbvie Inc., Bial Inc., Gerresheimer AG. CE received honoraria as a speaker from Abbvie Inc., Bial Inc., Bristol-Myers Squibbs Inc., Merck Inc., Synaptikon Inc. CE is in the steering board of Parkinson Netzwerke Deutschland.
AO received grants from the Koeln Fortune Program (grant-no. 329/2021, 142/2023, 145/2024, 15/2025), Faculty of Medicine, University of Cologne and the “Novartis-Stiftung für therapeutische Forschung”, all outside the submitted work.
AKF has received grants from the German Parkinson Society and STADAPHARM GmbH as well as honoraria from ProLog Wissen GmbH, Cologne, Germany; Seminar- und Fortbildungszentrum Rheine, Germany; LOGOMANIA, Fendt & Sax GbR, Munich, Germany; and LOGUAN, Ulm, Germany; dbs e.V., Moers, Germany. AKF is one of the authors of the cognitive intervention series “NEUROvitalis” but receives no corresponding honoraria.
