Abstract
Summary
This study is the first large-scale, head-to-head comparison suggesting that sodium-glucose cotransporter-2 inhibitors (SGLT2is) may offer greater neuroprotection against Parkinson's disease (PD) compared to metformin in patients with type 2 diabetes mellitus (T2DM). Utilizing a 20-year real-world dataset and propensity score matching, we found that SGLT2i users had a 28% lower adjusted hazard ratio (aHR) for PD (0.72; 95% CI, 0.62–0.84) and reduced all-cause mortality. Unlike previous studies suggesting a potential increased PD risk with SGLT2is, our robust study design, stringent exclusion criteria, and competing risk adjustments support a protective association. The findings highlight the need for further prospective research to explore the neuroprotective benefits of SGLT2is, which may justify prioritizing their use in T2DM patients at risk for neurodegeneration.
Background
Type 2 diabetes mellitus (T2DM) is linked to an increased risk of Parkinson's disease (PD), likely mediated by insulin resistance, inflammation, and mitochondrial dysfunction. While metformin has shown neuroprotective effects, sodium-glucose cotransporter-2 inhibitors (SGLT2is) have emerging benefits in neurodegeneration. This study provides the first real-world head-to-head comparison of SGLT2is and metformin on PD risk in T2DM patients.
Methods
Using the TriNetX platform, we analyzed a 20-year dataset (2005–2025) from 142 healthcare organizations, identifying 913,428 T2DM patients (96,018 SGLT2i, 817,410 metformin users). Patients with prior PD, neurodegenerative diseases, or exposure to neuroprotective/neurotoxic antidiabetic drugs were excluded. Propensity score matching (1:1) balanced cohorts across demographic, clinical, and pharmacological variables. Cox proportional hazards models estimated adjusted hazard ratios (aHRs), validated by positive and negative controls.
Results
SGLT2i use was associated with a 28% lower PD risk than metformin (aHR = 0.72; 95% CI, 0.62–0.84; p < 0.0001). Dementia, a positive control, also showed reduced risk (aHR = 0.73; 95% CI, 0.68–0.78; p < 0.0001), reinforcing the neuroprotective effect. Negative controls confirmed specificity. SGLT2i users had significantly lower all-cause mortality (aHR = 0.85; 95% CI, 0.83–0.89; p < 0.0001).
Conclusions
This first large-scale comparison suggests SGLT2is provide superior neuroprotection against PD compared to metformin in T2DM patients, warranting further investigation.
Plain Language Summary
Parkinson's disease is a brain condition that affects movement and is more likely to occur in people with type 2 diabetes. Some diabetes medications may offer protection for the brain, but it's not clear which ones work better. In this large real-world study using electronic health records from over 900,000 patients, we compared two common diabetes drugs: SGLT2 inhibitors and metformin. We found that people who took SGLT2 inhibitors had a significantly lower risk of developing Parkinson's disease compared to those who took metformin. This protective effect was especially clear in older adults, people with poor blood sugar control, and those with kidney problems. The results remained strong even after adjusting for many other health conditions, medications, and lifestyle factors such as smoking and alcohol use. Importantly, SGLT2 inhibitor users also lived longer, which could have affected how often Parkinson's disease was diagnosed. However, we accounted for this in our analysis and still found a consistent benefit. These findings suggest that SGLT2 inhibitors might not only help control blood sugar but also protect the brain in people with type 2 diabetes. More research is needed to understand how these drugs work on the brain and whether they should be considered as first-line treatment for patients at risk of neurodegenerative diseases like Parkinson's.
Keywords
Introduction
Type 2 diabetes mellitus (T2DM) is a growing global health burden, affecting millions and increasing the risk of neurodegenerative diseases, including Parkinson's disease (PD).1–5 Epidemiological studies have consistently demonstrated that T2DM is associated with an elevated PD risk, particularly in younger patients, those with longer diabetes duration, and individuals with more advanced disease.4,6,7 The pathophysiological connection between T2DM and PD is supported by evidence from epidemiological studies, as well as in vivo rodent models and in vitro neuronal studies, implicating insulin resistance, oxidative stress, neuroinflammation, mitochondrial dysfunction, ferroptosis, and α-synuclein aggregation as shared biological mechanisms.1,8,9 Notably, methylglyoxal, a reactive glycolytic byproduct and potent endogenous glycating agent, accumulates in patients with T2DM and may promote MPTP-like neurotoxicity, thereby contributing to dopaminergic neurodegeneration.1,8,9 Given the rising prevalence of both conditions, addressing this relationship is a public health priority.8–11
PD imposes a substantial socioeconomic burden, leading to significant healthcare costs, productivity loss, and declining quality of life.12,13 Strategies to mitigate PD risk in T2DM patients include optimal glycemic control and the potential repurposing of antidiabetic medications with neuroprotective properties.14–16 Metformin lowers blood glucose primarily by suppressing hepatic gluconeogenesis and improving peripheral insulin sensitivity. 17 These metabolic effects may indirectly reduce systemic inflammation, oxidative stress, and mitochondrial dysfunction—mechanisms also implicated in neurodegeneration. 18 Metformin has also been hypothesized to confer neuroprotective benefits through AMP-activated protein kinase (AMPK) activation, enhancement of mitochondrial function, and modulation of neuroinflammation and α-synuclein aggregation.19–21 However, observational studies examining the association between metformin use and PD risk have yielded mixed results. Ping et al. and Qin et al. reported increased PD risk with metformin monotherapy (OR 1.66 and HR 1.50, respectively),22,23 potentially linked to long-term vitamin B12 deficiency or reverse causality. In contrast, Wahlqvist et al. observed a reduced PD risk among metformin users (HR 0.62), 24 and several other studies have demonstrated an inverse dose-response association, suggesting even lower cumulative exposures may confer neuroprotection.25,26 These inconsistent findings may reflect a non-linear, dose-dependent relationship.23,25 Low cumulative exposure (<300 defined daily doses) has been associated with reduced PD risk, 25 while higher cumulative doses or prolonged use may attenuate benefits or increase risk.23,25 Biologically, mild AMPK activation at lower doses may promote neuroprotection,21,25,27 whereas prolonged exposure could lead to B12 depletion and mitochondrial complex I inhibition, both detrimental to neuronal health.23,25,27 Further studies are needed to clarify metformin's dose-response dynamics in neurodegenerative outcomes.
Sodium-glucose cotransporter-2 inhibitors (SGLT2is) are a newer class of antidiabetic agents with cardiometabolic benefits beyond glucose control.28,29 By inhibiting renal glucose reabsorption, they improve glycemic control, reduce body weight, and lower blood pressure.28,29 Recent observational studies and meta-analyses suggest that SGLT2is may also confer neuroprotective effects, particularly in lowering the risk of dementia and cognitive decline among patients with T2DM.30–35 These findings provide a rationale for evaluating their impact on PD, a neurodegenerative disorder with overlapping pathophysiologic pathways. Proposed mechanisms for neuroprotection include improved cerebral perfusion, suppression of neuroinflammation, enhanced mitochondrial function, and reduced oxidative stress.11,28,30,32,36 SGLT2is may also activate autophagy to clear α-synuclein aggregates and promote ketone body utilization as alternative neuronal fuels.37,38 Given metformin's established but inconsistent associations with PD risk,21,24–27,39–43 we conducted a retrospective cohort study to compare the incidence of PD among patients with T2DM who initiated SGLT2is versus metformin using a large real-world dataset. Our objective was to evaluate whether SGLT2is provide additional neuroprotective benefits relative to metformin.
Methods
Data source
This study utilized the TriNetX platform, a global federated health research network aggregating de-identified electronic health records (EHRs) from 142 healthcare organizations (HCOs) worldwide. 44 The dataset includes demographics, diagnoses, lab results, medications, procedures, and genomic data while adhering to HIPAA and GDPR privacy regulations to ensure patient anonymity.44–46
A cohort of 913,428 T2DM patients was identified, comprising 96,018 SGLT2i users and 817,410 metformin users, spanning January 20, 2005, to January 20, 2025. The dataset, covering hospitals, primary care, and specialty clinics, provided extensive real-world data for robust epidemiological analysis and rigorous evaluation of clinical outcomes across different antidiabetic treatments.44,45 The analysis was conducted using the TriNetX Analytics Platform on February 26, 2025, at 06:33:24 UTC, based on de-identified real-world data from 142 healthcare organizations worldwide.
Study population and design
This study included adults (≥18 years) with type 2 diabetes mellitus (T2DM; ICD-10-CM: E11) who were diagnosed between January 20, 2005, and January 20, 2025, and initiated treatment with either an SGLT2 inhibitor or metformin without prior exposure to either drug. To ensure data completeness, all patients were required to have at least 12 months of continuous clinical records prior to the index date, defined as the date of the first prescription. Individuals without sufficient baseline data prior to the end of the study period were excluded.
To minimize confounding, we excluded individuals with prior PD (ICD-10-CM: G20), secondary parkinsonism (G21), or anti-parkinsonian drug use (ATC: N04). Patients with neurodegenerative diseases (Alzheimer's, vascular dementia, or other dementias), substance-induced cognitive impairment, or prior glucagon-like peptide-1 receptor agonists (GLP-1 RAs, ATC code A10BJ), or dipeptidyl peptidase-4 inhibitors (DPP-4i, ATC code A10BH) exposure were also excluded due to potential neuroprotective or neurotoxic effects.
Exposure definitions
Patients newly prescribed SGLT2i (A10BK) or metformin (A10BA) were assigned to Cohort A and Cohort B, respectively. To ensure an incident user design, those with prior SGLT2i or metformin exposure or who switched treatments during follow-up were excluded. To minimize confounding, individuals with prior or concurrent GLP-1 RA or DPP-4i use were excluded, while first-generation antidiabetic agents (e.g., sulfonylureas, thiazolidinediones, alpha-glucosidase inhibitors, insulins) were adjusted for using propensity score matching (PSM) (Table 1). To ensure adequate medication exposure, patients required ≥1 year of continuous prescriptions with no extended refill gaps. An extended refill gap was defined as a period exceeding 90 consecutive days without prescription renewal after the expected coverage of the previous prescription. Those with only a single prescription and no continued use were excluded to prevent misclassification of short-term users.
Baseline characteristics of patients receiving SGLT2 inhibitors or metformin and the risk of Parkinson's disease, before and after propensity score matching.
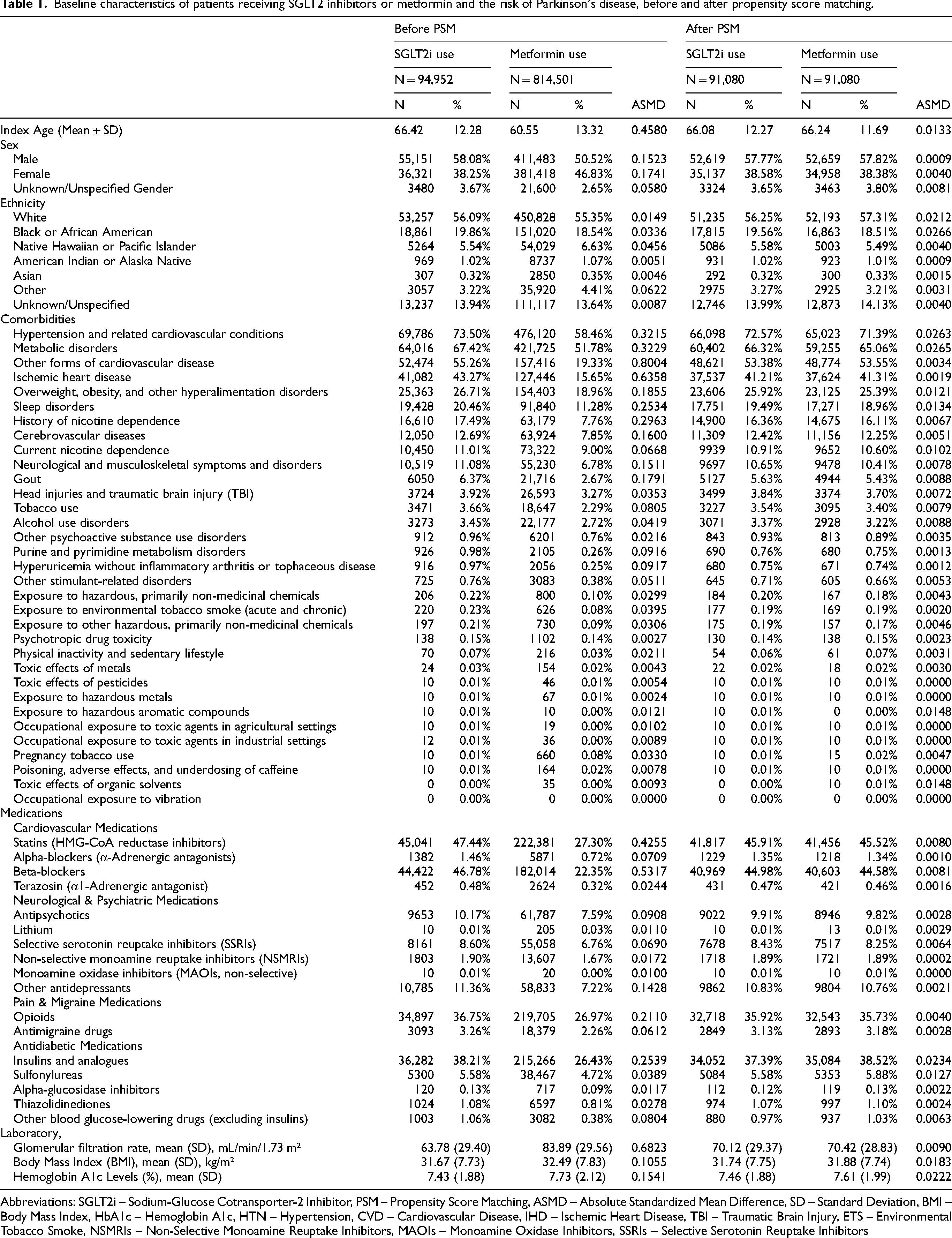
Abbreviations: SGLT2i – Sodium-Glucose Cotransporter-2 Inhibitor, PSM – Propensity Score Matching, ASMD – Absolute Standardized Mean Difference, SD – Standard Deviation, BMI – Body Mass Index, HbA1c – Hemoglobin A1c, HTN – Hypertension, CVD – Cardiovascular Disease, IHD – Ischemic Heart Disease, TBI – Traumatic Brain Injury, ETS – Environmental Tobacco Smoke, NSMRIs – Non-Selective Monoamine Reuptake Inhibitors, MAOIs – Monoamine Oxidase Inhibitors, SSRIs – Selective Serotonin Reuptake Inhibitors
Outcome measures
The primary outcome was the first recorded diagnosis of PD (ICD-10-CM: G20, G21), while all-cause mortality was analyzed as a secondary outcome to account for competing risks. Since SGLT2i users may have lower mortality rates, failing to adjust for this could underestimate their potential neuroprotective effect against PD.
To minimize reverse causation, patients diagnosed with PD within six months of treatment initiation were excluded. A total of 302 patients were excluded on this basis from the initial cohort to minimize the impact of reverse causality and immortal time bias. Follow-up began on day 181 post-index and continued until PD diagnosis, death, loss to follow-up, or study completion.
To validate our findings, we included dementia (ICD-10-CM: F01, F02, F03, G30) as a biologically plausible positive control, given its overlapping pathophysiology with PD and emerging evidence of reduced dementia risk associated with SGLT2i use in patients with type 2 diabetes.34,47 Negative controls included appendicitis, hematologic malignancies, common cold, burn injuries, and suicidal ideation—conditions with no plausible link to antidiabetic medications or PD risk. Additionally, a negative exposure control (vitamin C use)48,49 was assessed to rule out systematic biases in healthcare utilization and prescribing patterns. We acknowledge that over-the-counter use of vitamin C may not be comprehensively captured; however, its documentation in health system pharmacies or provider orders allows partial visibility within the TriNetX network. Vitamin C was used as a supplementary exposure control, rather than a definitive comparator, to help detect potential recording or utilization biases.
Statistical analysis
To minimize confounding and ensure comparability between treatment groups, we performed 1:1 nearest-neighbor PSM with a caliper of 0.2 standard deviations. Propensity scores were estimated using logistic regression incorporating a comprehensive set of covariates (Table 1), including demographic factors (age, sex, race/ethnicity), comorbidities (e.g., hypertension, ischemic heart disease, stroke), glycemic and renal markers (HbA1c, BMI, eGFR), and relevant medication use. All covariates listed in Table 1 were included in the model. Covariates were selected a priori based on clinical and epidemiologic evidence regarding PD risk factors and antidiabetic medication use, rather than through data-driven methods, to minimize overfitting and avoid collider bias. Multicollinearity was assessed using variance inflation factors (VIF), with no significant collinearity observed. 50 Patients with missing data on key covariates were excluded from the analysis. All continuous variables were summarized as mean ± standard deviation (SD), and categorical variables as counts and percentages. A complete-case approach was adopted due to the large sample size, which preserved statistical power while minimizing potential imputation bias. Neuroactive drugs (e.g., statins, beta-blockers, opioids, antipsychotics, benzodiazepines, antidepressants), environmental exposures (e.g., tobacco, alcohol, toxic agents), psychiatric disorders, and history of traumatic brain injury were also included as covariates due to their associations with PD risk.
Covariate balance was confirmed using absolute standardized mean differences (ASMDs), all <0.1 post-matching. To assess model robustness, we performed inverse probability of treatment weighting (IPTW), which yielded consistent results. Cox proportional hazards regression was used to estimate adjusted hazard ratios (aHRs) with 95% confidence intervals (CIs), with Schoenfeld residuals confirming the proportional hazards assumption. Cox models are particularly suited for long-term follow-up studies as they incorporate the dimension of time, estimating the instantaneous risk (hazard) of an outcome at any point during follow-up. This allows for more accurate modeling of chronic conditions like PD, which may develop years after initial exposure. In contrast, relative risks (RRs) were calculated as a simple measure of cumulative incidence and do not account for time-to-event or censoring. While RRs can provide an intuitive comparison of overall event proportions between groups, they are not intended for causal inference in the context of time-to-event data. To address potential reverse causation arising from the prodromal phase of PD, we conducted sensitivity analyses excluding patients diagnosed with PD within 6, 12, and 24 months after treatment initiation. We also performed competing risk analysis using Fine-Gray models and conducted subgroup analyses stratified by age, sex, glycemic control, renal function, and medication use. Positive and negative control outcomes were evaluated to detect potential residual confounding.
All statistical tests were two-sided, with significance set at p < 0.05. To account for multiple comparisons in secondary and exploratory analyses, the Benjamini-Hochberg false discovery rate correction was applied.
Results
Baseline characteristics of the study cohort
Table 1 summarizes the baseline characteristics of patients receiving SGLT2i or metformin, both before and after PSM. Before matching, the SGLT2i group was older (66.42 ± 12.28 vs. 60.55 ± 13.32 years; ASMD = 0.4580) and had higher comorbidity burdens, including hypertension (73.50% vs. 58.46%; ASMD = 0.3215), ischemic heart disease (43.27% vs. 15.65%; ASMD = 0.6358), metabolic disorders (67.42% vs. 51.78%; ASMD = 0.3229), and cerebrovascular disease (12.69% vs. 7.85%; ASMD = 0.1600). They also had higher nicotine dependence (17.49% vs. 7.76%; ASMD = 0.2963) and sleep disorders (20.46% vs. 11.28%; ASMD = 0.2534). Before matching, males comprised 58.08% of the SGLT2i group and 50.52% of the metformin group (ASMD = 0.1523). After PSM, sex distribution was well balanced (57.77% vs. 57.82% male; ASMD = 0.0009).
After PSM, a well-balanced cohort was achieved (ASMD < 0.1 for all variables), with 91,080 patients per group. The matched mean age was similar (66.08 ± 12.27 vs. 66.24 ± 11.69 years; ASMD = 0.0133), along with comorbidities such as hypertension (72.57% vs. 71.39%; ASMD = 0.0263), ischemic heart disease (41.21% vs. 41.31%; ASMD = 0.0019), and metabolic disorders (66.32% vs. 65.06%; ASMD = 0.0265).
Risk of PD and all-cause mortality
The incidence of PD was 240 out of 91,080 (0.26%) in the SGLT2i group and 772 out of 91,080 (0.85%) in the metformin group. Among PD cases, the mean time from treatment initiation to diagnosis was 987.4 ± 603.2 days for SGLT2i users and 944.7 ± 591.5 days for metformin users. For the primary outcome, the incidence of PD was significantly lower in the SGLT2i group compared to the metformin group (0.26% vs. 0.85%) (Table 2). The AHR for PD in SGLT2i users was 0.72 (95% CI, 0.62–0.84), indicating a 28% lower risk compared to metformin users. The RR was 0.31 (95% CI, 0.27–0.36), confirming a substantial reduction in PD incidence.
Risk of Parkinson's disease and all-cause mortality in patients receiving SGLT2 inhibitors vs. metformin after propensity score matching.

Abbreviations: SGLT2i – Sodium-Glucose Cotransporter-2 Inhibitor, PSM – Propensity Score Matching, AHR – Adjusted Hazard Ratio, CI – Confidence Interval
For the secondary outcome, all-cause mortality was significantly lower in the SGLT2i group compared to the metformin group (8.10% vs. 18.15%). The AHR for all-cause mortality was 0.85 (95% CI, 0.83–0.89), suggesting a 15% reduction in mortality risk among SGLT2i users. The corresponding RR was 0.45 (95% CI, 0.43–0.46), further supporting the survival benefit associated with SGLT2i treatment.
These findings highlight a potential neuroprotective effect of SGLT2is, with a lower risk of PD and a reduced competing risk of mortality compared to metformin.
Sensitivity analysis of reverse causation
In sensitivity analyses excluding PD diagnoses occurring within 6, 12, and 24 months after treatment initiation, the adjusted hazard ratios (aHRs) remained consistent: 0.72 (95% CI, 0.62–0.84), 0.73 (95% CI, 0.61–0.86), and 0.71 (95% CI, 0.58–0.87), respectively, supporting the robustness of the observed association. Full results are provided in Supplemental Table 1.
Kaplan-Meier analysis of PD incidence and overall survival
Figure 1 shows that PD incidence was significantly lower in the SGLT2i group than in the metformin group, with the survival curves diverging early and continuing to separate over time. The log-rank test confirmed a statistically significant difference, supporting a potential neuroprotective effect of SGLT2is.
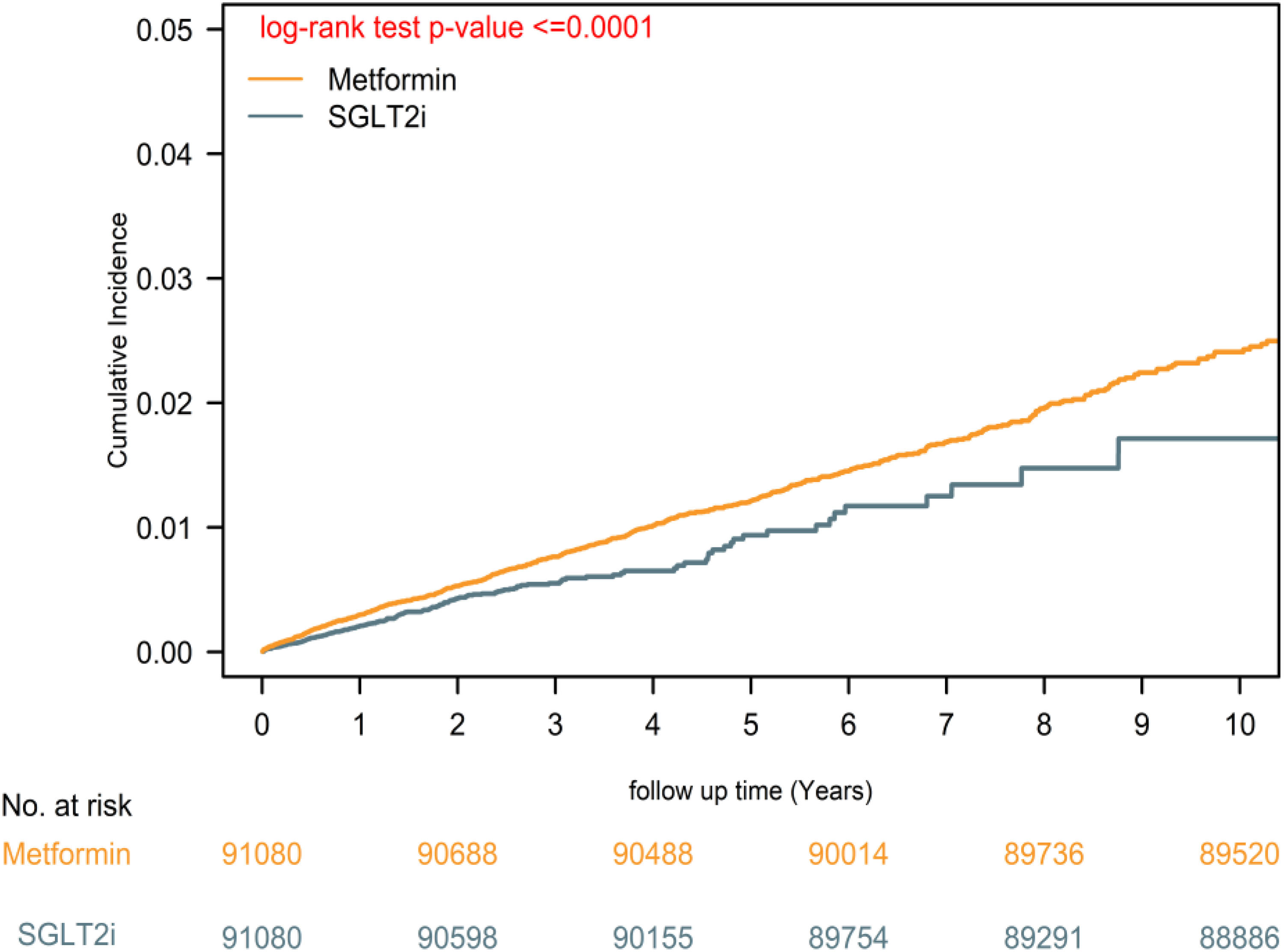
Kaplan–Meier estimates of Parkinson's disease incidence in patients receiving SGLT2 inhibitors vs. metformin. Notes: Although the matched cohort included the same number of individuals in both groups (n = 91,080), the metformin group experienced a substantially higher number of PD events (772 vs. 240). This accounts for the smoother appearance of the metformin curve. The SGLT2i curve appears more stepwise due to fewer observed events. The number-at-risk table confirms that population sizes remained balanced over time.
Supplemental Figure 1 demonstrates a significant survival advantage for SGLT2i users, with lower all-cause mortality compared to metformin. The log-rank test confirmed a statistically significant difference, indicating that SGLT2is reduce both PD risk and mortality, addressing competing risk concerns.
Subgroup analysis of PD risk
Figure 2 presents a subgroup analysis of the association between SGLT2i use and PD risk across key demographic and clinical variables. The protective effect of SGLT2i compared to metformin was observed in most subgroups, with adjusted hazard ratios (aHRs) consistently favoring SGLT2i.
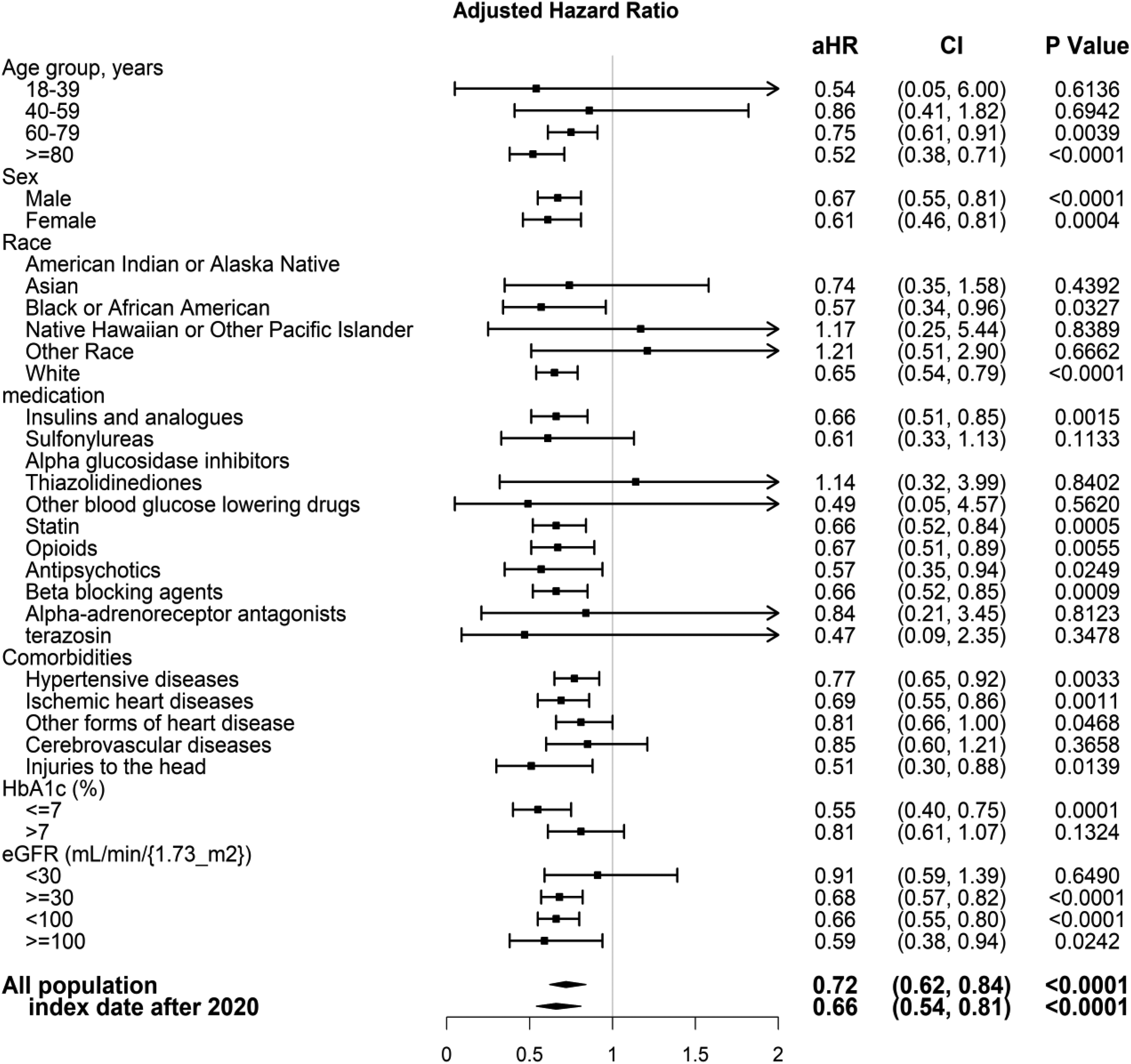
Subgroup analysis of SGLT2 inhibitors vs. metformin on Parkinson's disease risk. Abbreviations: aHR – Adjusted Hazard Ratio, CI – Confidence Interval, HbA1c – Hemoglobin A1c, eGFR – Estimated Glomerular Filtration Rate.
When stratified by ethnicity, significant associations were found in White (aHR, 0.65; 95% CI, 0.54–0.79) and Asian (aHR, 0.57; 95% CI, 0.34–0.96) patients. However, no significant association was observed in Black or African American (aHR, 1.17; 95% CI, 0.25–5.44) or American Indian/Alaska Native (aHR, 0.74; 95% CI, 0.35–1.58) subgroups. These nonsignificant results likely reflect limited statistical power due to small sample sizes or differential SGLT2i prescribing patterns, rather than a true absence of effect. The wide confidence intervals support this interpretation.
The association was particularly pronounced in older adults (≥60 years), individuals with HbA1c ≥ 7.5%, and those with impaired renal function (eGFR <60 mL/min/1.73 m²), suggesting stronger neuroprotective effects in clinically vulnerable populations. The protective association persisted regardless of sex or concurrent statin use, indicating robustness across diverse patient groups.
These findings highlight the potential generalizability of the neuroprotective benefit of SGLT2is while emphasizing the importance of evaluating effect modification across ethnic groups. Future studies using larger, multi-ethnic datasets are warranted to confirm these differential effects and explore underlying mechanisms.
Validation analysis using positive and negative controls
The primary outcome analysis confirmed that SGLT2i use was associated with a significantly lower risk of PD compared to metformin, consistent with the findings in the main analysis. The positive control outcome (dementia) showed a similar protective association, supporting the validity of the neuroprotective effect observed with SGLT2i.
For negative outcome controls (acute appendicitis, hematologic malignancies, common cold, burn injuries, and suicidal ideation or attempts), no significant associations were observed, reinforcing that the primary findings were not due to residual confounding or systematic biases. The negative exposure control (vitamin C use) also showed no association with PD risk, further supporting the specificity of the SGLT2i effect.
These findings strengthen the causal inference that SGLT2i may confer a protective effect against PD, independent of confounding factors or competing risks.
Discussion
This is the first large-scale real-world study to directly compare SGLT2is and metformin in relation to PD risk among patients with T2DM. Using a 20-year dataset, SGLT2i use was associated with a 28% lower risk of PD (aHR, 0.72; 95% CI, 0.62–0.84) and a markedly lower relative risk (RR, 0.31; 95% CI, 0.27–0.36) (Table 2). After PSM, baseline characteristics were well balanced, supporting the validity of the comparison. SGLT2is also conferred lower all-cause mortality (aHR, 0.85; RR, 0.45), suggesting a survival benefit that helps mitigate bias from competing risk.
Kaplan-Meier analysis (Figure 1) demonstrated an early and sustained divergence in PD incidence, favoring SGLT2is over metformin. While both RRs and aHRs indicated consistent neuroprotective associations of SGLT2is versus metformin, we observed numerical discrepancies between these measures, particularly for all-cause mortality. This reflects fundamental differences in interpretation: RRs summarize cumulative incidence over the entire follow-up, whereas aHRs from Cox models reflect instantaneous risk at any given time point. For PD, the early and persistent separation of Kaplan-Meier curves (Figure 1) yielded concordant estimates (RR = 0.31; aHR = 0.72). In contrast, survival curves for all-cause mortality remained similar for over three years (Figure S1) before diverging, resulting in a more modest aHR (0.85) despite a substantial RR (0.45). This temporal separation likely reflects non-proportional hazards, a common feature in long-term follow-up studies, and underscores the complementary value of reporting both RRs and aHRs in pharmacoepidemiologic research. Supplemental analysis (Figure 1S) confirmed a survival benefit, addressing competing mortality. Subgroup analyses (Figure 2) revealed significant PD risk reduction among patients ≥60 years, those with HbA1c ≥ 7.5%, and impaired renal function, suggesting greater neuroprotection in high-risk populations. Nonsignificant findings in younger subgroups likely reflect fewer PD events and limited power. Positive and negative control analyses (Figure 3) confirmed robustness—dementia risk was reduced, while unrelated outcomes showed no association. The use of vitamin C as a negative exposure control further supported specificity. These findings challenge metformin's presumed neuroprotective role and highlight SGLT2is as a potentially superior option for PD risk reduction in T2DM, warranting further investigation.
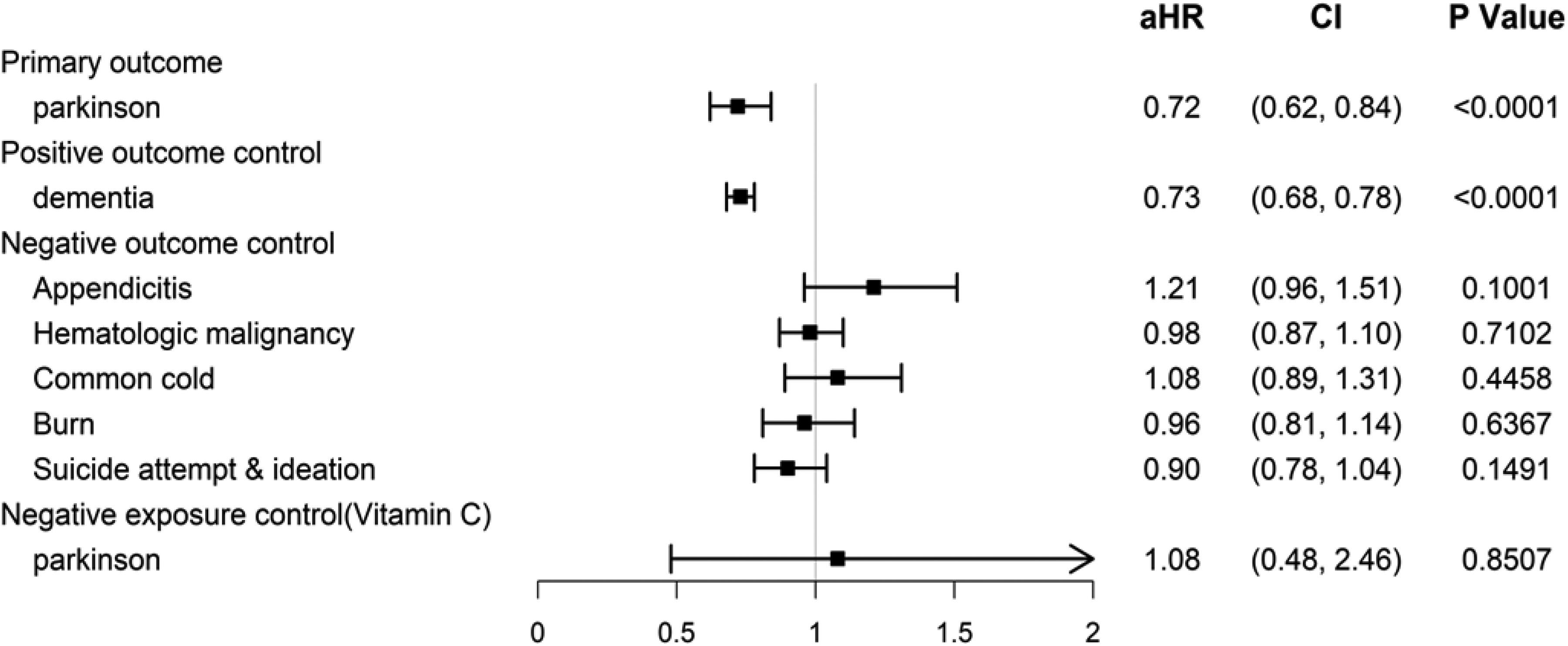
Subgroup analysis of SGLT2 inhibitors vs. metformin on primary, positive, and negative outcome controls, and negative exposure control. Abbreviations: aHR – Adjusted Hazard Ratio, CI – Confidence Interval.
SGLT2is may provide superior neuroprotection against PD in patients with T2DM compared to metformin due to their broader mechanistic effects beyond glycemic control.11,28,30,32,36 While metformin has well-documented neuroprotective properties, primarily mediated through AMPK activation, mitochondrial preservation, and anti-inflammatory effects,21,24–27,39–43 our findings suggest that SGLT2is confer additional benefits that may more effectively mitigate diabetes-related neurodegeneration. SGLT2is directly reduce oxidative stress, a key driver of PD pathology, by limiting reactive oxygen species (ROS) production, restoring mitochondrial function, and modulating NADPH oxidase activity.11,38,51,52 Furthermore, SGLT2is enhance autophagy-lysosomal pathways, crucial for clearing α-synuclein aggregates, a hallmark of PD.37,53–55 Unlike metformin, which primarily improves metabolic health, SGLT2is have been shown to increase ketone body production, providing an alternative and neuroprotective energy substrate for dopaminergic neurons.11,38,56,57 Importantly, our study demonstrated that even after widespread adoption post-2020 (Figure 2), SGLT2is continued to exhibit a stronger protective association against PD (aHR = 0.66, 95% CI: 0.54–0.81, p < 0.0001), suggesting that increased clinical familiarity and optimized prescribing further enhance their benefits. SGLT2i users exhibited a significantly lower risk of both PD (aHR = 0.72, 95% CI: 0.62–0.84, p < 0.0001) and dementia (aHR = 0.73, 95% CI: 0.68–0.78, p < 0.0001), reinforcing their broader neuroprotective benefits. Given their well-established cardiovascular, renal, and emerging neuroprotective benefits, 58 SGLT2is may warrant prioritization as a first-line therapy for T2DM patients at risk for neurodegeneration, pending further prospective studies to validate their potential in PD prevention.
While prior studies—such as Mendelian randomization analyses—have explored the neurodegenerative effects of SGLT1/2 inhibition, 59 they differ in methodology and do not reflect real-world exposure to SGLT2is. Our findings, based on large-scale observational data with robust confounder control, provide complementary evidence. Discrepancies may be due to confounding by indication,1,59 reverse causality from prodromal PD influencing treatment decisions, 2 and surveillance bias from more frequent monitoring in SGLT2i users. 6 We addressed these by implementing rigorous PSM, excluding PD diagnoses within six months of treatment initiation, accounting for competing mortality risks, and validating findings with positive and negative control outcomes. Unlike prior studies with heterogeneous designs and short follow-up, our 20-year real-world analysis supports SGLT2is as offering superior protection against PD compared to metformin, highlighting the need for prospective confirmation.
This study leverages a 20-year real-world dataset from 142 healthcare organizations, offering robust power and generalizability. A strict new-user design, exclusion of prior SGLT2i or metformin exposure, and propensity score matching with comprehensive clinical, metabolic, and lifestyle covariates minimized confounding. Use of an active comparator (SGLT2i vs. metformin) mitigated healthy user bias more effectively than comparisons with non-users. 60 Control outcomes (Figure 3), competing risk models, and subgroup analyses (Figure 2) further validated findings. Temporal stratification revealed a stronger protective effect post-2020 (aHR = 0.66; 95% CI, 0.54–0.81), likely reflecting improved prescribing practices. Adherence was ensured by requiring ≥1 year of continuous prescriptions without extended refill gaps, minimizing exposure misclassification and reinforcing the reliability of long-term effect estimates.
Despite rigorous adjustments, residual confounding cannot be excluded given the non-randomized nature of treatment assignment. Large-scale observational studies remain essential for real-world evidence, especially in pharmacovigilance and long-term safety evaluation. Reliance on ICD-10-CM coding may introduce misclassification, though this risk is minimized through validated case definitions, strict exclusions, and extended follow-up. The negative exposure control (vitamin C) may be under-recorded due to its over-the-counter status but serves as a useful proxy for detecting systematic biases in healthcare utilization. Future studies should incorporate biomarkers, neuroimaging, and genetic profiling to enhance diagnostic accuracy and elucidate SGLT2is’ neuroprotective mechanisms. Despite these limitations, our study offers one of the most comprehensive real-world assessments of SGLT2i use and PD risk, supporting the need for prospective validation.
Conclusions
Our study provides real-world evidence that SGLT2is are associated with a 28% lower risk of PD compared to metformin in T2DM patients. Leveraging a large-scale, 20-year dataset and rigorous PSM, our findings remained robust across sensitivity analyses, particularly in the post-2020 cohort. While RCTs are the gold standard for causal inference, ethical and feasibility constraints highlight the importance of real-world studies in informing clinical decisions. These results warrant further prospective investigations into the potential neuroprotective benefits of SGLT2is.
Supplemental Material
sj-docx-1-pkn-10.1177_1877718X251359391 - Supplemental material for SGLT2 inhibitors vs. metformin for Parkinson's disease risk reduction in type 2 diabetes
Supplemental material, sj-docx-1-pkn-10.1177_1877718X251359391 for SGLT2 inhibitors vs. metformin for Parkinson's disease risk reduction in type 2 diabetes by Mingyang Sun, Xiaoling Wang, Zhongyuan Lu, Yitian Yang, Shuang Lv, Mengrong Miao, Wan-Ming Chen, Szu-Yuan Wu and Jiaqiang Zhang in Journal of Parkinson's Disease
Footnotes
Abbreviations
Ethics approval and consent
This study utilized the TriNetX platform, which exclusively provides de-identified and aggregated electronic health record data. In accordance with the Health Insurance Portability and Accountability Act (HIPAA), such data do not allow for individual identification and thus do not meet the federal definition of human subjects research. Consequently, research conducted using this platform is exempt from Institutional Review Board (IRB) oversight and does not require informed consent.
Author contributions
Funding
This work was supported by the National Key Research and Development Program of China (Funding Number: 2023YFC2506900), which funded Zhang's research, and by the Lo-Hsu Medical Foundation, Lotung Poh-Ai Hospital, which supported the work of Szu-Yuan Wu (Funding Numbers: 11403 and 11404).
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Guarantor statement
Prof. Jiaqiang Zhang is the guarantor of this work and, as such, had full access to all the data in the study and takes responsibility for the integrity of the data and the accuracy of the data analysis.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
