Abstract
Background
Freezing of gait (FOG) is a common and disabling symptom in patients with Parkinson's disease (PD), significantly impairing motor function and quality of life. While traditional deep brain stimulation (DBS) of the subthalamic nucleus (STN) provides some benefits, its efficacy in alleviating FOG remains limited. Combined stimulation of the STN and the substantia nigra pars reticulata (SNr) has recently emerged as a potentially superior approach.
Objective
To compare the efficacy of STN-only stimulation and combined STN + SNr stimulation in improving FOG symptoms and quality of life in patients with PD.
Methods
This multicenter, prospective, randomized, crossover study was conducted between May 2020 and May 2024 and enrolled patients with PD and significant FOG. All participants received bilateral DBS electrode implantation. Each subject sequentially underwent both STN-only and combined STN + SNr stimulation conditions according to a randomized crossover schedule, with each stimulation period lasting for six months. Outcome assessments, including the Freezing of Gait Questionnaire (FOG-Q) and the Parkinson's Disease Questionnaire Summary Index (PDQ-SI), were conducted at baseline, 6, 12, and 18 months. Data were analyzed using mixed-model repeated-measures analysis of variance, followed by post hoc Bonferroni-adjusted pairwise comparisons to account for the crossover design.
Results
Compared to STN-only stimulation, combined STN + SNr stimulation resulted in significantly greater improvement in FOG-Q scores and PDQ-SI scores. The benefits were observed at each assessment following the switch to combined stimulation.
Conclusions
Combined STN and SNr stimulation was more effective than STN-only stimulation in alleviating FOG symptoms and improving quality of life in patients with PD. This dual-target DBS approach may represent a promising therapeutic strategy for managing refractory FOG in PD.
Keywords
Introduction
Parkinson's disease (PD) is a prevalent neurodegenerative disorder characterized by bradykinesia, tremor, rigidity, and various gait disturbances, among which freezing of gait (FOG) is particularly disabling.1,2 FOG is defined as a sudden, brief inability to initiate or maintain forward movement of the feet, often occurring during gait initiation, turning, or when navigating narrow spaces. 3 This symptom significantly increases the risk of falls and leads to substantial deterioration in quality of life for affected individuals.4,5
Although pharmacological therapy remains the mainstay of PD management, its efficacy in treating FOG is often limited, especially in advanced disease stages where patients may become resistant to dopaminergic medications.5–7 Deep brain stimulation (DBS) targeting the subthalamic nucleus (STN) is an established surgical intervention that can substantially improve tremor, rigidity, and bradykinesia by modulating abnormal basal ganglia circuitry.7,8 However, the impact of conventional STN-DBS on FOG is inconsistent, and a significant proportion of patients continue to experience FOG despite optimal stimulation.6,9
Recent research has explored the potential of dual-target DBS strategies, particularly the combined stimulation of the STN and the substantia nigra pars reticulata (SNr), to address refractory FOG. 10 The SNr, a major output nucleus of the basal ganglia, plays a critical role in gait regulation and locomotor control. 11 Dysfunctional SNr output has been implicated in the pathophysiology of FOG, as excessive inhibitory output from the SNr to brainstem locomotor centers may contribute to gait freezing. 12 It is hypothesized that concurrent stimulation of both STN and SNr may more effectively restore normal gait patterns by modulating both the input and output nodes of the basal ganglia-thalamo-cortical circuits. 13
Preliminary studies and small clinical series have suggested that dual-target DBS may provide greater improvement in FOG than conventional STN stimulation alone, though robust comparative data remain limited. Most existing research has focused on general motor outcomes or single-target stimulation, with few large-scale, systematic studies specifically evaluating the benefits of dual-target approaches for FOG. Thus, further investigation is needed to clarify the therapeutic value of combined STN and SNr stimulation in this challenging population.
The primary aim of this multicenter, prospective, randomized, crossover study is to compare the efficacy of combined STN and SNr stimulation versus conventional STN-only stimulation in alleviating FOG in patients with PD. Secondary objectives include evaluating the effects of these approaches on overall motor function, quality of life (QoL), and safety profiles. We hypothesize that dual-target stimulation will result in superior improvement in FOG and QoL compared to STN-only stimulation. By systematically evaluating these two approaches, this study seeks to provide evidence-based guidance for optimizing DBS strategies in PD patients with refractory FOG.
FOG remains a major unmet need in PD management. As an emerging strategy, combined DBS targeting the STN and SNr may offer a promising avenue for patients unresponsive to conventional treatment. The findings of this study may contribute to refining DBS protocols and improving clinical outcomes for individuals with PD.
Methods
Clinical data
This multicenter, prospective, randomized, crossover study included 24 patients diagnosed with Parkinson's disease (PD) and experiencing freezing of gait (FOG), who underwent DBS targeting the subthalamic nucleus (STN) and the SNr at Guangyuan Central Hospital, Dazhou Central Hospital, Guang'an People's Hospital, and Yibin First People's Hospital between May 2020 and May 2024. Detailed demographic and clinical characteristics of the participants are presented in Table 1.
Clinical characteristics and DBS treatment-related data.
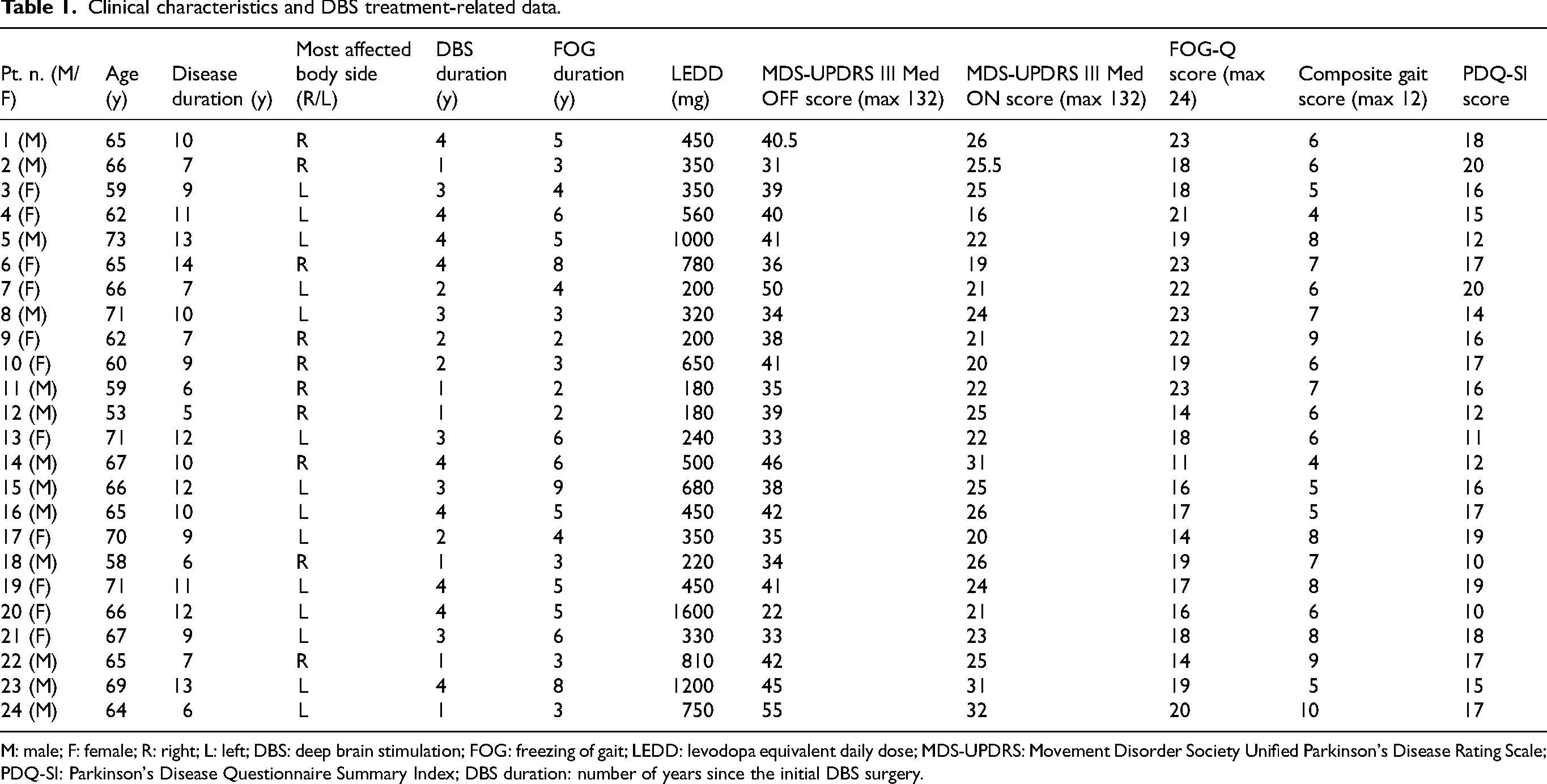
M: male; F: female; R: right; L: left; DBS: deep brain stimulation; FOG: freezing of gait; LEDD: levodopa equivalent daily dose; MDS-UPDRS: Movement Disorder Society Unified Parkinson's Disease Rating Scale; PDQ-SI: Parkinson's Disease Questionnaire Summary Index; DBS duration: number of years since the initial DBS surgery.
Inclusion criteria
(1) Diagnosis of Parkinson's disease according to the Movement Disorder Society Clinical Diagnostic Criteria; (2) Presence of significant FOG, defined as a Freezing of Gait Questionnaire (FOG-Q) score ≥10, with patients exhibiting off FOG, on FOG, or on-off FOG phenotypes; (3) Age 40–80 years and disease duration ≥5 years; (4) Clear surgical indication for DBS therapy, defined as disabling motor fluctuations, dyskinesias, or medication-refractory motor symptoms, and a positive response to levodopa challenge (>30% improvement in UPDRS-III score).
Exclusion criteria
(1) Presence of other severe neurological or psychiatric disorders, including: Cognitive impairment, defined as a Mini-Mental State Examination score <24;Major depressive disorder, defined as a diagnosis by Diagnostic and Statistical Manual of Mental Disorders, Fifth Edition (DSM-5) criteria. or a Hamilton Depression Rating Scale score >17; (2) Inability to tolerate surgery or high risk for postoperative complications; (3) Medical or neurological conditions other than PD that significantly interfere with gait; (4) Severe systemic diseases (e.g., cardiac, pulmonary, or hepatic dysfunction).
Study protocol
All enrolled patients underwent bilateral implantation of DBS electrodes targeting the STN, with electrode extension to the SNr. Intraoperative microelectrode recordings were used to validate target coordinates and ensure electrode coverage of both STN and SNr regions at different depths. Postoperative imaging confirmed accurate electrode placement. Electrode impedance and stimulation testing were performed to verify device functionality. All procedures were conducted by an experienced neurosurgical team following standardized protocols.
Detailed individual stimulation parameters and clinical outcomes for each participant in both intervention periods are provided in Table 2. This includes electrode contact configurations, voltage, frequency, pulse width, and corresponding changes in clinical scales.
Treatment effect data for Group A (maintaining the original target) and Group B (initiating dual-target stimulation).
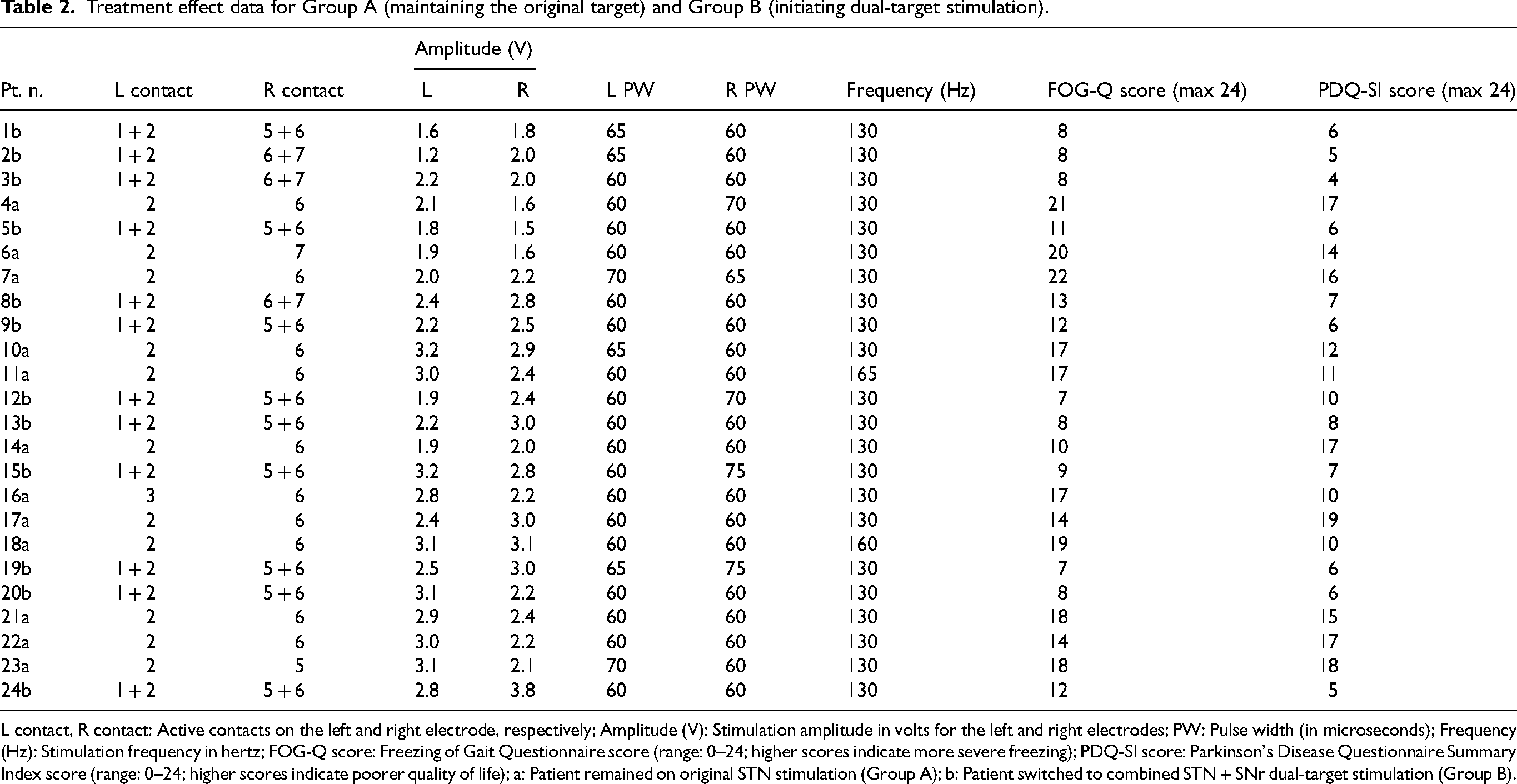
L contact, R contact: Active contacts on the left and right electrode, respectively; Amplitude (V): Stimulation amplitude in volts for the left and right electrodes; PW: Pulse width (in microseconds); Frequency (Hz): Stimulation frequency in hertz; FOG-Q score: Freezing of Gait Questionnaire score (range: 0–24; higher scores indicate more severe freezing); PDQ-SI score: Parkinson's Disease Questionnaire Summary Index score (range: 0–24; higher scores indicate poorer quality of life); a: Patient remained on original STN stimulation (Group A); b: Patient switched to combined STN + SNr dual-target stimulation (Group B).
Randomization and crossover design
At six months postoperatively, all patients were randomly assigned (1:1) to one of two groups by an independent third party using a computer-generated randomization table.
Group A: Received single-target STN stimulation. Group B: Received dual-target STN + SNr stimulation. Randomization details were blinded to both patients and evaluators.
At 12 months, the stimulation protocols were crossed over:
Group A switched to dual-target STN + SNr stimulation. Group B switched to single-target STN stimulation.
All patients were evaluated at baseline (post-surgery), 6 months, 12 months, and 18 months.
Outcome measures
Primary outcomes:
Severity of FOG, assessed by the FOG-Q score. Quality of life, evaluated by the Parkinson's Disease Questionnaire-39 (PDQ-39) Summary Index.
Secondary outcomes:
Motor symptoms, assessed by the Unified Parkinson's Disease Rating Scale Part III (UPDRS-III). Adverse events related to DBS.
All assessments were performed in the medication-off state by trained, blinded evaluators following standardized protocols. Only validated clinical scales were used; objective measurements with wearable devices were not employed in this study. Future studies may integrate wearable technology for additional quantitative assessment.
In addition to individual-level data, group-level summaries of stimulation parameters and associated clinical scores are shown in Table 3. This table highlights mean voltage, frequency, and pulse width settings across the two groups, as well as their average changes in FOG-Q, PDQ-39 SI, and UPDRS-III scores at each time point.
Stimulation parameters and clinical scores for Group A (dual-target activation) and Group B (single-target DBS).
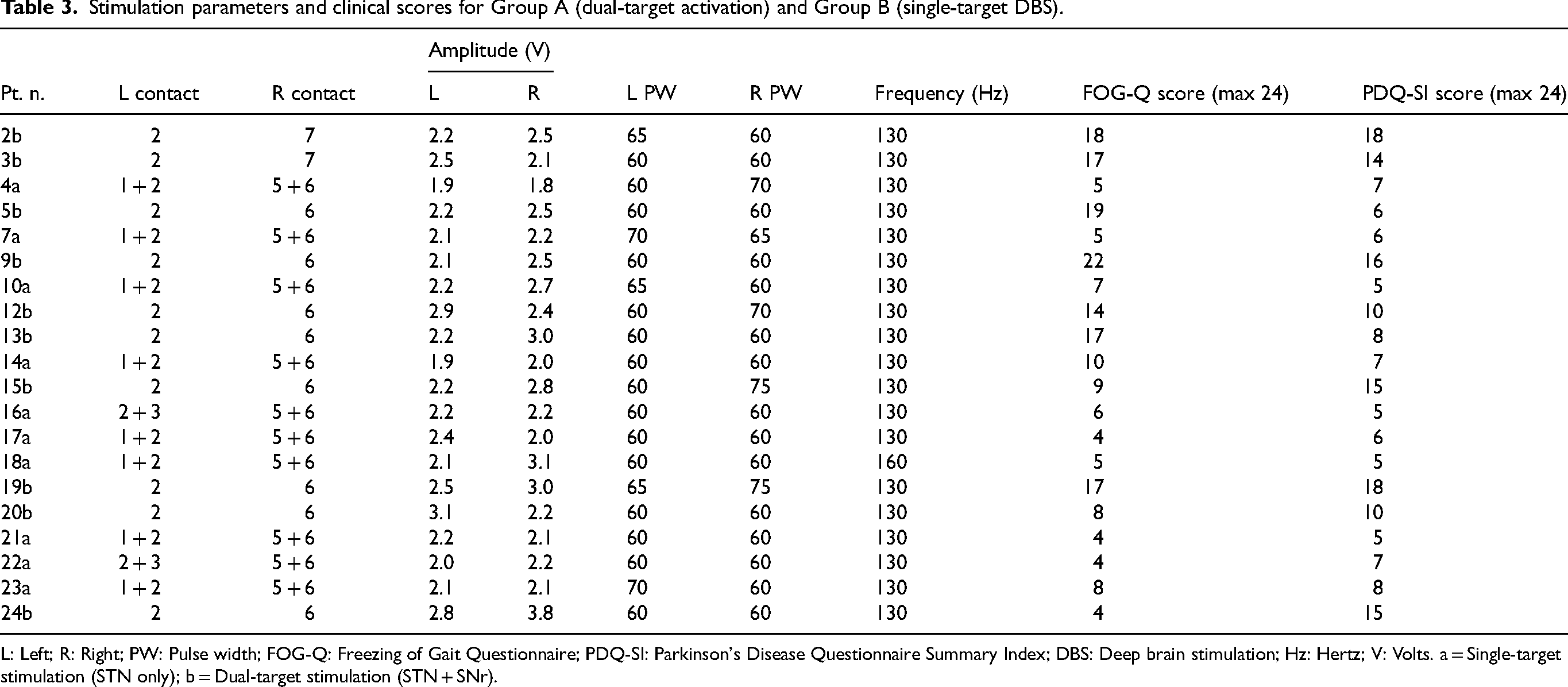
L: Left; R: Right; PW: Pulse width; FOG-Q: Freezing of Gait Questionnaire; PDQ-SI: Parkinson's Disease Questionnaire Summary Index; DBS: Deep brain stimulation; Hz: Hertz; V: Volts. a = Single-target stimulation (STN only); b = Dual-target stimulation (STN + SNr).
Statistical analysis
All statistical analyses were conducted using SPSS 27.0 (IBM Corp., Armonk, NY) and MATLAB R2024b (MathWorks Inc., Natick, MA). Continuous variables were expressed as mean ± standard deviation (SD) or median (interquartile range, IQR) based on data distribution. Between-group comparisons were performed using independent samples t-tests or Mann–Whitney U tests, while within-group comparisons across time points were assessed using paired t-tests or Wilcoxon signed-rank tests as appropriate.
Repeated-measures analysis of variance was used to evaluate the interaction effect between stimulation type (STN-only vs. STN&SNr), time (baseline, 6 months, 12 months, 18 months), and their interaction on primary and secondary outcomes. Post hoc pairwise comparisons were adjusted using Bonferroni correction.
To assess potential nonlinear relationships between stimulation parameters (e.g., voltage, frequency) and clinical outcomes (e.g., ΔFOG-Q score), curve fitting analysis was conducted using second-order polynomial regression models. Model fit was evaluated by the coefficient of determination (R²) and statistical significance of the regression coefficients.
Categorical variables, including adverse events, were analyzed using chi-square test or Fisher's exact test when appropriate. A two-tailed p-value < 0.05 was considered statistically significant.
Results
A total of 24 patients were enrolled in this study, of whom 20 (83.3%) completed all scheduled follow-up assessments. Four patients discontinued participation: two due to stimulation-related adverse events, one due to inability to complete gait assessments, and one due to poor tolerance to stimulation settings. All analyzed patients met the inclusion criteria, and there were no significant between-group differences in baseline demographic or clinical characteristics (Table 1).
Primary outcomes
Repeated-measures ANOVA was performed to examine the effects of stimulation mode (STN-only vs. STN + SNr), time (6, 12, and 18 months), and their interaction on FOG-Q and PDQ-39 Summary Index (PDQ-SI) scores.
For FOG-Q scores, there were significant main effects of stimulation type (F(1,19) = 16.21, p = 0.001) and time (F(2,38) = 11.92, p < 0.001), as well as a significant stimulation-by-time interaction (F(2,38) = 7.48, p = 0.002), indicating that dual-target stimulation yielded greater improvements in gait freezing across time compared with STN-only stimulation.
Similarly, PDQ-SI scores showed significant main effects of stimulation type (F(1,19) = 13.07, p = 0.002) and time (F(2,38) = 9.65, p = 0.001), along with a significant interaction effect (F(2,38) = 6.13, p = 0.005), reflecting superior quality-of-life outcomes with STN + SNr stimulation.
Group-level trends over time are detailed in Tables 2 and 3 and visualized in Figures 1–3. Notably, the FOG-Q score decreased more markedly under the dual-target condition, particularly at 12 and 18 months (Figures 1 and 2). Figure 3 presents boxplots demonstrating consistent reduction in FOG-Q scores across both stimulation paradigms.
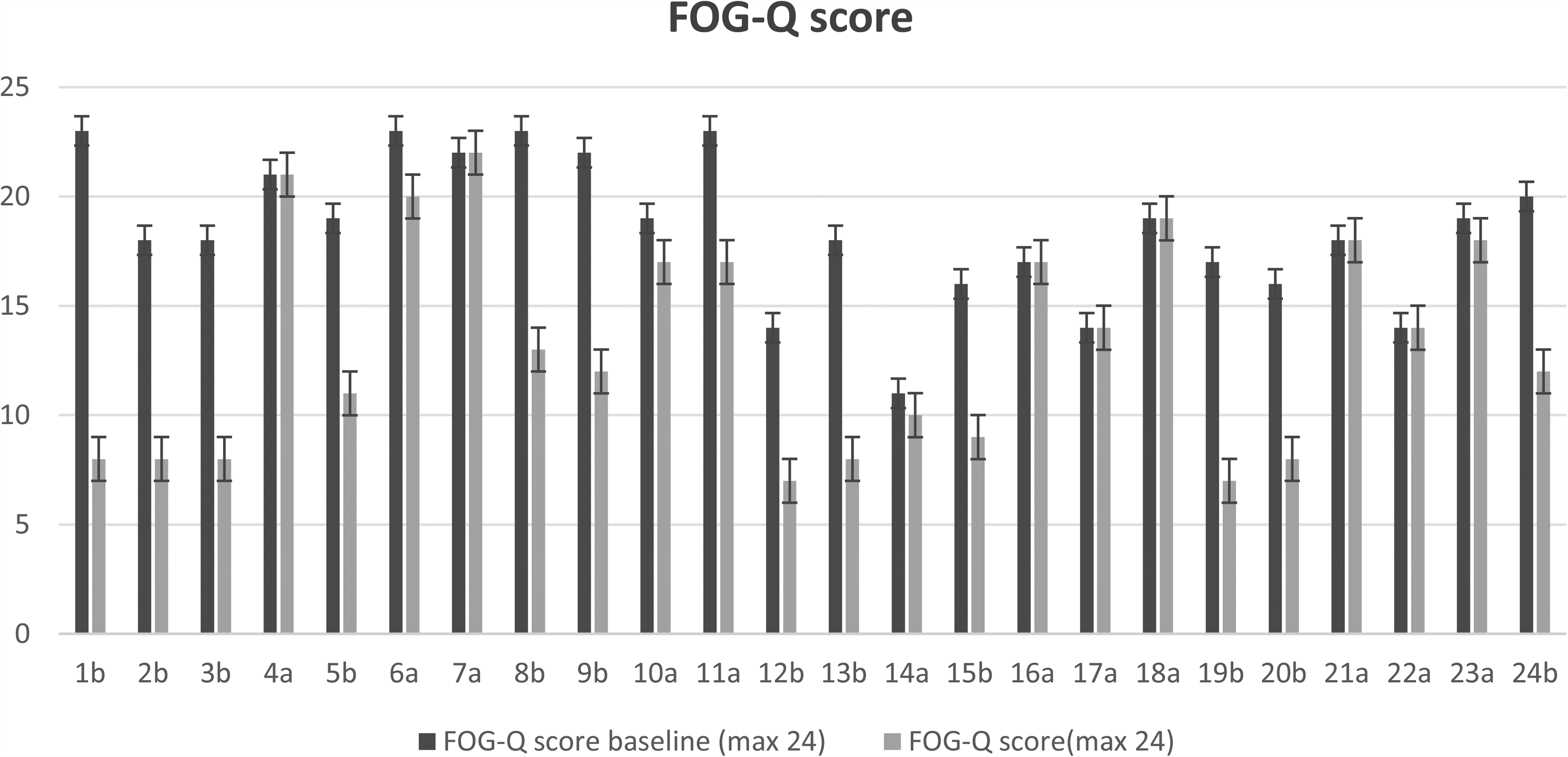
(a) Group A maintains the original target; (b) Group B initiates dual targets, improvement in FOG-Q scores.
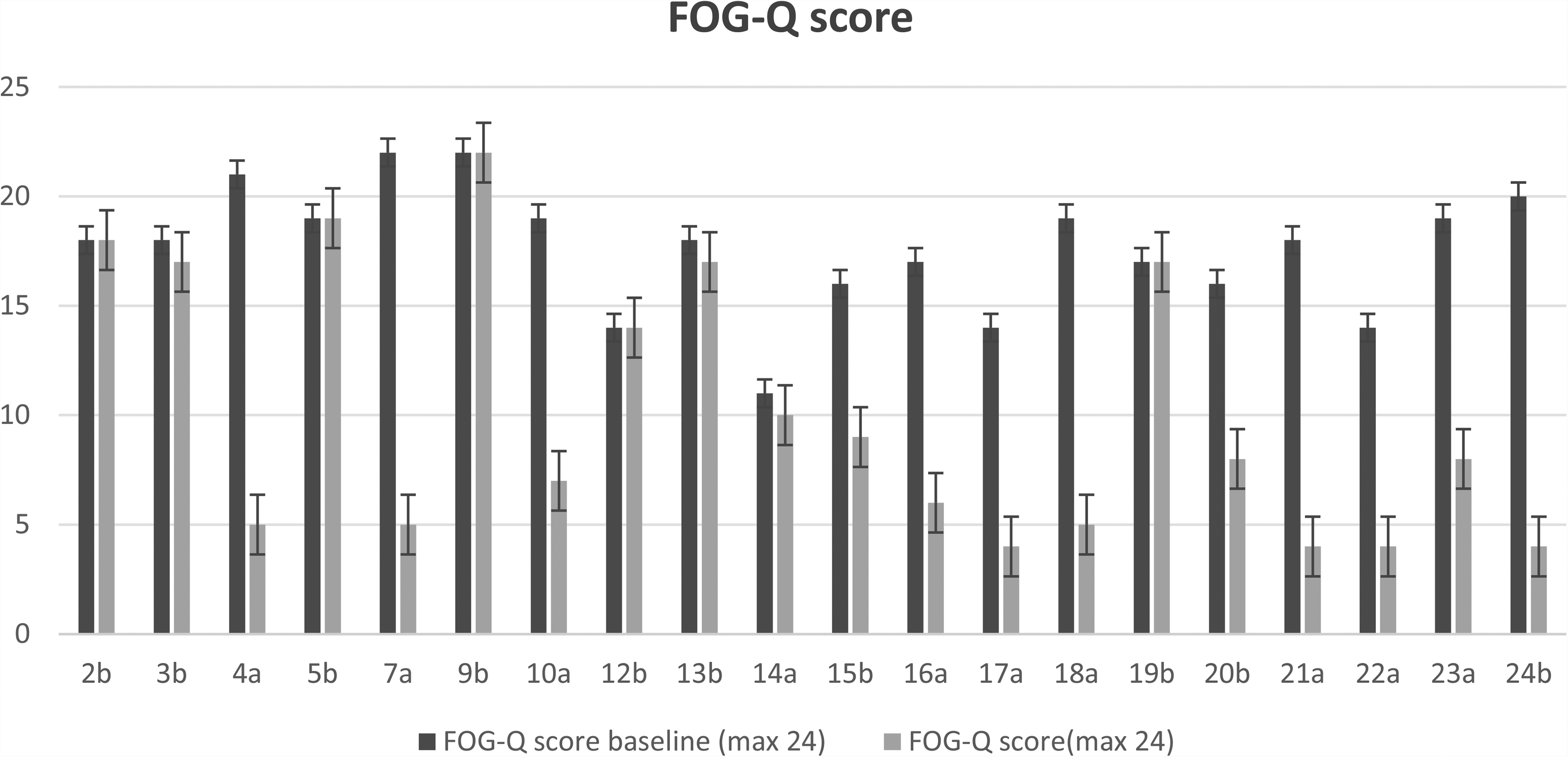
FOG Improvement Scores for Group A with Dual-Target DBS Activation and Group B with Single-Target DBS Activation.
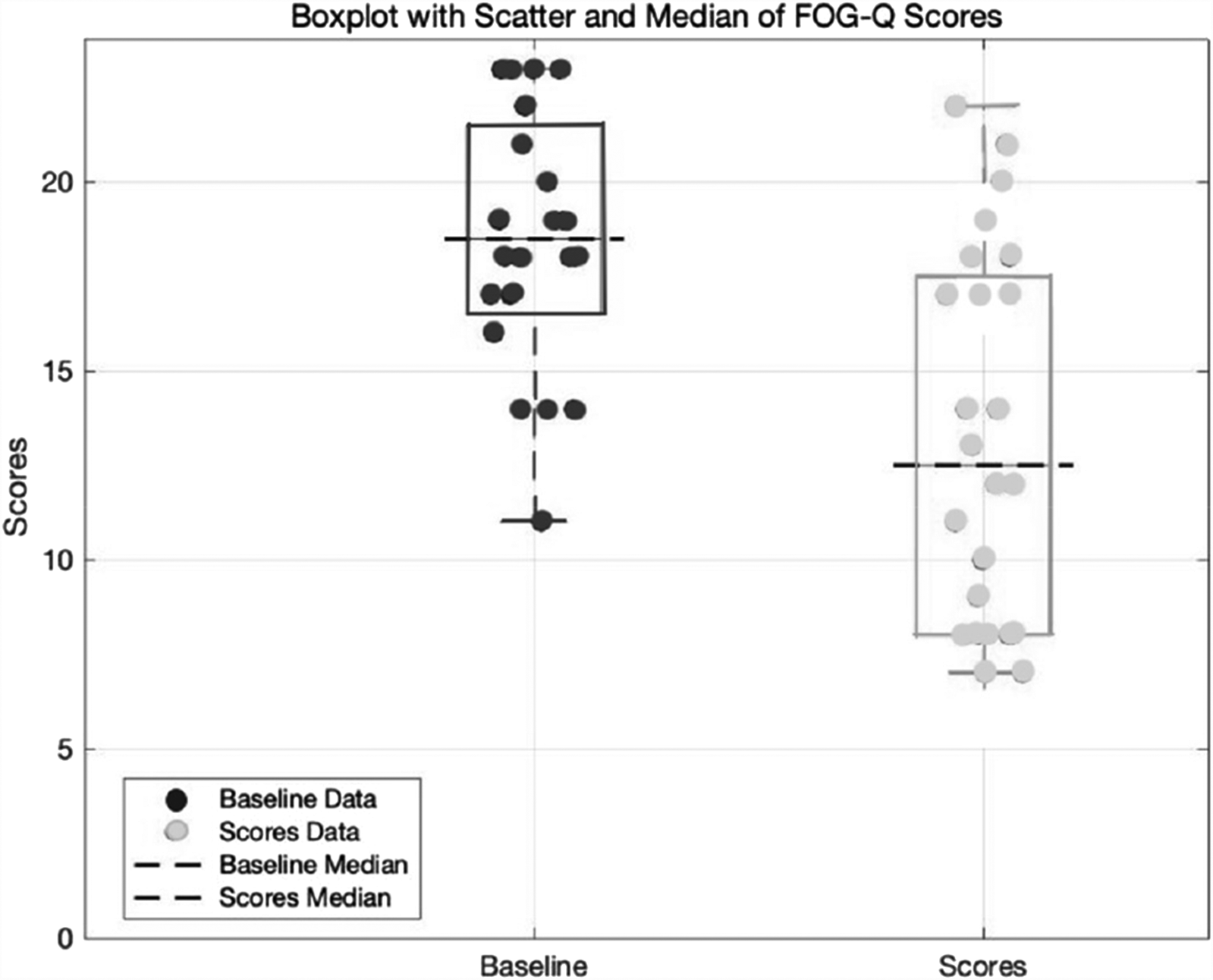
Overall Improvement in FOG-Q Scores
Pairwise comparisons and effect sizes
Post hoc pairwise comparisons (Bonferroni-corrected) revealed that the mean improvement in FOG-Q scores was significantly greater during STN + SNr stimulation compared to STN-only stimulation (mean difference: 4.7 points; 95% CI: 2.1–7.3; p = 0.001). For PDQ-SI, the corresponding mean difference was 3.2 points (95% CI: 1.0–5.4; p = 0.006). These results demonstrate a clinically meaningful and statistically significant benefit of dual-target stimulation over conventional single-target therapy.
Association between gait and quality-of-life outcomes
Correlation analysis demonstrated a moderate positive relationship between percentage improvements in FOG-Q and PDQ-SI scores (Pearson r = 0.59, p = 0.004), suggesting a linked but not identical trajectory of improvement in motor and quality-of-life domains (Figure 4). Subgroup variability was noted, highlighting the multifactorial nature of therapeutic response in advanced PD.
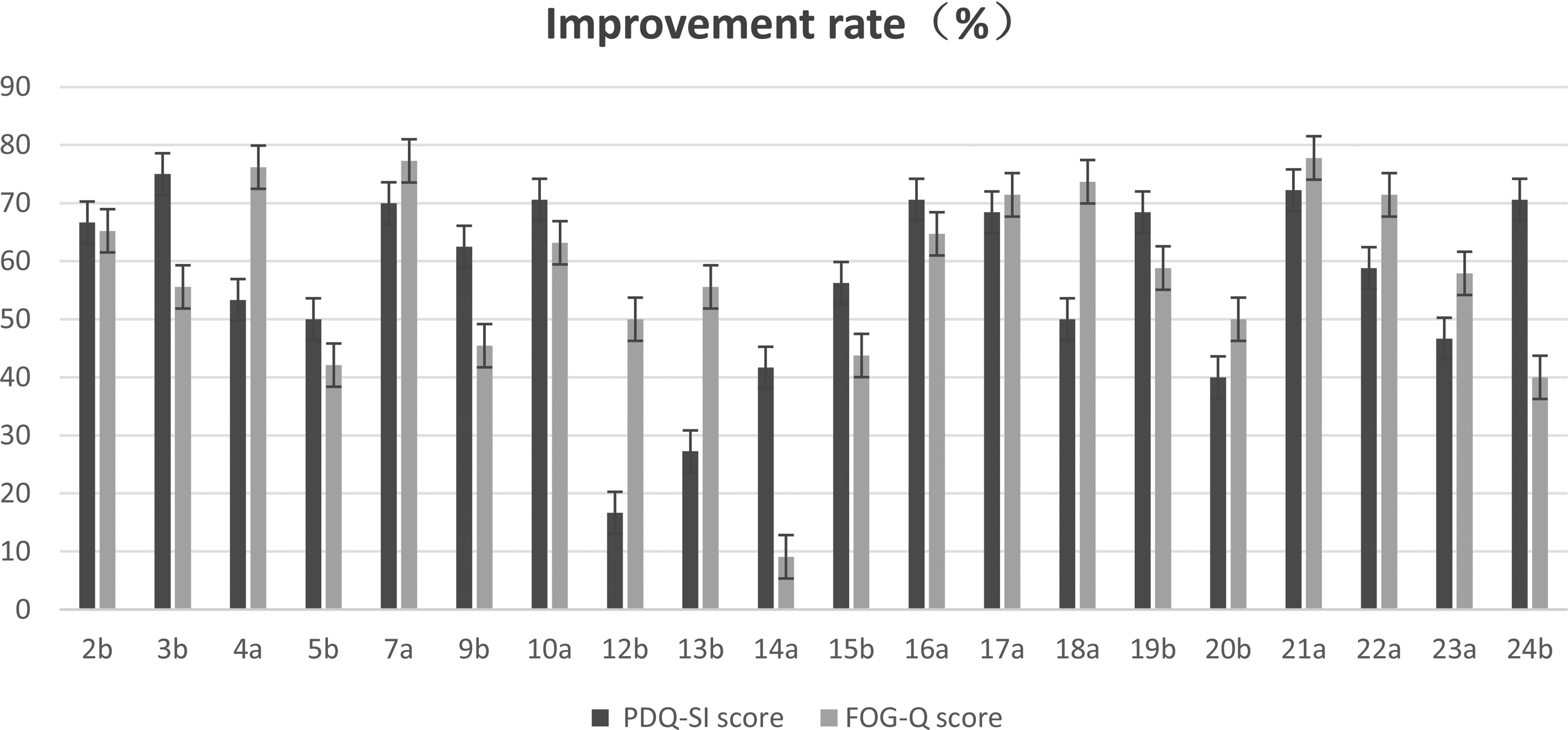
Overall Improvement Rates of FOG-Q and PDQ-SI
Adverse events
Among the 24 enrolled patients, four discontinued the study during follow-up. Two of these withdrawals were due to stimulation-induced adverse events: one patient developed persistent dysarthria refractory to reprogramming, and another experienced chronic dizziness attributed to dual-target stimulation. One patient was unable to tolerate parameter adjustments, and one withdrew due to logistical barriers in completing gait assessments.
Among the 20 patients who completed the crossover protocol, no serious or irreversible adverse events occurred. The incidence of adverse events did not differ significantly between stimulation conditions (Fisher's exact test, p > 0.05), as summarized in Table 4.
Changes in freezing symptom scores before and after Intervention.

DBS-STN: deep brain stimulation of the subthalamic nucleus; SNr: substantia nigra pars reticulata; SD: standard deviation.
Discussion
This randomized crossover trial provides compelling evidence that combined subthalamic nucleus (STN) and SNr deep brain stimulation (DBS-STN + SNr) yields superior therapeutic benefits for FOG in PD, compared to conventional STN-only stimulation. Repeated-measures analysis demonstrated significant main effects of stimulation type and time, as well as a robust stimulation-by-time interaction, with dual-target stimulation consistently resulting in greater FOG-Q and PDQ-39 Summary Index (PDQ-SI) score improvements.
The observed mean reduction of 4.7 points in FOG-Q scores with STN + SNr stimulation versus STN-only (95% CI: 2.1–7.3, p = 0.001) represents not only statistical significance but also a clinically meaningful absolute gain of 25.1% in efficacy. Notably, the decrease in post-intervention score variability (SD 4.1 vs. 5.5) suggests enhanced interindividual consistency of response under dual-target stimulation. These findings contribute novel data to a field in which prior studies have reported more modest improvements (28–38%) with single-target protocols.14–17 Thus, our results provide critical validation of multi-target stimulation as a promising therapeutic innovation in advanced PD.
From a mechanistic perspective, the enhanced efficacy of dual-target DBS may reflect synergistic modulation of distinct motor circuit components.16,18–20 The STN is involved in motor initiation and scaling, while the SNr regulates motor inhibition and brainstem locomotor centers.21–23 In PD, hyperactivity in the STN and disinhibition in the SNr may jointly contribute to the manifestation of FOG.24–27 Simultaneous stimulation of both targets may rebalance these pathological circuits, potentially restoring more physiologic gait dynamics.28,29 This hypothesis aligns with emerging neurophysiological and connectomic models of freezing, and underscores the need for future functional imaging and electrophysiological studies (e.g., local field potential recording, fMRI, or magnetoencephalography (MEG)) to elucidate the precise mechanisms underlying multi-target DBS efficacy.30,31
Our findings are consistent with recent literature supporting multi-target neuromodulation strategies. For instance, several studies have demonstrated benefit from combined stimulation of the STN and the globus pallidus internus in addressing complex motor symptoms in PD.5,6,32 However, studies specifically evaluating DBS-STN & SNr dual-target stimulation remain scarce. 33 Our work helps to fill this knowledge gap by systematically evaluating clinical outcomes using a prospective, crossover design, and by focusing on a highly disabling symptom—freezing of gait—that is often resistant to traditional DBS approaches.
It should be noted, however, that not all previous studies have found significant benefit with dual-target DBS.17,22,34 Discrepancies in the literature may stem from heterogeneity in sample selection, variability in follow-up durations, and differences in stimulation parameters or electrode placement. Furthermore, inconsistent control of confounding variables such as medication status, disease subtype, and duration may contribute to divergent results. Our study partially addresses these issues through a standardized surgical protocol, blinded outcome assessments, and crossover design, but additional multicenter trials with harmonized inclusion criteria and stratified analyses are needed to further validate our findings.
Despite its strengths, this study has several limitations. First, the sample size was modest, and although power analysis supported the cohort size, larger samples are needed to generalize findings and explore subgroup differences. Second, long-term outcomes beyond 18 months remain unknown. Third, we did not perform stratified analyses by PD subtype or disease duration, which may influence DBS responsiveness. Finally, although clinical scales such as the FOG-Q and PDQ-39 were used, objective gait metrics from wearable sensors or gait labs were not employed; future trials integrating such technologies may offer more granular insights.
From a clinical standpoint, our results suggest that DBS-STN + SNr combined stimulation represents a viable strategy for patients with refractory FOG, offering both enhanced efficacy and improved consistency of response. This approach may be especially valuable for patients who fail to respond adequately to conventional DBS or who exhibit complex gait phenotypes. As neuromodulation technologies evolve, individualized stimulation paradigms incorporating adaptive closed-loop DBS, patient-specific connectomics, and advanced electrode design may further enhance outcomes. Future research may explore closed-loop DBS systems capable of dynamically adjusting STN and SNr stimulation based on real-time gait biomarkers, potentially enhancing therapeutic efficacy for FOG.
Conclusion
This prospective, randomized crossover study demonstrates that combined STN and SNr stimulation significantly improves freezing of gait and quality of life in Parkinson's disease compared to conventional STN-only DBS. These findings provide strong clinical support for the use of dual-target neuromodulation strategies in advanced PD and offer a potential new therapeutic pathway for patients with FOG unresponsive to standard treatment. Future large-scale, multicenter trials with extended follow-up and objective gait quantification are warranted to confirm the long-term efficacy, refine patient selection criteria, and optimize stimulation protocols for broader clinical adoption.
Footnotes
Acknowledgements
We sincerely thank the medical teams at Guangyuan Central Hospital, Yibin First People's Hospital, Dazhou Central Hospital and Guang'an People's Hospital for their invaluable support in patient recruitment, clinical management, and data collection. Additionally, we appreciate the contributions of all participating patients and their families, whose trust and cooperation made this study possible.
Ethical considerations
This retrospective study was approved by the Ethics Committee of Guangyuan Central Hospital (Approval No. GYZX510824042406) and conducted in accordance with the principles of the Declaration of Helsinki. The requirement for informed consent was waived due to the retrospective nature of the study.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the Health Commission of Sichuan Province Medical Science and Technology Program (Grant No. 24WSXT042) and the Wu Jieping Medical Foundation (Grant No. 320.6750.2024-6-113).
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
The datasets generated and/or analyzed during the current study are available from the corresponding author upon reasonable request.
