Abstract
Background
Atypical parkinsonian syndromes are highly prevalent in the French West Indies (FWI), making up 70% of degenerative parkinsonisms and including “Caribbean Atypical Parkinsonism”. Environmental neurotoxins from Annonaceae plants are implicated. Despite close ties, parkinsonism data for French Guiana remain limited.
Objective
This study aimed to compare atypical parkinsonism frequencies between French Guiana and FWI, assess clinical characteristics in French Guiana, and evaluate potential environmental toxin exposure.
Methods
Degenerative parkinsonism patients were recruited from a community-based population in French Guiana and compared with a FWI cohort.
Results
Among 372 patients (67 from French Guiana, 305 from FWI), atypical parkinsonian syndromes accounted for 41.8% in French Guiana, lower than in FWI (66.2%, p < 0.001). In French Guiana, these syndromes were more common in males (sex-ratio: 3 vs. 1.22 in FWI, p = 0.044; adjusted p-value = 0.281) and often involved cerebellar symptoms (p < 0.001). Cases not fitting classical subtypes were classified as “other atypical parkinsonian syndromes” (35.7% in French Guiana, 41.6% in FWI), with a supranuclear palsy-like phenotype often presenting with additional rapid eye movement (REM) sleep behavior disorder, hallucinations, or orthostatic hypotension. Annonaceae consumption was higher in FWI (93%) than in French Guiana (79.2%, p < 0.001), while alcohol use was more common in French Guiana (p = 0.005).
Conclusions
Atypical parkinsonism in French Guiana resembles that in FWI but is less common, with an intermediate prevalence between Caucasian and Caribbean populations. Shared environmental factors, such as Annonaceae exposure, may contribute to this presentation, supporting the term “Caribbean Atypical Parkinsonism” for both regions.
Plain language summary
Atypical parkinsonian syndromes are a group of degenerative parkinsonian syndromes that share similarities with Parkinson's disease (PD) but are distinguished by features such as abnormal eye movements, cognitive decline and balance issues. In the French West Indies—particularly Guadeloupe and Martinique—atypical parkinsonian syndromes are more prevalent than PD. One suspected cause is the consumption of plants from the Annonaceae family, including soursop and sugar-apple, commonly consumed as fruits or in herbal infusions, which has been associated with the onset of atypical parkinsonian syndromes. Despite the deep historical and cultural connections between the French West Indies and French Guiana, both overseas French territories respectively in the Caribbean and in South America, the profile of degenerative parkinsonism in French Guiana remains underexplored. This study aimed to compare atypical parkinsonism between French Guiana and the French West Indies and to examine potential correlations between Annonaceae plant consumption and atypical parkinsonian syndromes in these regions. Sixty-seven patients with degenerative parkinsonism from French Guiana were compared to 305 patients from the French West Indies. Over 40% of the French Guiana cohort exhibited atypical parkinsonian syndromes, a prevalence intermediate between Caucasian and Caribbean populations. In French Guiana, these syndromes predominantly affected males, with more pronounced cerebellar dysfunction and abnormal eye movements. Annonaceae consumption was reported by approximately 80% of Guianese patients and was even higher in the French West Indies. This study highlights that atypical parkinsonian syndromes observed in the French West Indies are also prevalent in French Guiana, potentially associated with Annonaceae plant consumption.
Keywords
Introduction
Degenerative parkinsonism in the Caribbean has shown distinct phenotypic and anatomopathological features, with a notable predominance of atypical parkinsonian syndromes over typical Parkinson's disease (PD). In the French West Indies (FWI), specifically in Guadeloupe and Martinique, most parkinsonian syndromes are atypical, accounting for 66–74% of cases.1–4 Only about one-third of patients meet the diagnostic criteria for PD, 5 a stark contrast to other studies where atypical parkinsonian syndromes are reported at lower proportions (3–35%).6–9 Classically, atypical parkinsonian syndromes include four main disorders: progressive supranuclear palsy (PSP), multiple system atrophy, corticobasal degeneration, and dementia with Lewy bodies. These syndromes are characterized by poor or absent response to dopaminergic therapy, worse prognosis, and neurological “red flags” such as cognitive impairment or postural instability, that are rarely observed in PD at earlier stages of the disease, but quite frequent at advanced stages of PD.10–13
In the FWI, atypical parkinsonism, known as “Caribbean Parkinsonism” or “Caribbean Atypical Parkinsonism”, combines clinical features of various atypical parkinsonian syndromes subtypes. This nosological entity is endemic in both Guadeloupe1,3 and Martinique. 4 It is characterized by a parkinsonian syndrome associated with a variable combination of additional signs such as supranuclear oculomotor palsy, cognitive decline, hallucinations, rapid eye movement sleep behavior disorder (RBD), dysautonomia, and myoclonus, sometimes combined in the same patient. Approximately half of Caribbean Atypical Parkinsonism cases resemble PSP, commonly termed PSP-like. Pathologically, Caribbean Atypical Parkinsonism is classified as a tauopathy based on limited pathological data but exhibits features characteristic of synucleinopathies rather suggesting mixed neuropathological lesions.2,4,14 Environmental origins have been linked to chronic exposure to toxins from Annonaceae plants used in traditional teas. 15 Annonacin, a mitochondrial toxin found in these plants, has demonstrated neurotoxicity in both cell cultures and animal models.15,16 Chronic consumption of Annonaceae derivatives, leading to PSP-like syndromes, has been reported in regions where acetogenin-containing fruits are consumed. 17 Given the socio-cultural and pharmacopoeial ties between the FWI (including Guadeloupe and Martinique), and French Guiana (often termed French Amazonia), both overseas French territories respectively in the Caribbean and in South America, we hypothesized that a similar degenerative parkinsonism profile might be present in French Guiana and that its links to environmental risk factors could be comparable. However, French Guiana has geographical, demographic, and cultural differences with the FWI, and this hypothesis, although plausible, has yet to be tested in the Guianese population. Indeed, French Guiana is a French overseas territory located on the South American continent, between Brazil and Suriname. This territory has the highest gross national product per capita in Latin American, which attracts many immigrants. As a result of these migratory flows, 29% of the population and nearly half of the adults are of foreign origin, primarily from surrounding South American countries. The population is mainly composed of French Guianese Creoles, continental French and Haitian Creoles, most of whom reside in coastal regions. The communes in the interior of French Guiana—mostly surrounded by Amazonian Forest—are inaccessible by road. The populations residing there are essentially Amerindians or Maroons.18,19 French Guiana is also home to other communities including those of Southeast Asian descent, contributing to its ethnic diversity.20,21 In French West Indies, the majority of the population consists of individuals of mixed African and European ancestry, followed by continental French. Other ethnic groups such as Asians (including Tamil, East Indians and Chinese communities) are less represented. 22 French Guiana has a modern French universal health care system with a hybrid model combining public and private healthcare services, which is, by far, the highest health expenditure per capita in Latin America. 18
In this context, the main aim of this study was thus to compare the prevalence of atypical parkinsonism between French Guiana and the FWI. Secondary objectives included describing the clinical features of atypical parkinsonian syndromes in French Guiana and assessing exposure to environmental toxins potentially linked to degenerative parkinsonism in patients from both regions.
Methods
Study design
We collected data from a community-based population followed in the Department of Neurology at Cayenne General Hospital, French Guiana, and compared it with a cohort of 305 patients recruited in the Departments of Neurology at the University Hospitals of Martinique and Guadeloupe, as well as from the Rehabilitation Department of the University Hospital of Martinique (Supplemental Figure 1).4,23
All consecutive patients with degenerative parkinsonism over a period of five years (2012 and 2016) were included in both French Guiana and FWI. They were assessed by two neurologists specializing in movement disorders (AL, ER), from the University Hospital of Guadeloupe and la Pitié-Salpêtrière Hospital, respectively. In French Guiana, patients were evaluated by the same two specialists as part of regular specialized expert missions conducted during the study period. Parkinsonism was clinically diagnosed based on the United Kingdom Parkinson's Disease Society Brain Bank Diagnostic Criteria. 5 Patients with clinical or paraclinical data suggestive of symptomatic parkinsonism including post-traumatic, vascular, iatrogenic (particularly due to antipsychotics), or post-encephalitic forms, were excluded.
Data collection
Clinical assessment
The history and clinical examination followed a standardized protocol. Demographic data, medical history, age at onset, medication use, and clinical symptoms were collected. Motor symptoms were evaluated using the Unified Parkinson's Disease Rating Scale III.
A positive levodopa response was defined as either a subjective improvement of at least 50%, or a 50% improvement in the Unified Parkinson's Disease Rating Scale III score following an acute levodopa challenge. Non-motor symptoms were assessed including hyposmia, RBD using the Stiasny-Kolster score (cut-off ≥ 5), orthostatic hypotension (a systolic drop ≥ 30 mmHg or diastolic drop ≥ 15 mmHg), fluctuating cognition via the Mayo Fluctuation Scale (cut-off ≥ 3), and dementia using the Mattis Dementia Rating Scale (cut-off ≤ 123) or the Mini-Mental State Examination with education-adjusted cut-off scores.24,25 If the latter two scales could not be assessed, dementia was clinically diagnosed in the presence of a progressive cognitive decline that significantly interfered with normal social or occupational functioning, or with daily activities, consistent with established diagnostic criteria. Dementia was defined as early if documented within three years of onset. Disease severity and disability were evaluated using the Hoehn and Yahr scale (Unified Parkinson's Disease Rating Scale V) and the Schwab and England Activities Daily Living scale (Unified Parkinson's Disease Rating Scale VI).
Environmental questionnaire
An environmental questionnaire was administrated to assess tobacco, alcohol, coffee, and Annonaceae consumption in French Guiana and the FWI.
Participants were asked about their food habits regarding the consumption of three fruits from the Annonaceae family, available in the Caribbean islands and French Guiana: A. muricata (soursop, corossol, graviola, guanabana), A. squamosa (sweetsop, sugar apple, pomme-cannelle, saramuyo), and A. reticulata (custard apple, cachiman). Consumption of Annonaceae fruits (or fruit juices) and herbal teas was evaluated separately for each fruit over successive 10-year periods throughout the patient's life. Cumulative consumption was expressed in fruit-years, defined as the number of fruits or glasses of fruit juice consumed for each fruit (one glass of juice = one fruit) per day, multiplied by the frequency of consumption per year and the number of years of consumption. For example, a person who has eaten two fruits per day for 10 years, one fruit per day for 20 years, or one fruit every two days for 40 years would all have a cumulative consumption of 20 fruit-years. 3
For herbal teas, cumulative consumption was expressed in cup-years, where one cup-year corresponds to drinking one cup per day for one year. Finally, we combined both fruit/juice and herbal tea consumption by summing the above measures, expressing the result in an arbitrary unit corresponding to a fruit-or-cup-year unit. Dietary information was collected through a structured questionnaire developed in collaboration with dieticians and epidemiologists and has been used in previous studies.3,23 If needed, a family informant could assist the patient in answering the questions. Consumption of at least 10 fruit-years or cup-years was considered high consumption.
Classification of patients
Patients with degenerative parkinsonism were categorized as PD or atypical parkinsonian syndromes. The diagnosis of clinically established or probable PD was based on the MDS Clinical Diagnostic Criteria for Parkinson's Disease. 5 The diagnosis of atypical parkinsonian syndromes was based on the presence of red flags, such as early falls, postural instability, bulbar involvement, severe dysautonomia or dementia, poor levodopa response, vertical supranuclear ophthalmoplegia, selective downward jerk slowing, cerebellar syndrome, or pyramidal signs. 26 Unclassified cases were those where PD / atypical parkinsonian syndromes differentiation could not be established.
Patients with atypical parkinsonian syndromes were further divided into dementia with Lewy bodies, multiple system atrophy, PSP, and corticobasal degeneration subgroups following their respective diagnostic criteria.27–30 In patients with chronic alcohol exposure and cerebellar syndrome, additional supporting evidence for the diagnosis of multiple system atrophy (such as autonomic failure, lack of levodopa response and/or imaging findings) were sought to differentiate multiple system atrophy from PD. Cases not fitting these criteria were labeled as “other atypical parkinsonism”, with those exhibiting supranuclear vertical gaze palsy classified as PSP-like.
Statistical analysis
Statistical analyses were conducted using R (R Core Team (2024). _R: A Language and Environment for Statistical Computing_. R Foundation for Statistical Computing, Vienna, Austria. https://www.R-project.org/). Results are presented as means (standard deviation) or median (1st-3rd quartiles) for continuous variables and counts (percentages) for categorical variables. Comparisons between groups (FWI vs. French Guiana and PD vs. atypical parkinsonian syndromes) were made using unpaired Student's t-test or Wilcoxon rank sum test for continuous variables and chi-squared test or Fisher's exact test for categorical variables. A binary multivariate logistic regression model identified risk factors for atypical parkinsonian syndromes, excluding specific criteria of atypical parkinsonian syndromes such as cerebellar syndrome, supranuclear gaze palsy, and early dementia, as these criteria are characteristic of these syndromes. For multiple comparisons, the Benjamini-Hochberg procedure was used to calculate adjusted p-values. The significance threshold was set at p < 0.05.
Ethics
The study was approved by the French South-West and Outremer III ethics committee (2011/86). The project adhered to the principles outlined in the Declaration of Helsinki. All participants provided written informed consent.
Results
Characteristics of degenerative parkinsonism in French Guiana
Sixty-seven parkinsonian patients from French Guiana were assessed after a median disease duration of six years. The sex ratio was 1.31 (male/female), with a mean age of 70 ± 12 years at evaluation and a mean age of onset of 63 ± 13 years. Among these patients, 49.3% met the criteria for clinically established or probable PD, while 41.8% were classified as atypical. Six patients remained unclassified as the differentiation between PD and atypical cases could not be established due to insufficient and unreliable information regarding their medical history or difficulties in assessing their response to levodopa therapy (Table 1).
General and demographic characteristics of patients with degenerative parkinsonism in French Guiana and French West Indies.
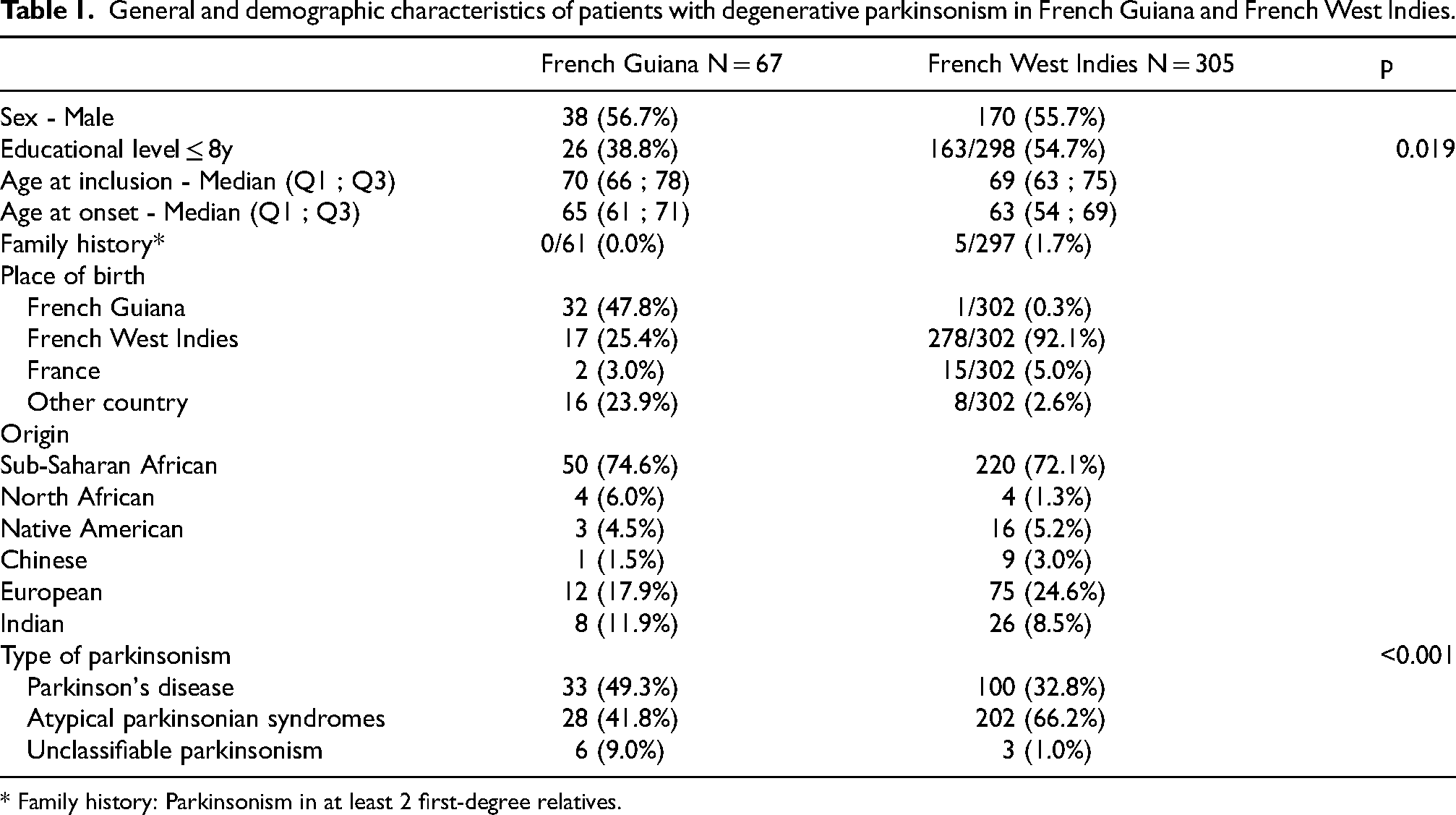
* Family history: Parkinsonism in at least 2 first-degree relatives.
There were no significant differences between PD and atypical groups regarding age at onset and educational level (Supplemental Table 1). However, the sex ratio was 0.83 for PD and 3 for atypical cases (p = 0.092). Vertical supranuclear palsy was observed in 37% of patients with atypical parkinsonism, and cerebellar syndrome in 42.9% of them (Table 2). A large proportion of atypical patients experienced falls (67.9%). Dementia was highly prevalent in atypical cases compared to PD patients (75.0% vs. 38.5%, p = 0.061), with early dementia present in 17.9% of those with atypical parkinsonism. Other non-motor symptoms, such as RBD and hyposmia, were as common in atypical patients as in those with PD.
Comparison of demographic and clinical characteristics of parkinsonism in French Guiana and French West Indies.
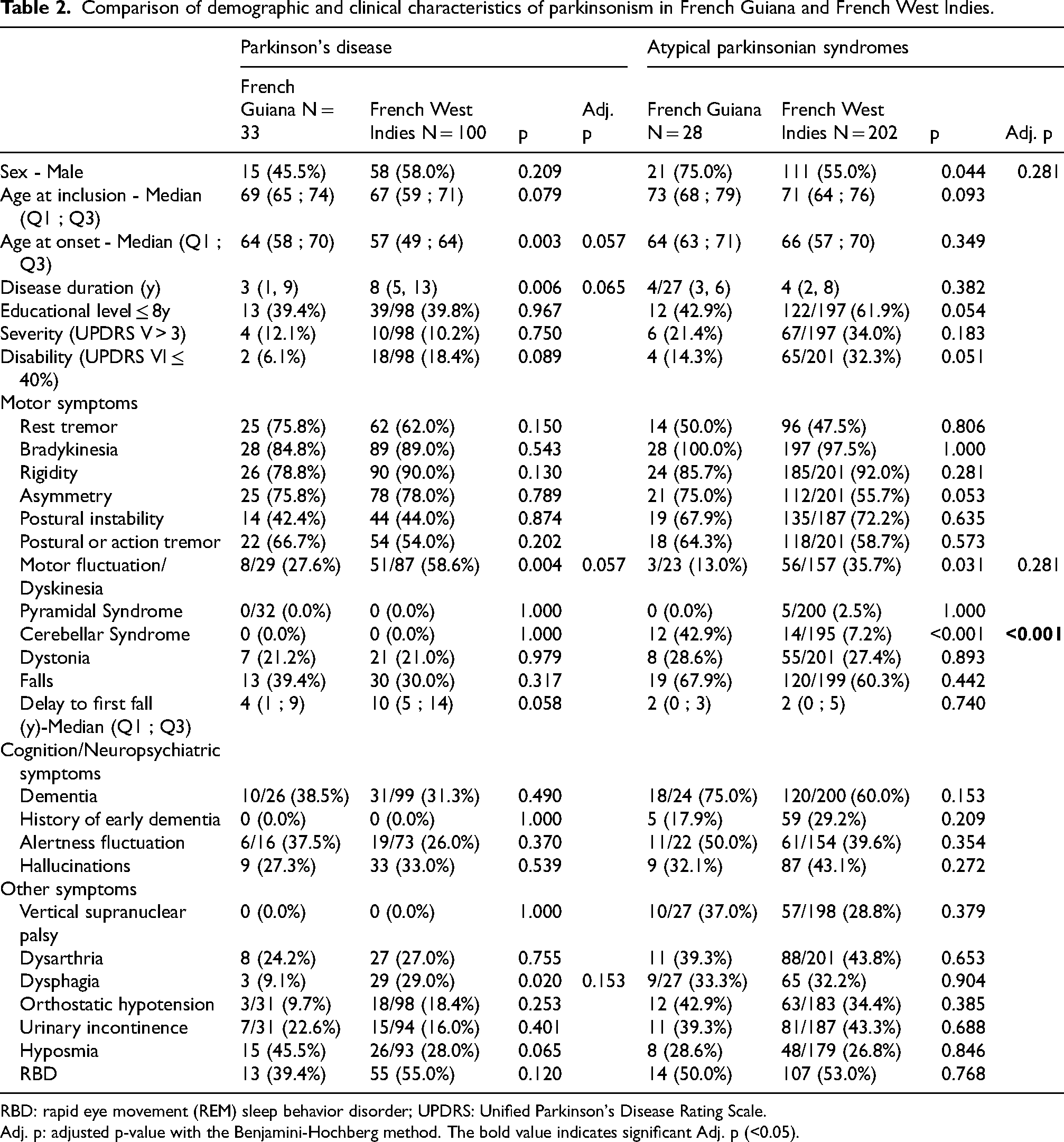
RBD: rapid eye movement (REM) sleep behavior disorder; UPDRS: Unified Parkinson's Disease Rating Scale.
Adj. p: adjusted p-value with the Benjamini-Hochberg method. The bold value indicates significant Adj. p (<0.05).
Comparison of general features of parkinsonian patients in French Guiana versus French West Indies
The 67 patients from French Guiana were compared with 305 patients from Guadeloupe and Martinique, who were included in the study during the same period (Table 1). There were no significant differences between the groups in terms of sex ratio and age at onset. However, Caribbean patients had a lower educational level (p = 0.019). Notably, 52.2% of Guianese patients were born outside French Guiana, with 25.4% from the FWI. The proportion of patients meeting the criteria for clinically established or probable PD was higher in French Guiana (49.3%) compared to the FWI (32.8%, p < 0.001). In contrast, patients with atypical parkinsonian syndromes constituted 41.8% of the Guianese cohort, whereas this proportion was higher in Martinique and Guadeloupe, at 66.2%.
Clinical characteristics of parkinsonian patients in French Guiana compared to the French West Indies
For PD, patients from French Guiana were older at onset (64 vs. 57 years; p = 0.003; adjusted p = 0.057) and the disease duration was shorter (3 vs. 8 years; p = 0.006; adjusted p = 0.065). There were fewer motor fluctuations/dyskinesia (27.6% vs. 58.6%; p = 0.004; adjusted p = 0.057) and less dysphagia (9.1% vs. 29%; p = 0.020; adjusted p = 0.153) than among patients from the FWI.
The proportion of male patients was higher among atypical cases in French Guiana, with a sex ratio of 3 compared to 1.22 in the FWI (p = 0.044). However, this difference was no longer significant after correction for multiple comparisons (adjusted p = 0.281) (Table 2). There were no significant differences in age at onset or educational level between the two regions. The clinical characteristics of patients with atypical parkinsonism were similar in both groups, except for a higher frequency of cerebellar syndrome and fewer motor fluctuations/dyskinesia in the Guianese cohort (p < 0.001 and p = 0.031, respectively). After correction for multiple comparisons, only the higher frequency of cerebellar syndrome remained significant (adjusted p < 0.001).
Focus on subgroups of atypical parkinsonian syndromes in French Guiana compared to the French West Indies
Among the 230 atypical parkinsonian patients from French Guiana and the French West Indies, 136 (56.6%) met the diagnostic criteria for a specific subtype of atypical parkinsonism. There was no significant difference in the distribution of subtypes between the two regions (Figure 1). The most prevalent form was multiple system atrophy, occurring in 39.3% of French Guiana patients and 31.2% of those from the FWI. This was followed by dementia with Lewy bodies at 17.9% and 19.8%, respectively, and PSP at 7.1% and 7.4% in French Guiana and the FWI. No patients were diagnosed with corticobasal degeneration in either cohort. When examining each subtype individually (Table 3), the groups were comparable between both regions, except for patients with multiple system atrophy, who exhibited a higher frequency of cerebellar component in French Guiana compared to the FWI (81.8% vs. 11.1%; p < 0.001; adjusted p < 0.001).
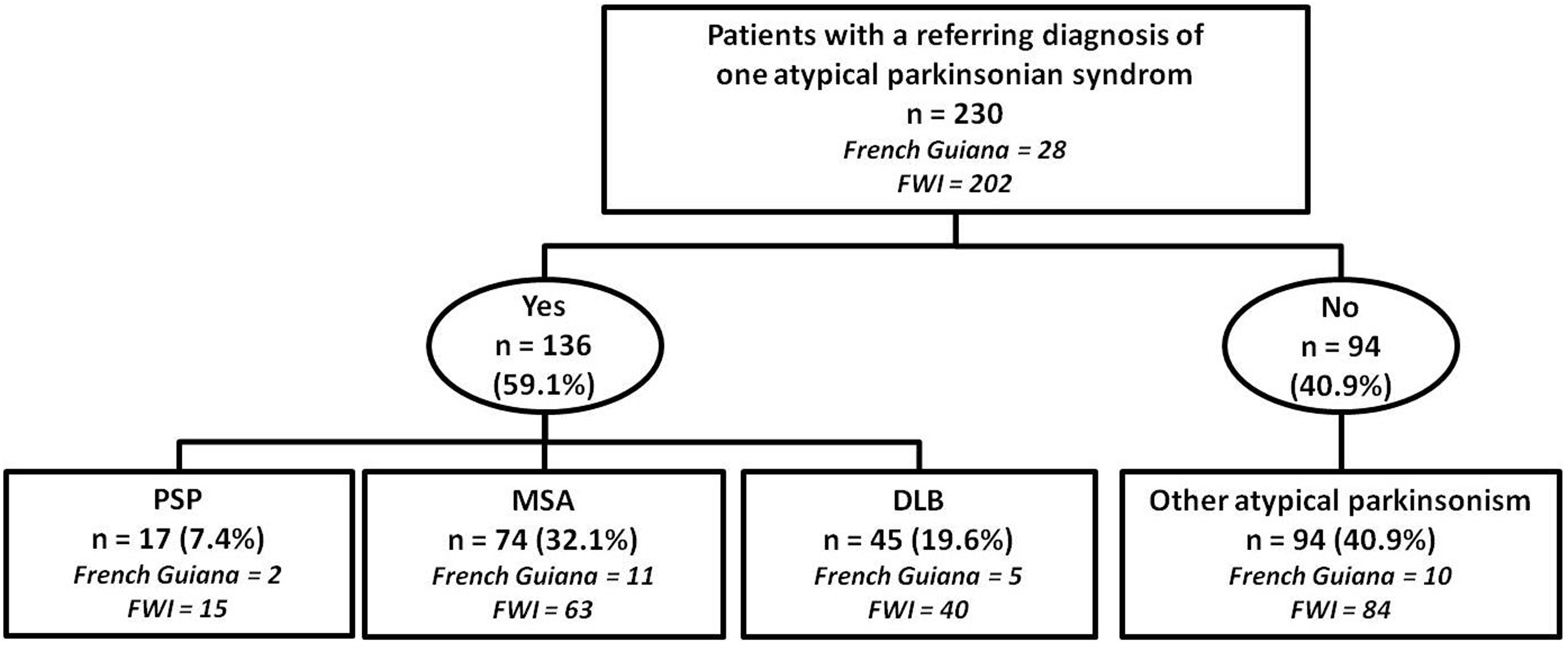
Distribution of the subgroups of atypical parkinsonian syndromes for the entire study cohort. DLB: dementia with Lewy bodies; FWI: French West Indies; MSA: multiple system atrophy; PSP: progressive supranuclear palsy.
Main clinical characteristics of atypical parkinsonism in French Guiana and French West Indies.
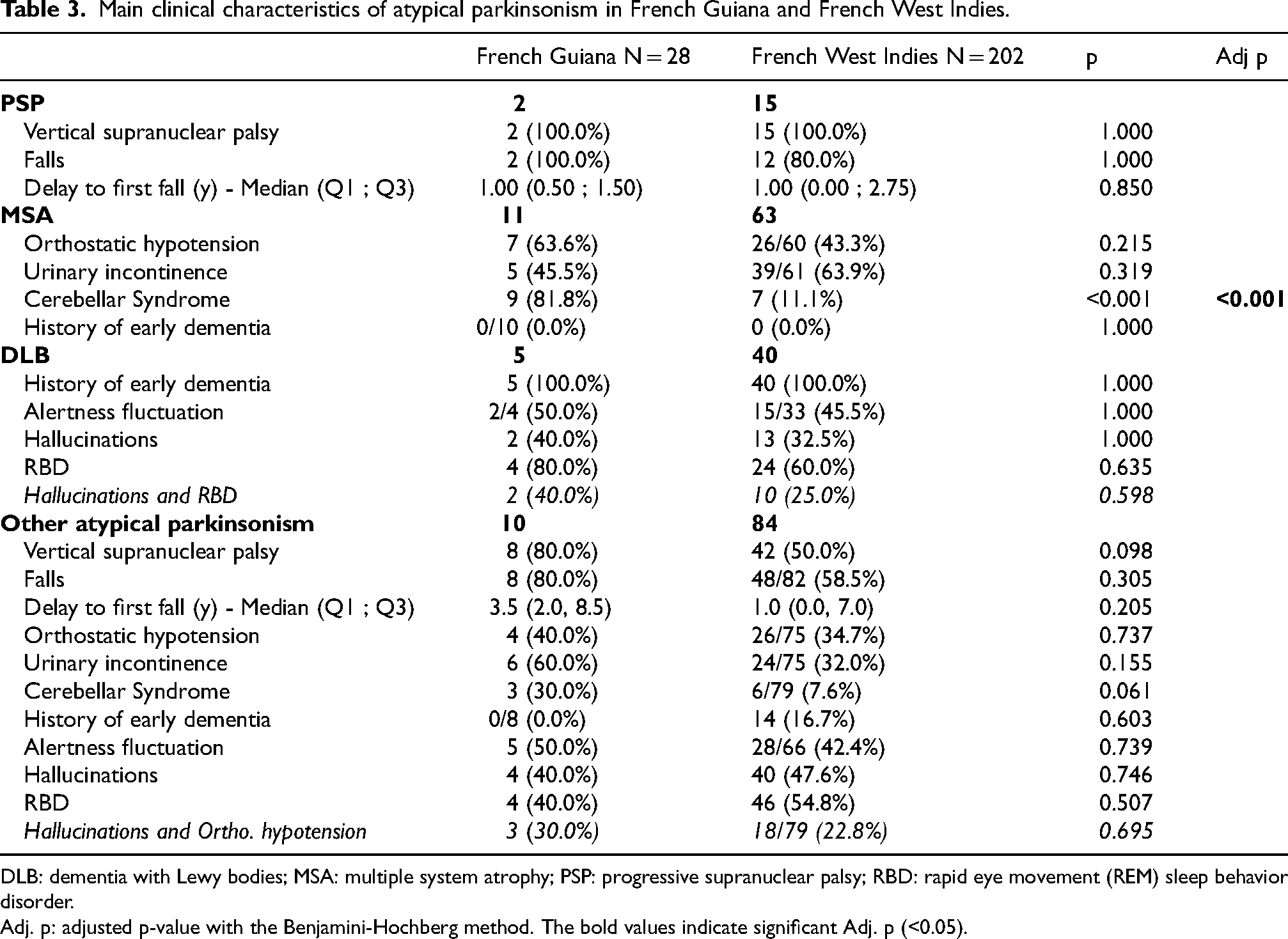
DLB: dementia with Lewy bodies; MSA: multiple system atrophy; PSP: progressive supranuclear palsy; RBD: rapid eye movement (REM) sleep behavior disorder.
Adj. p: adjusted p-value with the Benjamini-Hochberg method. The bold values indicate significant Adj. p (<0.05).
The remaining 94 patients (40.9%) were classified as “other atypical parkinsonism”, with 10 (35.7% of all atypical cases) from French Guiana and 84 (41.6% of all atypical cases) from the FWI (Supplemental Table 2). This ‘other atypical parkinsonism’ group showed similar clinical features between the regions (Supplemental Table 3), although PSP-like presentations were more common in French Guiana (80% vs. 50%) (Supplemental Table 2).
Findings from the environmental questionnaire in French Guiana and in the French West Indies
Findings from the environmental questionnaire on Annonaceae consumption are summarized in Supplemental Figure 2. Consumption of Annonaceae (fruit and teas of soursop and sugar-apple) was more frequent in the FWI, reported by 93% of parkinsonian patients compared to 74.2% in Guiana (p < 0.001), with the exception of sugar-apple herbal tea/infusion, which was significantly higher in Guiana (reported by 46.2% of patients) compared to the Caribbean (22.6%) (p = 0.001). In Guiana, 65.7% of patients reported no or very occasional alcohol consumption, compared to 84.4% in the FWI. Conversely, 34.3% of Guianese patients consumed alcohol regularly (weekly or daily) or excessively (parties), while only 15.6% of Caribbean patients did so (p = 0.005). Across all study participants, regular alcohol consumption was associated to the occurrence of cerebellar syndrome in patients with atypical parkinsonian syndromes (odds ratio [OR]: 2.67 [95% CI: 1.09
Discussion
Our findings revealed that atypical parkinsonian syndromes accounted for 42% of degenerative parkinsonism cases in French Guiana, an intermediate proportion between the predominantly PD populations of the United States and Europe (where atypical parkinsonian syndromes represent around 20–30% of degenerative parkinsonism) and the elevated rates observed in the French Caribbean (around 70%).6–9,31 Compared to its immediate neighboring countries, Brazil appears to be classified as a predominantly PD population, with PD representing nearly 70% of all parkinsonism cases.32,33 To our knowledge, no data are available on the clinical phenotype or frequency of PD and atypical parkinsonian syndromes in Suriname, the second neighboring country of French Guiana. Taken together, this reflects a significant regional variation in the prevalence of atypical parkinsonism, which has been well-documented in the FWI and is thought to be closely linked to environmental factors, particularly the consumption of Annonaceae plants. Of note, the difference in prevalence between French Guiana and the FWI is not proportional to the variation in Annonaceae exposure, suggesting the role of additional environmental factors in French Guiana and/or the FWI. This study not only broadens our understanding of degenerative parkinsonism in an under-researched area but also highlights the need for further investigation into how environmental factors may contribute to the global variability of parkinsonian syndromes.
In our Guianese cohort, 41.8% of patients had atypical parkinsonism, characterized by poor levodopa response with postural instability (68%), cerebellar syndrome (43%), and supranuclear gaze palsy (37%). 4 Non-motor symptoms included RBD (50%), dysautonomia (40%), hallucinations (32%), and early dementia (17.9%). Although losing statistical significance after adjustment for multiple comparisons, the sex ratio was higher in French Guiana (3:1) compared to the FWI (1.22:1), possibly reflecting selection bias or greater exposure to occupational hazards, such as pesticides and heavy metals prevalent in male-dominated industries. 34 Further studies are needed to clarify the role of environmental factors in neurodegenerative diseases in this population, particularly pesticides such as chlordecone, which has been implicated in promoting parkinsonism-like neurodegeneration with Tau lesions in midbrain cultures and C. elegans models. 35 The subtypes of atypical parkinsonism were similarly distributed in both regions, with multiple system atrophy being the most common, followed by dementia with Lewy bodies and PSP. However, patients with multiple system atrophy in French Guiana had a higher prevalence of cerebellar symptoms, which remained significant after adjustment for multiple comparisons. This higher prevalence of cerebellar symptoms in Guianese patients compared to FWI cases could potentially be linked to alcohol consumption or genetic factors in the context of substantial ethnic diversity. The association between regular alcohol consumption and the occurrence of cerebellar syndrome in patients with atypical parkinsonism, particularly those with multiple system atrophy, could not be assessed separately in Guianese, likely due to the limited sample size in this cohort. Notably, in the Pan-American multicenter cohort study of multiple system atrophy by Gatto et al., which included Latin American centers from Argentina, Chile, Mexico and Peru, the authors reported that the cerebellar phenotype of multiple system atrophy was predominant in non-Caucasians, particularly in the Mestizo population (defined as individuals of mixed European and Amerindian descent). These findings highlight the need for larger epidemiological studies to investigate genetic, epigenetic, and environmental risk factors, aiming to better elucidate the pathogenesis of these diseases in this region. 36
Similar to Caribbean patients with atypical parkinsonism, a substantial proportion of Guianese patients (35.7%) exhibited atypical features that did not align with classical criteria, frequently presenting a ‘PSP-like phenotype’ with supranuclear palsy, RBD, hallucinations, and orthostatic hypotension. Such atypical presentations, with characteristics deviating from established criteria, have also been reported in the United Kingdom. 37 For instance, Afro-Caribbean patients in the United Kingdom showed a bradykinesia-dominant disease, with axial and segmental rigidity, early postural instability, and neuropsychiatric symptoms such as complex visual hallucinations. Patients of Indian origin displayed a mixed presentation, including bradykinesia, resting and postural tremor, and a relative intolerance to levodopa, coupled with early onset of axial dyskinesias. 37 These mixed phenotypes might represent a pathological continuum between classical tauopathies and synucleinopathies, potentially shaped by a unique genetic background and shared environmental factors distinct to both the French Caribbean and Amazonian regions. Indeed, recent findings suggest that concomitant pathologies, even at low levels, can synergistically drive the cognitive impairment and potentially additional features in aging individuals and patients with degenerative parkinsonism.38,39
Our environmental questionnaire revealed a high rate of Annonaceae consumption in French Guiana, similar to the FWI, with 25.8% of patients frequently consuming soursop and 12.3% consuming sugar-apple. Although we did not directly assess the correlation between Annonaceae intake and the risk of developing an atypical parkinsonian syndrome due to the study design, these findings are consistent with previous epidemiological1,3,23 and experimental studies40–42 that link annonacin, a neurotoxin found in Annonaceae, to degenerative parkinsonism. 43 A connection between soursop consumption and the high incidence of atypical parkinsonism in Guadeloupe has been proposed.1,2 In vivo studies on animal models have shown that annonacin acts as a mitochondrial complex I inhibitor, penetrating the brain parenchyma and inducing dopaminergic neuron degeneration. 41 Additionally, in vitro research has demonstrated that annonacin can trigger tauopathy, similar to that observed post-mortem in patients in Guadeloupe, causing cellular toxicity, abnormal tau protein redistribution, and impaired mitochondrial transport. 42
The findings of our study must be considered in light of some limitations. The higher education level in the Guianese cohort may suggest a selection bias related to healthcare access. Other recruitment biases could also affect the representativeness of our study population, which in turn may impact the robustness of our statistical analysis. Limited access to healthcare services, particularly for patients residing in remote regions in French Guiana, such as the villages in the interior, could further contribute to selection bias. 18 This barrier to care may be even more pronounced for individuals with more severe forms of atypical parkinsonian syndromes, potentially limiting the generalizability of our findings to the entire population with degenerative parkinsonism in French Guiana. Additionally, the retrospective design of the study poses challenges in assessing certain aspects, particularly disease progression and dietary habits, which are susceptible to recall bias. Additionally, the reliance on clinical assessments for diagnosing PD versus atypical parkinsonian syndromes may have resulted in misclassification, particularly given the absence of detailed neuropsychological evaluations, oculomotor recordings and systematic brain imaging in Guianese patients. To establish the diagnosis of dementia, we also used the Mattis Dementia Rating Scale and the Mini-Mental State Examination in our study, which are the most commonly used scales among French clinicians. However, the French consensus procedure for assessing cognitive function in PD recommended the Montreal Cognitive Assessment (MoCA) as a screening test for cognitive impairment or dementia in PD, but this recommendation was published after the period of our study. 44 Additionally, one limitation of our study is the use of the previous diagnostic criteria for dementia with Lewy bodies, multiple system atrophy and PSP. In order to apply the new criteria,45–47 several elements are missing in our data to reclassify the patients accordingly. It is obviously conceivable, especially with expanded phenotypic spectrum of PSP under the new criteria, that probably a certain percentage of cases of our cohort classified as “other atypical parkinsonism” could fit the diagnosis of specific phenotypes of PSP. However, we cannot determine these percentages with certainty. A particular focus on the clinical and paraclinical features of the specific subgroup with “other atypical parkinsonism” would undoubtedly be of paramount importance in further studies to better understand this entity within the ever-expanding scope of atypical parkinsonism. Furthermore, due to the study's observational design, the small sample size, and the lack of control group to further evaluate the potential contribution of dietary habits, we cannot definitively establish a causal link between Annonaceae consumption and atypical parkinsonism in French Guiana. Additionally, the observational design of this single-center study makes the generalizability of our findings challenging. Finally, for statistical analysis, given the number of comparisons performed, it is important to emphasize the need for caution in interpreting the associations found.
Despite its limitations, our study offers an in-depth view of degenerative parkinsonism in French Guiana, revealing a notably high proportion of atypical parkinsonian syndromes and underscoring the potential role of environmental factors. These findings suggest that the concept of ‘Caribbean Atypical Parkinsonism’ could be expanded to the Amazonian region to encompass French Guiana. However, larger prospective studies are essential to validate these results and to further investigate the complex interaction between genetic predispositions and environmental exposures in this region.
Supplemental Material
sj-docx-1-pkn-10.1177_1877718X251359212 - Supplemental material for Atypical parkinsonian syndromes in French Guiana: Similarities and differences with Caribbean variants
Supplemental material, sj-docx-1-pkn-10.1177_1877718X251359212 for Atypical parkinsonian syndromes in French Guiana: Similarities and differences with Caribbean variants by Amina Nasri, Hugo Chaumont, Souraya Arnaud, Benoit Tressieres, Mamadou Sow, Angéla Lackmy, Régine Edragas, Mathieu Nacher, Bertrand de Toffol, Emmanuel Roze and Annie Lannuzel in Journal of Parkinson's Disease
Footnotes
Acknowledgments
We thank all the individuals, their caregivers, and their families who participated in the study.
Ethical considerations
The study was approved by the French South-West and Outremer III ethics committee (2011/86). The project adhered to the principles outlined in the Declaration of Helsinki.
Consent to participate
All participants provided written informed consent.
Consent for publication
Informed consent for publishing was obtained from all participants.
Funding
This work was funded by the French Ministry of Health (Interregional Hospital Clinical Research Program 2011-A01259-32), Guadeloupe Region and the European Union through REG-MND (Registre Guadeloupéen des Maladies Neurodégénératives; 2019-FED-118).
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Datasets/data availability statement
The data supporting the findings of this study are available on request from the corresponding author. The data are not publicly available due to privacy or ethical restrictions.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
