Abstract
Parkinson's disease (PD) is a progressive neurodegenerative disorder clinically defined by the presence of cardinal motor features like bradykinesia, rigidity and resting tremor. The mainstay of PD treatment is pharmacological dopamine substitution and responsiveness of motor symptoms to levodopa is a supporting diagnostic feature of this illness. Although the armentarium of drugs to treat the motor symptoms of PD and their efficacy on a variety of motor problems are overall impressive, significant unmet needs persist, particularly in the advanced stages of PD. These include situations of poorly responsive tremor, motor complications associated with sustained levodopa exposure, disorders of gait and balance as well as impairments of posture, speech and swallowing. This review provides an overview of available treatment strategies for these areas of unmet need and emerging therapies under development. Future perspectives include novel pharmacological targets, new modes of drug delivery, combination therapies and the integration of real-time monitoring and wearable technologies to tailor treatments.
Plain language summary
Parkinson's disease (PD) is a progressive neurological condition that causes motor symptoms such as tremor, slow movements (bradykinesia), and muscle stiffness (rigidity). It develops as dopamine-producing neurons in the brain gradually deteriorate, leading to significant movement problems. Current treatments aim to manage symptoms, with levodopa being the most effective medication for restoring dopamine and improving motor function. Other treatments include dopamine agonists, MAO-B inhibitors, COMT inhibitors, and device-based interventions like continuous drug delivery systems and deep brain stimulation. However, in advanced stages of the disease, many patients develop symptoms that no longer respond well to these therapies. This review highlights the persistent challenges of drug-resistant motor symptoms. The key message of this review is that while current treatments have greatly improved early and mid-stage PD care, new therapeutic approaches are urgently needed for drug-resistant symptoms.
Keywords
Introduction
Despite the prominent role non-motor symptoms play both in the evolution of disability with advancing Parkinson's disease (PD) and notwithstanding their significance as early disease manifestations that may antedate the appearance of motor signs PD is commonly considered a paradigmatic movement disorder. 1 Current clinical diagnostic criteria are anchored on the presence of cardinal motor features and symptomatic therapies are centered on improving and maintaining motor control. 2 Striatal dopamine replacement with levodopa is still paraphrased as the ‘gold standard’ for symptomatic efficacy of pharmacotherapies even after more than 50 years following its first introduction as a PD drug. 3 In addition, numerous well designed trials have established the symptomatic efficacy of dopamine (DA) agonists and monoamine oxidase type B (MAO-B) inhibitors to improve motor symptoms of PD and these and other agents like catechol-O-methyltransferase (COMT) inhibitors and non-dopaminergic drugs like amantadine, istradefylline or zonisamide are effective in reducing the severity of levodopa-related motor complications. 4 The introduction of device aided therapies like deep brain stimulation (DBS) and continuous drug-delivery approaches have further increased therapeutic options in advanced stages of PD. 5
Although the efficacy of PD pharmacotherapy is overall impressive, there are still significant areas of unmet need in the medical management of PD motor symptoms. With the exception of insufficient drug-responsiveness of PD tremor, which can occur already at the initiation of treatment, most of the unmet needs in the drug treatment of PD motor symptoms relate to later disease stages. They include the pharmacological control of levodopa-response fluctuations and drug-induced dyskinesias and to an even greater degree the control of levodopa-resistant motor manifestations of advanced PD (see Figure 1).
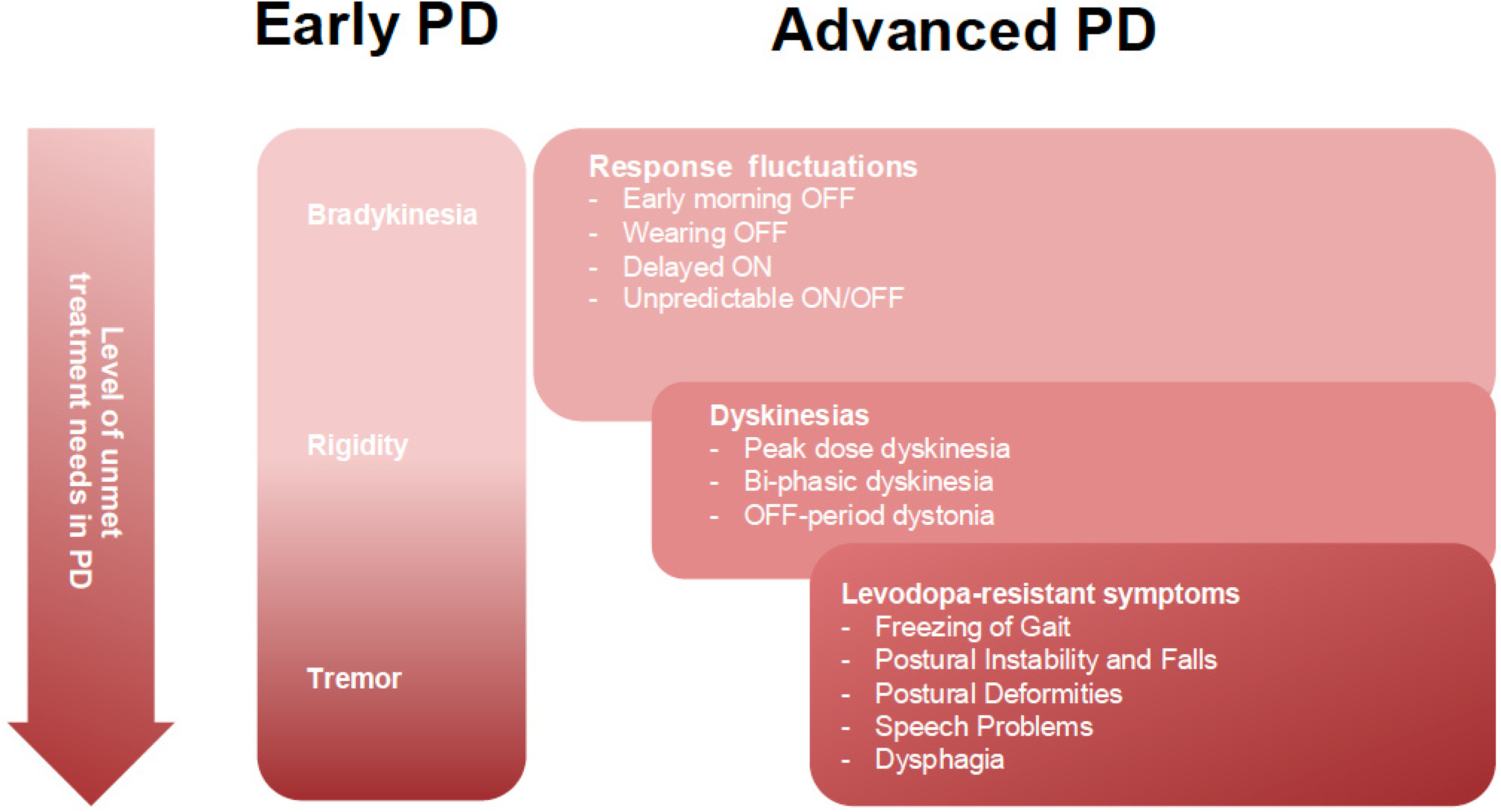
Treatment needs to control PD motor symptoms.
Management of PD tremor
The presence of classical asymmetric 4–6-Hz resting tremor is part of clinical diagnostic criteria for PD as is the responsiveness of the cardinal motor features to levodopa. 6 The response of tremor to levodopa is often perceived as less consistent than that of bradykinesia and rigidity, although in clinical practice underdosing of levodopa or non-adherence are common reasons for poor tremor control. Levodopa doses to achieve tremor control may be higher than those needed for sufficient improvement of bradykinesia, although a recent placebo-controlled trial in subjects with early PD has found similar effect sizes of levodopa for the three cardinal motor domains of bradykinesia, tremor and rigidity. 7 The first step in improving tremor control in early PD should therefore include dose titration of levodopa as well as ensuring proper gastrointestinal absorption, and also trials of dopaminergic, e.g., MAO-B inhibitors, or non-dopaminergic adjuncts, e.g., anticholinergics in younger patients without cognitive impairment or propranolol in those with prominent action tremor or bothersome tremor increase under conditions of stress. 8 If tremor is a disabling part of motor fluctuations, the treatment target is reduction in off-time (see below). In a subgroup of patients, however, PD tremor can be genuinely resistant to dopaminergic or other drug therapies, and this scenario is an important indication for deep brain stimulation (DBS) surgery or MR-guided focussed ultrasound (MRgFUS) thalamotomy or even unilateral surgical thalamotomy where MRgFUS or DBS are not available. 9
The reasons for an insufficient response of PD tremor to dopaminergic treatment are incompletely understood but may involve increased tremor-related cerebellar activity in non-responders versus responders. 10
Challenges and future perspectives
While disabling drug-resistant tremor in PD is usually responsive to non-pharmacological therapies like DBS and MRgFUS or surgical thalamotomy these therapies are invasive and carry a risk of serious complications. Furthermore, DBS procedures are usually not an option for elderly subjects beyond the age of 70 and MRgFUS is not possible in subjects with high skull thickness. There is thus a major need for the development of efficacious pharmacological options for affected patients. Ways forward should include digital monitoring of a subject's individual tremor response pattern to enable optimisation of drug combinations and dosing. In addition, there is still a need for more basic research to better understand the exact targets and mechanisms by which non-dopaminergic agents like clozapine or anticholinergics exert their beneficial effects in patients whose tremor is poorly controlled by dopaminergic drugs. Novel pharmacological targets to be explored include L-type Ca-channels which are involved in the regulation of basal-ganglia-thalamo-cortical as well as cerebello-thalamo-cortical activity as well as subunit-specific modulators of gamma-aminobutyric acid type A (GABA-A) receptors.11,12 Until further progress is made in the pharmacological therapy of dopaminergic-resistant PD tremor assistive devices may enhance function in upper limb motor activities of daily living in those who decline or are ineligible for surgical therapies.13,14
Levodopa-related motor complications
The majority of PD patients successfully treated with levodopa eventually experience some degree of loss of motor control due to the development of drug-related response complications both in terms of response fluctuations and the evolution of drug-induced involuntary movements. These are major determinants of disability in mid-stage PD and usually persist into later stages.
Response fluctuations
Fluctuations in the level of drug response usually become first apparent within the first 3 years of chronic exposure to levodopa, but focused screening in research settings suggests that predictable declines in the level of motor control in the early morning hours prior to the first drug administration of the day as well as towards the end of interdose intervals during the day may affect up to 30% of subjects even in the first two years after starting levodopa.15,16 Importantly, these response fluctuations include variation in both motor control and also the severity of non-motor features of PD. 17
Fluctuation patterns depend on underlying mechanisms which include the short half-life of levodopa leading to plasma level oscillations that eventually translate into fluctuations of striatal synaptic dopamine concentrations, but also erratic gastrointestinal transport an absorption (see Figure 2).
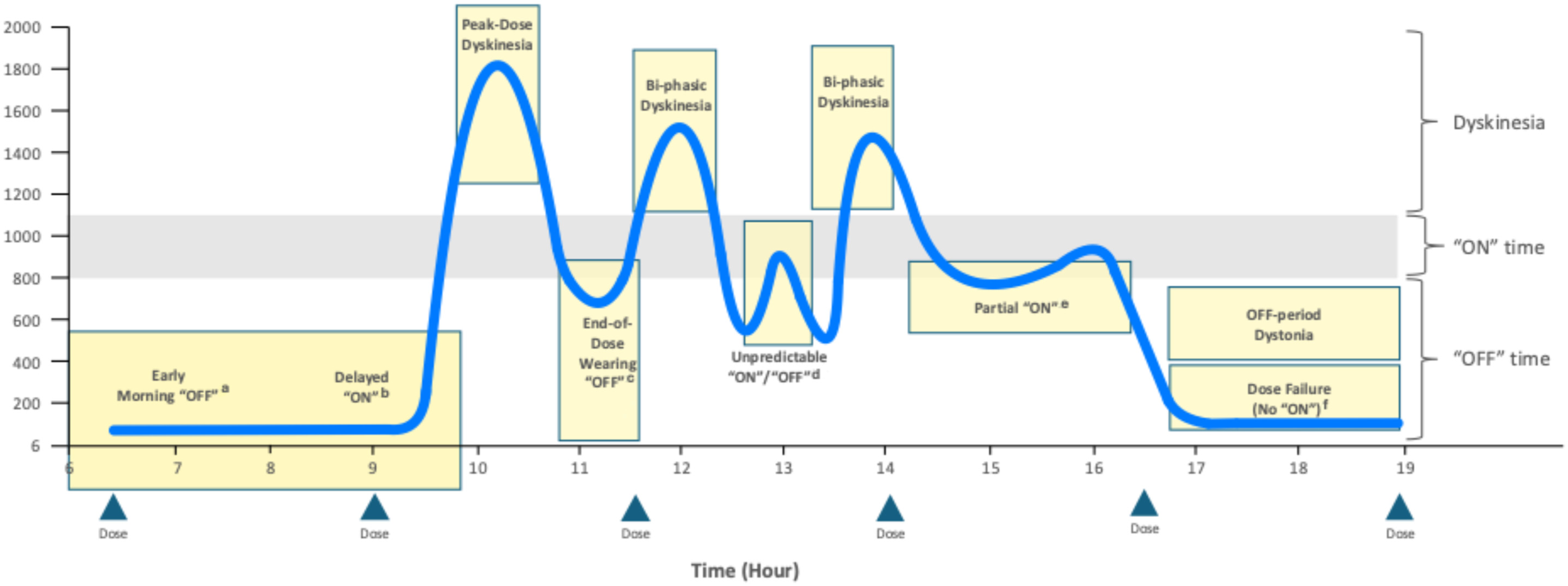
Clinical motor complication patterns and their relationship to levodopa plasma levels. Modified from Isaacson et al. 18
Pharmacological strategies to reduce OFF-time and increase ON-time include increases in dose size and dosing frequency of standard levodopa preparations as well the use of extended-release formulations or non-oral delivery modes of levodopa (Table 1). Well-designed randomized placebo-controlled trials have shown ON-time gains over placebo ranging from 0.5 to 0.9 h with extended-release formulations, and around 1.8 h with continuous intestinal or s.c. infusions of levodopa.19–23 Similar results were obtained in a controlled trial of subcutaneous waking-day infusions of apomorphine. 24 Trials of MAO-B inhibitors like rasagiline or safinamide have shown increased ON-time or reduced OFF-time by slightly less than one hour compared to placebo while adjunct COMT inhibitors have reduced OFF-time by about one hour in studies with opicapone and entacapone and by up to 90 min in trials with tolcapone.25–28 Effect sizes have been somewhat larger in trials of adjunct dopamine agonists therapy. 25 On-demand dopaminergic rescue medications like subcutaneous or sublingual apomorphine and inhaled levodopa have all shown efficacy in aborting OFF-periods within 15 to 30 min but controlled trials assessing their overall effect on daily OFF-time are lacking. 29
Effect sizes of symptomatic drug therapies for levodopa-related motor complications in PD (data from controlled studies).

Some non-dopaminergic adjuncts have also shown efficacy in reducing OFF-time in fluctuating PD with an effect size of 1 h versus placebo for an extended-release formulation of amantadine and around 45 min for istradefylline.25,30
Drug-induced dyskinesias
Levodopa-induced dyskinesias (LIDs) may manifest in synchrony with response fluctuations but are a later event in about one third of patients and may also rarely present before fluctuations. 31 Depending on their occurrence in different phases of the levodopa response cycle LIDs are classified into ON-period, biphasic and OFF-period types (see figure 2).32,33 The most common presentation involves choreic movements of the limbs during ON-periods, often more pronounced on the side initially and more severely affected by parkinsonian signs, while ON-period LIDs of the craniocervical region are dystonic as is OFF-period dystonia, which usually affects the distal lower limbs.34,35
Studies in in unselected samples of PD patients treated with levodopa for an average of six years have found an overall prevalence of LIDs of 30% of which severity was rated as ‘mild’ in 68% and as ‘moderate’ or ‘severe’ in 30%. 36
Risk factors for the development of LIDs include cumulative levodopa dose, longer PD duration at the time of starting levodopa, younger age at onset, female gender and low body weight. 37 Pathogenetic mechanisms are still incompletely understood but involve striatal dopaminergic denervation with loss of presynaptic dopamine storage capacity, discontinuous drug delivery and resulting ‘pulsatile’ dopamine receptor stimulation due unphysiological surges of synaptic dopamine levels and dysregulated dopamine release from serotonergic terminals, which eventually leads to a cascade of maladaptive neuronal responses resulting in altered signaling in basal ganglia-cortical motor networks. 38
In contrast to the multitude of pharmacological interventions with well-established efficacy in reducing OFF-time and increasing ON-time (Table 1) there are only three drug interventions which have been shown to reduce the severity of LIDs in randomized placebo-controlled trials. 4 Amantadine is a drug with multiple pharmacological actions of which antagonism at N-Methyl-D-Aspartate (NMDA) glutamate receptors is believed to underly its antidyskinetic efficacy. 39 The effect size of adjunct amantadine in reducing the severity of LIDs has consistently ranged between 40% and 60% across several controlled trials.40,41 A similar magnitude of effect has also been demonstrated in a randomized trial comparing levodopa-carbidopa intestinal gel infusions with oral therapy. 42 Clozapine has been shown to reduce the duration of ON-time with dyskinesias in a small 10-week placebo-controlled trial involving a total of 50 subjects but carries a risk of drug-induced agranulocytosis requiring regular blood count monitoring. 43
Treating motor complications: challenges and future perspectives
While the pharmacological armentarium to treat levodopa-related response fluctuations may seem impressive there are also significant unmet needs. First, across all controlled trials performed in levodopa-treated patients to date, regardless of type of intervention, reductions in OFF time in comparison to placebo have at best been in the order of two hours and reductions from baseline have generally been less than 60% - even with device-aided continuous drug delivery techniques. 44
Reasons behind this therapeutic gap are multifactorial and include gastrointestinal absorption and transport issues with oral levodopa-based therapies, limitations of the effects of adjunct enzyme inhibitors on peripheral or central pharmacokinetics of levodopa or dopamine, limited effect sizes of non-dopaminergic strategies or blood-brain-barrier transport and central pharmacodynamic issues with non-oral continuous dopaminergic drug delivery - all of which result in failure to achieve continuous striatal dopamine receptor stimulation. 45 In addition, most of the dopaminergic strategies to treat motor fluctuations carry a risk of inducing or worsening drug-induced dyskinesias, with the possible exception of continuous infusions of apomorphine or levodopa. Current continuous dopaminergic infusion therapies themselves are suboptimal regarding ease of use, device-sizes, invasiveness with intestinal infusions or limited skin tolerability with subcutaneous approaches. In addition, device-aided drug delivery systems generally require some level of caregiver support and the implementation of patient support services.
Finally, there is currently no pharmacological therapy with proven efficacy in preventing the development of levodopa-related motor complications, which calls for continued effort in research and drug development to close this gap. One obvious objective for future trials is to test the preventive effects of the early use of drug combinations of levodopa with agents that have proven efficacy in reducing motor complications like COMT inhibitors or amantadine or even combinations of more than 2 agents. Positive signals for potential efficacy of the COMT inhibitor opicapone or the combination of levodopa with amantadine have recently been published. 46 The same applies to trials testing the long-term outcome of new extended-release formulations of levodopa.
The D1-agonists tavapadon has shown efficacy in increasing ‘On’-time without dyskinesias as an adjunct to levodopa and novel long-acting dopamine agonists with enhanced efficacy and improved safety remain on the agenda of PD drug development. 47
The search for new modes of easy-to-use continuous drug-delivery has includes oral solutions of levodopa-carbidopa delivered with osmotic–mini-pumps attached to the teeth (DopaFuse), for which a recent phase 2 trial has shown reductions in plasma level variation and clinical response fluctuations. 48 Non-oral dopaminergic infusion therapies will need to progress towards further enhancing solubility of dopaminergic agents like apomorphine and levodopa to enable similar mini-pump technology as is routine practice in other indications like insulin delivery in type 1 diabetes. This could significantly increase ease of use and open the door to trials of continuous delivery modes in early PD to test their preventive effects regarding the development of motor complications.
Invasive delivery of key enzymes of dopamine synthesis like tyrosine hydroxylase, GTP-cyclohydroxylase or aromatic amino acid decarboxylase directly into the putamen has provided signals of efficacy in phase 2 clinical trials and could conceptually enable more physiological dopamine substation and forestall the development of motor complications. The latter is also one of the driving concepts behind cell-based therapies. 49
Despite disappointing results of previous trials of serotonergic agonists and various glutamatergic, adrenergic, Adenosine 2A or opioid receptor antagonists as well as different metabotropic Glutamate receptor 4 or 5 allosteric modulators to treat drug-induced dyskinesias the example of amantadine should encourage further exploration of non-dopaminergic targets.50,51
A modulator of GPR6, a G-protein coupled receptor expressed in striatal projection neurons of the indirect striato-pallidal pathway, has recently shown efficacy to reduce OFF-time in a placebo-controlled phase 2 trial. 52 Also inhibitors of phosphodiesterase (PDE), which play a role in basal ganglia dopaminergic signaling, have shown efficacy in preclinical models of LID. 53
Combination therapies could also be a way to enhance effect sizes of antidyskinetic drug treatment. A recent proof-of concept study has tested the effect of a fixed combination of two serotonergic drugs – the 5-HT1A agonist buspirone and the 5-HT1B/1D agonist zolmitriptan – and found significant reductions in dyskinesia score after single dose levodopa challenges without compromise of ON-motor scores during a 7-day treatment period (NCT03956979, NCT04377945). 54 Promising results have also been reported for a phase 2 trial of the D3 dopamine receptor antagonist mesdopetam, which showed dyskinesia reduction without compromise of motor control (NCT04435431). 55
Gait and balance problems
Subtle abnormalities of gait are among the early motor signs of Parkinson's Disease and usually respond well to dopaminergic replacement therapies. With advancing disease, gait abnormalities progress in severity and freezing of gait and falls become major sources of disability with limited drug options for their treatment
Freezing of gait
Freezing of gait (FoG) is a debilitating motor complication affecting approximately one-third of patients with PD. 66 It is characterized by sudden, transient episodes of an inability to initiate or continue walking, often triggered by specific scenarios such as step initiation, turning, or navigating through narrow spaces. Episodes typically last a few seconds but can occasionally extend beyond 30 s, significantly increasing the risk of falls and subsequent injury. 67 This makes FoG a critical determinant of reduced quality of life in PD patients. In levodopa-treated patients with motor fluctuations, FoG can be restricted to OFF-periods or persist when patients are fully ‘ON’ (ON-period freezing). Rarely, ON-period freezing can occur as a levodopa-induced phenomenon. 68
The precise mechanisms underlying FoG remain poorly understood. 69 It likely arises from transient disruptions in the locomotor circuitry involving cortical and striatal cholinergic, serotonergic, noradrenergic, and GABAergic projections. Identified risk factors for FoG include longer disease duration, higher levodopa doses, male sex, depression, and cognitive decline. 70
In clinical practice, pharmacological management of FoG mainly relies on optimization of dopaminergic therapy and the reduction of motor fluctuations to control-OFF-period freezing.
Nevertheless, FoG can remain refractory to adjustments in dopaminergic medication and, in some cases, may even be induced by levodopa itself or dopamine agonists, necessitating a reduction in dopaminergic dosage in such situations. 71
While there is some evidence that exercise-based approaches, assistive devices, and neuromodulation including brain and spinal cord stimulation can improve FoG in PD patients, only few RCTs have assessed different types of pharmacological therapies and results have generally yielded mixed results.72–75
Continuous, device-aided drug delivery may be an effective treatment for PD patients suffering from FoG with poor response or refractory to oral medication.76,77
Evidence for efficacy was found in studies investigating the combination of L-threo-3,4-dihydroxyphenylserine (L-DOPS), a precursor of noradrenaline, with the COMT inhibitor entacapone, which showed improvement in FoG, particularly in levodopa-resistant cases. 78
While the efficacy of amantadine remains controversial noradrenergic agents, such as atomoxetine and methylphenidate have shown promise in advanced PD patients receiving subthalamic stimulation.39,79
Positive effects have also been observed in small studies using adenosine A2A receptor antagonists like caffeine and istradefylline. 80
Furthermore, in a post-randomization phase of the initial DATATOP trial, comparing selegiline versus placebo in patients already on levodopa, those participants continuing selegiline treatment were less likely to develop FoG compared to those switched to placebo after 2 years of follow-up. 81 Consistent with this observation, adjunct MAO-B inhibitors like rasagiline or safinamide have also been reported to reduce ON-state FoG in levodopa treated patients with motor fluctuations.27,82
A 10-week randomized trial of the oral nicotine formulation NC001, showed significant reductions in FoG compared to placebo, with improvements attributed to selective nicotinic receptor stimulation. 83
Local injections of botulinum toxin into the calf muscles and cannabinoids have been tested as well, but failed to show efficacy on FoG.84,85
Postural instability and falls
More than one third of a large series of ambulatory PD patients with a mean disease duration of close to eight years reported falls in the preceding three months and around 70% of individuals with PD with a falls history experience recurrent falls. 86
The frequency of falls in PD is nearly double that of the general older population and approximately half of the falls in PD subjects require medical care due to falls related injury.
Risk factors for recurrent falls in PD include age and disease duration as well as other disease related factors like freezing of gait, dyskinesias, orthostatic hypotension, cognitive decline, and visuospatial dysfunction. Co-morbidities like osteo-arthritis or sedating medication also play important roles.
Although levodopa and other dopaminergic drug therapies may reduce falls risk by improving mobility including gait and balance, no studies have directly assessed their beneficial impact on falls and levodopa-induced dyskinesias are a known risk factor for falling.
Cholinergic dysfunction at the level of the basal forebrain and the pedunculo-pontine nucleus is believed to be a major contributor to postural impairment and falls in PD. 87 This has prompted interest in cholinesterase inhibitors as a treatment for gait and balance problems and the available evidence suggests a reduced risk for falls in PD patients treated with these agents. 88 Consistent with this, the oral nicotine formulation NC001 was shown to significantly reduce falls and FoG in a 10-week, double-blind, placebo-controlled trial. 83
A 10-week placebo-controlled trial of the adrenergic agonist droxidopa in patients with neurogenic orthostatic hypotension reported a significant reduction in fall frequency and fall-related injuries compared to placebo, which was not explained by the effect on blood pressure. 89
Challenges and future perspectives
Although freezing of gait, postural instability, and falls remain a major area of unmet medical need, there is a striking lack of drug trials targeting these issues. Only two studies targeting falls in PD are currently ongoing, including a phase 2 trial testing the efficacy pirepemat (IRL752) (NCT05258071), and a phase 3 trial assessing the efficacy of transdermal rivastigmine (NCT04226248).90,91
Ways forward must include a better understanding of the molecular signaling changes in the neural circuits regulating gait and balance, that characterize their dysfunction in PD. Trial designs in human intervention studies will also have to be improved regarding sufficient sample sizes and trial durations, and should include patient-based objective outcome measures using wearable technologies and real-time gait and falls monitoring to enhance sensitivity and objectivity. Combination of multimodal approaches of pharmacological and non-pharmacological interventions like medication, gait training, cognitive therapies, and exercise programs, may offer synergistic benefits and improve outcomes.
Postural deformities
Postural abnormalities (PAs) in PD encompass a spectrum of axial deformities, ranging from mild stooped posture to severe conditions like camptocormia, Pisa syndrome, scoliosis, and antecollis. These deformities are present in approximately 20% of PD patients, with subtype-specific rates of 19.6% for scoliosis, 10.2% for camptocormia, 8% for Pisa syndrome, and 7.9% for antecollis. 92 Their development is associated with advanced age, longer disease duration, greater disease severity, and the akinetic-rigid subtype of PD. 93
The pathophysiology of PAs is heterogeneous and multifactorial, and a range of mechanisms may contribute—either alone or in combination—to their onset and progression. These include muscular rigidity, axial dystonia, myopathy-related weakness, proprioceptive deficits, and structural spinal changes which can all contribute to the development of these deformities. 93
Axial dystonia is considered a significant contributor to deformities such as camptocormia and Pisa syndrome. Pharmacological approaches to the management of PAs in PD produce inconsistent results. Oral or intestinal gel delivery of levodopa has been shown to improve camptocormia, dropped head, and Pisa syndrome in some studies.94,95 Similarly, continuous subcutaneous apomorphine infusion has been associated with improvement in trunk flexion, 96 and selegiline has improved thoracolumbar anteflexion. 97
Axial rigidity, a core motor symptom of PD, is likely a major contributor to the development or worsening of postural abnormalities, especially when acting in conjunction with other mechanisms. Pharmacological interventions that target rigidity—such as levodopa, apomorphine, and selegiline—have been associated with improvements in posture, including camptocormia, Pisa syndrome, and dropped head.94–97 These benefits are thought to result from a reduction in rigidity and enhancement of motor control, although their effectiveness may vary depending on the type and severity of the underlying deformity.
Nevertheless, there is also evidence for the potential of dopaminergic drug treatment to worsen or trigger PAs, including dopamine agonists, levodopa, rasagiline as well as amantadine. 98
The exact mechanism behind these effects is unclear and with the exception of dopamine agonist treatment causality has not always been clearly established, but dystonia induced by dopaminergic stimulation may play a role.
In other cases, muscle weakness and focal myopathy, particularly affecting the paraspinal muscles, have been implicated in the development of PAs such as camptocormia. 91 These cases may be less responsive to dopaminergic therapy and might benefit more from strength-based physiotherapy or other supportive interventions.
Furthermore, proprioceptive deficits and altered sensorimotor integration are also believed to play a role, contributing to difficulties in maintaining upright posture. 91 Over time, these functional abnormalities may be compounded by structural spinal changes, making deformities more fixed and less responsive to therapy. This may also explain why treatments such as physical therapy, orthotic devices, or posture-correction braces often offer only temporary relief, with uncertain long-term benefits.
In addition to dopaminergic agents, non-dopaminergic pharmacologic approaches have shown promise in isolated cases. For instance, istradefylline has demonstrated some benefit in open-label observations as have botulinum toxin (BoNT) injections for focal dystonic components.99,100
Subthalamic nucleus deep brain stimulation (STN-DBS) has been reported to improve camptocormia or lateral trunk flexion in some series, but overall, the effects of DBS on PAs is heterogenous. 101 Physical therapies as well as orthotic devices and posture-correction braces can offer temporary relief, but their long-term benefits remain uncertain.
Challenges and future perspectives
The mechanisms underlying the development of PAs with advancing PD are poorly understood and this is a major obstacle for the development of preventive or symptomatic therapies. In the absence of specific targets for pharmacological interventions efforts should focus on the development and testing of preventive approaches like combinations of optimized medical therapy of motor symptoms with exercise-based protocols to enhance postural control. Along these lines a randomized trial by Zhang et al. is currently evaluating Zhan Zhuang Qigong for Pisa syndrome. 102 Emerging technologies, such as instrumented feedback systems, may hold promise for enhancing real-time postural training and prove useful both as symptomatic and preventive strategy. Testing the efficacy of preventive strategies against the development poses major challenges, mainly in relation to trial duration, and will require long-term pragmatic trials of combinations of pharmacological and non-pharmacological hybrid strategies.
Speech problems
Speech problems, particularly hypokinetic dysarthria, affect up to 90% of patients with PD, significantly impairing communication and social interaction. 103
Hypokinetic dysarthria is characterized by reduced vocal loudness (hypophonia), monotonic tone, imprecise articulation, and decreased intelligibility, all of which worsen with disease progression. 104
The underlying neural mechanisms of speech impairment in PD include dopamine deficiency leading to hypokinesia of laryngeal and respiratory muscles, while non-dopaminergic mechanisms contribute to sensory processing abnormalities, internal cueing difficulties, and impaired self-monitoring of speech output. 105
Levodopa and other dopaminergic therapies usually improve vocal loudness and prosody in PD and detailed assessments have shown improvements in tongue agility, loudness, and articulatory speed during prosodic tasks. 106 However, speech dysfunction increases with advancing disease, to include dysfluency, motor blocks, and palilalia. 106 Levodopa-induced dyskinesias can exacerbate dysarthria and dysprosody and long-term cumulative levodopa exposure has been associated with speech dysfluency and impaired articulation and rhythm. Nonetheless, levodopa still improves some speech related motor control aspects like maximum phonation time or loudness even in advanced PD, however its overall effect on voice quality is limited in this patient population. 107 STN-DBS, while potentially beneficial, carries a risk of stimulation induced dysarthria. 108
Challenges and future perspectives
Similar to other drug-refractory motor symptoms of advanced PD the neural basis of dysfunctional speech output remains incompletely understood. Optimizing dopaminergic basal ganglia neurotransmission is the only well-characterized pharmacological approach to treat speech impairments in PD. The development of automated speech analysis tools has shown promise for capturing subtle changes in speech and monitoring medication effects and may aid the development of personalized treatment regimens. As for disorders of gait, balance and posture these will likely require combinations with non-pharmacological interventions, such as speech and respiratory training programs.
Dysphagia
Dysphagia eventually affects over 80% of PD patients and can be responsible for major adverse health outcomes resulting from impaired medication intake, malnutrition, dehydration and aspiration pneumonia. 109
The pathophysiology of dysphagia in PD is characterized by impairments affecting all phases of swallowing and is driven by both dopaminergic and non-dopaminergic dysfunctions. Beyond motor impairment cognitive and sensory dysfunction contribute significantly to swallowing difficulties. 110
Current pharmacological management mainly involves dopaminergic medications, and a recent small retrospective investigator-blinded study also showed significantly improved measures in the oral and pharyngeal swallowing phases of the MAO-B inhibitor safinamide using video fluoroscopic swallowing assessments in nine PD subjects. 111
Overall, current evidence suggests that dopaminergic treatments may be more effective in the early stages of PD, whereas their impact diminishes as the disease progresses. 112
Botulinum toxin injections targeting upper esophageal sphincter dysfunction may offer benefits for select patients, but this approach requires specialized expertise and careful patient selection.
Non-pharmacological approaches like neurorehabilitation including swallowing therapy, compensatory strategies like bolus modification, and video-assisted exercises, have demonstrated some efficacy, but the validity of these results is often limited by small sample sizes and lack of control groups. 112
Non-invasive brain stimulation techniques, such as repetitive transcranial magnetic stimulation (rTMS) and transcutaneous electrical stimulation (TES), have been explored with limited and inconsistent effects on dysphagia measures. 113
Challenges and future perspectives
Despite the high prevalence and major impact of dysphagia on functional capacity in advanced PD, there is a striking lack of high-quality randomized controlled trials, and there is no pharmacological treatment specifically approved to treat dysphagia in PD. Currently, one phase 3 study assessing the effect on dysphagia of nicergoline, an ergot derivative, is under investigation in patients with PD or stroke (NCT05551182).
The limited responsiveness to dopaminergic drugs highlights the need to further elucidate the neuropharmacology of swallowing and the pathways and mechanisms contributing to swallowing dysfunction. High-quality randomized controlled trials with standardized and sensitive swallowing assessments across different disease stages are needed to evaluate both existing and novel therapies. Integrating advanced technologies may provide valuable insights into the dynamics of swallowing and help personalize treatment strategies.
Conclusions
PD stands out among the neurodegenerative diseases by the availability of highly effective symptomatic therapies. Clinical trials performed over the last 3 decades have resulted in a large armentarium of pharmacological agents with established efficacy to treat the cardinal motor features of the disease as well as the motor complications that can develop with sustained exposure to levodopa. As a result of this therapeutic progress in the medical management of PD there has been an impressive increase in quality of life and functional independence and also in the numbers of years individuals live with PD. While overall positive, this development has also meant that patients as well as their carers and treating physicians increasingly face the challenges of progression into the late stages of this illness which include a variety of disabling motor problems which poorly respond to currently available drug therapies. They include gait and balance problems, falls, postural deformities, dysarthria and dysphagia. The underlying pathophysiology and the signaling changes in the networks subserving these motor functions are incompletely understood making targeted drug development challenging. Progress in this difficult therapeutic arena of PD drug treatment can only be made by advancing our understanding of the molecular underpinnings of dysfunction in the relevant motor circuits to enhance drug discovery. Ultimately, the refractory motor problems of late-stage PD are a call to action regarding the development of disease-modifying therapies in order to forestall or prevent their development.
Footnotes
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: BH reports honoraria from Novartis AG, AbbVie, Bial and grants from the Austrian science fund (FWF) outside the submitted work.
Werner Poewe has received lecture fees and honoraria for consultancy in relation to clinical drug development programs from AbbVie, AC Immune, Alterity, BIAL, Boehringer, Britannia, Lilly, Eisai, Lundbeck, Roche, Takeda, Britannia, Eisai, Roche, Stada, and Zambon; grant support from The Michael J. Fox Foundation and the EU FP7 & Horizon 2020 programs; and safety monitoring board membership for UCB. He has leadership roles in the Movement Disorder Society, Austrian Society of Neurology, and Austrian PD Society.
Data availability statement
This is a review article and does not involve original research data. All data presented or analyzed in this review are derived from publicly available sources, including published research articles, and clinical trial databases. Any references to specific studies or datasets are properly cited in the manuscript. Further details regarding the sources of information can be provided upon request.
