Abstract
A definitive diagnostic test for Parkinson's disease (PD) remains elusive, so identification of potential biomarkers can facilitate diagnosis and early intervention. Two dogs were trained to distinguish between dry skin swabs obtained from people with Parkinson's (PwP) and control participants. After 38–53 weeks of training on 205 samples, the dogs were tested in a double-blind trial using 60 control and 40 target (drug-naïve PwP) samples. They each showed high sensitivity (70% and 80%) and specificity (90% and 98%). This supports previous findings that dogs can be trained to reliably detect the odor of PD.
Plain language summary
There is currently no single, reliable test for Parkinson's disease. Understanding the biological changes that happen in the body may help early diagnosis. Dogs have an incredible sense of smell and have been trained to detect a variety of diseases. Here we have trained two dogs to tell the difference between the oily skin secretions, known as sebum, from people with Parkinson's and those without. After their training, the dogs were tested using 60 new control and 40 target skin swabs. The dogs correctly identified 70% and 80% of the Parkinson's samples and correctly ignored 90% and 98% of the control samples. These results support earlier research showing that dogs can be trained to reliably detect the smell of Parkinson's disease.
Introduction
Identifying diagnostic biomarkers of Parkinson's disease (PD) is the subject of much ongoing research, 1 particularly those that may predict development, or help to diagnose disease earlier. 2 One early non-motor symptom is seborrheic dermatitis: excessive sebum (a waxy, oily biofluid) excreted from the sebaceous glands in patients with PD.3,4 This is associated with lipid dysregulation 5 and could potentially be used as an indicator for the disease.6,7 Sebum has been noted to have a distinctive odor in repeated blinded studies with Joy Milne, who has hyperosmia. 8 Dogs have been trained to detect the scent of many human diseases9–13 from prostate cancer 14 to malaria 15 and Covid-19. 16 In a prospective, diagnostic case-control study, 17 three trained dogs were tested on sebum samples from PD patients, and matched controls. Using the diagnostic criterion of two of the three dogs alerting, sensitivity and specificity rates of 91% and 95% to medicated and 89% and 86% to drug naïve samples, respectively were achieved. Holt and Johnston 17 saw similarly high levels in a mixed-breed cohort of 23 home-owned dogs (86% sensitivity and 89% specificity). The authors concluded that trained dogs could be a useful, quick non-invasive and cost-effective method to identify patients with PD, but they provided limited information on the dog's training and testing.
In the current study, we further tested the capacity of trained dogs to detect PD using sebum swabs. Since the dogs are effectively the measuring instrument, it is important to provide full details of their training, calibration and testing. To simulate real operations, unlike Gao et al., 18 we did not use precisely matched-control samples. However, since it is vital to minimize all confounding factors, 19 we trained dogs using samples taken shortly after diagnosis and a control group ranging from healthy individuals to patients with other neurological conditions. Dogs were trained by a professional dog trainer (MS) using an established protocol 14 and then tested during double-blind trials.
Methods
Ethical approval was obtained by the NHS Health Research Authority (IRAS project ID: 191917, REC reference: 15/SW/0354). Parkinson's disease participants (diagnosed by a Movement Disorder specialist as fulfilling the UK Brain Bank criteria 20 ) were recruited from 25 different NHS. 6 Dry skin swabs were collected as described in Supplemental Material 1 and by Hoare et al. 21 Control samples were collected from people accompanying people with Parkinson's (PwP) to clinics and from volunteers attending the Medical Detection Dog Centre.
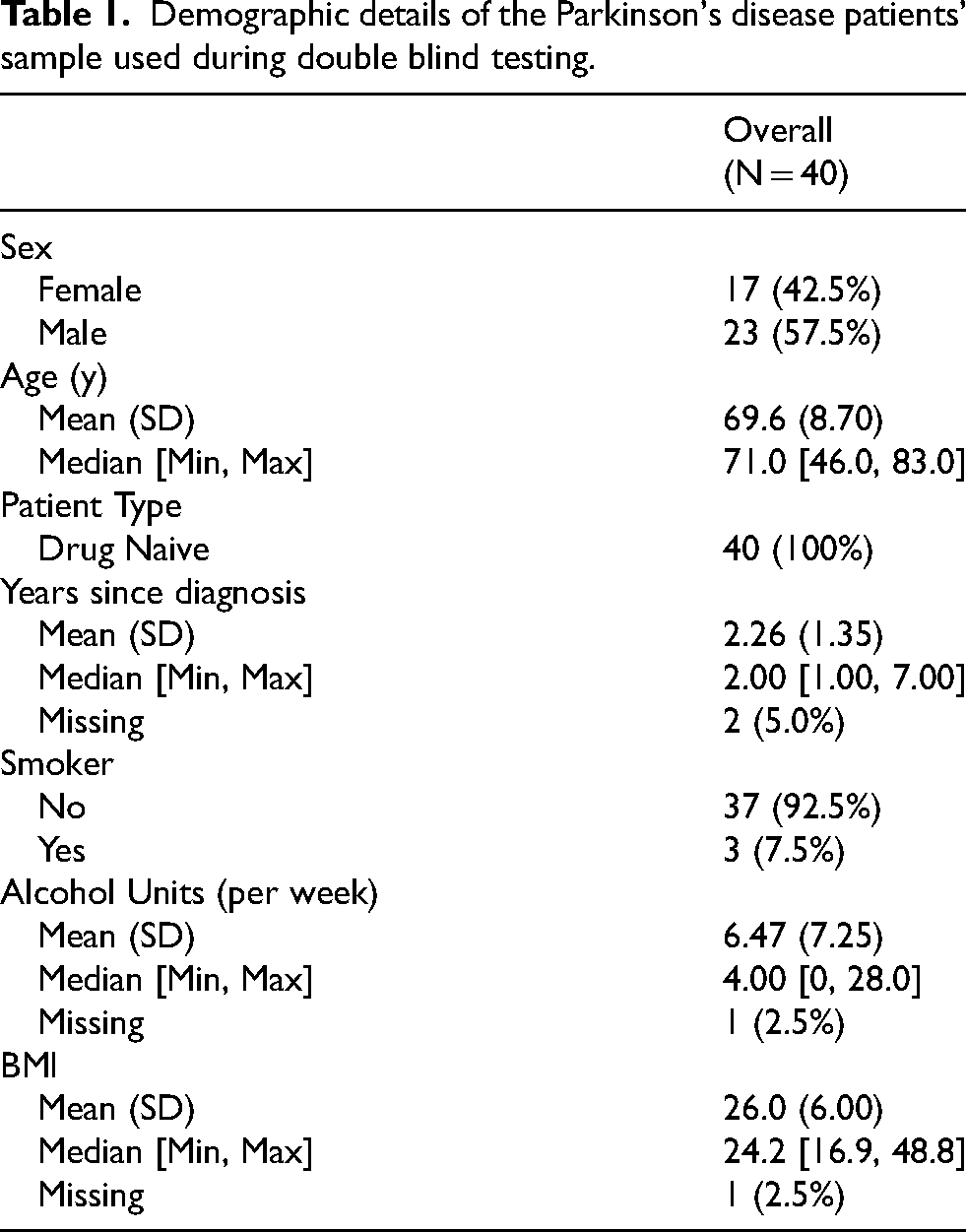
From the sample bank, 130 samples from PwP (Table 1) and 175 from controls were selected to match demographics (defined as age, Body Mass Index [BMI], gender, smoking status) as closely as possible. These were divided into 205 training and 100 testing samples. Those used for testing were all drug naïve (not known to have any medication administered for PD), while several medicated patients were used in the latter stages of training. All samples were cut in half ahead of storage. Duplicates were either both used for training, or one used in training, and the other as a “filler” control during testing. The training samples were labeled, but the test samples were given only a code, so testing was totally blind.
Demographic details of the Parkinson's disease patients’ sample used during double blind testing.
After initial screening of ten dogs, five were deemed to have potential aptitude and commenced initial training. As training increased in difficulty over time (Supplemental Material 2), three of the dogs failed to learn, and were rejected. Only the data from the two remaining dogs a two-year old male Golden Retriever (Dog 1) and a three-year old male Labrador×Golden Retriever (Dog 2) are presented. After 38–53 days of training the dogs proceeded to a double-blind randomized controlled trial and were presented with 100 previously unseen samples; from drug naïve individuals with confirmed PD (n = 40) and controls (n = 60). Due to the scarcity of controls, we used an additional 100 duplicates of control samples encountered by the dogs during training as “fillers” to facilitate presenting lines of four samples. The dogs’ responses to these were not included in any analyses.
Samples were presented in the same way as during training, in a four-stand line. In each “line” there was either none or one positive sample. Both the trainer/handler and experimenter were blind to the presence and position of any targets. Ten control and five positive samples from the training set were used for calibration, to confirm the dog was focused on the scent. Calibration runs could be requested at any time, were unblinded and their results were not included in the analyses.
The regular trainer guided the dog to search each line of samples. Dogs were permitted to pass over each line a maximum of four times. Once the handler was confident of the dog's choice, a decision was communicated to the experimenter. For each sample the dogs’ behavior was coded as Indication, Hesitation, Interest, No Interest or Not Searched, with Hesitation, Interest, and No Interest all treated as negative responses for analysis.
Each line was also presented in reverse order so that samples for which no decision was made were re-presented. Then any unsearched samples were collected together in new lines, until a decision had been made for all samples.
A blinded experimenter entered the responses into the database. The software revealed whether the response was correct, and if so, the trainer rewarded their dog. If any sample was incorrect, no reward was given. The database was password protected and only the trial statistician (SM) knew both passwords prior to trial completion.
We used the first encounter with each of the 100 novel samples to calculate sensitivity (proportion of target samples found) and specificity (proportion of control samples not responded to) for each dog. We also tested whether there was any association between the errors made by each dog, using Fisher’s exact tests.
Results
During testing both dogs discriminated between the PD positive and control samples they had not encountered before (p<0.0001), with sensitivities of 70.0% and 80.0% and specificities of 90.0% and 98.3% (Table 2).
Sensitivity and specificity of two dogs in double-blind trials and agreement between them. p-values are from Fisher's exact tests for correlation between the dogs' responses, and numbers in brackets are the expected numbers if there were no correlation or 95% confidence intervals for sensitivity and specificity. p < 0.0001 for differences in response rates between positive and control samples for both dogs.

Agreement between the two dogs was higher than expected by chance for positive samples: and for control samples was close to expectation (Table 2).
Discussion
This study confirms previous findings,17,18 that dogs can be trained to discriminate between PwP and control sebum swab samples. Although only two dogs from the initial ten trialed were deemed suitable to complete full training, their subsequent performance demonstrates it's possible for dogs of the correct aptitude to learn the task.
Dogs achieved high sensitivity and specificity, showing there is an olfactory signature distinct to patients with PD. Sensitivity levels of 70% and 80% are well above chance, considerably higher than other medical conditions (e.g., 41% bladder cancer 14 ) and comparable to malaria detection. 15 The other controlled study examining detection of PD by dogs, obtained higher sensitivity (91%), but similar specificity (95%) as seen here (98% and 90%).
There are multiple plausible reasons for the difference in sensitivity between the two studies; Gao et al. used consensus responses of two dogs as the detection criterion, 18 while our analysis took each dog's responses individually. Gao's testing protocol used PD samples and perfectly matched-controls, while our controls were not matched which may make the task harder, but we believe more authentic. Gao et al. also presented a known PD target to dogs ahead of each testing run, so they were able to “match-to-source”, which may improve detection but may not be practical in operational settings. Gao's dogs were trained over two years, while ours were less than a year, although the number of samples encountered as well as other details were not described by the Gao et al., so cannot be compared. However, the studies together suggest that refinements to training and sample presentation in the future could lead to further increases in detection accuracy.
In our study, while the specificity of Dog 2 was 98% reflecting very few false alerts, and similarly the dog showed the highest sensitivity (80%), Dog 1 showed slightly lower specificity (90%) and sensitivity of only 70%. These differences may reflect personality variations 22 or subtle differences in training. Agreement between dogs was higher than would be expected by chance; suggesting some samples were easier to identify than others. Initial inspection of samples which both dogs incorrectly identified suggested no obvious commonalities, but further research may help reveal relevant factors.
In conclusion, the performance of dogs trained on PD dry skin swabs here and in the past studies is encouraging. Since increased sebum secretion has been noticed before motor signs of PD23,24 and we have recently shown that individuals with iRBD have a distinct volatile signature and odor, 7 early canine detection of biomarkers in sebum could potentially aid existing methods. Laboratory tests have reported accuracies of up to 98%,25,26,27 but have yet to be implemented in any healthcare setting. SPECT scans have similar accuracy,28,29 but are rarely used as a primary diagnostic. Hence, while we do not envisage dogs being diagnostic, they could potentially, with refinements, help to validate methods of clinical utility (as we have previously demonstrated with a hyperosmic individual5–7) and aid rapid screening and diagnosis. This may in turn lead to opportunities for earlier intervention and be especially beneficial for hard to diagnose cases of PD. Longitudinal studies of dogs’ capacity to alert prior to formal diagnosis are now required.
Supplemental Material
sj-docx-1-pkn-10.1177_1877718X251342485 - Supplemental material for Trained dogs can detect the odor of Parkinson's disease
Supplemental material, sj-docx-1-pkn-10.1177_1877718X251342485 for Trained dogs can detect the odor of Parkinson's disease by Nicola Rooney, Drupad K Trivedi, Eleanor Sinclair, Caitlin Walton-Doyle, Monty Silverdale, Perdita Barran, Tilo Kunath, Steve Morant, Mark Somerville, Jayde Smith, Julie Jones-Diette, Jenny Corish, Joy Milne and Claire Guest in Journal of Parkinson's Disease
Footnotes
Acknowledgements
We thank the Michael J Fox Foundation (grant ref:12921) and Parkinson's UK (grant ref: K-1504) for funding this study. This work was also supported by an EPSRC DTA grant to the School of Chemistry, which has funded the PhD project of E.S., and a BBSRC DTP grant which has funded the PhD project of C.W.D. (BB/R505869/1). We also thank our recruitment centres for their enthusiasm and rigor during the recruitment process. We are grateful to all the participants who took part in this study as well as PIs and nurses across all the recruiting centres. DT is thankful for CAMS, UK funding to support his time for this research.
ORCID iDs
Ethical considerations
Ethical approval was obtained by the NHS Health Research Authority (IRAS project ID: 191917, REC reference: 15/SW/0354).
Consent to participate
Written informed consent was obtained from all participants.
Consent for publication
Written consent explicitly stated publishing anonymised data.
Funding
We thank the Michael J Fox Foundation (grant ref:12921) and Parkinson's UK (grant ref: K-1504) for funding this study. This work was also supported by an EPSRC DTA grant to the School of Chemistry, which has funded the PhD project of E.S., and a BBSRC DTP grant which has funded the PhD project of C.W.D. (BB/R505869/1). We also thank our recruitment centres for their enthusiasm and rigor during the recruitment process. We are grateful to all the participants who took part in this study as well as PIs and nurses across all the recruiting centres. DT is thankful for CAMS, UK funding to support his time for this research: This work was funded by Michael J Fox Foundation and Parkinsons UK.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability statement
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
