Abstract
Background
Cognitive impairment and dementia are common findings in patients with Parkinson's disease (PD). However, the long-term effects of subthalamic deep brain stimulation (STN-DBS) on cognition remain unclear.
Objective
We report short- and long-term effects of STN-DBS on cognition in PD.
Methods
We analyzed neuropsychological data before STN-DBS surgery, 3-month post-surgery, 1-year post-surgery and in a long-term follow-up (8–15 years post-surgery) to examine the effects of STN-DBS on cognition.
Results
81 patients with a mean disease duration of 13.0 years were examined before surgery. 50.6% were identified with mild cognitive impairment (MCI), having a mean disease duration of 14.2 years. Pre-surgical PD-MCI was not associated with clinically diagnosed dementia (PD-D) before death or before long-term follow-up (OR 0.8, 95% CI 0.3–2.2, p = 0.714), but disease duration at the time of surgery was associated with development of PD-D (OR 1.2, 95% CI 1.1–1.3, p = 0.005). Verbal fluency declined significantly 3 months after surgery, while other domains remained unaffected. In neuropsychological testing at long-term follow-up (N = 29), global cognitive impairment or dementia was found in 19 patients. The presence of depressive symptoms before surgery was associated to PD-D at long-term follow-up. Death before long-term follow-up was more common in patients with pre-surgical MCI than in patients with normal cognition.
Conclusions
Influence on cognition was described in a short- and long-term follow-up study up to 15 years after STN-DBS surgery in PD. Disease duration, but not pre-surgical MCI was associated with development of dementia. Impaired verbal fluency was observed both in a short- and long-term follow-up.
Plain language summary
People with Parkinson's disease (PwP) can be treated with a surgical procedure called deep brain stimulation (DBS), when oral medication is no longer effective. In DBS, electrodes are implanted in a deep part of the brain affected by the disease. Stimulating this area can improve motor symptoms. Cognitive impairment and dementia are common in later stages of Parkinson's disease (PD). However, the long-term effects of DBS on cognition remains unclear. Our study reports cognition over time in PwP with DBS. We followed 81 PwP for up to 15 years after DBS. We examined cognition using tests covering the different cognitive domains and mood. Participants were tested before surgery, 3 months after surgery, 1 year after surgery and 8–15 years after surgery. We found that cognition remained relatively spared short-term, but that a test targeting language was affected. At the long-term follow-up assessment, cognitive impairment was common. We found that the PwP who had developed some cognitive changes before DBS did not have an increased risk of developing dementia, compared to PwP having normal cognition before surgery. We discovered that a longer disease duration at the time of surgery, was associated with developing cognitive impairment and dementia long-term. The presence of depressive symptoms was not affected by DBS, but in long-term follow-up examination, it was more common in people with dementia. We conclude that DBS does not increase the risk of dementia in PwP, and that dementia is related to disease duration and the natural progression of PD.
Keywords
Introduction
Deep brain stimulation of the subthalamic nucleus (STN-DBS) is a widely-established treatment of advanced, levodopa-responsive, fluctuating Parkinson's disease (PD). 1 While several short-term follow-up studies of patients treated with STN-DBS showed continuous effect on motor symptoms, a progression of non-motor symptoms, including development of dementia was found.2,3 Cognitive changes are common in PD and several studies have shown that mild cognitive impairment (MCI) can be seen early in the course of the disease and may be an indication of the development of dementia.4–7 As the disease progresses, most patients develop dementia.8,9 Depression is also a common finding amongst patients with PD throughout disease development and has a substantial impact on patients’ quality of life.10,11
Little is known of the long-term effects of STN-DBS on cognition, and an evaluation must take into consideration the progression of disease as well as the background risk of dementia. Patients undergo neuropsychological examination to rule out dementia in the evaluation for DBS surgery, allowing some cognitive impairment. 12
The aim of this study is to evaluate long-term cognitive changes following STN-DBS through an analysis of neuropsychological data before and after surgery including a long-term follow-up assessment after more than 8 years.
Methods
This study is an exploratory study based on a clinical follow-up of consecutive patients in one DBS Center. When the DBS treatment was initiated in Copenhagen, a protocol was performed based on the Core Assessment Program for Surgical Interventional Therapies (CAPSIT) criteria. 12 In the first years after the treatment protocol was initiated, data before surgery, 3 months after surgery and 1 year after surgery were collected systematically and saved in clinical files. The patients were evaluated using the Unified Parkinson's Disease Rating Scale (UPDRS) and had neuropsychological assessments. In addition, clinical and demographic data were collected. After 3 years, the 3 months evaluation was omitted, wherefore only some patients participating in our study had a 3-month post-surgical evaluation of neuropsychology. Because of low attrition to the Center, almost all patients were followed to death, or the time of this long-term follow-up study, which aimed to re-examine the patients more than 8 years after surgery. The patients who could not participate because of dementia or death, were included with clinical data from patient files. Description of the protocol and the effects of STN-DBS on motor symptoms and activity of daily living for this patient population have been reported by Thomsen et al. (2020). 13 The timeline of the study is depicted in Figure 1.
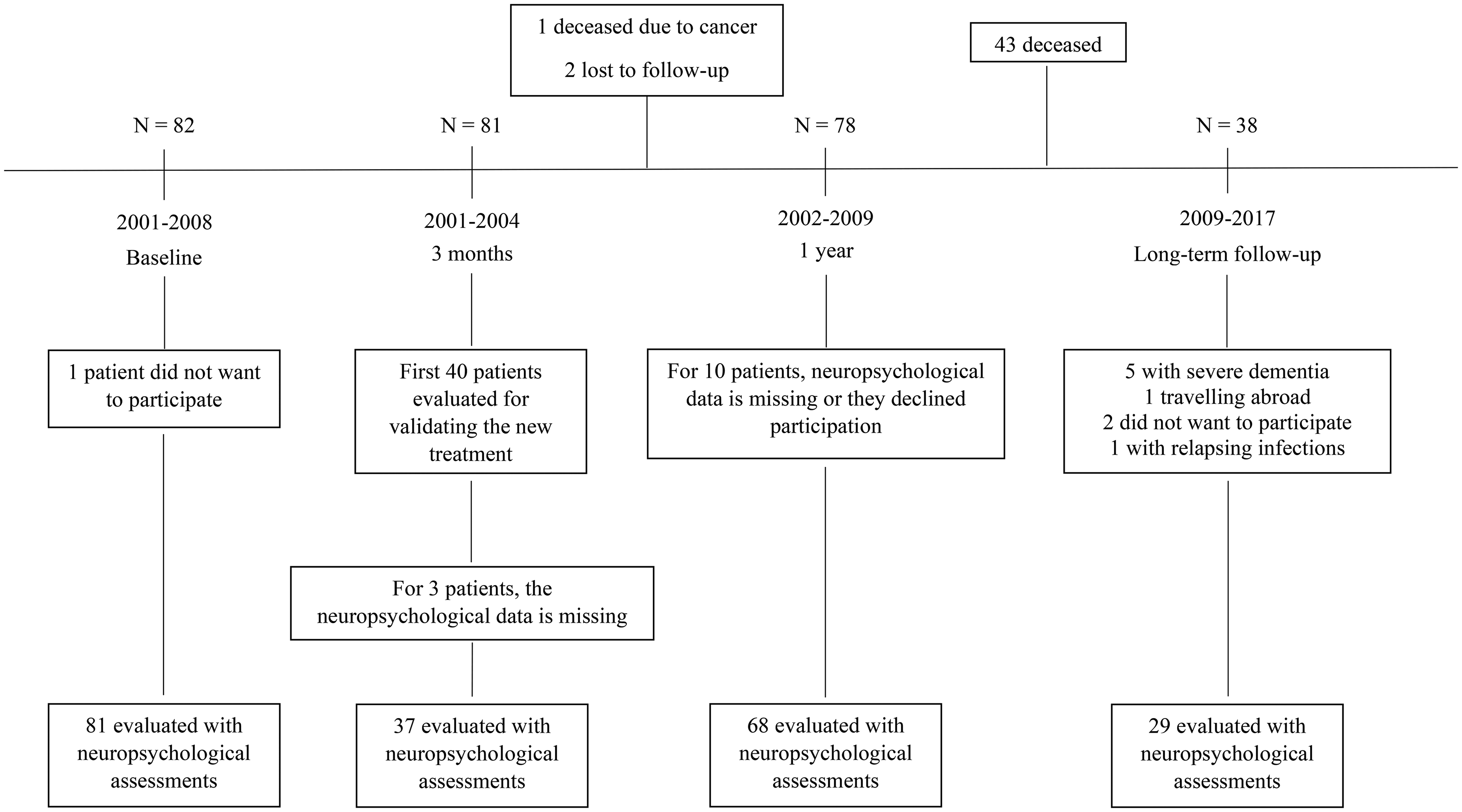
Timeline. Figure 1 shows a timeline of the study. The timeline includes the number of patients presented (N) at each follow-up point and the number of patients included for neuropsychological evaluation. The timeline accounts for reasons why patients were not included for evaluations or lost to follow-up.
We report here neuropsychological data from this population. We curated clinical medical records from the time of the pre-operative examination to the time of the long-term follow-up, or to the death of the patient. Patients selected for a pre-operative evaluation had been examined by their neurologist to rule out clinically diagnosed dementia. In addition, patients with significant depressive or other psychiatric disturbances requiring further treatment were not candidates for surgery, while patients with well-treated depression or hallucinations were able to proceed with surgery. After inclusion for STN-DBS, neuropsychological examinations were carried out by neuropsychologists to secure that an uncovered dementia was not missed before surgery.
From 2001 to 2008, a total of 82 patients with PD were treated with bilateral STN-DBS surgery at Copenhagen University Hospital. A total of 81 patients (females n = 31, males = 50) were included in this study. The long-term follow-up examination was carried out 8–15 years (mean 11.6 years) after surgery (July 1, 2017). By then, a total of 43 patients were deceased, 38 patients were still alive, and of those, 29 were included for the long-term follow-up neuropsychological examination (Figure 1).
In this exploratory report, we follow the changes in cognition after STN-DBS in 81 PD patients up to 15 years after surgery. From patient files, it was noted if patients were given a clinical diagnosis of dementia by their neurologist either based on examinations or based on the initiation of treatment with an acetylcholine-esterase inhibitor. In this study, we define dementia as clinically diagnosed dementia by the treating neurologist, henceforth termed Parkinson's disease dementia (PD-D). We do not use the results of the neuropsychological tests at each follow-up point to diagnose patients with PD-D dementia but describe the results of the tests. The neuropsychological tests at each follow-up point are used to define patients with normal cognition (NC) or MCI. We have divided the baseline group into two groups, whether they had NC or MCI before STN-DBS, to assess long-term outcomes in the two groups.
Surgical procedure
The patients were included in accordance with the inclusion criteria for DBS surgery suggested in the CAPSIT protocol. 12 They were diagnosed with levodopa-responsive PD with a disease duration of at least 5 years, motor fluctuations and levodopa-induced dyskinesia despite optimized medical therapy, a Hoehn and Yahr scale ≥3.5 and were found neither demented nor clinically depressed.
Exclusion criteria were lack of response to medication, contraindications to surgery, dementia or significant psychiatric disorders. Medicine-induced hallucinations were treated, and the patient was excluded from surgery if symptoms persisted after treatment.
The surgical procedure is described in Thomsen et al. (2020). 13 Also see the Supplemental Material.
Neuropsychological tests
The cognitive test battery included an evaluation of memory, attention, concentration, language, executive function, visuo-spatial function and depressive symptoms. The cognitive evaluation was sought to be performed in the best medicated state, “best ON”.
The neuropsychological test battery that was used primarily, included the same neuropsychological tests for all patients, but was changed slightly during the follow-up period due to new neuropsychologists in the department. The test battery performed at long-term follow-up was based on the same tests being used before surgery.
MCI was determined according to level I criteria described by Litvan et al. (2012). 14 Impairment was demonstrated by performance of 1 standard deviation (SD) below age, gender and culturally appropriate norms. MCI was defined by impairment on a scale of global cognitive abilities or on at least two neuropsychological tests, represented by either two impaired tests in one cognitive domain or one impaired test in two different cognitive domains. 14
The premorbid intellectual level was estimated with the Danish Adult Reading Test (DART 45), which is similar to the National Adult Reading Test in a revised version with increased reliability (maximum score 45). 15
The Mattis Dementia Rating Scale (MDRS) 16 and/or the Mini-Mental State Examination (MMSE) 17 were used as a global test for dementia.
Basic attention and concentration were assessed by the Wechsler Adult Intelligence Scale-revised (WAIS-R Digit Span), which was scored as one summary value that is age-normed. 18 Delayed Repetition of Digits and Serial Digit Addition, the latter being similar to Paced Auditory Serial Addition Test (PASAT). 19 From 2004 and onwards (1-year evaluation) some psychologists chose to include the Symbol Digit Modalities Test (SDMT) 20 and continuous subtraction (100–7) to examine attention and concentration, while the Street Completion Test and the Clock-Drawing Test were used to examine visual perceptual functioning. 21 Attention and working memory were also examined with an untimed Simple Stroop Color-Word. 22 In the untimed Simple Stroop Color-Word test the patient is shown small cards sequentially, and the task is to name the color of the print on the card. The first 10 cards contain colored crosses, then follow 40 incongruently colored color names. For 5 words the colors of the print matched the word, which increased the likelihood of an error. The score was the number of correct named colors for the 40 color-words. The lack of timing made the test less susceptible to non-executive factors, e.g., general slowing of speed.
Memory tests included Serial Learning of 10 words with selective reminding, 23 which had a high load on attention and strategy, and the Category Cued Recall (CCR) part of the Double Memory Test, 24 where results were recorded as the number of correct immediate recall and the number of correct recalls after a 30-min delay. The results of Serial Learning were recorded as the number of errors during learning and as 10 min delayed recall.
The executive function was tested by either Wisconsin Card Sorting Test (WCST) with the modifications suggested by Nelson 25 or verbal fluency test. For elaboration, maintenance and shift of mental sets the WCST was included. Verbal Fluency was assessed for category (number of animals mentioned in 1 min), literal (number of S-words mentioned in 1 min) and alternating (number of alternating A- and F-words mentioned in 1 min). General problem solving ability was assessed with WAIS-R Picture Arrangement 18 that was split into 2 subsets with equal and un-equal numbered items respectively to obtain parallel sets suitable for retesting (score 0–10).
The Montgomery-Åsberg Depression Rating Scale (MADRS) 26 was used to screen patients for depressive symptoms. At 1-year evaluation the Major Depression Inventory Scale (MDI) 27 was sometimes used instead of MADRS. The MADRS or MDI were not used to diagnose clinical depression but solely used to screen for depressive symptoms, as they might affect cognitive test results.
At long-term follow-up the test used for assessment were MDRS, Serial Learning, WAIS-R Digit Span, CCR (immediate and delayed recall), Delayed Repetition, Delayed Serial Addition, Picture Arrangement, Simple Stroop Color-Word, Verbal Fluency with Category Word Fluency, Literal Word Fluency and Alternating Word Fluency, WCST, MMSE, and MADRS. These tests were also the tests most commonly used at baseline, 3 months and 1 year follow-up.
Lower education was defined as completed primary school and/or high school or dropped out of school, medium education included a short-term or medium-term higher education programs, and high education included long-term higher education programs.
Statistics
Long-term outcomes were compared to baseline using the Wilcoxon signed-rank test with continuity correction for matched pairs. The statistical tests were two-tailed and the level of significance was calculated as a probability value (p) < 0.05. For analysis of patient demography an unpaired two-sample t-test was used to compare the mean of two independent groups when the assumptions of normal distribution and homogeneity in variances were met and Pearson's Chi-squared test with Yates’ continuity correction was used for testing independence between two categorical variables.
Multivariate linear regression was carried out to adjust for potential confounders of each neuropsychological test. Confounders were sex, age at surgery and level of education.
Logistic regression with conversion to PD-D as outcome was used to calculate odds ratio (OR) with a 95% confidence interval (CI). The variable of interest was pre-surgical MCI and disease duration with and without adjustment for age at surgery, sex and level of education.
Depressive symptoms rates were compared by Pearson's Chi-squared test.
The correlation between depressive symptoms and dementia at long-term follow up was analyzed by logistic regression for calculation of p-value, OR and 95% CI with and without adjustment for age, sex and educational level.
Data reported are values adjusted for age, sex, and education. All statistical analyses and graphical depictions were performed in the software RStudio version 1.1.383 (R Core Team (2019). R: A language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria.).
Ethical considerations
The study is approved by the regional ethics committee, H-15007736. Informed consent was obtained for all participants who were alive at the time of the study. It was approved by the committee to use data from the files from deceased patients.
Results
Of the 81 included patients, 37 had a 3-month post-surgery assessment, 68 had a 1-year post-surgery assessment and 29 patients participated in a long-term follow-up study (Figure 1). Table 1 shows the demographic data and characteristics of the patients at baseline, 3 months, 1 year and long-term, and the characteristics of the patients not available for long-term follow-up. The group of patients available for long-term follow-up were younger and had a shorter duration of disease at the time of surgery compared to the patients not available for long-term follow-up.
Patients demographics and characteristics.
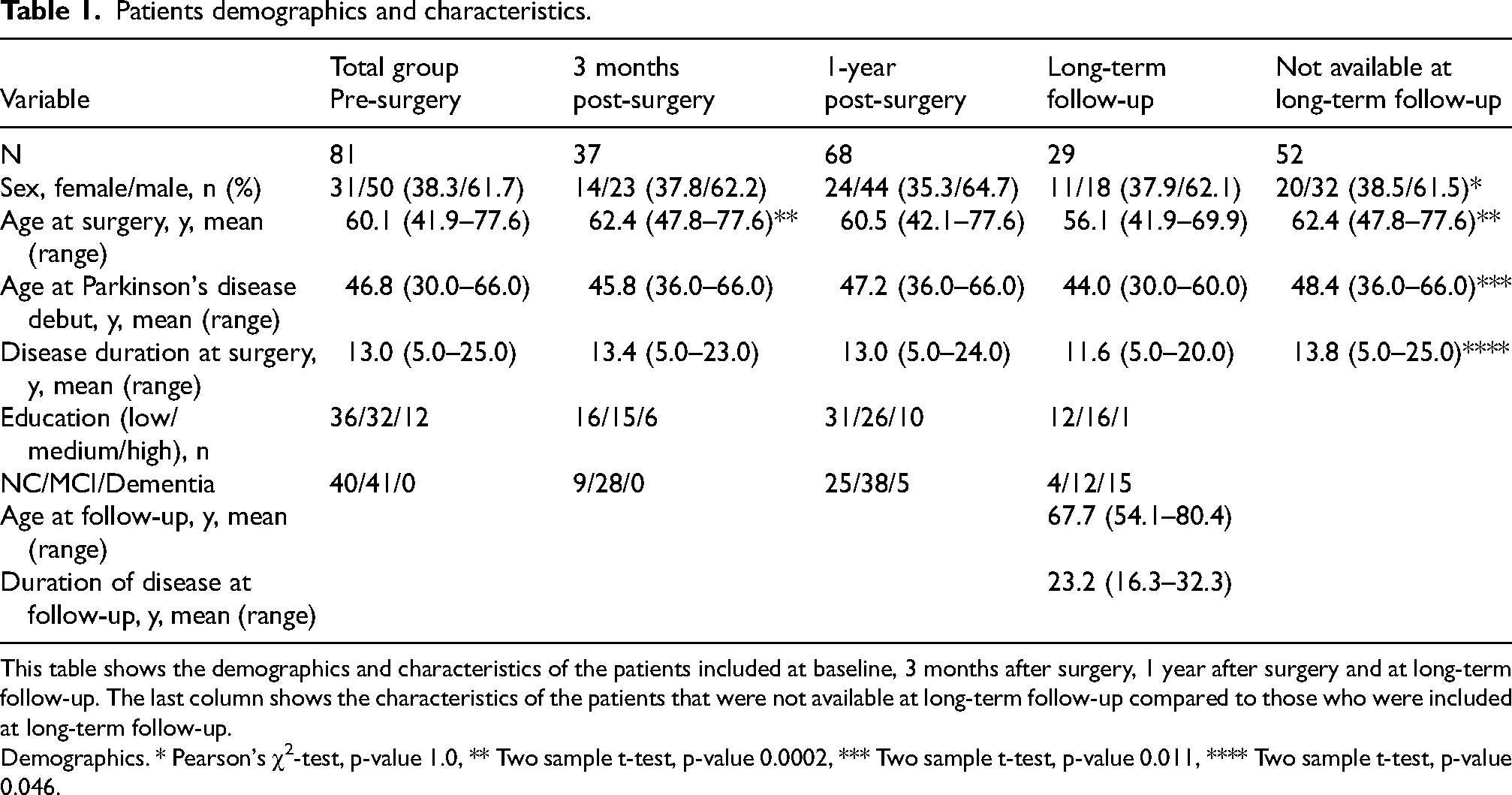
This table shows the demographics and characteristics of the patients included at baseline, 3 months after surgery, 1 year after surgery and at long-term follow-up. The last column shows the characteristics of the patients that were not available at long-term follow-up compared to those who were included at long-term follow-up.
Demographics. * Pearson's χ2-test, p-value 1.0, ** Two sample t-test, p-value 0.0002, *** Two sample t-test, p-value 0.011, **** Two sample t-test, p-value 0.046.
Table 2 shows comparison of pre-surgical tests to post-surgical tests. At 3-months evaluation verbal fluency for category, literal and alternating word significantly declined after STN-DBS surgery compared to the pre-surgical state. Other domains tested at 3 months showed some impairment, but not statistically significant (Table 2).
Neuropsychological test results pre- and post-surgery (3 months, 1-year and long-term follow-up).
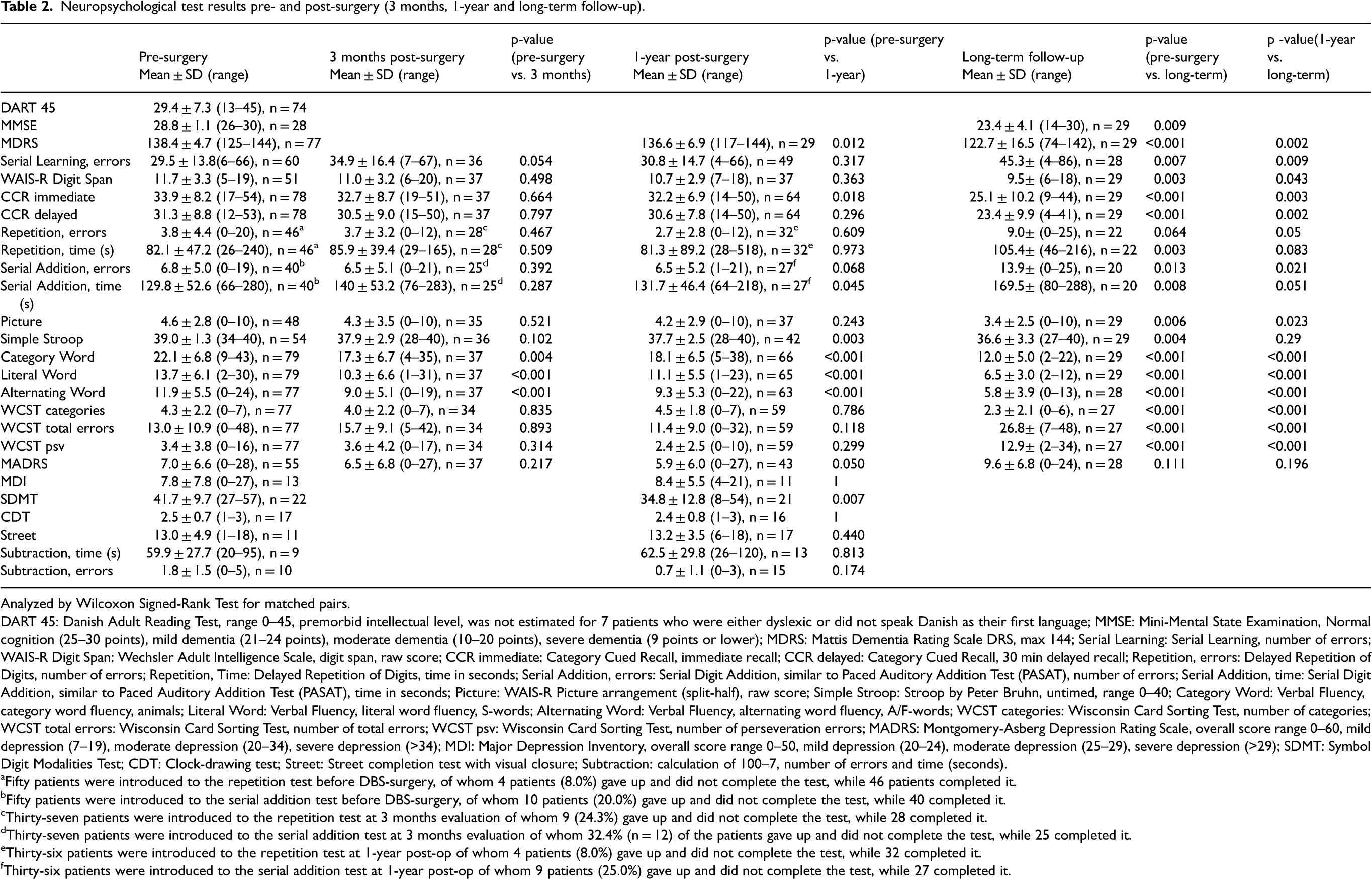
Analyzed by Wilcoxon Signed-Rank Test for matched pairs.
DART 45: Danish Adult Reading Test, range 0–45, premorbid intellectual level, was not estimated for 7 patients who were either dyslexic or did not speak Danish as their first language; MMSE: Mini-Mental State Examination, Normal cognition (25–30 points), mild dementia (21–24 points), moderate dementia (10–20 points), severe dementia (9 points or lower); MDRS: Mattis Dementia Rating Scale DRS, max 144; Serial Learning: Serial Learning, number of errors; WAIS-R Digit Span: Wechsler Adult Intelligence Scale, digit span, raw score; CCR immediate: Category Cued Recall, immediate recall; CCR delayed: Category Cued Recall, 30 min delayed recall; Repetition, errors: Delayed Repetition of Digits, number of errors; Repetition, Time: Delayed Repetition of Digits, time in seconds; Serial Addition, errors: Serial Digit Addition, similar to Paced Auditory Addition Test (PASAT), number of errors; Serial Addition, time: Serial Digit Addition, similar to Paced Auditory Addition Test (PASAT), time in seconds; Picture: WAIS-R Picture arrangement (split-half), raw score; Simple Stroop: Stroop by Peter Bruhn, untimed, range 0–40; Category Word: Verbal Fluency, category word fluency, animals; Literal Word: Verbal Fluency, literal word fluency, S-words; Alternating Word: Verbal Fluency, alternating word fluency, A/F-words; WCST categories: Wisconsin Card Sorting Test, number of categories; WCST total errors: Wisconsin Card Sorting Test, number of total errors; WCST psv: Wisconsin Card Sorting Test, number of perseveration errors; MADRS: Montgomery-Asberg Depression Rating Scale, overall score range 0–60, mild depression (7–19), moderate depression (20–34), severe depression (>34); MDI: Major Depression Inventory, overall score range 0–50, mild depression (20–24), moderate depression (25–29), severe depression (>29); SDMT: Symbol Digit Modalities Test; CDT: Clock-drawing test; Street: Street completion test with visual closure; Subtraction: calculation of 100–7, number of errors and time (seconds).
Fifty patients were introduced to the repetition test before DBS-surgery, of whom 4 patients (8.0%) gave up and did not complete the test, while 46 patients completed it.
Fifty patients were introduced to the serial addition test before DBS-surgery, of whom 10 patients (20.0%) gave up and did not complete the test, while 40 completed it.
Thirty-seven patients were introduced to the repetition test at 3 months evaluation of whom 9 (24.3%) gave up and did not complete the test, while 28 completed it.
Thirty-seven patients were introduced to the serial addition test at 3 months evaluation of whom 32.4% (n = 12) of the patients gave up and did not complete the test, while 25 completed it.
Thirty-six patients were introduced to the repetition test at 1-year post-op of whom 4 patients (8.0%) gave up and did not complete the test, while 32 completed it.
Thirty-six patients were introduced to the serial addition test at 1-year post-op of whom 9 patients (25.0%) gave up and did not complete the test, while 27 completed it.
In the 1-year evaluation the mean of MDRS was 136.6, and significantly lower (p = 0.01) than the mean of MDRS before surgery (Table 2). The immediate recall of CCR (p = 0.018), Stroop (p = 0.003) and SDMT (p = 0.007) and Verbal fluency (p < 0.001) were also significantly lower 1 year after surgery. Other tests remained statistically unaffected 1 year after surgery (Table 2). A general decline in performance was observed 3 months after STN-DBS surgery including verbal fluency (category, literal and alternating word), serial learning, CCR, repetition, serial addition and WCST (see Supplemental Figures). However, the patients performed better overall in the above-mentioned tests 1 year compared to 3 months after surgery, although not statistically significant.
Cognitive tests were significantly impaired at the long-term follow-up assessment compared to both baseline and 1 year after surgery (Table 2). Patients tended to perform significantly worse in all tests at long-term follow-up compared to 1-year assessment, except when looking at number of errors in Delayed Repetition of Digits and Stroop test. The decline in performance was most pronounced for MDRS, CCR, verbal fluency and WCST, p < 0.001 (see Table 2 and Supplemental Figures).
The data from the patients, divided in two groups based on whether they had PD-NC or PD-MCI at baseline in neuropsychological testing, is shown in Table 3 and Figure 2. At baseline, 49.4% of the patients (n = 40) showed PD-NC, while 50.6% (n = 41) showed PD-MCI (Table 3). PD-MCI multiple-domains was seen in 31 patients, while PD-MCI single-domain was seen in 10 patients. Sex and age of disease onset did not differ between PD-MCI patients and PD-NC patients at baseline. However, PD-MCI patients had a longer duration of disease compared to PD-NC patients.
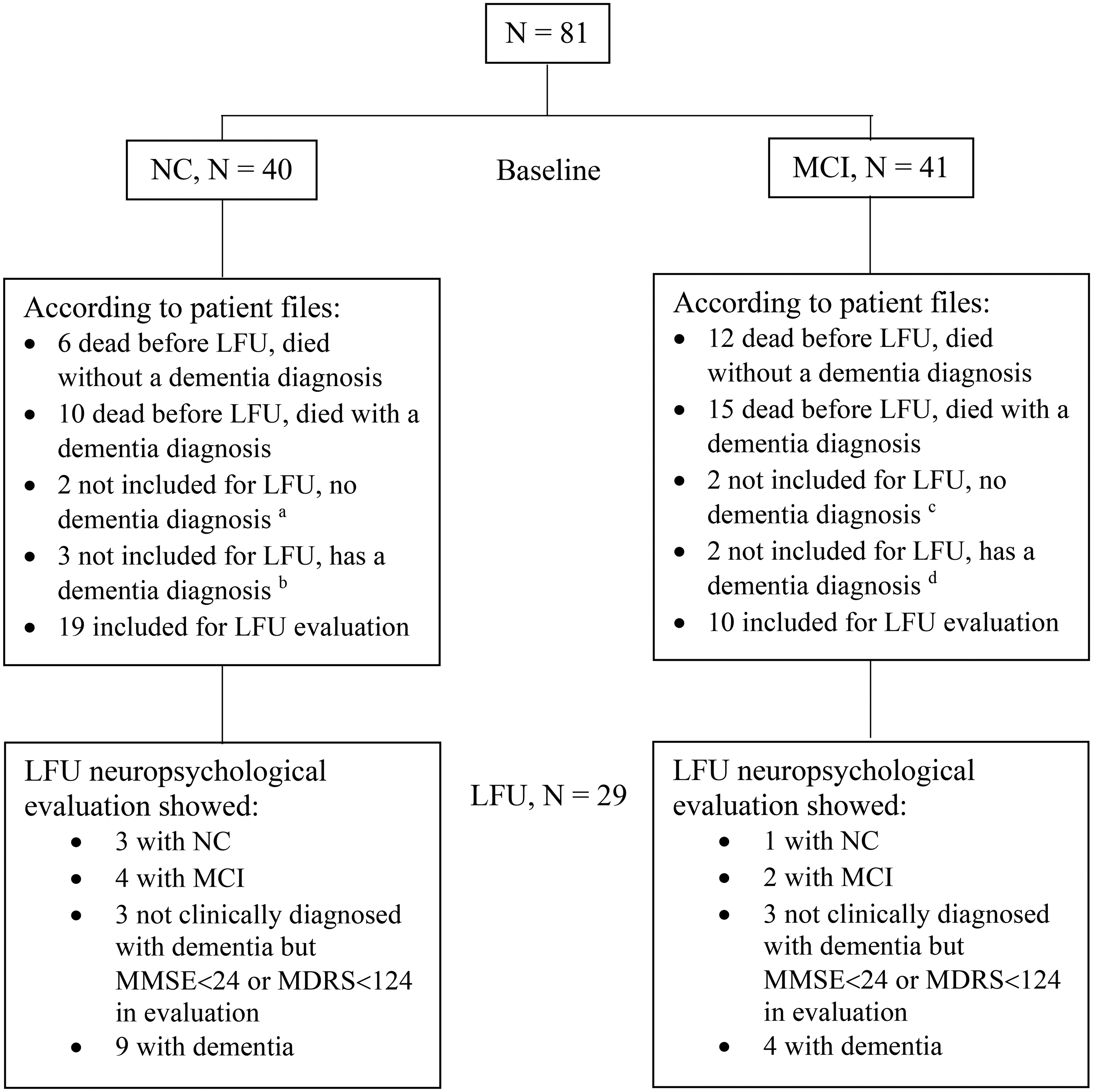
Normal cognition vs. mild cognitive impairment. Figure 2 shows long-term results of patients with either normal cognition (NC) or mild cognitive impairment (MCI) at baseline. a1 did not want to participate, reason is not stated and 1 could not participate because of severe ill spouse. bdementia too severe for participation. c1 with relapsing infections, 1 traveling abroad. ddementia too severe for participation. N: number; LFU: long-term follow-up; MMSE: Mini-Mental State Examination, Normal cognition (25–30 points), mild dementia (21–24 points), moderate dementia (10–20 points), severe dementia (9 points or lower); MDRS: Mattis Dementia Rating Scale, max score 144.
Patient demography before surgery.
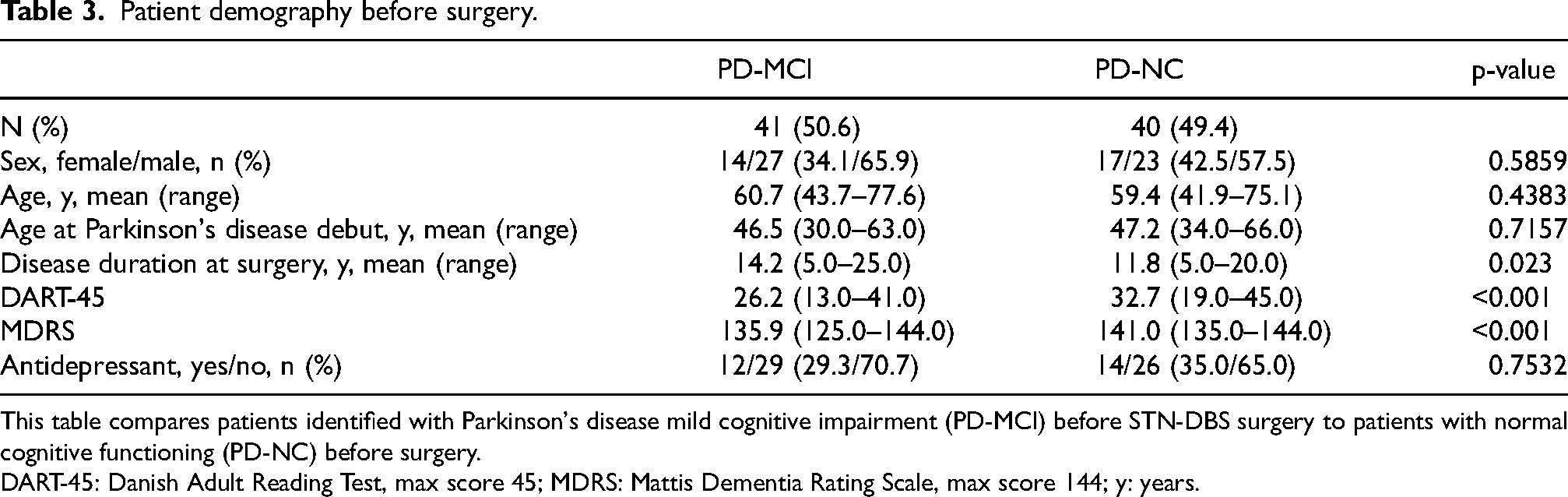
This table compares patients identified with Parkinson's disease mild cognitive impairment (PD-MCI) before STN-DBS surgery to patients with normal cognitive functioning (PD-NC) before surgery.
DART-45: Danish Adult Reading Test, max score 45; MDRS: Mattis Dementia Rating Scale, max score 144; y: years.
The premorbid intellectual level measured by DART-45 and the MDRS score were significantly lower in patients with PD-MCI compared to patients with PD-NC (Table 3).
At the 3-month post-surgery evaluation, 75.7% (n = 28) showed signs of PD-MCI, of whom 26 were characterized as PD-MCI multiple domain and 2 were characterized as PD-MCI single domain, while 24.3% (n = 9) of the patients were characterized as PD-NC. Five patients with PD-NC pre-surgically had developed PD-MCI. Three patients who did not participate in a 3-month post-surgery evaluation had developed PD-D about 3 months after STN-DBS, according to patient files, all of whom were characterized as having PD-MCI before surgery.
In the 1-year post-surgery evaluation PD-MCI accounted for 55.9% (n = 38) of the patients, clinically diagnosed PD-D 7.3% (n = 5), and PD-NC 36.8% (n = 25). One patient, not participating in the 1-year post-surgery evaluation, had developed PD-D according to patient files. Patients with PD-MCI multiple domains accounted for 89.5% of the patients with MCI, while single domain PD-MCI accounted for 10.5%.
Figure 2 shows long-term results of patients with either PD-MCI or PD-NC at baseline. At the long-term follow-up study 44.8% (n = 13/29) were clinically diagnosed with PD-D according to patient files, 9 of those had PD-NC before surgery and 4 had PD-MCI before surgery. Five patients were not eligible for clinical long-term follow-up assessments because of severe dementia but were still alive when the long-term follow-up study was initiated. See also Figures 1 and 2.
The four patients with PD-NC at long-term follow-up were all women. One patient performed well in all tests, two of the patients performed worse in verbal fluency and one patient performed worse in Serial Learning compared to age, gender and culturally appropriate norms. The four patients had a mean age of 60.4 years at long-term follow-up and a mean age at surgery of 47.4 years, while the other patients available for long-term follow-up study had a mean age of 68.8 years and a mean age at surgery of 57.5 years. By the time of STN-DBS surgery the four patients had a mean disease duration of 8.5 years, while the rest participating in long-term follow-up had a mean disease duration of 12.0 years. The mean score of DART-45 was 29.3 and the mean score of MDRS before STN-DBS surgery was 141.3. Three of the four patients had PD-NC before STN-DBS. The patient with impaired Serial Learning at long-term follow-up had PD-MCI prior to STN-DBS as the test scores of Serial Learning and CCR were impaired. This patient performed well in CCR at long-term follow-up.
Table 4 shows data for logistic regression analysis. No correlation was found between PD-MCI before surgery and the development of PD-D before death or before long-term follow-up (OR 0.8, 95% CI 0.3–2.2, p = 0.714). 51.2% of the patients identified with PD-MCI before STN-DBS developed PD-D before death or before long-term follow-up (n = 21/41), while 55% of pre-surgical PD-NC patients developed PD-D (n = 22/40) (Figure 2).
Logistic regression analysis.
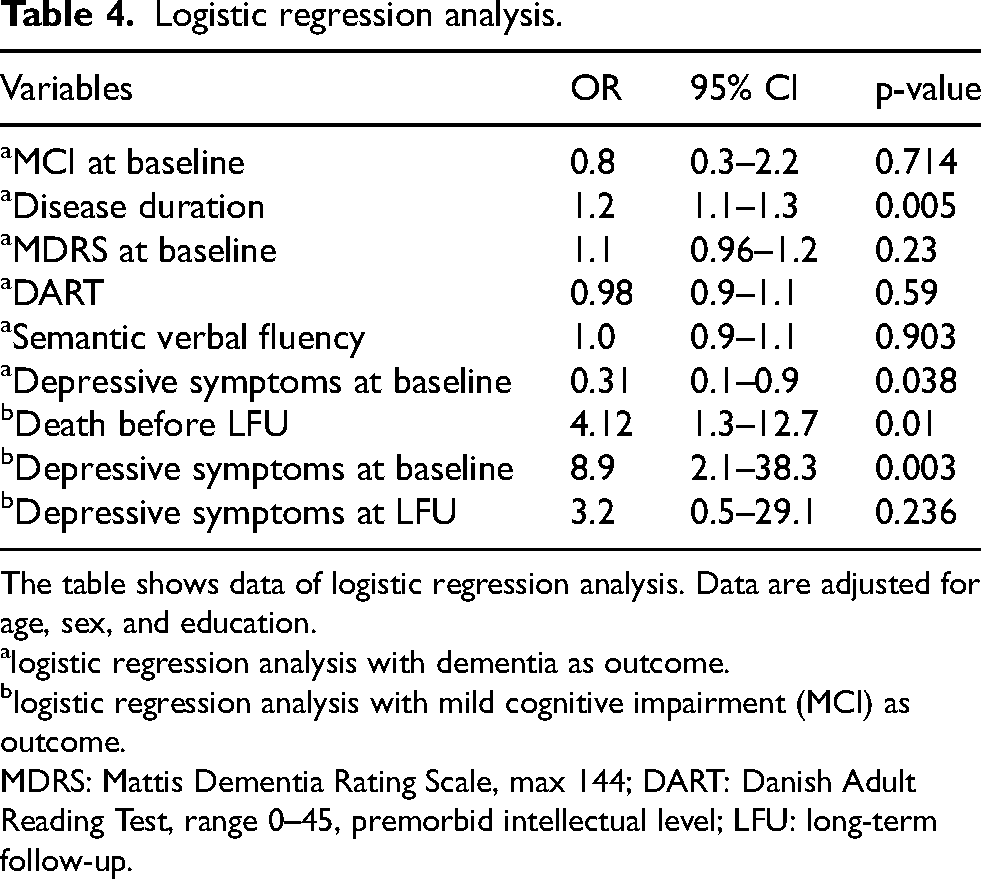
The table shows data of logistic regression analysis. Data are adjusted for age, sex, and education.
logistic regression analysis with dementia as outcome.
logistic regression analysis with mild cognitive impairment (MCI) as outcome.
MDRS: Mattis Dementia Rating Scale, max 144; DART: Danish Adult Reading Test, range 0–45, premorbid intellectual level; LFU: long-term follow-up.
Logistic regression showed an association between PD-D and years of disease duration with adjustment for sex, age at surgery and education (OR 1.2, 95% CI 1.1–1.3, p = 0.005) (Table 4). Neither the pre-surgical MDRS score (OR 1.1, 95% CI 0.96–1.2, p = 0.23) nor the premorbid intellectual level measured by DART were correlated with the development of PD-D (OR 0.98, 95% CI 0.9–1.1, p = 0.59), both analyses adjusted for sex, age at surgery and disease duration (Table 4).
No correlation was found between development of PD-D and impairment in specific cognitive tests before surgery. Posterior-cortical cognitive deficits at baseline were not predictive of PD-D long-term (p = 0.903 for semantic verbal fluency, Table 4). Age at surgery was not predictive of the development of PD-D at long-term follow-up (p = 0.162).
Death before long-term follow-up was more common in MCI compared to the pre-surgical PD-NC group (p = 0.01, OR = 4.12, 95% CI 1.3–12.7) adjusted for sex, age at surgery, disease duration at surgery and level of education, see also Figure 2 where it states that 27 patients with MCI at baseline (27/41) are deceased before long-term follow-up (12 died without a dementia diagnosis, 15 died with a dementia diagnosis), while 16 patients with NC at baseline (16/40) are deceased before long-term follow-up (6 died without a dementia diagnosis, 10 died with a dementia diagnosis).
When adjusting for sex and age with linear regression, patients with a higher education performed significantly better in DART score (p < 0.001), MDRS score (p < 0.001), number of errors in serial learning (p = 0.027), CCR immediate recall (p = 0.029), serial repetition time (p = 0.044), serial addition time (p = 0.043), verbal fluency for category word (p < 0.001), literal word (p = 0.002), and alternating word (p = 0.005), number of categories in WCST (p = 0.017) and MMSE score (p = 0.028) at baseline. However, the MDRS score was significantly influenced by the DART score and not the level of education when the correlation between MDRS score and DART score was analyzed (p < 0.001, 95% CI 0.13–0.44). Women had a higher MMSE score than men pre-surgically (p = 0.015, 95% CI 0.23–1.92 adjusted for age and education).
Women performed better than men at long-term follow up in Serial Learning (p = 0.037), WAIS-R Digit Span (0.015), and CCR (p = 0.038 for immediate recall and p = 0.042 for delayed recall). Performance did not differ among sex in other tests. Patients of higher education were faster than patients of medium and lower education in Serial Addition (p = 0.015). Age, sex, and education did not seem to influence other test scores at long-term follow-up (data not shown), but all three factors were considered as potential confounders and henceforth included for logistic regression.
The mean intake of antiparkinsonian dopaminergic medication (converted to levodopa equivalent daily dose, LEDD) was 1014 (range 190–2404) mg before surgery, 446 (range 0–1160) mg in the group evaluated after 3 months, 430 mg (range 0–1090) after 1 year and 616 (range 0–2367) mg after 8–15 years. These data were previously reported by Thomsen et al. (2020). 13
The questionnaires MADRS or MDI were used to rate depressive symptoms. Before surgery 33.8% (n = 23) showed depressive symptoms rated with MADRS or MDI. At 3 months follow-up 37.8% (n = 14), at 1-year follow-up 25.9% (n = 14) and at long-term follow-up 64.3% of the patients (n = 18) had ratings indicative of depressive symptoms (Table 2). The scores were significantly higher at long-term follow-up compared to pre-surgical state (p = 0.012) and 1 year after surgery (p = 0.002) (Table 5).
Depressive symptom rate.

The table shows the depressive symptom rates resulted from the questionnaires MADRS or MDI at baseline/pre-surgery, 3 months after surgery, 1 year after surgery and at long-term follow-up.
Compared to pre-surgery by Pearson's Chi-squared test.
Compared to 1-year post-surgery by Pearson's Chi-squared test.
The use of antidepressants was not correlated to MCI before surgery or by follow-up assessments (Table 3).
Before surgery, patients with PD-MCI were more likely to meet the cut-off for possible depressive symptoms by MADRS or MDI (OR 8.9, 95% CI 2.1–38.3, p = 0.003). PD-MCI before surgery was not associated with rating of depressive symptoms at long-term follow-up (OR 3.2, 95% CI 0.5–29.1, p = 0.236). Patients with depressive symptoms, according to MADRS or MDI, before surgery were more likely to have PD-D long-term compared to patients without depressive symptoms according to MADRS or MDI scores (OR 0.31, 95% CI 0.1–0.9, p = 0.038).
At long-term follow-up, almost all patients with PD-D had depressive symptoms according to MADRS or MDI. Only 2 patients with PD-D did not show signs of depressive symptoms, hence it was not possible to analyze these data. We did not find higher MADRS or MDI scores amongst patients with MCI compared to the four patients with NC at long-term follow-up.
Discussion
We have presented a long-term follow-up study of a group of patients with PD and bilateral implantation of electrodes in STN (STN-DBS). The patients were thoroughly examined with neuropsychological examinations before surgery and followed for up to 15 years. We have shown that STN-DBS was associated with isolated changes in performance on select cognitive tests, but we failed to show that presence of MCI at the time of STN-DBS was significantly associated with development of dementia at long-term follow-up. The loss of subjects before long-term follow-up, due to death, might have affected this result. It was previously reported from this cohort, using a survival analysis, that 43 patients out of 81 were clinically diagnosed with dementia before death or before the long-term follow-up assessment with a mean disease duration of 20.5 years, 13 and we have reported that 25 patients among the 43 patients who died before the time of long-term follow-up had developed PD-D. The prevalence of PD-D in our follow-up was thus consistent with previously published data on natural PD progression28,29 which may suggest that development of PD-D in our cohort is correlated to the expected progression of the disease.
Previous studies of cohorts treated with STN-DBS have shown similar results. Bove et al. (2020) reported that the prevalence and incidence of dementia was not higher in 175 PD patients 1 year, 5 years and 10 years after STN-DBS surgery than those reported in the general PD population, 3 and Scelzo et al. (2019) found lower rates of PD-MCI at 1-year follow-up in 91 patients treated with DBS compared to 91 medically-treated patients, and found the risk of dementia to be similar in the two groups. 30 A review in 2019 concluded that the incidence and severity of dementia among PD patients receiving DBS were comparable to medically treated PD patients. 31
On the other hand, in this small sample, disease duration was associated with development of dementia at long term follow-up. Our study showed, that 50.6% of the patients had PD-MCI before STN-DBS surgery with a mean disease duration of 14.1 years. This is in line with other studies, although the frequency varies somewhat from 75.7% after 11.3 years of disease duration, 4 63.1% with 10.6 years of disease duration 6 to 23% with 14.2 years of disease duration. 7 While many cohort studies have found MCI to be a risk factor for PD-D,32,33 in our cohort the development of PD-D was only correlated to disease duration and signs of depressive symptoms according to ratings using MADRS or MDI before surgery. In a cohort study (the CamPaign study) where 142 patients with PD were followed longitudinally for the first 10 years after diagnosis, it was found that age, the degree of motor impairment and ‘posterior-cortical’ cognitive deficits as opposed to executive problems at baseline were predictive of a higher risk of development of dementia. 34 We did not see a correlation between development of clinical diagnosed dementia and age or specific cognitive tests before surgery. Unlike the CamPaign cohort, the group of patients included for DBS already had a disease duration of a mean of more than 10 years. Therefore, the group can be considered to already having shown to be prone to a more favorable mean prognosis than the cohort that was followed from debut. This is corroborated by a study by Kim et al. (2014), where 103 patients were followed for 7 years after DBS, and the development of dementia was predicted by executive function before surgery. 6
Although we did not find the prevalence of clinically diagnosed dementia to be higher than expected, we found significant changes in select cognitive tests after surgery. A tendency for larger cognitive changes 3 months after surgery, compared with 1 year follow-up was observed. While this is an exploratory result in a small sample, it is worth mentioning, as it illustrates that short-term results obtained within months after surgery may be influenced by the procedure, and it is important to also look at more long-term data. A risk of cognitive changes following STN-DBS has been described in several studies with shorter follow-up.4,6,7,35–37 Both an effect of surgery itself and the localization of lead implantation through the caudate or the ventricular wall38,39 and an inherent effect of high-frequency stimulation depending on the localization in STN have been shown.40,41 In addition, pre-operative patient-related factors, such as age, LEDD and axial symptoms were shown to explain 23% of the variance of post-operative decline in a comparison with a similar group with best medical treatment by Daniels et al. (2010). 42 As in these earlier studies, we found a cognitive decline after surgery, especially in the executive domain, that did not, however, predict later dementia.
The most widely described cognitive change after STN-DBS is verbal fluency. We found a significant decline in verbal fluency just 3 months post-surgery, while other domains remained relatively unaffected. Interestingly, verbal fluency improved at 1-year follow-up, although it was still significantly below pre-operative level. The decline in verbal fluency 3 months after surgery might suggest a relation to the surgical procedure and the positioning of the electrode, or it could be related to reduced dopaminergic medication levels following STN-DBS, which in our cohort was reduced 56% already 3 months after surgery. Tiedt et al. found that dopaminergic medication affected phonemic verbal fluency with improved word production and increased error-rates, while no significant difference was observed between the control group and the PD group in the OFF condition. 43 A study by Witt et al. suggested a relation to localization of the electrode in surgery with a significant decline in semantic verbal fluency in a group of PD patients treated with STN-DBS compared to a PD-control group on best medical treatment. 38 Mikos et al. reported a correlation between volume of tissue activated and phonemic verbal fluency decline and concluded that the worsening of verbal fluency performance was correlated to stimulation of larger ventral parts of STN. 44 Importantly, it was shown in an 8-year follow-up in a group of patients eligible for surgery, where both patients who accepted and received STN-DBS and the group who declined surgery were examined, that verbal fluency among patients who received STN-DBS was significantly worse at 8 years after initiation of the treatment than the group of patients who declined surgery. 45
Depression or significant depressive symptom are associated with worse performance on cognitive tests,11,46,47 and therefore MADRS or MDI were included in the neuropsychological test battery. We report, that MADRS or MDI ratings indicative of depressive symptoms before DBS-surgery were more likely to be associated with a development of dementia long-term compared to low MADRS or MDI rating scores. Depressive symptoms pre-surgically were associated to PD-MCI both pre-surgically and long-term. The rates of depressive symptoms were significantly higher at long-term follow-up than before surgery and patients with clinically diagnosed dementia were more likely to also have depressive symptoms. Okun et al. (2009) found that DBS regardless of target resulted in a mild improvement of mood 7 months post-surgery, 48 and Ardouin et al. (1999) saw an improvement of mood 6 months after STN-DBS. 49 Reduced depressive symptoms up to 2 years after STN-DBS was confirmed by a review 50 and a meta-analysis. 36 However, long-term effects of DBS on mood is less clear. We saw a slight improvement in ratings of depressive symptoms 1-year post-surgery although not statistically significant. The higher prevalence of depressive symptoms 8–15 years after STN-DBS surgery might be associated with the higher prevalence of dementia at long-term follow-up.
Limitations
Limitations of this study include a missing control group, a relatively small sample size and that some of the tests used were time based, e.g., the PASAT test. Due to the motor dysfunction of PD patients, it should be considered whether time-based tests should be included for neuropsychological assessment of PD patients. Parts of the dataset are curated from clinical files, causing some missing data. This study investigates associations of MCI to development of dementia in patients who were alive at long-term follow-up. This causes a high risk of selection bias, as the loss of subjects due to death might affect the results. The patients who died before long-term follow-up could have been more likely to have developed dementia. However, we believe that the patients at long-term follow-up are representative for those who survive and therefore clinically interesting; important to report descriptively and when possible, also statistically. As in other explorative studies, there is a risk of type 1 as well as type 2 errors.
Conclusion
Overall, in this small sample of patients treated with STN-DBS, disease duration and the presence of ratings of depressive symptoms before surgery were associated with the later development of clinically diagnosed dementia, while importantly, the presence of MCI before surgery was not associated with a clinically diagnosed dementia more than eight years after surgery. Several limitations are present in this small clinical study making it difficult to draw a conclusion, but the study is meant to be hypothesis-generating, and results should be further tested in other studies.
Supplemental Material
sj-docx-1-pkn-10.1177_1877718X251334049 - Supplemental material for Mild cognitive impairment is not predictive of dementia up to 15 years after subthalamic deep brain stimulation in Parkinson's disease
Supplemental material, sj-docx-1-pkn-10.1177_1877718X251334049 for Mild cognitive impairment is not predictive of dementia up to 15 years after subthalamic deep brain stimulation in Parkinson's disease by Sine Fjeldhøj, Birgitte Liang Chen Thomsen, Palle Møller Pedersen, Steen Rusborg Jensen, Anders Clausen, Merete Karlsborg, Bo Jespersen, Ove Ketil Bergdal and Annemette Løkkegaard in Journal of Parkinson's Disease
Footnotes
Acknowledgments
We are sincerely grateful to the patients participating in this study and for making it possible to examine the effects of STN-DBS on cognition and mood both short-term and long-term. We are also sincerely grateful to the staff at Copenhagen University Hospital, Bispebjerg, and Copenhagen University Hospital, Rigshospitalet, for their work during the STN-DBS surgery and the neuropsychological testing, and for following the patients through the years. Statistical consultation was provided by Clinical Epidemiologist, MSc, Ph.d Matilde Winther-Jensen (Center for Clinical Research and Prevention, Bispebjerg and Frederiksberg Hospital).
Ethical considerations
The study was approved by the regional ethics committee, H-15007736, on July-01-2015. Informed consent for participation and anonymized publication of data was obtained for all participants who were alive at the time of the study. It was approved by the committee to use data from the files from deceased patients, with the need for written informed consent waived.
Funding
This study was funded by the Danish Parkinson Association and the Copenhagen University Hospital, Bispebjerg.
Conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article. Disclosure for AL: Boston Scientific, Course attendance
Data availability
The data that support the findings of this study are available from the corresponding author, AL, upon reasonable request.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
