Abstract
There is a distinct lack of consensus on the most effective treatments for neurodegeneration with brain iron accumulation. This is due to the rarity of the disease, its phenotypic variability, and the multiplicity of pathophysiological mechanisms. Our team has already proposed the use of conservative iron chelation in cases of neuroferritinopathy, with interesting results. However, no mention has yet been made of the treatment of parkinsonism-dystonia related to VAC14 gene mutations. The case reported here illustrates clinical stability after 2 years of conservative iron chelation, with an improvement in radiological images.
We report the clinico-radiological evolution of a patient with atypical parkinsonism-dystonia associated with brain iron accumulation (neurodegeneration with brain iron accumulation; NBIA) and biallelic mutation of the VAC14 gene involved in childhood-onset striato-nigral degeneration (SNDC) 1 who was treated at Lille University Hospital with iron-chelating treatment (deferiprone) over a period of 2 years.
The VAC14 protein is an assembly protein, capable of dimerization, and the VAC14 homodimer can assemble with FIG4 phosphoinositide 5-phosphatase and FYVE proteins to form a ternary complex. This ternary complex regulates and turns over phosphatidylinositol 3,5-bisphosphate (PI(3,5)P2). 2 A defect in PI(3,5)P2 causes neurodegeneration in humans and mice. 3 PI(3,5)P2 plays a role in inhibiting fusion, and promoting fusion of intrasynaptic vesicles. 4 Postmortem studies show localized neuronal death in the putamen, caudate nucleus, pallidum, and substantia nigra in these patients. 5
Our patient was a 32-year-old male with no relevant medical history or family history. His psychomotor development as a child was normal and he had completed a vocational certificate in cookery. Examination at age 32-years revealed parkinsonism that had started with akinesia and tremor of the right lower limb at rest when he was 21 years old, which had spread progressively to all four limbs, associated with a slight postural component (see Supplemental Video 1). The patient had hypokinetic dysarthria, slight camptocormia, small-step walking, and loss of arm swing, without postural instability. He also suffered from apraxia of eye opening with severe blepharospasm and oral-lingual-facial praxis disorders. No pyramidal syndrome or cerebellar syndrome was identified, but a slight convergence defect and saccadic eye pursuits were noted. The patient did not suffer from depression, any sleep disorder or hallucinations, but did have a slight non-invasive anxiety disorder. He often complained of cramps in his lower limbs related to distal dystonia. A Montreal cognitive assessment (MoCA) gave a score of 25/30 at age 32, with subcortico-frontal cognitive dysfunction and disruption of temporo-limbic networks in the dominant hemisphere.
Brain magnetic resonance imaging (MRI) acquisitions were performed on 3T system (Philips, Achieva dStream, Best, The Netherlands). For a qualitative analysis of NBIA-associated brain changes, the protocol included a 3D T1 TFE (turbo-field echo) sequence (voxel: 1 mm isotropic, TE/TR = 4.6/9.8 ms, flip angle = 8°) to assess brain atrophy, a 3D FLAIR sequence (voxel: 1 mm isotropic, TE/TR/TI = 333/8000/2500 ms, flip angle = 90°) to assess deep gray nuclei and white matter signal changes and SWI (susceptibility weighted imaging) sequence (voxel: 0.6 × 0.6 ×1.2 mm, TEs/TR = 5.6, 11.8, 18.0, 24.2/27.6, flip angle = 17°) to assess iron overload.
For a quantitative analysis of brain iron overload, we computed R2* maps from a 3D multiecho gradient echo sequence (voxel: 1 × 1 × 2 mm, TEs/TR = 4.2, 9.5, 14.7, 20.0, 25.3, 30.5, 35.8, 41.0/54, flip angle = 16 = 5°).
Visual analysis of MR images demonstrated changes generally expected in NBIAs, 6 namely a marked hyposignal on SWI and FLAIR indicative of a regional iron overload in the pallidum and substantia nigra, as well as a pallidal atrophy. Dopamine transporter (DAT) single-photon emission tomography (SPECT) with (123) Ioflupane was normal. Cerebral computed tomography showed no calcification of the basal ganglia. Analyses of the cerebrospinal fluid, fundus, and electroneuromyogram were normal. Biologically, a broad iron, copper, vitamin, and metabolic panel was normal.
Levodopa was ineffective, as were trihexyphenidyl and clonazepam for dystonia. Botulinum toxin injections were administered every 3 months for blepharospasm, with partial efficacy. Two variants of unknown significance were identified at the heterozygous state in exon 11 (c.1283T > A, p.(Leu428His)) and exon 12 (c.1319C > T, p.(Thr440Met)) of the VAC14 gene following exome sequencing. Familial segregation analyses demonstrated the heterozygous status.
In the absence of any recognized effective therapy, 7 and even though no primary abnormality of iron metabolism was detected, compassionate treatment with deferiprone (30 mg/kg/day) was introduced at age 32.
Deferiprone is a small, lipophilic, blood-brain barrier iron chelator. Its affinity for iron is weaker than that of transferrin, thus it will not chelate unless transferrin is already saturated. 8 Its use in neuroferritinopathy and pantothenate kinase-associated neurodegeneration has already been demonstrated.9,10 The idea of using deferiprone in VAC14 stems from the major lipid peroxidation found in all NBIAs, leading to ferroptosis, which is improved by iron chelators, hence the idea of offering this therapy to our patient. 11
Clinical and biological tolerance of treatment was excellent. Between age 30–32, before he was treated, his MDS-UPDRS III score increased from 45/132 to 51/132 in 2 years, whereas after treatment had been started, between age 32–34, there was an improvement from 51/132 to 46/132 (rigidity, akinesia of left lower limb and postural stability). Although the scope of the conclusions drawn from this score is limited, treatment appears to have altered the course of his disease (see Supplemental Table 1). However, there was a slight increase in memory difficulties, executive function disorders, and psychomotor slowing (MoCA 22/30) at age 34. Between age 30–32, quantitative R2* MRI maps demonstrated a 33% and 21% reduction of iron overload in the pallidum and substantia nigra, respectively 12 (Figure 1).
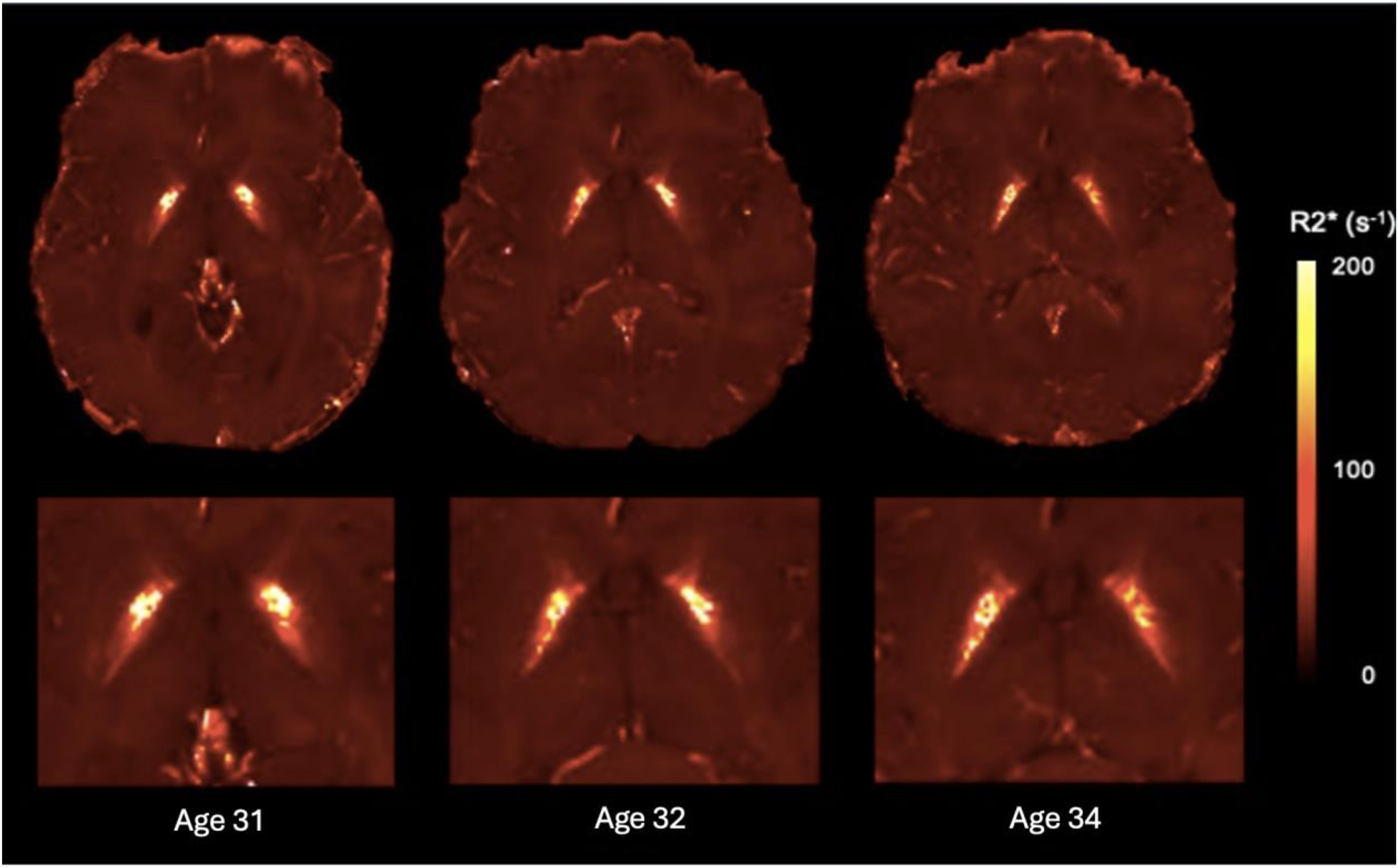
Evolution of iron load in brain structures evaluated from R2* magnetic resonance imaging. CSF: cerebrospinal fluid.
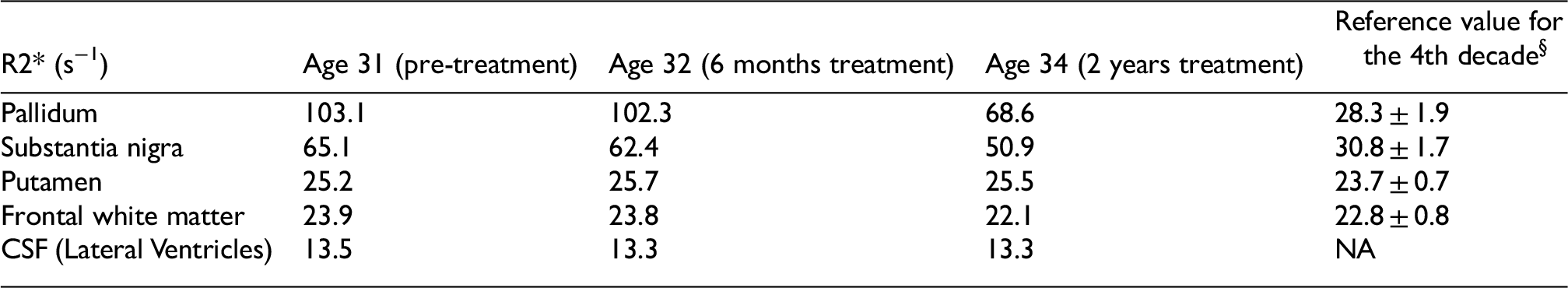
To our knowledge, this is the first case of medical treatment of SNDC to be described.7,13 Despite the late onset and slow progression of the disease, our patient had marked iron load. The stabilization of symptoms and MDS-UPDRS-III in this context is promising but requires further validation in other patients.
Known variants cause parkinsonism-dystonia phenotypes in children, 5 whereas our case concerned a variant of undetermined significance, but whose clinical symptoms were similar to those in children, with a later age of onset (21 years vs. 3 years on average).5,14 In addition, another case of a patient with the same symptoms as our patient has already been published, with another variant of undetermined significance (in different exons from our case) and a later age of onset.13,15
Supplemental Material
Supplemental Material
sj-docx-1-pkn-10.1177_1877718X251331820 - Supplemental material for Conservative iron chelation for VAC14: Two-year clinical-radiological follow-up
Supplemental material, sj-docx-1-pkn-10.1177_1877718X251331820 for Conservative iron chelation for VAC14: Two-year clinical-radiological follow-up by Thomas Ollivier, David Devos, Gregory Kuchcinski, Luc Defebvre, Vincent Huin and Eugenie Mutez in Journal of Parkinson's Disease
Footnotes
Acknowledgments
We would like to particularly thank the patient who agreed to share his personal experience after 2 years of deferiprone treatment. Without his consent, this case report would not have been possible. This contribution is a further step towards improving the therapeutic management of NBIA. Naturally, we thank Val Kerr (Hopwood) for English editing.
Ethical considerations
Ethics committee approval was not required for this case report.
Consent to participate
Our patient has provided written consent for the use of his clinical data and video material.
Consent for publication
Our patient has provided written consent for the use of his clinical data and video material.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the University of Lille and the Lille university Hospital (CHU Lille). This work was also supported by grants from the “France Alzheimer” and “France Parkinson” Charities.
Conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability
Data sharing is not applicable to this article as no datasets were generated or analyzed during the study.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
