Abstract
Background
Experimental and observational studies suggest that β-adrenoreceptor drugs (β2-agonists/β-antagonists) are associated with Parkinson's disease (PD) risk. Previous epidemiological studies may be hampered by reverse causation/confounding.
Objective
We examined the association of β-adrenoreceptor drugs with PD incidence, while addressing reverse causation and confounding in the E3N cohort study (2004–2018) using a new-user design.
Methods
Incident β2-agonists/β-antagonists users were identified through drug claims databases. Incident PD was ascertained using multiple sources and validated by experts. Drugs-PD associations were assessed using time-varying Cox proportional hazards models adjusted for multiple confounders. Main analyses used a 5y-exposure lag to address reverse causation; sensitivity analyses used a 2y-lag or no lag. We set up a nested case-control study to compare trajectories of β2-agonists/β-antagonists prescriptions before diagnosis using logistic mixed models.
Results
Analyses for β2-agonists were based on 81,890 women; 15,169 started using β2-agonists and 579 developed PD. PD incidence was 36% lower (hazard ratio = 0.64, 95% confidence interval = 0.41–0.98; p-trend = 0.04 for the number of claims) in users of long-acting/ultra-long-acting β2-agonists (LABAs/ultra-LABAs) compared to never users. There was no significant association for β2-agonists overall and short-acting β2-agonists. Analyses for β-antagonists were based on 75,896 women; 13,081 started using β-antagonists and 552 developed PD. PD incidence was similar in ever and never users in analyses with a 5y-lag but was higher in ever than never users in analyses with 2y-lag or no lag.
Conclusions
Incident use of LABAs/ultra-LABAs is associated with lower PD incidence in women. Conversely, the association between β-antagonists and PD in women is likely due to reverse causation.
Plain language summary
There is an urgent need to identify neuroprotective drugs that could be used to delay or prevent the onset of Parkinson's disease. Repurposing existing drugs may represent a rapid and accessible solution for identifying new treatment strategies for Parkinson's disease. β-adrenoreceptor drugs include two main classes of drugs. β2-agonists activate β-receptors and are used to treat asthma and chronic obstructive pulmonary disease; tremor is one of their side-effects. β-antagonists block β-receptors and are mainly used for cardiovascular disease; propranolol, which is often used to treat essential tremor, belongs to this group. Previous studies on the association between β-adrenoreceptor drugs and the risk of Parkinson's disease provided inconsistent findings. There are two major difficulties for interpreting them. First, patients with Parkinson's disease have lower rates of smoking while patients with chronic obstructive pulmonary disease have higher rates of smoking; this difference could explain that some studies showed a lower risk of Parkinson's disease in persons treated by β2-agonists. Hence, smoking must be carefully considered in analyses of the relation between β2-agonists and Parkinson's disease. Second, Parkinson's disease develops over many years and symptoms that precede the diagnosis of Parkinson's disease may lead to more frequent medical contacts and/or changes in prescriptions of drugs before the diagnosis. Hence, studies with a long follow-up allowing to examine the role of drugs prescribed long before the Parkinson's diagnosis are necessary. In this new research, we examined the association of β-adrenoreceptor drugs with Parkinson's disease risk in a large cohort of French women followed for 15 years for whom we had detailed information on numerous characteristics, including smoking. This study showed that users of long-acting/ultra-long-acting β2-agonists had 36% lower Parkinson's risk compared to never users, after accounting for smoking and other characteristics, while there was no difference for β2-agonists overall or for short-acting β2-agonists. Alternatively, there was an increased risk of Parkinson's disease in users of β-antagonists, propranolol in particular, compared to never users, that is likely explained by β-antagonists prescribed shortly before the diagnosis due to tremor preceding diagnosis. Additional studies are needed to understand the mechanisms underlying the association for long-acting/ultra-long-acting β2-agonists and to examine whether they could slow disease progression in patients with Parkinson's disease.
Introduction
Experimental models suggest that β2-adrenoreceptor stimulation may play a beneficial role in Parkinson's disease (PD). 1 Pooled results from eight observational studies on the association of β2-agonists (whose main indication is asthma) with PD showed lower risk in ever vs. never users (risk ratio [RR] = 0.87, 95% confidence interval [CI] = 0.78–0.97) with significant heterogeneity (I²=87%, p-heterogeneity < .001). 2 The association was stronger in 2 cohort studies (RR = 0.78, 95% CI = 0.54–1.13) than in 6 case-control studies (RR = 0.91, 95% CI = 0.82–1.01) but the difference was not statistically significant (P = 0.42). One case-control study showed an inverse association between β2-agonists and PD only in persons without type 2 diabetes (T2D). 3
Conversely, a pooled analysis (10 studies) showed that ever use of β-antagonists (main indications: hypertension, tachycardia, myocardial infarction) was associated with increased PD risk (OR = 1.19, 95% CI = 1.05–1.35) with significant heterogeneity (I²=92%, p < 0.001). 2 This association was driven by propranolol (non-selective β-antagonist). 2 The association was observed in cohort studies (n = 2; RR = 1.78, 95% CI = 1.24–2.57) but not in case-control studies (n = 8; RR = 1.09, 95% CI = 0.97–1.22) with a statistically significant difference (p = 0.01).
Reverse causation due to prodromal symptoms 4 and confounding represent important issues for the interpretation of previous studies. Studies with a long follow-up are needed to address reverse causation, in order to discard exposures during the prodromal phase. Several previous studies on the association of β2-adrenoreceptor drugs with PD had an incomplete consideration of these issues.
We examined the association of β-adrenoreceptor drugs with PD in a large cohort of French women followed for 15y, while taking into account the potential for reverse causation and confounding.
Methods
Participants
E3N is a French cohort study of 98,995 women, born between 1925–1950, recruited in 1990, and affiliated with a French national health insurance plan covering mostly teachers (Mutuelle Générale de l’Education Nationale). 5
Participants (∼20% of invited women) completed a self-administered questionnaire (lifestyle, medical history) at baseline (1990) and every 24–36 months thereafter. Eleven waves of data are available (last, Q11–2014; average response rate at each questionnaire = 80%). Only 3% of E3N participants are lost to follow-up. 5 Vital status is available until the end of follow-up and causes of death until 31/12/2014.
Since 01/01/2004, drug and medical consultation claims databases are available for 95% of participants and include all outpatient reimbursements. For each drug delivery, databases include the Anatomical Therapeutic Chemical (ATC) code, purchase date, number of tablets/package, and dose/tablet.
All participants signed informed consent, in compliance with rules of The French National Commission for Data Protection and Privacy which approved the study. The study protocol is registered at clinicaltrials.gov (NCT03285230).
Exposure
We used drug claim databases to assess use of β2-agonists/β-antagonists identified by Anatomical Therapeutic Chemical codes (Supplemental Table 1) between 01/01/2004–31/12/2018.
We distinguished β2-agonists depending on their duration of action: short-acting SABAs have the shortest half-life and are for immediate symptomatic relief, while long-acting LABAs/ultra-LABAs provide prolonged treatment due to longer half-life. Due to a small number of participants exposed to ultra-LABAs, we combined LABAs + ultra-LABAs.
For β-antagonists, we distinguished propranolol from other non-selective β-antagonists and selective β1-antagonists (Supplemental Table 1).
Outcome
Our approach to ascertain PD is described in detail elsewhere. 6 Potential PD patients were identified through self-reported doctor-diagnoses of PD, antiparkinsonian drug claims (ATC-N04), and death certificates. Potential PD patients were contacted to confirm the diagnosis. For women who confirmed a PD/parkinsonism diagnosis and those who could not be contacted, we obtained medical records to adjudicate PD status (definite, probable, possible, no PD). 7 We retained as PD cases for the analyses those with definite/probable PD and excluded those with possible PD. Participants with essential tremor, restless leg syndrome, or other causes of parkinsonism were considered not to have PD and censored at the end of their follow-up.
When no medical records were available (38%), we predicted PD status using an algorithm based on drug claims and medical visits and validated against a clinical diagnosis (94% sensitivity, 88% specificity). 6
Year of PD diagnosis was set as the year of diagnosis (from medical records) or, in decreasing order of priority, self-reported year of diagnosis, year of first use of antiparkinsonian drugs, and year of the first questionnaire where PD was self-reported.
PD incidence rates in E3N are in agreement with those observed in women from Western Europe (Global Burden of Disease, 1992–2018) in favor of the validity of our approach. 6
Covariates
A detailed description of covariates is available as Supplemental Methods. Analyses were adjusted for PD risk factors (rural residence, education, age at menarche, parity, artificial menopause, physical activity, caffeine intake, smoking, body mass index [BMI], lipophilic statin use, type 2 diabetes, hyperuricemia), main indications of β-adrenoreceptor drugs (β2-agonists: asthma, chronic obstructive pulmonary disease [COPD]; β-antagonists: hypertension, cardiovascular disease [CVD]), and inhaled corticosteroids (ICSs; frequently co-prescribed with β2-agonists).
Use of medical services should be considered when evaluating associations of drugs/comorbidities with PD 8 ; analyses were adjusted for the time-varying number of medical consultations during the preceding six months.
Statistical analysis
Analyses were performed using SAS9.4 (SAS Institute Inc., Cary-NC) and Stata17 (StataCorp, College Station-TX). Two-tailed p-values ≤0.05 were considered statistically significant.
Analyses were based on a new user design; prevalent users of drugs were excluded from the analyses (Supplemental Methods). Flowcharts are available in Figure 1 and Supplemental Figures 1 and 2.
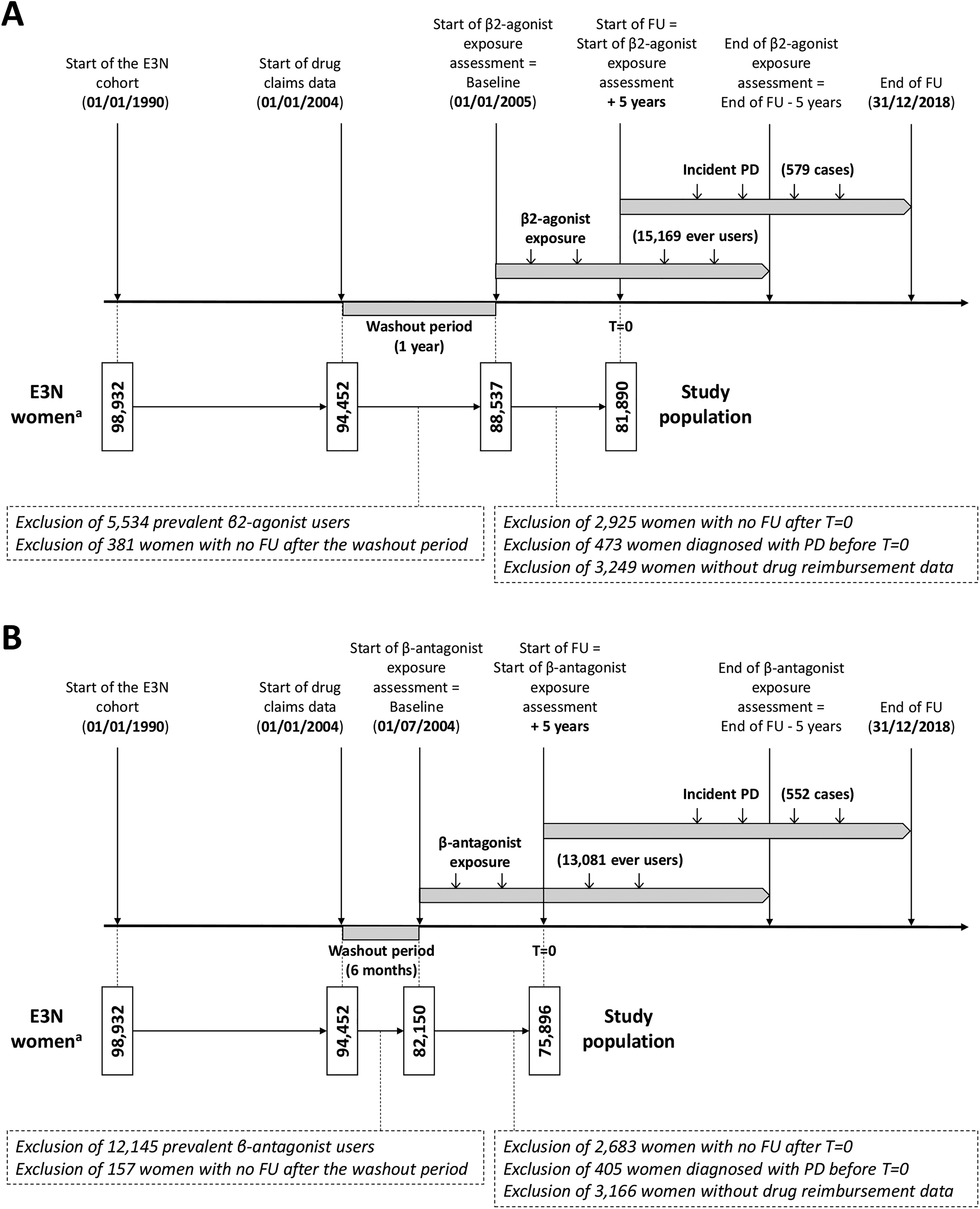
Study design for the analysis of the relation of time-varying use of β2-agonists (A) and β-antagonists (B) with the incidence of Parkinson's disease with a 5y exposure lag. FU: follow-up; PD: Parkinson's disease. aOf the 98,995 E3N women recruited in 1990, we excluded 50 women with possible Parkinson's disease and 13 women with an unknown date of Parkinson's disease diagnosis.
Nested case-control study: trajectories of β-adrenoreceptor drug prescriptions
We examined trajectories of β2-agonists/β-antagonists prescriptions prior to PD diagnosis within a nested case-control study (20 controls/case). To be selected as controls, participants had to be PD-free at the year of diagnosis of the matched case (index year, Y0), and have the same age at Y0 (incidence density sampling). 9
Trajectories of the frequency of ≥1 annual prescription of β2-agonists/β-antagonists prior to Y0 were estimated using a mixed logistic regression model with a backward timescale and a random intercept and slope (unstructured variance). Time was modelled using restricted cubic splines. We tested whether trajectories were different in cases and controls by including an interaction term (PD × time). The model was adjusted for age at Y0 (linear + quadratic terms). We plotted annual marginal predictions (95% CI) and case-control differences.
Cohort study: β-adrenoreceptor drugs and PD incidence
We estimated hazard ratios (HRs, 95% CI) using the Cox proportional hazards regression model for time-varying covariates with age as the time scale. Main analyses included a 5y exposure lag; hazard at time t is a function of exposure up to 5y earlier. We selected this lag based on previous studies10,11 while retaining a sufficient number of cases (Supplemental Methods).
Potential confounders were considered as fixed or time-varying (Supplemental Methods) and lagged in the same way as drugs. Specific categories were created for missing values of categorical covariates so that participants with missing values for covariates were retained in the analyses and all analyses are based on the same number of participants.
We performed analyses for time-varying ever drug use and cumulative number of drug claims (β2-agonists: 0/1/2/≥3; β-antagonists: 0 + three groups based on tertiles). For subgroup analyses of β2-agonists/β-antagonists, all subgroups were included in the same model (mutually adjusted).
For β2-agonists, we built four sequential models: Model 1 (M1) used age as the time scale; M2 was further adjusted for baseline type of residence, education, parity, age at menarche, and caffeine consumption, and time-varying smoking, BMI, physical activity, menopause, number of medical consultations, T2D, hypertension, CVD, hyperuricemia, and lipophilic statins; M3 was further adjusted for time-varying ever ICSs use; M4 was further adjusted for main indications (asthma/COPD). For β-antagonists, M1/M2 were the same as those used for β2-agonists and M3 was further adjusted for main indications (hypertension/CVD).
Sensitivity analyses
For β2-agonists, we performed analyses for ever use of individual drugs used by ≥1000 participants.
Main analyses were adjusted for time-varying ever/former/current smoking (available for >99% of participants). Given the importance of smoking as a confounder, we examined the impact of alternative smoking definitions (time-varying pack-years of smoking, 22% missing; baseline smoking) and restricted analyses to never smokers throughout the follow-up.
Given a previous report of an inverse β2-agonists-PD association restricted to persons without T2D, 3 we tested the β2-agonists × T2D interaction and performed analyses restricted to participants without T2D. Our study lacked statistical power for analyses in diabetic participants.
For both types of β-adrenoreceptor drugs, we performed analyses with a 2y-lag or no lag, and analyses with a 5y-lag including prevalent users for comparison to main analyses. For β2-agonists, we repeated our analyses with a 7y-lag.
Statistical power calculations
We computed the statistical power of the study for different HR values based on the observed probability of the event and frequency of exposures of interest using the SAS9.4 PROC POWER procedure with the coxreg option for Cox proportional hazards model. Our study had 80% power to detect a HR of 0.77 for all β2-agonists and of 1.32 for β2-antagonists (Supplemental Figure 3).
Results
β2-agonists
Study population
Table 1 shows participants’ baseline characteristics, overall and according to β2-agonists use at the end of the follow-up (never/1 prescription/>1 prescription). Of 81,890 participants (baseline age = 63.3y), 15,169 (18.5%) started using β2-agonists over the follow-up (Figure 1A). Of them, 7647 (50.4%) had >1 prescription: compared to never-users, they were older and had lower education and physical activity, earlier age at menarche, more children, higher BMI, and more frequent medical contacts and comorbidities, including asthma/COPD; they consumed more caffeine and were more often current smokers, post-menopausal, and ICS or lipophilic statin users. They also had more frequent asthma/COPD history than one-time users.
Baseline participants’ characteristics, overall and by β2-agonist exposure at the end of the follow-up.
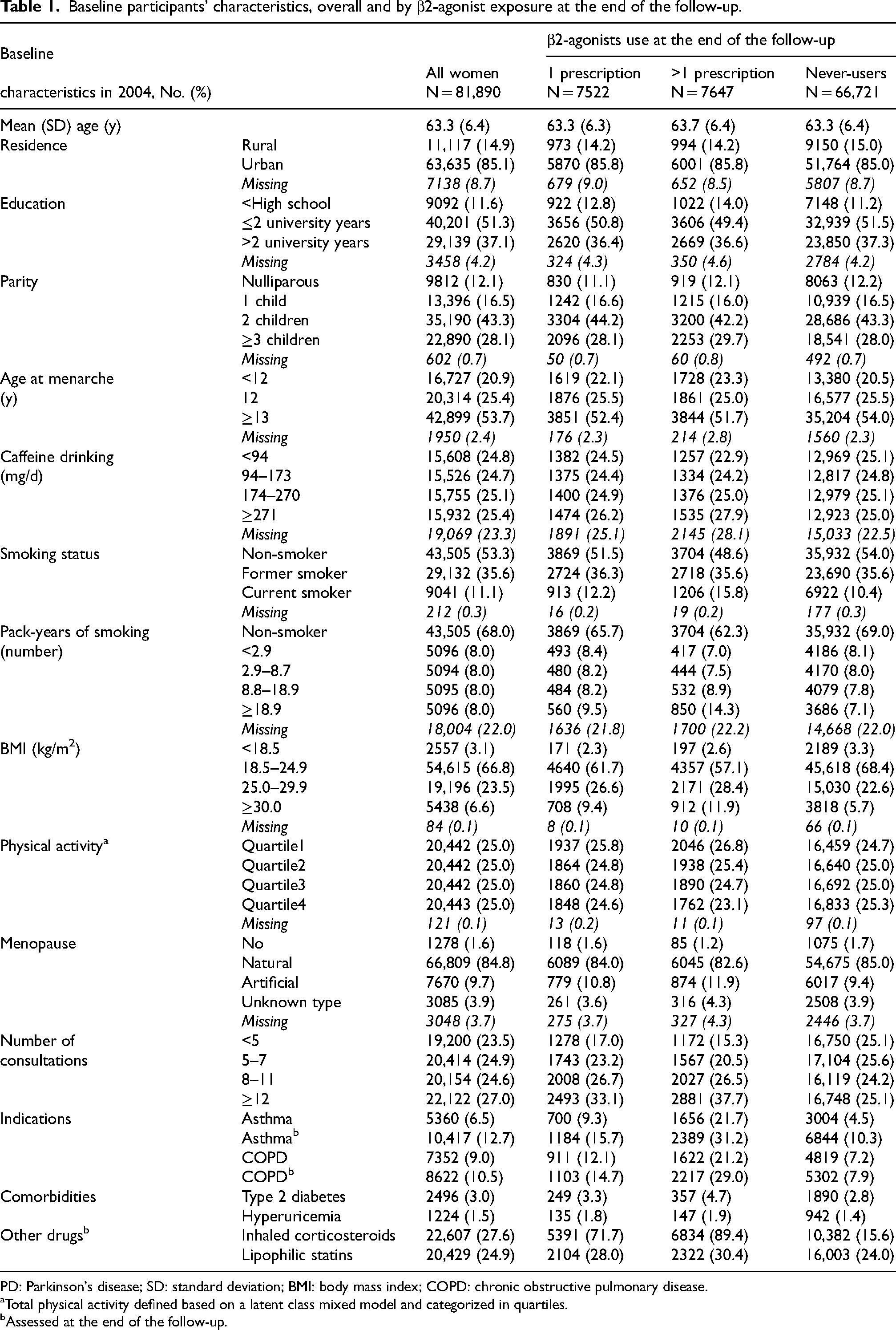
PD: Parkinson's disease; SD: standard deviation; BMI: body mass index; COPD: chronic obstructive pulmonary disease.
Total physical activity defined based on a latent class mixed model and categorized in quartiles.
Assessed at the end of the follow-up.
Between 01/01/2005–31/12/2018, 788 participants developed PD. After excluding those who developed PD within the first 5y, 579 participants developed PD between 01/01/2010–31/12/2018. Compared to those who did not (Supplemental Table 2), participants who developed PD were older, more frequently post-menopausal, less often obese, had more children, and consumed less caffeine; they smoked less frequently and had COPD less often. Asthma was not associated with PD.
Trajectories of β2-agonists prescriptions
We included 788 incident PD patients and 15,634 controls. The frequency of ≥1 annual prescription of β2-agonists increased over time in cases and controls, and tended to be lower in cases than controls up to 5y before diagnosis (Figure 2A1, A2). Although not significantly (pPD × time = 0.62), the frequency tended to increase slightly more steeply in cases than in controls, so that the case-control difference tended to decrease over time.
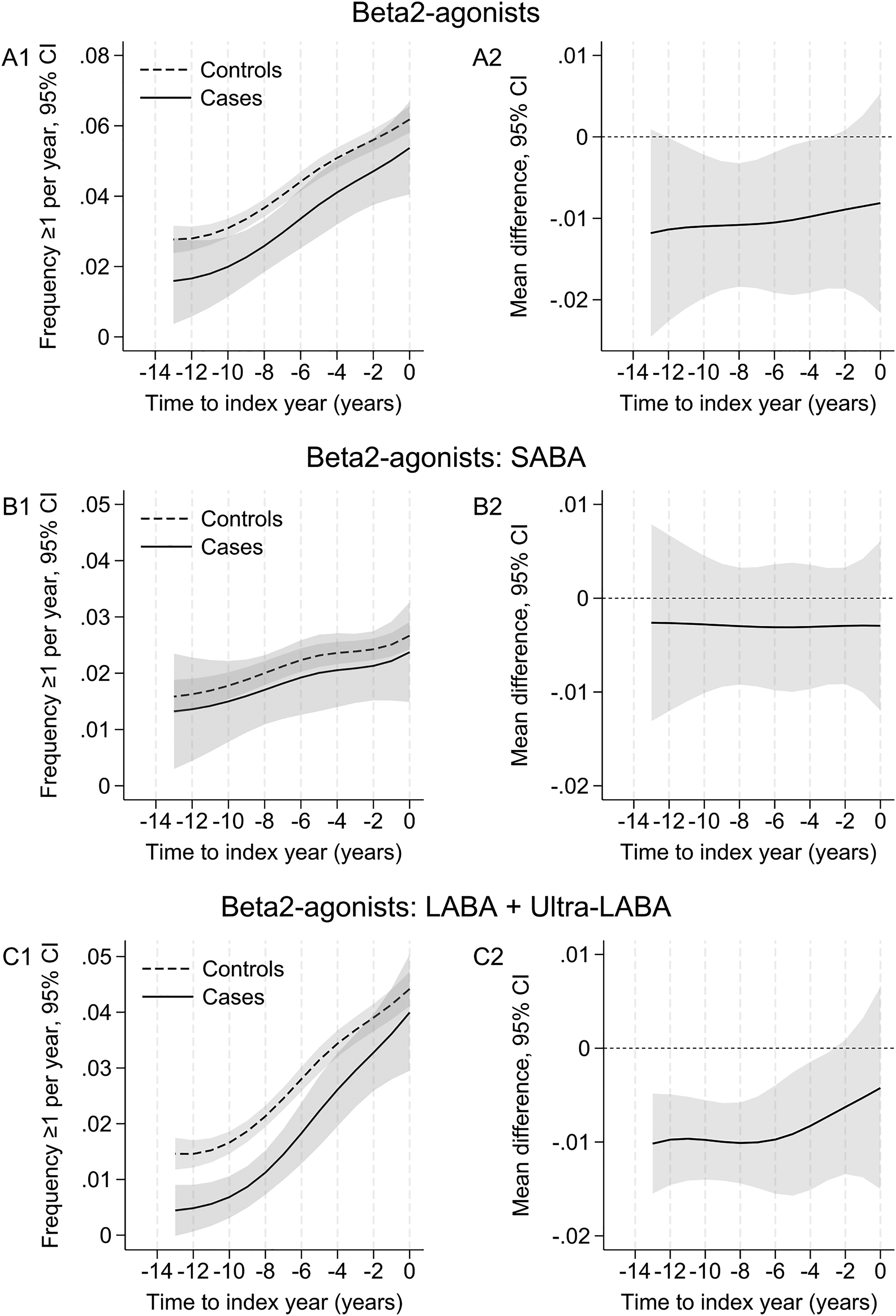
Trajectories of prescriptions of β2-agonists in cases and controls before the index date. SABA: short-acting β2-agonist; LABA: long-acting β2-agonist; ultra-LABA: ultra-long-acting β2-agonist. The case-control sample included 788 incident PD patients and 15,634 controls; 770 patients were matched to 20 controls, 15 to 10–19 controls, and 2 to <10 controls. The figures show estimates (95% confidence intervals, CI) of the frequency of ≥1 annual prescriptions of (A1) β2-agonists, (B1) SABAs, and (C1) LABAs/ultra-LABAs in cases and controls aged 75 years old at the index date (Y0 = 0), based on marginal predictions of a mixed logistic regression model with time in years (backward scale) coded as restricted cubic splines (3 knots at −12, −5, and −1 years). Plots A2, B2, and C2 show the corresponding average annual differences between cases and controls; differences whose CI do not include 0 are statistically significant (p ≤ 0.05).
There was no significant case-control difference for SABAs (pPD × time = 0.97; Figure 2B1, B2). For LABAs/ultra-LABAs (Figure 2C1, C2), the frequency of ≥1 annual prescription was significantly lower in cases than controls up to 5y before diagnosis, after which frequencies tended to converge in both groups; case-control differences were more pronounced compared to SABAs (pPD × time = 0.093).
β2-agonists and PD incidence
In M1 (Table 2), ever users of β2-agonists had lower PD incidence compared to never users (HR = 0.75, 95% CI = 0.56–0.99), with a statistically significant linear trend for the number of claims (p-trend = 0.03). The association was no longer significant after adjusting for confounders, ICSs, and asthma/COPD (HRM4 = 0.79, 95% CI = 0.57–1.11; p-trend = 0.12).
Association of time-varying β2-agonist use with the incidence of Parkinson's disease: analyses with a 5-year exposure lag.
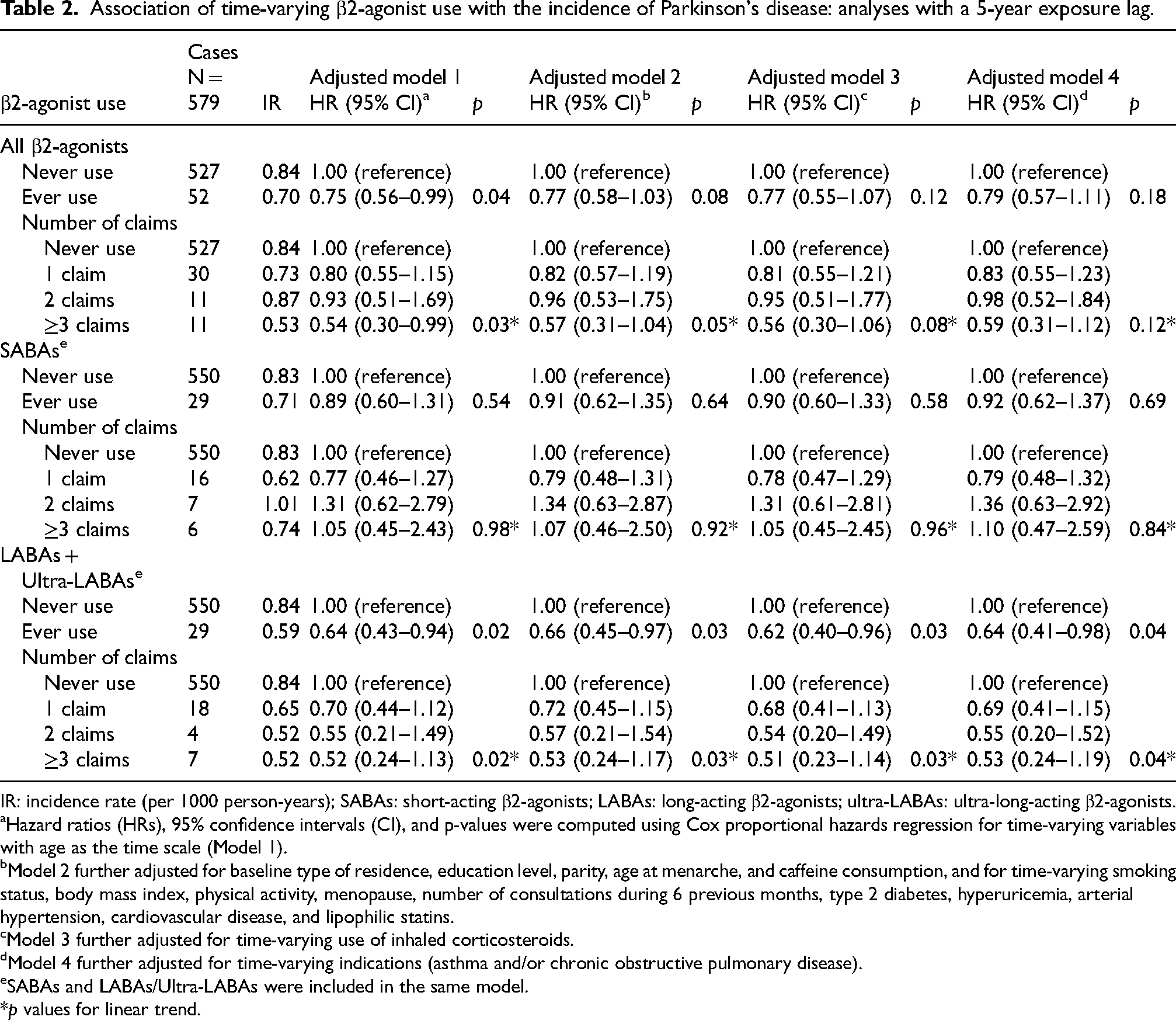
IR: incidence rate (per 1000 person-years); SABAs: short-acting β2-agonists; LABAs: long-acting β2-agonists; ultra-LABAs: ultra-long-acting β2-agonists.
Hazard ratios (HRs), 95% confidence intervals (CI), and p-values were computed using Cox proportional hazards regression for time-varying variables with age as the time scale (Model 1).
Model 2 further adjusted for baseline type of residence, education level, parity, age at menarche, and caffeine consumption, and for time-varying smoking status, body mass index, physical activity, menopause, number of consultations during 6 previous months, type 2 diabetes, hyperuricemia, arterial hypertension, cardiovascular disease, and lipophilic statins.
Model 3 further adjusted for time-varying use of inhaled corticosteroids.
Model 4 further adjusted for time-varying indications (asthma and/or chronic obstructive pulmonary disease).
SABAs and LABAs/Ultra-LABAs were included in the same model.
*p values for linear trend.
Among β2-agonist users, 8479 (55.9%; 29 PD patients) women used SABAs and 10,555 (69.6%; 29 PD patients) used LABAs/ultra-LABAs. In M1 (Table 2), ever users of LABAs/ultra-LABAs had lower PD incidence compared to never users (HR = 0.64, 95% CI = 0.43–0.94), with a statistically significant linear trend for the number of claims (p-trend = 0.02); the mean time between first exposure to LABAs + Ultra-LABAs and PD diagnosis was 7.7y (minimum = 5.1/maximum = 13.2) and the mean number of drug claims in PD patients with >1 claim was 10 (minimum = 2/maximum = 66). This association remained significant in M4 (HR = 0.64, 95% CI = 0.41–0.98; p-trend = 0.04).
One-time users are more likely not to have used the drugs they purchased or may use drugs for different indications compared to recurrent users; in addition, one-time exposure is unlikely to have a causal effect. Therefore, we also compared participants with >1 claim to those with 1 claim (HRM4 = 0.77, 95% CI = 0.36–1.66, p = 0.51) or 0 + 1 claim combined (HRM4 = 0.57, 95% CI = 0.30–1.09, p = 0.088). Similar results were obtained in analyses restricted to LABAs (data not shown). There was no association for SABAs.
Sensitivity analyses
Salbutamol was the most frequent SABAs (N = 6,977, 8.5%); formoterol was the most frequent LABA/ultra-LABA (N = 7,185, 8.8%), followed by salmeterol (N = 4,637, 5.7%). The strongest inverse associations were seen for formoterol (HRM4 = 0.62, p = 0.07) and salmeterol (HRM4 = 0.64, p = 0.15), although not statistically significant (Supplemental Table 3).
The association of LABAs/ultra-LABAs with PD became weaker as the lag decreased (Supplemental Table 4). The HRM4 for ever use of LABAs/ultra-LABAs was 0.73 (95% CI = 0.52–1.01) with a 2y-lag and 0.86 (95% CI = 0.65–1.14) without a lag. Alternatively, with a 7y-lag, there was a significant inverse association between LABAs/ultra-LABAs and PD of a similar size compared to analyses with a 5y-lag (HRM4 = 0.56, 95% CI = 0.31–0.99; Supplemental Table 5).
Analyses adjusted for baseline smoking status or pack-years (baseline/time-varying) yielded findings consistent with main analyses (Supplemental Table 6). Analyses restricted to never smokers yielded consistent HRs, although with wider CIs due to smaller sample size (Supplemental Table 7).
There was no significant LABAs/ultra-LABAs × T2D interaction (p = 0.75). The inverse association between LABAs/ultra-LABAs and PD was present in participants without T2D (HRM4 = 0.63, 95% CI = 0.40–0.98, p = 0.04).
In analyses with a 5y-lag including prevalent users (Supplemental Table 8), the association between LABAs/ultra-LABAs and PD was weaker compared to our main analyses and not statistically significant (HRM4 = 0.83, 95% CI = 0.59–1.16; p-trend = 0.21).
β-antagonists
Study population
Of 75,896 participants, 13,081 (17.2%) started using β-antagonists (Table 3, Figure 1-B); 10,176 (77.8%) had >1 prescription: compared to never-users, they were older and had lower education and physical activity, more children, higher BMI, and more frequent medical contacts and comorbidities (hypertension/CVD); they were more often diabetic and lipophilic statin users; they had CVD/hypertension history more often than one-time users.
Baseline participants’ characteristics, overall and by β-antagonist exposure at the end of the follow-up.
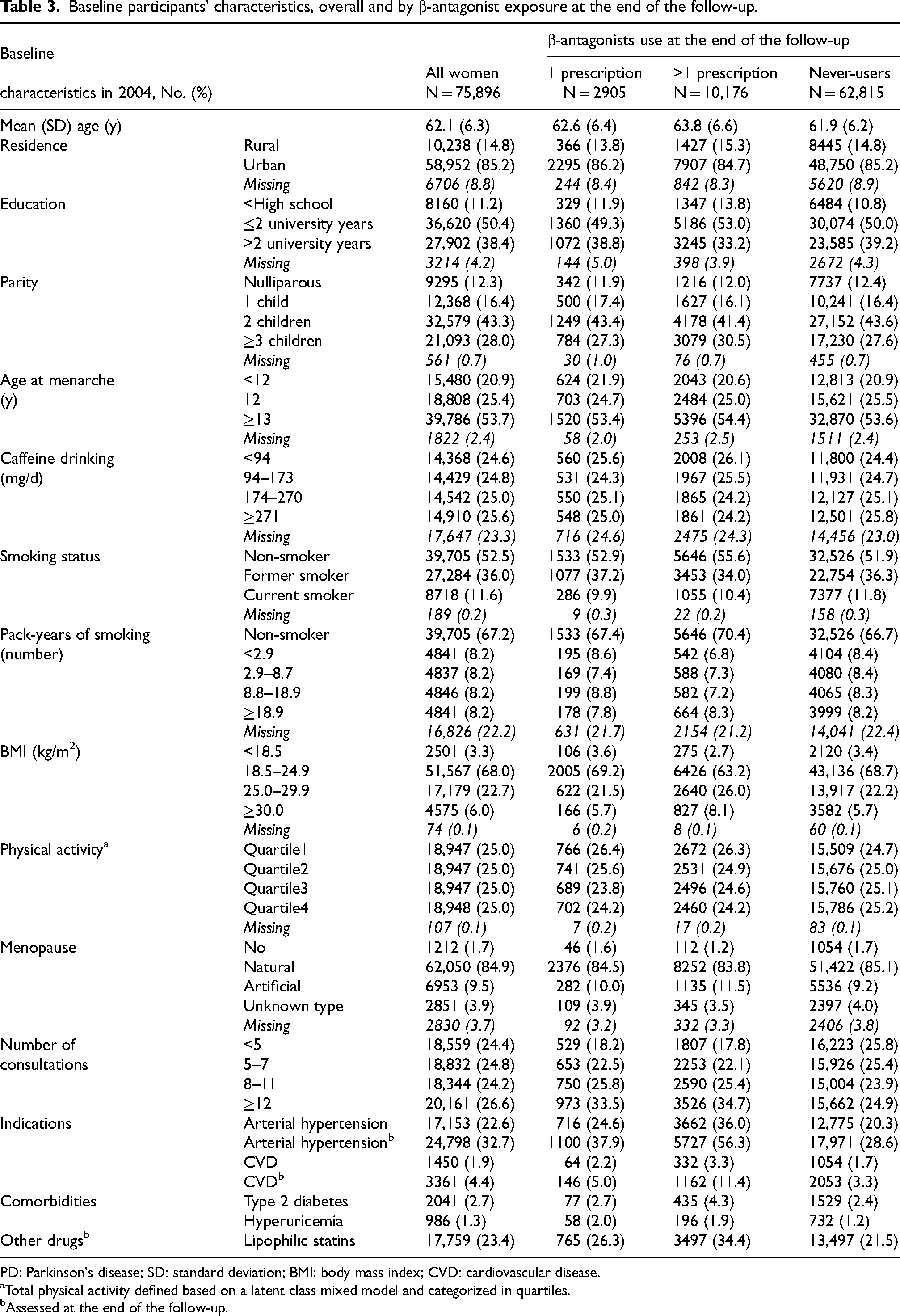
PD: Parkinson's disease; SD: standard deviation; BMI: body mass index; CVD: cardiovascular disease.
Total physical activity defined based on a latent class mixed model and categorized in quartiles.
Assessed at the end of the follow-up.
Between 01/07/2004–31/12/2018, 730 participants developed PD. After excluding those who developed PD within the first 5y of follow-up, 552 participants developed PD between 01/07/2009–31/12/2018.
Trajectories of β-antagonist prescriptions
The case-control sample included 730 incident PD patients and 14,541 controls. The frequency of ≥1 annual prescription of β-antagonists was initially similar in patients and controls; it increased at a steeper rate in patients than in controls, and became significantly higher in patients than controls ∼5 y before the index date (Figure 3A1/B1; pPD × time = 0.052). This pattern was mainly driven by propranolol (Figure 3B1/B2; pPD × time < 0.001). A similar pattern was present for less common non-selective β-antagonists (pPD × time = 0.001; Figure 3C1/C2). Selective β1-antagonists were more frequently prescribed to patients than controls in the 4–8y prior to diagnosis, but the difference decreased and there was no difference at Y0 (Figure 3D1/D2; pPD × time = 0.024).
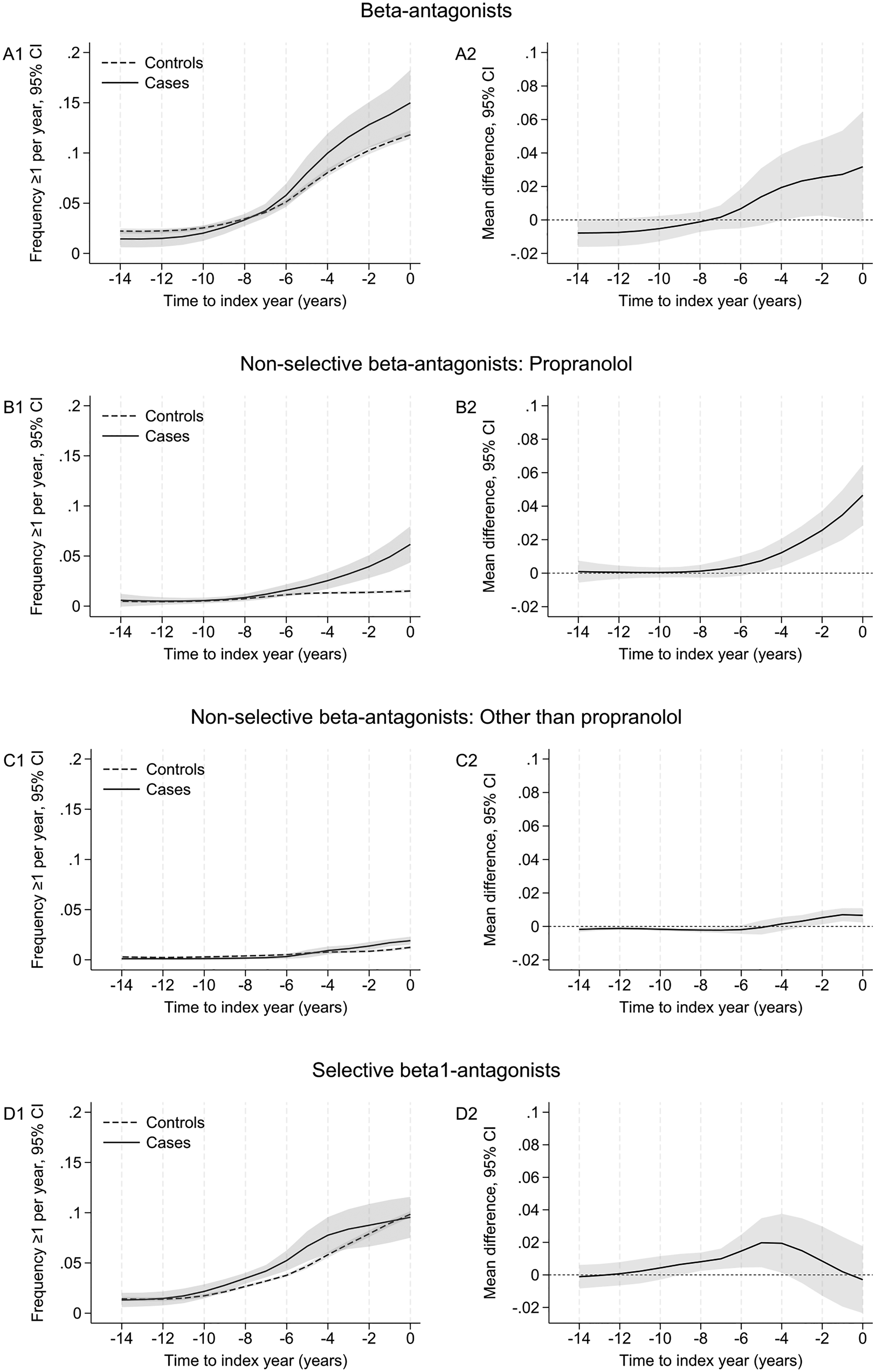
Trajectories of prescriptions of β-antagonists in cases and controls before the index date. The case-control sample included 730 incident PD patients and 14,541 controls; 722 patients were matched to 20 controls, 7 to 10–19 controls, and 1 to <10 controls. The figures show estimates (95% confidence intervals, CI) of the frequency of ≥1 annual prescriptions of (A1) β-antagonists, (B1) propranolol, (C1) other non-selective β-antagonists, and (D1) selective β1-antagonists, in cases and controls aged 75 years old at the index date (Y0 = 0), based on marginal predictions of a mixed logistic regression model with time in years (backward scale) coded as restricted cubic splines (3 knots at −12, −5, and −1 years). Plots A2, B2, C2, and D2 show the corresponding average annual differences between cases and controls; differences whose CI do not include 0 are statistically significant (p ≤ 0.05).
β-antagonists and PD incidence
Ever β-antagonists users tended to have increased PD risk, but the association was not statistically significant (Table 4, HRM3 = 1.24, 95% CI = 0.96–1.59; p-trend = 0.42).
Association of time-varying β-antagonist use with the incidence of Parkinson's disease: analyses with a 5-year exposure lag.
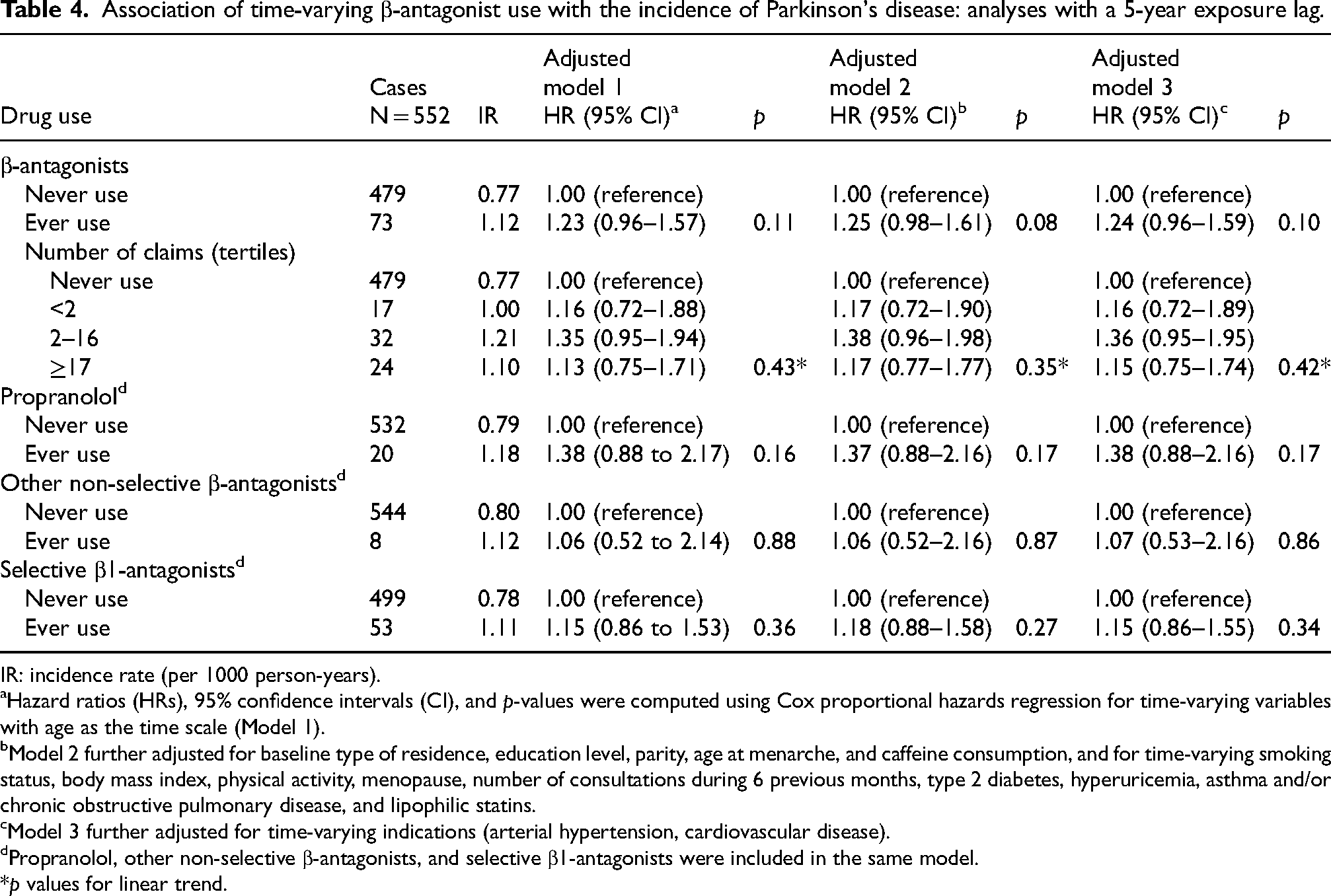
IR: incidence rate (per 1000 person-years).
Hazard ratios (HRs), 95% confidence intervals (CI), and p-values were computed using Cox proportional hazards regression for time-varying variables with age as the time scale (Model 1).
Model 2 further adjusted for baseline type of residence, education level, parity, age at menarche, and caffeine consumption, and for time-varying smoking status, body mass index, physical activity, menopause, number of consultations during 6 previous months, type 2 diabetes, hyperuricemia, asthma and/or chronic obstructive pulmonary disease, and lipophilic statins.
Model 3 further adjusted for time-varying indications (arterial hypertension, cardiovascular disease).
Propranolol, other non-selective β-antagonists, and selective β1-antagonists were included in the same model.
*p values for linear trend.
Among β-antagonist users, 3140 (24.0%) women used propranolol, 1519 (11.6%) other non-selective β-antagonists, and 10,037 (76.7%) selective β1-antagonists. The highest HR was observed for propranolol (HRM4 = 1.38, 95% CI = 0.88–2.16), but associations were not statistically significant.
Sensitivity analyses
The association between β-antagonists and PD increased as the lag decreased (Supplemental Table 9). β-antagonists were associated with increased PD incidence in analyses without a lag (HRM4 = 1.46, 95% CI = 1.22–1.74; p-trend = 0.02) and with a 2y-lag (HRM4 = 1.43, 95% CI = 1.17–1.75; p-trend = 0.03). In analyses without a lag, only propranolol was associated with PD (HRM4 = 2.29, 95% CI = 1.77–2.97); in analyses with a 2y-lag, the association between propranolol and PD decreased (HR = 1.65, 95% CI = 1.18–2.29) and selective β1-antagonists were also associated (HR = 1.34, 95% CI = 1.07–1.69).
Compared to main analyses in incident users, results were similar in analyses with a 5y-lag including prevalent users (Supplemental Table 10).
Discussion
In this large cohort study of women, incident use of LABAs/ultra-LABAs but not SABAs (nor β2-agonists overall) was associated with lower PD incidence in analyses with a 5y-lag. Incident use of β-antagonists was not associated with increased PD incidence in analyses with a 5y-lag, but was positively associated in analyses without a lag or with a 2y-lag, especially for propranolol.
β2-agonists
Supplemental Table 11 summarizes the main characteristics of previous studies. The latest meta-analysis reported a significant inverse association between β2-agonists and PD. 2 Three case-control12–14 and two cohort studies15,16 reported an inverse association, while two case-control studies17,18 and one cohort study 19 did not. One case-control study reported an inverse association in non-diabetics only. 3 Five additional studies have been published since. One cohort study (6y follow-up, baseline β2-agonists use) showed no association, 20 while three cohort studies (4–16y follow-up) reported lower incidence for time-varying use.21–23 Another case-control study did not show any association in patients with asthma/COPD. 24
The higher lipophilicity of LABAs/ultra-LABAs compared to SABAs allows them to bind lung β2-adrenoreceptors for a longer time. 25 Higher lipophilicity could allow LABAs/ultra-LABAs to penetrate more easily the blood-brain barrier and exert potential neuroprotective effects. 26 The meta-analysis found statistically significant inverse associations for two SABAs (salbutamol, 4 studies; terbutaline, 2 studies), and one LABA (formoterol, 2 studies), but not for another LABA (salmeterol, 2 studies). 2 Another case-control study found an association for cumulative duration of SABAs use but not for LABAs. 27 A case-control study with an 8y-lag reported an inverse association for formoterol + budesonide. 28 In one cohort study, ultra-LABAs displayed the strongest association. 21 We found an association for LABAs/ultra-LABAs; among individual drugs, the strongest associations were observed for two LABAs (formoterol, salmeterol) but were not statistically significant, due to insufficient statistical power.
Reverse causation due to prodromal PD represents a major issue for causal inference. Tremor is a common β2-agonists side-effect; therefore, PD patients with prodromal tremor 29 may be less likely to be prescribed β2-agonists before diagnosis or more likely to discontinue them. Six previous studies did not perform lagged analyses and 4 used lags <5y (Supplemental Table 11). Of 5 studies with lags ≥5y, one reported significant inverse associations with 5/8/10y-lags, 23 and another with 2/5y-lags 21 ; another study reported inverse associations for salbutamol (significant) and formoterol (non-significant) with 2/5/8y-lags 13 ; of two other studies, one reported an inverse association with β2-agonists (non-significant) 14 and the other no association. 12 Our main analyses used a 5y-lag. If prodromal tremor explained the inverse β2-agonists-PD association, we would expect it to become weaker as the lag increased; however, the HR did not decrease with a 7y-lag. Contrary to our hypothesis, β2-agonists prescriptions tended to increase slightly faster in cases than controls prior to diagnosis; the higher frequency of medical contacts of PD cases compared to controls in the years before diagnosis could lead to more frequent β2-agonists prescriptions. 11 Consequently, the association between LABAs/ultra-LABAs and PD decreased in analyses with a 2y-lag or no lag.
Confounding represents an important issue for the interpretation of previous studies. Smoking is inversely associated with PD, 30 can exacerbate asthma, and is a major risk factor of COPD. Hence, lack of (or imperfect) smoking adjustment may confound the association of β2-agonists with PD. The association between smoking and PD in our study is consistent with the literature. 30 Main analyses were adjusted for time-varying smoking status; adjusting for baseline smoking or pack-years or analyses restricted to never smokers yielded consistent findings. Of 15 previous studies (Supplemental Table 11), three adjusted for smoking,12,13,22 whereas others did not,14,18,19,23,24,27 adjusted for proxies,3,15,17,20,21 or did not provide information. 16 Future studies should carefully assess the role of smoking to reduce the risk of confounding-by-indication; larger studies in never smokers would be useful.
Our analyses were adjusted for other frequently co-prescribed drugs, and main β2-agonists indications (asthma/COPD). Previous studies showed weak positive associations between asthma and PD.19,22,31–33 Hence, confounding by asthma would not lead to an inverse association between β2-agonists and PD.
Analyses including prevalent users yielded weaker associations compared to main analyses. The new-user-design allows to exclude prevalent users who may be survivors in better health (prevalent user bias), to estimate more accurately the cumulative number of claims, and to measure confounders before treatment starts. 34 It was previously used to examine the association between statin initiation and colorectal cancer, another long latency condition.35,36 Studies have also examined the association between statins 37 or phosphodiesterase-5 inhibitors 38 initiation and dementia risk, or drugs associated with lower dementia incidence in PD. 39 However, we acknowledge that persons who initiate β2-agonists later in life may be different compared to those who start them earlier. Additional studies that compare the impact of including prevalent and incident users are warranted.
β-antagonists
β-antagonists were positively, but not significantly, associated with PD in analyses with a 5y-lag. The association increased and became statistically significant in analyses with shorter lags. It was mainly driven by propranolol; selective β1-antagonists were also associated with increased risk in analyses with a 2y-lag, although weaker compared to propranolol. The annual frequency of propranolol prescriptions started to increase at a markedly steeper rate in cases compared to controls 5y before diagnosis. There was some indication of changes before diagnosis for other non-selective and β1-selective antagonists, but differences were smaller and less consistent.
Reverse causation represents a major concern for analyses on β-antagonists, because propranolol is the most frequent treatment of essential tremor (ET). PD and ET can coexist and PD can be misdiagnosed as ET 40 ; action tremor is possible in PD and improved by β-antagonists. 40 Therefore, PD patients are more likely to use β-antagonists before PD diagnosis. Some previous studies performed lagged analyses and generally showed that associations between β-antagonists and PD strengthened as lags decreased.12,13,23,41 In some studies, associations remained statistically significant in analyses with lags >5y13,42; they were based on healthcare databases in which diagnosis date can be captured imprecisely and ET may be misclassified as PD. In one case-control study, 17 the OR (18-month lag) for propranolol decreased from 2.24 (95% CI = 1.90–2.64) to 0.97 (95% CI = 0.80–1.18) after adjustment for prodromal tremor. In addition, there was a similar pattern for primidone, another drug used for ET, suggesting that the association between β-antagonists and PD is explained by tremor rather than β-adrenergic antagonism.
Our findings strongly support the hypothesis that reverse causation explains the association of β-antagonists with PD due to prescriptions for prodromal tremor.
Strengths and weaknesses
In addition to the cohort study design, large sample size, and long follow-up, additional strengths pertain to time-varying exposures to address immortal time bias, 43 and our approach to address reverse causation. Our study is the first to have examined pre-diagnostic trajectories of β-adrenoreceptor drugs to highlight changes in prescriptions in the years before PD diagnosis. In addition, all previous studies on β-adrenoreceptor drugs and PD were based on healthcare databases. Compared to prospective cohort studies, these databases allow to identify larger number of patients and provide estimates that are more precise. On the other hand, they have two major drawbacks. 44 Information on confounders (i.e., smoking, physical activity, BMI) are often not available. Only 3/15 studies on β2-agonists adjusted for smoking,12,13,22 while we included numerous confounders. Their second limitation pertains to PD ascertainment. A systematic review on validity of algorithms to ascertain PD using routinely collected data reported large variability. 45 In a high-quality study, the positive predictive value (PPV) of <1 hospital PD admission was 71% (95% CI = 58–81). 46 Another study reported a 46% PPV (95% CI = 43–50); PD ascertainment using ICD codes overestimated the number of diagnoses by 73%. 47 Hence, studies based on healthcare databases are prone to diagnostic misclassification which can bias associations. In our study, the majority of PD diagnoses were validated based on medical records, allowing to differentiate PD from ET and other causes of parkinsonism. 6
Our study has limitations. First, participants are mostly health-conscious women not representative of the general population. However, since PD incidence rates were similar to expected rates, 6 selection into the cohort does not appear to have led to lower rates. E3N participants are educated and motivated women who provide high quality information in questionnaires with high response rates. 5 We acknowledge that we included only women, which hampers generalizability to men; however, women represent an understudied population in PD. Second, subgroup analyses and analyses according to number of claims need to be interpreted cautiously due to the small number of exposed PD patients. Our sample size is insufficient to estimate associations in strata defined by β2-agonists indications. Third, although the association of LABAs/ultra-LABAs with lower PD incidence was statistically significant, the HR had a relatively wide CI. Fourth, given the new-user design, the duration of exposure before PD diagnosis was relatively limited and might encompass the prodromal period in some patients. Another possible explanation could be that exposure to LABAs + Ultra-LABAs delays PD onset in persons with prodromal symptoms rather than prevents it. Finally, given its observational nature, our study does not prove causality.
In conclusion, our findings are consistent with the hypothesis that LABAs/ultra-LABAs may be associated with lower PD risk in women, although the difference between analyses including prevalent users or not calls for additional studies that examine this issue. The mechanisms underlying this association remain to be elucidated. Conversely, our findings provide strong evidence that the association between β-antagonists and PD in women is likely due to reverse causation.
Supplemental Material
sj-docx-1-pkn-10.1177_1877718X251330993 - Supplemental material for Use of β-adrenoreceptor drugs and Parkinson’s disease incidence in women from the French E3N cohort study
Supplemental material, sj-docx-1-pkn-10.1177_1877718X251330993 for Use of β-adrenoreceptor drugs and Parkinson’s disease incidence in women from the French E3N cohort study by Thi Thu Ha Nguyen, Agnès Fournier, Émeline Courtois, Fanny Artaud, Pascale Tubert-Bitter, Gianluca Severi, Pei-Chen Lee, Emmanuel Roze, Ismaïl Ahmed, Anne CM Thiébaut and Alexis Elbaz in Journal of Parkinson's Disease
Footnotes
Acknowledgments
We thank Isabelle Degaey, MD, for her contribution to the validation of diagnoses. We thank Raphaëlle Varraso, PhD, for her advice regarding the identification of women with asthma in E3N. The authors would like to acknowledge all women enrolled in the E3N cohort for their continued participation. They are also grateful to all members of the E3N study group and to the neurologists and general practitioners who provided medical documentation. The work reported in this article was performed during A.F.'s term as a Visiting Scientist at the International Agency for Research on Cancer.
ORCID iDs
Ethical considerations
The E3N cohort study is conducted in agreement with the French ethical standards for studies in human subjects.
Consent to participate
All participants signed informed consent, in compliance with rules of The French National Commission for Data Protection and Privacy which approved the study. The study protocol is registered at clinicaltrials.gov (NCT03285230).
Consent for publication
All participants gave their approval for publication of results of analyses performed in E3N.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This project was funded by the Michael J Fox foundation for Parkinson's research and the France Parkinson association. This work was realized with data of the E3N cohort (Inserm) and supported by the Mutuelle Générale de l’Education Nationale (MGEN), Gustave Roussy Institute, and French League against Cancer for the constitution and maintenance of the cohort. This work has benefited from State aid managed by the National Research Agency (ANR) under the program “Investment in the future” bearing the reference ANR-10-COHO-0006, as well as a subsidy from the Ministry of Higher Education, Research and Innovation for public service charges bearing the reference N°2102918823, 2103236497, and 2103586016, and from Iresp (Institut de recherche en santé publique). T.T.H.N. was supported by post-doctoral grants from the Michael J Fox foundation and the France Parkinson patient's association. E.C. was supported by a post-doctoral grant from the Michael J Fox foundation.
Conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability
E3N data are available to bona fide researchers for any type of health-related research, which is in the public interest. Data are made available under managed access owing to governance constraints and need to protect the privacy of participants. Raw data requests should be submitted through the E3N website (www.e3n.fr) or sent to contact@e3n.fr and will be reviewed by the E3N Access Committee (![]() ).
).
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
