Abstract
Dual tasking involves the simultaneous execution of two actions. In the context of healthy aging and neurodegenerative disorders, such as Parkinson's disease (PD) engagement in dual tasking frequently results in impaired gait or upper limb performance, thereby affecting functional independence. Transcranial electrical stimulation is a non-invasive technique able to modulate brain activity, which might represent a potential tool for reducing dual task interference. The goal of this review is to provide a comprehensive summary of the most recent findings about the use of transcranial electrical stimulation in improving dual tasking in the elderly and people with PD, including considerations about the optimal stimulation parameters. Differences in terms of stimulation protocols emerged across the included studies. Among transcranial electrical stimulation techniques, transcranial direct current stimulation (tDCS) was the most frequently employed. Currently, using tDCS to target dorsolateral prefrontal cortex either alone or in a multi-site fashion, along with a concurrent complex task, appears to be the most promising method for reducing dual task interference. Nevertheless, the lack of control over interindividual variability, the heterogeneity in outcome measures assessing dual tasking, and the variations in protocol elements like the frequency and the number of sessions prevented us from drawing definitive conclusions about the best paradigm.
Keywords
Introduction
Dual tasking, defined as the ability to simultaneously perform two tasks with distinct goals, 1 is a fundamental aspect of daily activities (e.g., chatting while cooking) and plays a vital role in ensuring safe mobility (e.g., talking on the phone while walking along the street). However, with aging and degeneration, extensive evidence indicates that engaging in dual task (DT) leads to decreased performance on one or both tasks, a phenomenon known as dual task interference (DTI). 2
Notably, performing attention-demanding tasks while walking deteriorates gait and balance performance, thereby increasing the risk of falls and this effect is more pronounced in individuals with neurological diseases, such as Parkinson's disease (PD). 3
In this context, a motor-cognitive DT occurs when a cognitive assignment (e.g., verbal fluency, counting backward) is added during gait. 4 Conversely, a motor-motor interaction occurs when a secondary motor action, such as carrying a cup of water or crossing obstacles, is simultaneously performed during walking.
Regarding upper limb functions, DTI is typically evaluated by combining fine motor skills (such as writing or typing) with a secondary task that engages cognitive processes such as working memory and attention (e.g., verbal fluency or serial subtraction task - SST). Also in this context, evidence indicates that dual tasking ability deteriorates with age, particularly in neurological conditions, and that DTI is associated with cognitive functions and daily functioning.
A common method for quantifying DTI is the Dual Task Cost (DTC), which is defined as the relative change from a performance executed in single-task respect to a DT condition and is calculated as follows: [(Dual-task performance – Single-task performance)/Single-task performance] X 100.5–7
Given the constant requirement to engage in DT during daily life activities, there is an urgent need to identify more effective interventions for healthy older adults and individuals with PD to overcome DT impairments. There are some data related to rehabilitation programs. Evidence on the effectiveness of physiotherapy and exercise in older adults has yielded inconclusive results in improving DT ability (for a review, see Law et al., 2014 8 ). Notably, due to safety concerns, training DT was often excluded from clinical practice in the past. However, recent research supports the importance of including DT training in rehabilitation programs. 9 Indeed, specific training based on motor-cognitive exercises showed some promising results in both the elderly without cognitive impairments 8 and individuals with PD, 10 despite the high heterogeneity of the therapeutic intervention. A recent systematic review with meta-analysis conducted on PD subjects found significant improvements in DT spatiotemporal parameters (i.e., gait speed, stride length, and cadence) when comparing DT training to single-task training, further supporting DT training use. 11
Furthermore, in recent years, the possibility of improving DT by applying transcranial electrical stimulation (TES) has gained attention whether delivered alone or paired with training.
Considering these premises, this review aims to provide a comprehensive summary of the latest findings about the use of TES in improving DT performance in the healthy elderly and patients with PD, with the aim of finding the best application approach (i.e., dose, target location, offline or online).
Here, we first summarize the evidence on dual-task impairments in the elderly and in PD patients, including the neuroanatomical network underlying dual-task performance and information about network dysfunctions linked to aging and neurodegeneration. Then, we will provide a brief overview of the basic functioning of TES. Finally, we will review studies addressing the use of TES to improve DT performance of both the upper and lower limbs in the elderly and in PD patients, critically discussing data to obtain information to be used in clinical practice.
Dual task in the healthy elderly and in Parkinson's disease
“Stops walking when talking” was the study by Lundin-Olsson et al. showing for the first time the attentional trade-off between gait and cognitive performance in elderly people during DT. 12 Starting from this seminal paper, the following decades have seen a growing interest in the interaction between dual tasking and aging, unveiling a correlation between cognitive and executive function deficits and DT motor performance. 13 Impairments in dual tasking among older adults have been mainly attributed to the physiological degeneration associated with aging, affecting primarily the white matter of the frontal regions. 14 This degeneration contributes to a spectrum of cognitive deficits ranging from impaired attention, memory, and particularly executive functions. 15 Executive functions encompass, among others, all the processes required to organize goal-directed behavior in the presence of potential distractions (such as in DT).16,17 The brain regions primarily involved in executive control are the prefrontal ones, which act as a relay station for cortical areas and subcortical structures, thus monitoring and controlling ongoing behavior. 16 Evidence showed that the elderly increasingly rely on cognitive control provided by prefrontal areas in executing simple tasks (e.g., walking, standing) and even more in complex tasks (e.g., DT walking). Specifically, older adults exhibit augmented activity in the dorsolateral prefrontal cortex (DLPFC) compared to young adults already during simple walking and even more during DT walking.18,19 This indicates that the elderly must rely on increased compensatory attentional control to correctly execute even simple motor tasks such as normal walking or quiet standing.18,19 These deficits are also evident in a significant increase in DTC in older adults during complex walking, 20 which in turn has been proposed as a predictor for increased fall risk, primarily attributable to the aforementioned reduction of cognitive/attentional capabilities. 21 It is important to highlight that increased cortical activity in non-motor regions may also be interpreted as a maladaptive processing, rather than a compensatory response, as previously demonstrated when studying the relationship between arousal and task performance. 22
In PD, a progressive neurodegenerative disorder characterized by impairments in both motor and non-motor functions, difficulties in performing DT are even more frequently reported than in the elderly. This applies to DT involving both gait and upper limb performance. During DT gait, individuals with PD show reduced step length,23–26 reduced gait velocity,23,24,26,27 and increased gait variability28–30 leading to an increased risk of falling.3,29–32 Regarding upper limb performance, impairments in handwriting 33 may be exacerbated in DT scenarios.34,35 Notably, the decline in DT performance may become more severe with the presence of freezing of gait (FOG) or specific PD phenotype (i.e., postural instability gait disorders), where cognitive and behavioral deficits might further affect DT execution.36,37
The pathophysiological basis of DT impairment in PD is not completely known, but two main theories are proposed, both involving DLPFC involvement, via the cognitive basal ganglia loop. The “loss of segregation theory” states that dopamine deprivation disrupts the parallel architecture of cortico-striatal circuits, which is organized into multiple loops (e.g., motor, cognitive, and motivational)38–41 allowing two activities to be executed simultaneously.42,43 The loss of parallel organization in PD44,45 might result in dysfunctional overlaps between motor and cognitive processes during DT. 46 The “neural bottleneck theory” is based on the greater dopamine depletion in PD in the dorsal-posterior putamen, which controls simple response selection and automatic movement47,48 respect to the ventral-anterior putamen.49,50 In PD patients, functional connectivity within the motor system shifts from the posterior to the anterior putamen,43,51,52 leading to a computational overlap between simple response selection and cognitive control processes.53,54 This might result in a neural bottleneck that prevents motor and cognitive tasks from being performed concurrently.
Recent evidence also suggests the role of cholinergic hypofunction in DT impairment occurring with aging and PD. In rats, Sarter and collaborators, found that loss of cortical cholinergic inputs impairs the attentional processing of gait, depriving the striatal circuitry of this information, used for selecting and sequencing motor actions. Thus, dual cholinergic–dopaminergic loss attenuates the supervision of striatal circuitry and thereby “unmasks” the consequences of striatal dopaminergic denervation on gait. 55 In humans, studies employing transcranial magnetic stimulation,56–58 functional magnetic resonance imaging59,60 and positron emission tomography imaging, 61 have established a connection between lower cholinergic activity and more pronounced DT gait deterioration in PD and the elderly.
Transcranial electrical stimulation
Transcranial direct current stimulation (tDCS), transcranial alternating current stimulation (tACS), and random noise stimulation (excluded from this review) are non-invasive methods used to modulate brain activity, belonging to the broader field of TES. By applying low-intensity (1–2 mA peak-to-peak) monophasic (tDCS) or biphasic (tACS) currents to the scalp, these techniques can adjust the neuronal resting state through hyperpolarization or depolarization of the neuronal membrane. 62 The effect on membrane potential varies with current polarity: anodal setup (anode on the target area and cathode as reference) induces depolarization, while cathodal setup (cathode on the target area and anode as reference) induces hyperpolarization. 63 Notably, tACS can also influence neuronal synchronization by adjusting the firing frequency of neurons according to natural brain rhythms (delta, theta, alpha, beta, and gamma). 64 This occurs because the alternating current can be set to various oscillation frequencies, modulating the discharge phase of the underlying neuronal cells. 65
Over the years, various TES montages have been developed, incorporating electrodes of different sizes, simultaneous stimulation of multiple regions (multisite TES), or configurations designed to deliver focal stimulation to enhance both inter- and intracortical networks, as in high-definition TES (hd-TES). 63 Moreover, most studies have applied TES protocols at rest (i.e., offline), evaluating the effects of stimulation in a pre/post-experimental design. However recent research has focused on combining stimulation with simple or complex tasks (i.e., online), such as standing, walking, or dual tasking (some examples will be provided in this review).
Numerous studies have examined the impact of TES on a multifaceted array of brain functions such as cognition (e.g., Antal et al., 2022 63 and Rauh et al., 2023 66 ), motor learning (e.g., Hu et al., 2022 67 ), motor performance (for a review on PD, see Goodwill et al., 2017 68 ) on different populations ranging from healthy subjects to patients suffering from stroke or affected by neurodegenerative diseases. However, the effects of TES on these functions are not completely clear, because TES protocols are affected by a high variability related not only to the technical aspects of the protocol itself but also to other factors.
First, interindividual variability in TES response is one of the biggest issues to be faced when designing a stimulation protocol. The strength and the distribution of the electrical field depend on several aspects such as the skin and skull thickness, the head and cerebrospinal fluid volumes, and the age of the subjects. 69 Moreover, also hormonal levels, genetics, gender, and pathophysiological substrates contribute to increasing the response variability to TES, 70 as well as the brain's neural state 71 and the possibility of activating the peripheral nerves under the active electrode.71,72 A possible way to control at least for some of these parameters is to use computer modeling software that can simulate the distribution (e.g., superficial area, depth of stimulation) and intensity of the induced electrical field in the brain by considering average data of skull and skin thickness and brain structure templates. 73
Another aspect that contributes to the variability of TES efficacy and that should be carefully controlled is the effects of medication. In this review, we considered studies involving the elderly (where in most of the studies the presence of drugs affecting the central nervous system was controlled – see Supplemental Material) and PD patients who were under dopaminergic therapy. The state of the dopaminergic system has been shown to affect neuroplasticity mechanisms driven by tDCS (for a review, see Nitsche et al., 2012 74 ). PD patients have shown deficits in neuroplasticity mechanisms that might be partly restored by L-Dopa, 75 meaning that the efficacy of TES might be related to the ON-phase, and hence, this aspect should be thoroughly considered when interpreting studies’ results. The same concept can be applied to other drugs affecting neuroplasticity such as cholinergic, serotonergic, or adrenergic drugs (Nitsche et al., 2012 74 ). Here, to partially control for these limitations, we evaluated the methodological strength of the included articles by applying a checklist proposed by Buch and collaborators, specifically for non-invasive brain stimulation techniques. 76
Methods
For this review, an independent online search on MEDLINE (PubMed) database was conducted to identify eligible English-language publications. The search terms included a combination of “dual-task”, and “transcranial direct current stimulation” or “transcranial alternating current stimulation”, and “Parkinson disease” or “elderly”. The publication dates ranged from January 2013 to December 2023.
Study selection criteria and screening process
Randomized controlled trials (RCTs) or crossover trials assessing the use of TES to improve DT performance were eligible. Studies were selected based on the following a priori inclusion criteria: i) involving the healthy elderly and/or patients with PD; ii) applying TES for improving DT performance; iii) reporting data on DT performance (e.g., DTC and/or DT parameters). The exclusion criteria encompassed review articles, data published as abstracts or conference papers, and studies applying tDCS as an add-on effect to training with multiple sessions.
Data collection
The following data were extracted from the included studies: i) study aim; ii) sample size; iii) study design; iv) number of TES sessions and washout; v) duration and intensity of stimulation; vi) modality (online/offline); vii) DT type; viii) main study results on DT. In addition, for studies involving individuals with PD, the following information was also extracted: i) Hoehn & Yahr (H&Y) stage; ii) disease duration; iii) disease severity (MDS-UPDRS part III and total score); iv) Levodopa equivalent daily dose (LEDD) and v) pharmacological condition of participants during the task (i.e., ON and/or OFF phase).
Methodological strength of included articles
To evaluate the methodological strength of the included articles, we applied a checklist proposed by Buch and collaborators, 76 specifically assessing, reporting, and controlling for: i) experimental design factors (e.g., blinding and control use), ii) participant factors (e.g., demographics, handedness, medication), iii) stimulation factors (e.g., electrode size and position, stimulation characteristics), and iv) analysis and statistics factors (e.g., reporting of effect-size(s), data available in public repositories).
Results
Our search strategy identified a total of 39 studies (17 for the elderly and 22 for PD patients), of which 23 were excluded after reading titles and abstracts. The remaining 16 studies underwent a comprehensive full-text assessment and were deemed eligible for this review (7 for the elderly and 9 for PD patients). The main findings of our search were presented in four sections describing the impact of TES on gait and balance DT and upper limb DT in the elderly and PD individuals. Demographic data of older adults are reported in the text, whereas characteristics of individuals with PD are summarized in Table 1. Details and main results of the included articles are reported in Table 2 (elderly) and Table 3 (PD), and in the Supplemental Material. Results of the effectiveness of neuromodulation on DT performance on different cortical and subcortical sites are depicted in Figure 1.
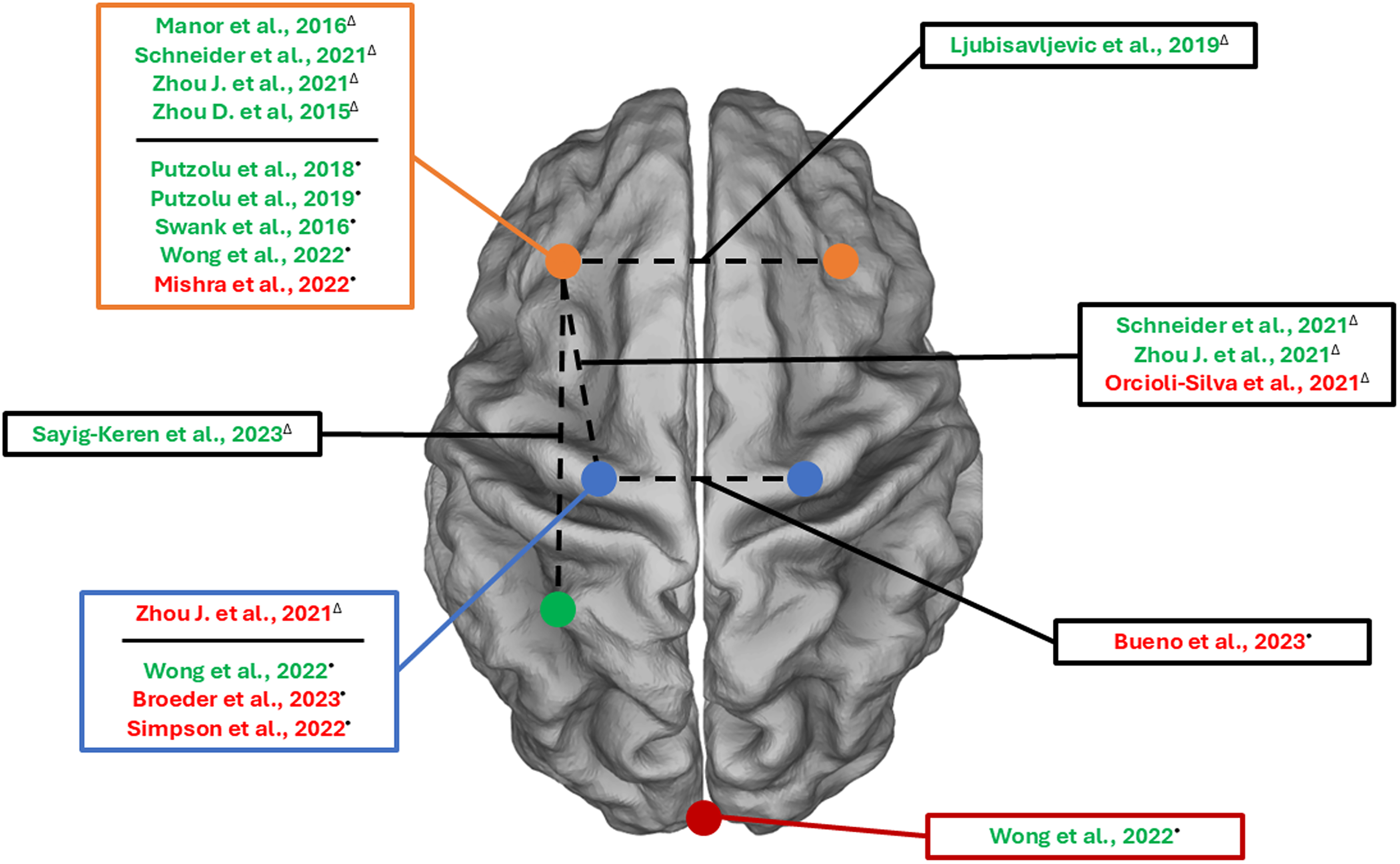
The figure shows the cortical regions stimulated in the studies included in the present review.
Demographic and clinical characteristics of PD participants at baseline.

Values are presented as mean ± standard deviation. * Indicates values presented as median (1st quartile, 3rd quartile). ° Indicates disease duration expressed in months. y: years; M: male; F: female; H&Y: Hoehn and Yahr; MDS-UPDRSIII: Movement Disorder Society-Unified Parkinson's Disease Rating Scale – part three; MDS-UPDRS TOT: Movement Disorder Society Unified Parkinson's Disease – total score; LEDD: Levodopa equivalent daily dose; mg: milligrams.
Studies characteristics and results for the elderly.

n°: number; DT: dual task; atDCS: anodal transcranial direct current stimulation; DLPFC: dorsolateral prefrontal cortex; age: mean age, reported as mean ± standard deviation; GPT: Grooved Pegboard Test; SST: serial subtraction task; DTC: dual task cost; M1: primary motor cortex; PFC: prefrontal cortex; N.S.: non-significant; tACS: transcranial alternating current stimulation; VR: virtual reality; SM1: primary sensorimotor cortex; CI: complexity index. Primary outcome measures are shown in bold in the Results column.
Studies characteristics and results for PD.
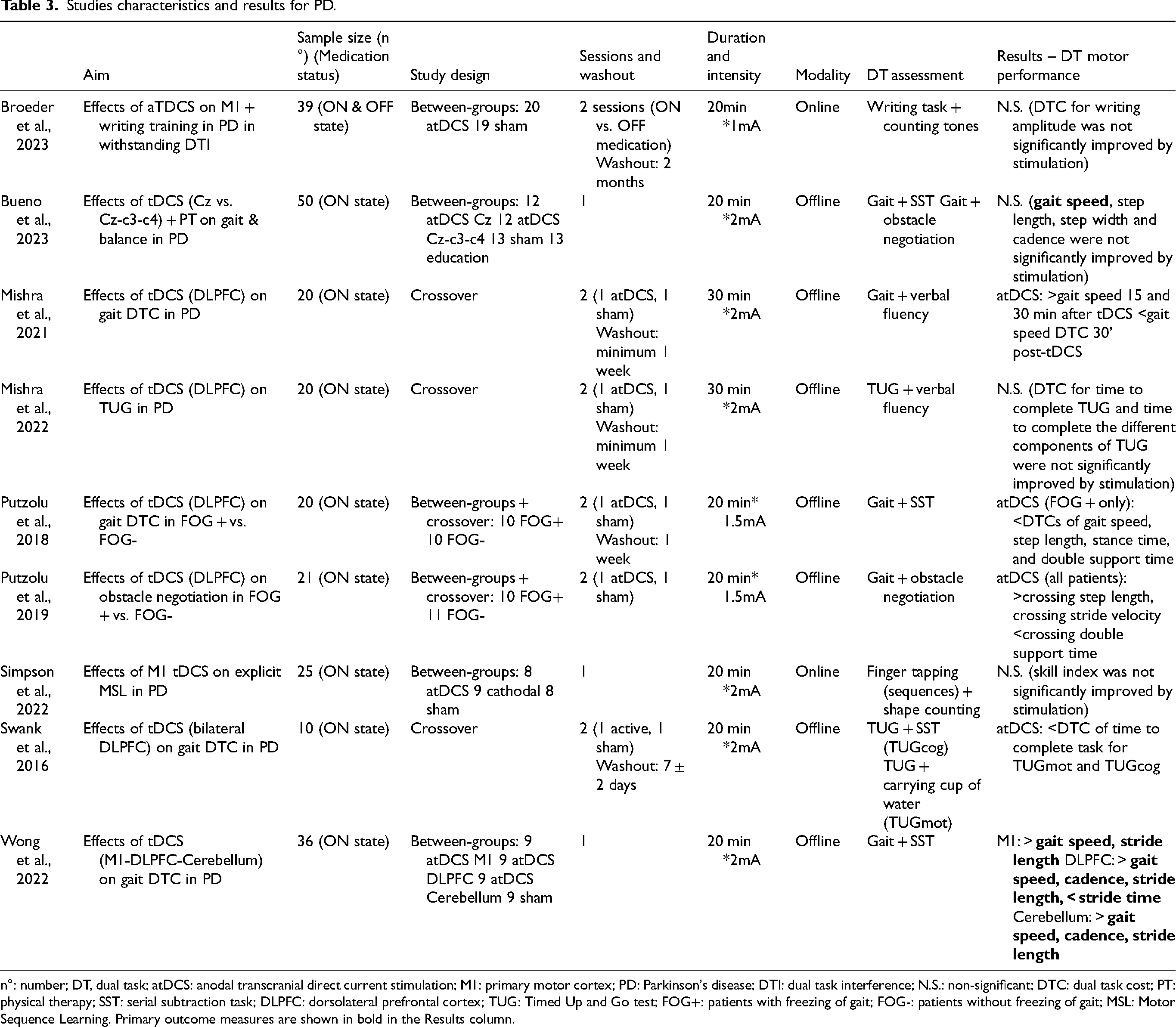
n°: number; DT, dual task; atDCS: anodal transcranial direct current stimulation; M1: primary motor cortex; PD: Parkinson's disease; DTI: dual task interference; N.S.: non-significant; DTC: dual task cost; PT: physical therapy; SST: serial subtraction task; DLPFC: dorsolateral prefrontal cortex; TUG: Timed Up and Go test; FOG+: patients with freezing of gait; FOG-: patients without freezing of gait; MSL: Motor Sequence Learning. Primary outcome measures are shown in bold in the Results column.
Impact of TES on gait/balance DT in the elderly
Of the six included studies evaluating the efficacy of TES on gait and balance DT in the elderly (total enrolled participants = 204), two delivered the stimulation over DLPFC alone, three delivered a multisite stimulation (for details see below) and one implemented a tACS protocol. Results of the studies will be presented beginning with DLPFC stimulation protocols in order of complexity (e.g., single site, multisite), followed by protocols involving sensorimotor areas or other TES protocols (i.e., tACS).
Single site tDCS over DLPFC. Two studies performed a single site tDCS stimulation over DLPFC and both showed positive results in terms of dual tasking behavioral measures. Specifically, Zhou D. et al. showed an increased balance control on DT standing task after atDCS over the left DLPFC (lDLPFC). 77 Consistently, Manor and colleagues found that atDCS over the lDLPFC induced an improvement in balance and gait speed in terms of a reduced DTC during dual tasking conditions. 78
Multisite TES over DLPFC and/or sensorimotor areas. Positive effects on DT performance after prefrontal stimulation were reported also in other studies, which implemented more composite stimulation protocols such as multisite tDCS and online stimulation. More in detail, three studies implemented multisite tDCS stimulation protocols over the DLPFC and sensorimotor areas (M1 or SM1), while only one tested a theta tACS multisite stimulation protocol over the DLPFC and the parietal cortex (PC). Starting from the tDCS protocols, Zhou J. et al. found a reduction of DTC in both standing (i.e., reduced DTC for postural sway speed and area) and walking (i.e., reduced DTC for gait speed) conditions after the ‘DLPFC’ and ‘DLPFC + SM1’ stimulation protocols. 79 Similarly, Schneider et al. found a reduction of DTC on gait parameters (for details, see Supplemental Material), when multisite tDCS (DLPFC + M1) was paired with complex walking (i.e., treadmill walking + virtual reality) compared to sham stimulation or tDCS during quiet sitting. 80 On the other hand, Orcioli-Silva and colleagues did not retrieve any substantial modifications in DTC for gait parameters following a multisite stimulation over M1 and left prefrontal cortex (PFC) combined with a treadmill walking at self-paced speed plus a serial subtraction task. 81
Lastly, Sayig-Keren and collaborators implemented a theta (i.e., 6 Hz) tACS multisite protocol (M1 – PC), and compared it with an atDCS protocol over lDLPFC. The authors found a positive effect of multisite theta-tACS in terms of reduction of DTC for gait speed and step length when compared to baseline and sham stimulation, but significant differences were not retrieved in comparison with atDCS over lDLPFC. 82
Impact of TES on gait/balance DT in PD
Of the seven included studies evaluating the efficacy of TES on gait and balance DT on PD (total enrolled participants = 177) six studies delivered the stimulation over DLPFC, four of which evaluated tDCS delivered in single-site to lDLPFC, one compared the effects of DLPFC tDCS to other cortical areas, and one study evaluated the effect of bilateral tDCS over DLPFC. Only one study evaluated the effects of tDCS on motor areas alone. Results of the studies will be presented in order as follows: i) Single site tDCS over DLPFC; ii) tDCS over bilateral DLPFC or sensorimotor areas.
Single site tDCS over DLPFC. Three out of four studies evaluating single-site tDCS effects over DLPFC reported significant changes in DT performance. Mishra and collaborators 83 demonstrated an increase in DT gait speed and number of words generated per minute and a reduction in gait speed DTC post-stimulation. A study of our group demonstrated that DTCs of gait speed, step length, stance time, and double support time significantly improved only in the group of PD patients with FOG. 7 In contrast, the effects on motor-motor interactions of DT gait, with a specific focus on obstacle negotiation performance, 84 showed improvements in step length, stride velocity, and double support time during obstacle crossing without differences between freezers and non-freezers. Moreover, these changes were positively correlated to the Hoehn and Yahr stage and the disease duration, supporting the idea that participants with worse clinical conditions benefited more from tDCS application. Conversely, a second study by Mishra and colleagues revealed that DT Timed Up and Go performance was not changed by tDCS over DLPFC. 85
tDCS over bilateral DLPFC or sensorimotor areas. Swank and co-workers targeted bilateral DLPFC, with the idea of simultaneously increasing extracellular dopamine levels in the striatum via cathodal stimulation over the right DLPFC and promoting functional network connectivity with anodal stimulation over the left DLPFC. 86 Results showed that the DTC of time to complete the task was diminished for both TUG while carrying a full cup of water and TUG while counting backward after active tDCS, with a greater impact on the latter task. Moreover, the DTC of the number of correct calculations was reduced after real stimulation.
Besides DLPFC, other stimulation targets have been studied. Indeed, in a recent RCT of Wong and colleagues, 87 36 PD patients were recruited, divided into 4 groups depending on the target site and experimental condition (i.e., M1, DLPFC, cerebellum, and sham), and stimulated for 20 min at 2 mA during their ON state. In DT, DLPFC tDCS intervention was able to increase gait speed, cadence, and stride length, reduce stride time, and increase cortical inhibition. Cerebellum tDCS significantly increased gait speed, cadence, and stride length, whereas M1 improved only gait speed and stride length.
In another investigation targeting Cz only or a larger region of the motor cortex (C3-Cz-C4) 88 prior to physical therapy (focused on improving gait and balance), no effects of active tDCS were found. However, it is important to highlight that the main aim of this study was not specifically to enhance DT gait performance but rather to improve overall walking and balance. This could have also influenced the target area selection.
Impact of TES on upper limb DT in the healthy elderly
One study investigated the effect of tDCS with different online montages (unilateral and bilateral over DLPFC) in a manual dexterity task in single and dual-task conditions, showing a reduced DTC in the concurrent cognitive task when anodal tDCS was applied over DLPFC. No effects were found in the manual dexterity task. 89
Impact of TES on upper limb DT in PD patients
Two studies investigated the impact of tDCS targeting M1 on DT during tasks performed with the upper limbs in PD, however without finding significant differences neither in writing amplitude and its DTC 90 nor on skill index after a motor sequence learning task. 91
Methodological strength of papers included in this review
The checklist of Buch and collaborators was applied to evaluate the methodological strength of the works included in this review and is reported in detail in the Supplemental Material.
All studies involving the elderly were sham-controlled and double-blinded. As for the sample size, three out of seven studies performed a power analysis, based on previous studies or pilot studies by the same group.79,81,82 All studies reported the age, gender, and medical condition of participants. Prescribed medication was not reported only by one study. 81 Concerning the stimulation protocols, all studies consistently reported electrode position, stimulation intensity, duration, and number of sessions. Importantly, all studies specified also the time between sessions (or washout time), which is critical information, especially considering the presence of crossover studies (for more information, see Table 2). The current density was not reported by three studies.77,78,82
Regarding PD patients, all the studies were sham-controlled and the majority included a double-blind design. Only 3 out of nine studies included a sample-size estimation.87,88,90
Concerning participant factors, all the included studies correctly reported the demographical characteristics of participants, medication status, and neuropsychological evaluation. All studies except one 84 correctly specified information about the washout time (for more information see Table 3). Stimulation factors, electrode position, stimulation intensity, duration, and number of sessions were correctly reported in all the studies. The current density was stated in only two studies.90,91
Factors were considered as controlled when there was: i) methodological consistency between groups or conditions (e.g., using the same equipment); ii) randomization or matching; iii) statistical analysis (e.g., between-groups comparison or used as a covariate). Participant factors were not controlled on average, in half of the studies, whereas stimulation factors were correctly controlled in most of the studies. Interestingly, only one study controlled for time interval between sessions 90 and two studies for subjects’ attention during testing.83,85
Discussion
The main aim of this review was to explore the effects of transcranial electrical stimulation on dual tasking in the elderly and in patients affected by PD.
General considerations
Among the studies included in this manuscript, differences were observed in terms of stimulation protocols, although generally the classical TES parameters were followed by all authors. Indeed, the current intensity was delivered in the range of 1-to-2 mA for a duration ranging from 20 to 30 min. One exception was reported in the study by Orcioli-Silva where the intensity of stimulation was set at 0.6 mA due to the reduced dimensions of the electrodes (i.e., 3 × 3 cm). Most studies in the elderly and in PD applied stimulation offline, with two exceptions made by Schneider et al. and Orcioli-Silva et al. in the elderly.
TES to improve dual task in the elderly and in PD: DLPFC as the best target?
Most of the studies showed an improvement in DT performance by using TES targeting the DLPFC, a crucial region in the executive network and therefore actively involved in dual tasking.
Of the studies targeting DLPFC that showed an enhancement of lower limb DT performance, all performed a single stimulation session. Three out of these studies involved the elderly79,80,82 whereas 5 studies involved PD patients, all delivering offline tDCS.7,83,84,86,87
In these studies, positive effects were reported in several domains ranging from postural control to gait, while limited evidence was retrievable for manual tasks in both PD and the elderly cohorts.
Regarding standing posture, which in terms of complexity might result in a simple and automatic task that is hardly affected by concurrent cognitive involvement, it is worth remembering that it requires multisensory integration. 92 The multisensory integration process results in a complex pattern of oscillatory movements of the center of pressure (COP), which decreases in complexity with senescence, reflecting an alteration in the ability to cope with perturbations. 77
Zhou D. et al. 77 and Manor et al. 93 applied offline tDCS protocols and evaluated quiet standing in ST and DT conditions in the elderly. First, confirming previous findings (see Kang et al., 2009 94 ) they showed how performing a simple cognitive task during quiet standing already reduces the complexity of the COP patterns, hence presenting a cost in terms of dual tasking performance. Secondly, and most importantly for the aim of the present review, they both showed that a single session of tDCS applied over DLPFC was able to increase the complexity pattern of the postural sway under dual tasking conditions 77 and to reduce the DTC associated with the concurrent execution of a cognitive task during quiet standing. 78
Similarly, but with a different approach, Zhou J. and colleagues found overlapping results but also with a multisite montage involving both prefrontal and primary sensorimotor areas. 79 Interestingly, their results showed that the efficacy of the stimulation apparently relies on the involvement of prefrontal regions, as no significant balance or gait improvements were observed in dual tasking when stimulating the sole sensorimotor area. 79 Notably, the same applies also in the case of upper limb DT performance as shown by Ljubisavlejvic on the elderly and by the two studies applying tDCS on M1 in PD. Here, an effect in improving upper limb DT performance was found only in the case of DLPFC stimulation (as in Ljubisavlejvic et al., 2019 89 ) but not in the case of M1 alone, where no effect on DT was found for PD patients.90,91
The effect exerted by tDCS on prefrontal regions on postural control might be explained in terms of an increased capacity of efficiently recruiting available cognitive resources, also referred to as ‘cognitive reserve’, which, in some ways, counterbalances the physiological cognitive decline observed in aging.14,15,78
Related to DT gait performance, the results shown in the elderly by Manor and Zhou J. in the previously cited works,78,79 reported a significant reduction in DTC during DT walking in addition to DT quiet standing. In addition, significant improvements in dual tasking gait were consistently observed also in people with PD.
Specifically, in the context of two offline studies targeting DLPFC to enhance motor-cognitive interaction,83,87 and one study applying a DLPFC bihemispheric protocol to improve both motor-cognitive and motor-motor walking tasks, 86 positive changes in gait spatiotemporal parameters or their DTC, were identified. Further, individuals with PD were shown to be able to improve obstacle-crossing performance after DLPFC atDCS, irrespective of the presence of FOG. 84 Previous research by our group 84 demonstrated that only PD patients with FOG improved motor-cognitive DTCs following DLPFC stimulation. Although DLPFC contribution during DT gait is not ruled out in the PT patients without FOG, this result suggests that DLPFC is more implicated in gait deterioration under dual-task in PD patients with FOG, making this area particularly susceptible to TES modulation. 7 However, the presence of FOG should be specifically addressed in future studies.
The speculation of DLPFC being more implicated in DT gait deterioration in PD patients with FOG can be traced back to the theories that explain DT worsening of gait performance in PD, which were mentioned in the ‘Introduction', specifically the “loss of segregation theory” in the basal ganglia. Recent evidence suggests that loss of segregation in the basal ganglia is particularly evident in patients with FOG, leading to the freezing phenomenon when competing, yet concurrent inputs (cognitive, sensorimotor, and emotional), are processed. 95 Indeed, the clinical characteristics of FOG (which precipitates in crowded places or when PD patients are under pressure) favor this hypothesis.
Lastly, related to DLPFC TES and DT gait in the elderly, it is worth mentioning the pilot study by Sayig-Keren et al., 82 where they aimed to modulate the activity of the prefrontal-parietal network via theta tACS (i.e., 6 Hz) and compared the outcomes with a classical tDCS protocol. Their results failed to prove significant differences between the theta-tACS and the tDCS protocols but highlighted the importance of a more thorough exploration of the correct tACS parameters in boosting dual tasking. Regarding this aspect, it is important to emphasize that research is going toward a more individualized approach for TES by studying protocols based on structural magnetic resonance (for exploring the anatomical features) and electroencephalography (to determine the correct frequencies of stimulation for tACS). 96 However, these methods are nowadays extremely expensive and need expertise that might be out of hand for clinicians.
Improvements in DT performance observed after DLPFC stimulation in the elderly and in PD patients may be attributed to the role of this area in the DT execution. Indeed, as largely described in the ‘Introduction’ section, DLPFC represents the connector between the striatum and frontal regions, and it is well-known that frontostriatal deficits may cause executive dysfunctions, that are frequently associated with impairments during DT walking in the elderly and in PD.14,97 A first possible mechanism underlying anodal tDCS effectiveness over DLPFC could be linked to the subthreshold increase of the resting membrane potential, resulting in temporary changes in cortical excitability, that serve as a booster for neuronal activity, with a consequent improvement in DT execution. 98 Moreover, modulating cortical excitability in individuals with PD through DLPFC tDCS may be associated with dopamine release, potentially accounting for the positive effects on motor function.99–102
Interestingly, one study in the elderly used a different stimulation approach combining the effects of a multisite lDLPFC and M1 and concomitant DT performance (online tDCS protocol). 80 The combination of the multisite tDCS and a complex walking (i.e., treadmill walking + virtual reality as in Mirelman et el., 2016 103 ) task resulted in the improvement of gait parameters. 80 As previously stated, activation of the DLPFC appears to be critical for improving dual tasking performance; however, the combination of TES with a functional task appears to have an even higher influence on performance. If we consider that in dual tasking there is a strong executive component controlled by the prefrontal cortex and motor areas, the association of multisite TES and a complex motor-cognitive task might result in a reduced DTC. In the context of concurrent complex tasks involving DT paired with TES, a possible explanation of the positive effects might rely on the concept “cells that fire together wire together”, 104 which in turn is based on the concept of synaptic plasticity first introduced by Donald Hebb. In the Hebbian theory, the repetitive stimulation of postsynaptic neurons by the hand of presynaptic neurons induces a synchronized firing activity, which results in an increase of the postsynaptic neuron efficacy and hence in long-lasting neuroplastic changes.105,106 Assuming these premises, it appears plausible that the association of (i) a non-invasive brain stimulation method such as TES on a cortical region involved in a task and (ii) the execution of the task might result in an augmented effect of the stimulation, which in turn reverberates its effects on behavior.
Limitations and future directions
Some considerations about the limitations of the studies included in this review are worthy to be made, to understand and speculate on what might be the direction of future studies.
First, while many DT paradigms were used, most of the studies assessed the DTC or a comparison of DT performance between the active and sham stimulation sessions, leading to a wide range of primary outcomes. For evaluating motor-cognitive interactions, SST was the most used secondary task whereas motor-motor interactions were assessed with obstacles negotiation or by carrying a cup of water. Unfortunately, the lack of uniformity between studies regarding the choice of DT paradigms and DT evaluation parameters limits us from drawing conclusive findings on this topic. An example of the importance of the selection of the most appropriate concurrent task in a DT paradigm comes from the two studies by Orcioli-Silva and Mishra and Trasher81,85 included in this review. In the first study, the authors employed a promising online (i.e., during a treadmill walk) multi-site (DLPFC + M1) paradigm. However, they selected a digit vigilance task as the secondary cognitive task, which may not have been sufficiently challenging or sensitive to detect changes in DT performance, particularly when combined with treadmill walking, which inherently induces a rhythmic gait pattern. As for the second study, the lack of significant effects found by Mishra and collaborators could potentially be due to the multiple repetitions of TUG within a short timeframe, which might have created a “ceiling effect”. In contrast, cognitive tasks changed each time (i.e., verbal fluency with different letters), probably leading patients to prioritize cognitive task performance, thus adopting the “posture second” prioritization strategy. 107
Related to outcome measures, not only DTC, that expresses the beneficial or detrimental effect on performance5–7 but also other outcomes that are generally evaluated in DT studies could be potential measures in future studies to evaluate the effect of TES on DT performance. We specifically refer to i) the Attention Allocation Index (AAI – Siu et al., 2007 108 ), which evaluates the amount of allocation of attention to a predetermined prioritized task, representing a measure of cognitive flexibility, and ii) the psychological refractory period, a measure of reaction time generally used in psychological studies. 109
Concerning the subjects’ number and characteristics, most of the studies lack an a priori power-based analysis that should be conducted to determine an adequate sample size for generalizing results. Researchers should also explore the effects of tDCS concerning specific subject characteristics, such as the presence of cognitive deficits (e.g., mild cognitive impairment) or, in the case of PD, the presence of FOG or different motor and non-motor phenotypes.
Regarding TES protocols, the methodological consistency of the included studies has been evaluated with the checklist by Buch and colleagues, resulting in a generally good description of the stimulation protocols. Anyway, some limitations emerged, especially in terms of stimulation parameters and location. First, most of the included studies did not report the current density, which makes it harder to understand whether the cortical areas underlying the electrodes were efficiently stimulated. It has been reported that the current density has to be above 0.017 mA/cm2 to modulate cortical activity, 110 otherwise, the effect of the stimulation may be variable and not strictly related to the protocol itself. Concerning target areas, positive effects have been retrieved for DLPFC, but also TES on the cerebellum demonstrated improvements in dual-tasking in PD. 87 Further research should explore the contribution of the cerebellum in DT and investigate it as a potential target for TES. Furthermore, a more focal stimulation that targets primarily the DLPFC should be used in future studies, by applying, for example, the 4 × 1 high-definition electrode montage, which consists of an ‘active’ central electrode (anode) over the target area and four ‘return’ electrode (cathodes) arranged circularly around the central one. 111 Although other montage options can be considered, 112 this montage has shown an increased spatial focality compared to the rectangular electrodes frequently used, resulting in a more precise stimulation and a lesser dispersion of electrical field in the surrounding areas.111,113 Furthermore, always in terms of TES protocols, the fact that we found only one study 82 investigating a protocol other than tDCS (without finding differences between theta-tACS and tDCS) highlights the need to properly understand the correct stimulation parameters for tACS (e.g., frequency and phase) to efficiently enhance neuronal activity in the target areas. A possible option in terms of a frequency band to explore might be the gamma band (i.e., > 30 Hz) which has demonstrated prokinetic 114 and neuroplastic effects 115 in healthy and PD, and the ability to entrain endogenous cortical oscillations in areas involved in higher cognitive tasks. 116
Finally, for PD patients, future research should consider using TES to improve DT performance during different disease stages and compare effects in various medication states (i.e., ON vs. OFF).
Conclusions
Succeeding in performing two tasks simultaneously is fundamental for conducting everyday life activities, and this ability is frequently impaired in the elderly and in patients with PD, causing secondary problems such as increased falls.
In this review, we summarized findings obtained in the last 10 years, regarding the effects of TES in improving DT execution in the elderly and in patients with PD. Although it appears clear that other studies are undoubtedly needed to prove the efficacy of TES in modifying dual tasking performance in these populations, we can say with a confident margin of certainty that the most promising area to enhance dual-tasking is DLPFC. Although TES might represent a potential tool for improving DT performance, a definite conclusion might not be drawn considering the high heterogeneity in the outcome measures (i.e., DTC vs. spatiotemporal parameters under DT performance) and in the stimulation protocols. Only by enhancing the quality of the studies involving TES, we could be able to maximize our capability to rule out confounding factors, understand the real contribution of specific cortical areas to a task, and hence implement stimulation protocols in rehabilitation programs for PD patients and the elderly.
Supplemental Material
sj-docx-1-pkn-10.1177_1877718X251327758 - Supplemental material for Recent advances of transcranial electrical stimulation in healthy aging and Parkinson’s disease: Effects on dual tasking
Supplemental material, sj-docx-1-pkn-10.1177_1877718X251327758 for Recent advances of transcranial electrical stimulation in healthy aging and Parkinson’s disease: Effects on dual tasking by Martina Putzolu, Alessandro Botta, Carola Cosentino, Susanna Mezzarobba, Gaia Bonassi, Elisa Ravizzotti, Sara Terranova, Giovanna Lagravinese, Elisa Pelosin and Laura Avanzino in Journal of Parkinson's Disease
Footnotes
Acknowledgments
The authors have no acknowledgments to report.
ORCID iDs
Ethical considerations
Not applicable.
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Work supported by #NEXTGENERATIONEU (NGEU) and funded by the Ministry of University and Research (MUR), National Recovery and Resilience Plan (NRRP), project MNESYS (PE0000006) – A Multiscale integrated approach to the study of the nervous system in health and disease (DN. 1553 11.10.2022)
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: Martina Putzolu reports financial support provided by the Ministry of University and Research (MUR).
Alessandro Botta reports a relationship with the EU Joint Programme for Neurodegenerative Disease Research that includes: employment.
Gaia Bonassi reports a relationship with the Ministry of University and Research (MUR) that includes: employment.
S. Terranova, C. Cosentino, S. Mezzarobba, and E. Ravizzotti, have nothing to declare.
L. Avanzino has received speaker honoraria from Zambon and Bial and received research supports (Grants) from the EU Joint Programme for Neurodegenerative Disease Research (JPND) (2022 call) and Michael J. Fox Foundation (Fall 2022 biomarkers to support therapeutic trials program). She received research support from the FRESCO Foundation and the Italian Ministry of Health (Ricerca Finalizzata 2022 and 5X1000).
E. Pelosin is part of an Advisory Board for the M.J. Fox Foundation. She has received grants from the Italian Ministry of University and Research (PRIN 2022) and Michael J. Fox Foundation (Fall 2022 biomarkers to support therapeutic trials program), and research supports from the Italian Ministry of Health (Ricerca Finalizzata 2022 and 5X1000).
Data availability
Data sharing is not applicable to this article as no datasets were generated or analyzed during this study.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
