Abstract
The accurate diagnosis of synucleinopathies—neurodegenerative diseases marked by misfolded α-synuclein protein aggregates, such as Parkinson's disease, dementia with Lewy bodies, and multiple system atrophy—remains a critical challenge. Conventional clinical criteria, frequently verified only through postmortem examination, results in diagnostic delays that impede timely intervention. Seeding amplification assay (SAA) has emerged as a promising diagnostic tool, offering high sensitivity for detecting α-synuclein aggregates even in early disease stages. While SAA enables early diagnosis by amplifying misfolded α-synuclein in biological samples, several barriers exist, including a lack of assay standardization, technical complexity, and difficulty differentiating among synucleinopathies. Additionally, the current SAA is primarily qualitative, limiting their ability to correlate with disease severity or progression. This review addresses these limitations by examining pre-analytical and analytical factors influencing SAA performance and exploring emerging quantitative approaches. Recent advancements include the integration of SAA with quantitative methodologies, which hold promise for enhanced diagnostic accuracy and clinical applicability. SAA's potential as a diagnostic and monitoring tool is significant and can be further improved by validation in longitudinal studies. The clinical implementation of SAA could revolutionize the early detection and management of synucleinopathies, ultimately improving patient outcomes through earlier diagnosis and tailored therapeutic strategies.
Plain language summary
Diagnosing Parkinson's disease, dementia with Lewy bodies, and multiple system atrophy can be challenging. These conditions, called synucleinopathies, are caused by harmful changes in a protein called α-synuclein that causes it to clump or aggregate. Doctors often rely on looking for symptoms of the disease to form a diagnosis, but confirmation usually happens only after death when the brain regions affected can be tested for the presence of α-synuclein aggregates. This results in challenges in providing timely treatment. A new lab test called the seeding amplification assay (SAA) shows promise for detecting changes in α-synuclein early, even before symptoms appear. SAA works by amplifying small amounts of this misfolded protein in samples from patients, making it easier to detect. However, the test has limitations. It is complex, not standardized across labs, and struggles to tell apart the different synucleinopathies. SAA also provides “yes” or “no” answers and cannot yet quantify the levels of the α-synuclein aggregates present and how severe or advanced the disease is. This review explores ways to improve SAA. It looks at how factors like sample handling and lab techniques affect results and highlights new methods that make SAA more accurate and quantitative. By combining SAA with technologies that can quantify protein levels more precisely, and studying SAA results of patients over time, the test could help doctors diagnose these diseases earlier and track their progression. Early diagnosis means patients could receive treatments tailored to their needs sooner, improving their quality of life.
Introduction
Neurological disorders represent a growing global health concern, particularly neurodegenerative diseases such as Parkinson's disease (PD), dementia with Lewy bodies (DLB), and multiple system atrophy (MSA) collectively known as synucleinopathies. 1 These diseases share a common pathological hallmark: the accumulation of misfolded α-synuclein protein in the affected regions of the brain, forming aggregates that disrupt neuronal and glial functions and contribute to disease progression. Synucleinopathies can present with a broad range of symptoms, from motor impairments in PD to cognitive and autonomic dysfunctions in DLB and MSA, making early and accurate diagnosis critical yet challenging.2–4
Traditional diagnostic methods rely primarily on clinical criteria, often confirmed postmortem, which delays diagnosis and limits the potential for early intervention. PD diagnosis centers on motor symptoms like bradykinesia, supported by response to dopamine therapy, and is guided by the UK Parkinson's Disease Society Brain Bank Criteria and tools like the MDS-UPDRS.5,6 DLB, in contrast, often presents with early cognitive decline, fluctuating cognition, visual hallucinations, and REM sleep behavior disorder, with parkinsonism following later; sensitivity to antipsychotics and DaT-SPECT imaging can aid differentiation from PD. 7 MSA is divided into MSA-P (parkinsonian) and MSA-C (cerebellar) based on symptomatology, with rapid progression, poor response to dopamine, and autonomic failure as key features. 8 Despite these criteria, symptoms overlap complicates accurate diagnosis, and clinical diagnosis alone is insufficient because these symptoms typically emerge only after substantial neurodegeneration has already occurred. By the time motor and cognitive symptoms become evident, significant neuronal loss and α-synuclein accumulation have often progressed, limiting therapeutic efficacy. 4 This delay highlights the need for early biomarkers that can identify pathology at its onset, ideally before major degeneration sets in. Biomarkers with high sensitivity and specificity are crucial for enabling timely intervention and improving the prognosis of patients with synucleinopathies.
Over recent years, protein-based biomarkers, particularly α-synuclein, have gained attention for their potential to directly reflect the pathological processes underlying synucleinopathies. While earlier assays, such as enzyme-linked immunosorbent assays (ELISA), were employed to detect α-synuclein in biofluids, they lacked the sensitivity required for early disease detection.9–12 Immunohistochemistry, though invaluable for postmortem confirmation, does not facilitate ante-mortem diagnosis. Advances in molecular diagnostics have introduced more sensitive techniques with seeding amplification assay (SAA) emerging as a transformative tool to detect α-synuclein aggregates. By exploiting the self-propagating nature of α-synuclein aggregates, SAA amplify minute amounts of misfolded α-synuclein present in biological samples, allowing for highly sensitive detection, even in early disease stages, as well as in biofluids such cerebrospinal fluid (CSF) where they are present in extremely low concentrations.13,14 This high sensitivity positions SAA as a potentially valuable biomarker for early detection and differentiation among synucleinopathies.
From its inception SAA, then known as RT-QuIC, by Atarashi et al. in 2011 has been marked by significant milestones, each contributing to the current understanding of misfolded proteins in neurodegenerative diseases 15 and making it one of the most studied diagnostic methods for synucleinopathies to date. The principle of the assay relies on amplifying pathological “misfolded” α-synuclein seeds, which induce the aggregation of recombinant α-synuclein under optimized conditions, mimicking disease-specific protein misfolding. The α-synuclein seeds present in the samples are elongated using recombinant α-synuclein, which increases the overall number of seeds in the sample. Simultaneously, larger aggregates are fragmented into smaller, self-propagating seeds continuing the amplification process. This amplification enables the detection of seeds using specific fluorescent dyes, such as thioflavin T, which bind selectively to amyloid structures. Theoretically, a higher number of seeds leads to quicker aggregation. 16 The assay generates a fluorescence signal as its readout, correlating with the formation of amyloid fibrils over time. It measures the presence and quantity of seeding-competent α-synuclein aggregates, providing insights into the pathological burden. A positive result is determined when the fluorescence signal exceeds a predefined threshold within a set timeframe, while negative samples remain below this threshold. Assay performance is evaluated through metrics like sensitivity, specificity, and reproducibility, with variability and inconsistencies stemming from factors such as sample handling, substrate quality, and inter-laboratory differences. 17
Extensive reviews and meta-analyses have thoroughly evaluated SAA's sensitivity, specificity, and diagnostic performance across biofluids, along with discussions on their technical adaptations and clinical applications.18,19 A recent systematic review and meta-analysis by Grossauer et al. in 2023 assessed the pooled diagnostic accuracy of SAA using CSF samples in differentiating synucleinopathies from controls to be at 88% sensitivity and 95% specificity. 20 These studies collectively highlight the advancements SAA have brought to the field and their ongoing optimization for non-invasive samples like blood and skin biopsies. However, significant challenges remain in translating SAA from a research tool to a standardized clinical diagnostic. Challenges in assay standardization and difficulty in distinguishing between synucleinopathies limit SAA's diagnostic reach. Additionally, the qualitative nature of current SAA restricts its ability to correlate with clinical features, such as disease severity or progression, further complicating its use as a comprehensive diagnostic tool. This review addresses the primary limitations of SAA in clinical practice and research. By examining these challenges, we aim to clarify the current capabilities of SAA and propose future directions to enhance its diagnostic accuracy and utility across diverse clinical contexts.
Current limitations and challenges of SAA in clinical and research applications
The advantages of SAA are numerous. They provide a higher sensitivity compared to other diagnostic methods (up to atto-gram levels) and can detect misfolded proteins at early disease stages, potentially aiding in earlier diagnosis and intervention.21,22 The applicability of the technique with a variation of biological samples also presents a valued advantage.21,23,24 However, despite their potential, SAA is not without limitations. For one, the requirement for specialized equipment and technical expertise can make it less accessible, and variations in assay protocols can lead to inconsistencies in results. Further intertwined are challenges of specificity, sensitivity, applicability across diverse PD subgroups, and broader clinical implementation. Each limitation underscores an opportunity towards refining, optimizing, and potentially transforming concept of SAA from a specialized, research-centric technique to an easily accessible, clinically integral part of PD diagnostics and management.
Standardization and reproducibility across laboratories
Variations in α-synuclein SAA protocols have led to inconsistencies in results and interpretation, such as differing diagnostic sensitivities and specificities reported for PD and DLB, challenges in distinguishing between synucleinopathies like multiple system atrophy and PD, and variability in assay performance across laboratories. These variations could be introduced due to technical variabilities such as differences in the preparation of recombinant α-synuclein, the composition of reaction mixtures, sample processing methods, and experimental settings and detection parameters.25–27 Such inconsistencies can lead to variability across plates, batches, and laboratories, limiting the consistency and broader applicability of these techniques due to differences in recombinant protein preparation or reaction conditions.19,25 These inconsistencies emphasize the need for standardized practices to improve reproducibility across laboratories.
Protocols for α-synuclein SAA differ based on several factors, including reaction buffer pH, incubation times, temperature, shaking speed, the presence of specific detergents such as sodium lauryl sulfate (SDS), and the use of beads, all of which can significantly influence the assay's sensitivity and specificity.16,28–31 Bellomo and colleagues underscored this variability by examining the effects of monomeric α-synuclein concentration, glass bead presence, pH, human CSF, and detergents on α-synuclein aggregation kinetics. 25 Key findings indicated that adding glass beads accelerated aggregation and improved consistency across samples, likely by promoting more effective seeding through surface interactions, while lower monomer concentrations enhanced sensitivity by extending the differentiation between seeded and unseeded samples. The study also found that lower pH levels promoted faster aggregation by reducing electrostatic repulsion, and that CSF addition delayed aggregation, likely due to interference from CSF proteins. The inclusion of SDS dramatically increased aggregation speed but heightened the risk of spontaneous aggregation, potentially affecting assay specificity. These findings highlight the importance of precise parameter control to ensure reliable diagnostic results across different laboratory settings. 25
Building on Bellomo's findings, the study by Mammana et al. explored additional preanalytical factors, focusing on blood contamination, freeze-thaw cycles, and detergent choice. 26 Their analysis showed that even minimal blood contamination (≥0.01%) could impact aggregation kinetics, often leading to inconsistent results, underscoring the importance of rigorous sample preparation. Variability in freeze-thaw cycles was also found to compromise α-synuclein stability, affecting reproducibility; standardizing freeze-thaw protocols could mitigate these impacts. Additionally, the presence of non-ionic detergents like Tween 20 and Triton-X was shown to increase aggregation, leading authors to recommend excluding these additives to maintain assay integrity. Further, the quality and concentration of α-synuclein monomers, the substrate in the reaction, directly influence the kinetics of aggregation, with optimal conditions leading to faster and more robust amplification of α-synuclein aggregates. Variability in the behavior of monomeric recombinant protein in SAA represents a significant limitation. This variability arises from several factors, including differences in preparation and purification methods, the potential presence of impurities, and the stability of the protein during storage and handling as highlighted in an extensive study by Janarthanam and Clabaugh et al. 27 They showed that factors such as ionic strength and pH can markedly alter the aggregation kinetics and consistency of the assay. Additionally, inconsistencies in protein storage conditions or repeated freeze-thaw cycles may lead to partial aggregation of the monomers, further compounding variability and reducing assay reproducibility. The presence of impurities or inappropriate substrate forms such as trace levels of aggregated, truncated α-synuclein or unrelated proteins in the starting material, could act as unintentional seed, inhibitors or competitors in the aggregation process hindering the assay's performance and leading to unreliable results.26,27,32 Taken together, the studies illustrate the substantial role preanalytical and analytical factors play in SAA performance, advocating for standardized protocols that control for these variables—such as buffer composition, sample handling, α-synuclein monomer substrate batches and reaction additives. Such standardization is essential to ensure diagnostic reliability, facilitate comparability across studies, and support the clinical integration of SAA as diagnostic tools for synucleinopathies.
SAA is technically demanding, often requires specialized equipment and highly trained personnel to ensure accurate execution. The technical complexity and resource intensiveness of SAA can limit their adoption, particularly in clinical laboratories with limited equipment or personnel. For smaller or resource-limited labs, these technical requirements make it difficult to adopt SAA as part of routine diagnostics. The need for skilled operators also presents a challenge, as these assays require precise handling, and minor procedural deviations can alter assay sensitivity or lead to false positives. In the clinical setting, standard diagnostic labs may lack the personnel training or equipment needed to perform SAA, limiting their integration into mainstream diagnostics.
Differential diagnosis of synucleinopathies using SAA
Majority of studies using SAA for diagnosing different synucleinopathies have shown a high degree of sensitivity and specificity determining the clear presence or absence of α-synuclein seeds in the different biological samples of individuals with evident diseases and in prodromal stages, where it is assumed that α-synuclein pathology is less. The concept that diverse structural forms of the same misfolded protein could explain the variability in clinical presentations across a range of synucleinopathies is supported by a body of research.33–35 SAA measures the critical trait of proteopathic seeding and offers a pathway for confirming the presence of proteopathic seeds directly in accessible biological fluids or tissues. Utilizing parameters like kinetics and peak fluorescence levels, it is feasible to distinguish between different strains of the same protein, as illustrated in studies on prion disease.36,37 However, the interpretation of SAA results can be complicated by variations between different studies, as well as the impact of reaction conditions and the recombinant substrates employed, thereby limiting the current applicability of these techniques.
Thioflavin-T (Th-T) is a benzothiazole dye that is commonly used to detect amyloid fibrils, a form of protein aggregation. It binds to beta-sheet rich structures, leading to an increase in fluorescence, and is often used in assays to detect and quantify protein aggregation.30,38 However, due to the structural differences in α-synuclein aggregates between MSA, DLB and PD, Th-T-based assays might not be as effective in all types of α-synuclein strains. 35
MSA is known to be associated with the formation of glial cytoplasmic inclusions, predominantly comprised of α-synuclein. These inclusions are believed to have a different conformation compared to the Lewy bodies (LBs) found in PD, which also consist of aggregated α-synuclein. In a study by Schweighauser et al. (2020), it was shown that α-synuclein aggregates derived from MSA brain tissue were less efficiently seeded and had different characteristics compared to those derived from PD brain tissue. 35 Furthermore, the MSA-derived aggregates showed less Th-T fluorescence, indicating that they might have a different amyloid structure, which could lead to them being missed or under-detected by Th-T-based assays. In response to this, a study evaluated 168 different reaction buffers with various pH levels and salts, using characterized brain homogenates from MSA and PD patients, to improve the reliability of the assay. 39 These findings highlight the complexity of α-synuclein pathology in synucleinopathies and underscore the necessity for further research and development of more sensitive and specific assays for the detection and differentiation of α-synuclein strains associated with different diseases.
The diagnostic landscape for neurodegenerative diseases is further complicated by the prevalence of co-pathologies. Studies have demonstrated that LBs pathology, a hallmark of α-synuclein aggregation, is frequently found in patients with Alzheimer's disease and other types of dementia, even in cases without clinical symptoms of PD.40–43 These co-pathologies can distort the interpretation of SAA results across neurodegenerative disorders. Positive results yielded are based solely on the presence of α-synuclein and cannot be interpreted to be a diagnosis of one particular neurodegenerative disease. Recent studies have noted that in populations with high co-morbidity rates, such as elderly individuals, SAA positivity may not reliably correlate with PD-specific symptoms.44,45 This reinforces the need for SAA to be interpreted in conjunction with clinical criteria and additional biomarkers, particularly for patients with cognitive impairment or non-classical motor presentations. The Queen Square Brain Bank (QSBB) criteria and Movement Disorder Society (MDS) diagnostic guidelines emphasize the importance of observing symptoms over time, such as persistent motor asymmetry or response to levodopa, in order to improve diagnostic specificity and reduce the likelihood of false positives from incidental pathologies.6,46
Lack of quantification of α-synuclein seeds using SAA
SAA has primarily been utilized in a qualitative manner to detect the presence of misfolded α-synuclein aggregates generating binary results of positive or negative for α-synuclein seeds. The assay typically measures the time it takes for a reaction to occur, lag phase, and the intensity of the fluorescent signal generated by the binding of the dye Th-T to β-sheet-rich aggregates, which provides an indirect measure of the amount of α-synuclein seeds present in the sample.30,38 Quantifying α-synuclein seeds in biological samples would provide valuable and accurate insights for the diagnosis and monitoring of synucleinopathies.
Efforts to quantitate α-synuclein seeds have involved modifications and innovations in assay protocols. One prevalent method for quantification in SAA is kinetic analysis, where the progression of fibril formation is tracked over time. By evaluating the time required to reach a specific fluorescence threshold (time-to-threshold, Ttt), researchers can infer the quantity of α-synuclein seeds present; shorter Ttt values are indicative of higher seed concentrations. 47 This method was originally developed for quantifying prion proteins and has been adapted for use in amyloid SAA. 15
Building on this, the End-Point Quaking-Induced Conversion (EP-QuIC) method presents an alternative approach, wherein samples undergo a fixed incubation period, followed by fluorescence intensity measurement. The seeding activity is then estimated based on the end-point fluorescence values, providing a snapshot of the α-synuclein seeds present. This method has proven its efficacy in differentiating samples with varying concentrations of prion seeds and has been adapted for α-synuclein quantification. 48
One approach has been to utilize standard curves generated from synthetic α-synuclein fibrils, which allows for the conversion of assay readouts into an approximate concentration of α-synuclein seeds. 20 However, this method assumes that the seeding activity of synthetic fibrils is comparable to that of fibrils present in biological samples, which may not always be the case due to potential differences in their structural properties. Additionally, the presence of inhibitors or enhancers of seeding activity in biological samples can further complicate the interpretation of results.
In efforts to provide a more direct method of quantifying α-synuclein seeds, Majbour et al. established a novel approach combining SAA and oligomers-specific ELISA to provide a measurable test readout that could more accurately and robustly reflect disease severity in patients with PD (Figure 1). 49 Results of their work had shown successful direct quantification seeded α-synuclein in brain homogenate and CSF samples by testing SAA products in oligomers-specific ELISA. Quantified α-synuclein seeds were capable of discriminating between PD and controls at an early timepoint to the SAA alone and were also found to significantly correlate with PD motor severity scores, MDS-UPDRS III and Hoehn and Yahr (H&Y), in two independent CSF cohorts. While this robustly quantified SAA products from PD, DLB with high specificity and sensitivity, the methodological process is complex due to the running of two separate assays to generate results. Development of a more simplified approach streamlining this methodology into a single assay are currently ongoing by the same group. 50
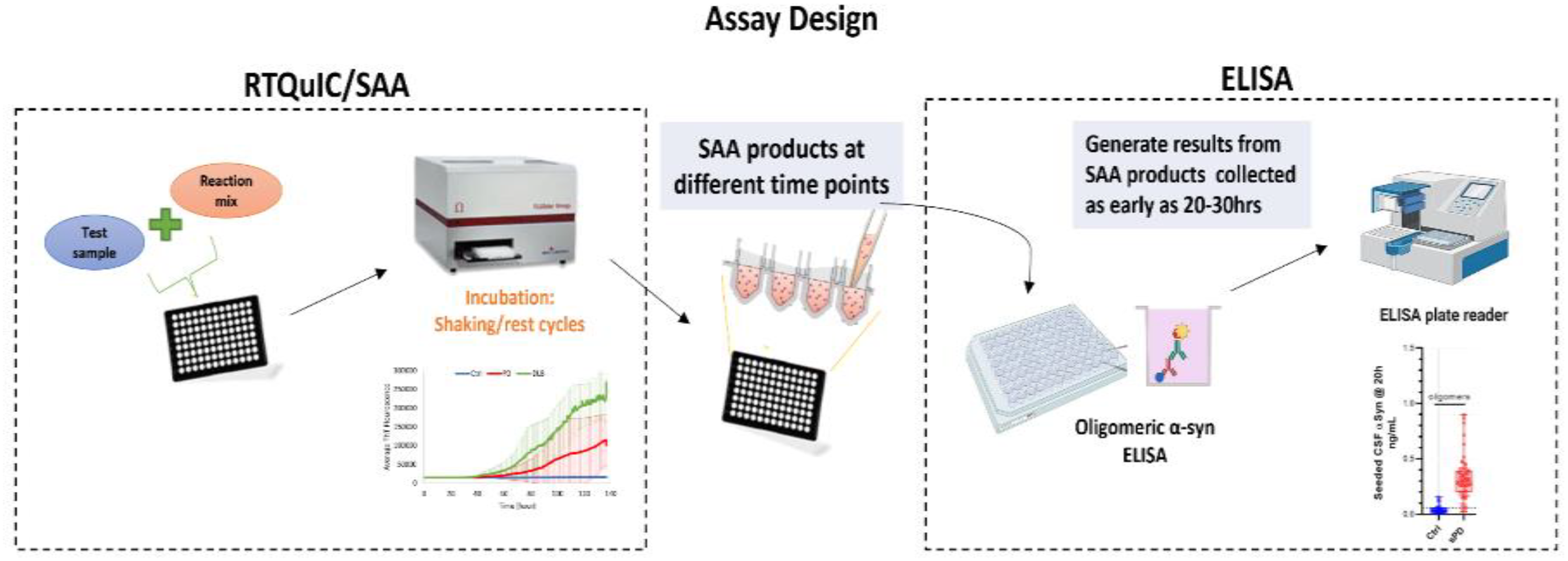
Assay design of novel approach combining two methodologies, SAA and ELISA, for quantifying α-synuclein seeds.
Real-Time Fast Amyloid Seeding and Translocation (RT-FAST) is a newer method that leverages nanopipette technology for single-molecule α-synuclein quantification. 51 By combining rapid seed amplification with real-time translocation, RT-FAST can detect α-synuclein seeds at concentrations as low as 2 pM, enabling detailed monitoring of seed dynamic. RT-FAST's high sensitivity makes it ideal for tracking changes in α-synuclein aggregates in patient samples over time, advancing its potential use in monitoring disease progression and treatment response. Recent work by Gilboa et al. (2024) introduced compartmentalized digital SAA, where α-synuclein aggregates are isolated into microwells, droplets, or hydrogel microcapsules. 52 This method allows for precise, single-aggregate quantification by capturing seeds individually and analyzing aggregate characteristics. The study enhanced sensitivity further by pre-capturing aggregates on antibody-coated magnetic beads, concentrating pathological α-synuclein seeds before quantification. This compartmentalization strategy quantifies α-synuclein more accurately. It also allows for detailed morphological analysis. This advancement could enhance the assay's diagnostic and therapeutic applications, particularly in clinical contexts that require high sensitivity.51,52
Despite these promising advancements in quantifying α-synuclein seeds, further validation is necessary. This includes kinetic analyses, hybrid assays, and digital methods like RT-FAST and compartmentalized SAA. We need to ensure these approaches reliably capture disease-relevant aggregates across diverse patient populations. Continued research and refinement are crucial to simplify these methods for routine clinical use. It is essential that quantitative SAA provide consistent and reproducible measures of α-synuclein pathology; this will make them suitable for diagnosis, monitoring, and treatment evaluation in synucleinopathies.
Lack of correlation between SAA results to clinical variables
In the process of diagnosing synucleinopathies and other neurodegenerative diseases, evaluating clinical assessments, including motor and cognitive scales and their correlation to SAA kinetic parameters is crucial. Among these, the MDS-UPDRS-III motor scale, cognitive tests like the Mini-Mental State Examination score (MMSE) and the Montreal Cognitive Assessment (MoCA), are frequently employed.28,53 While SAA has shown exceptional proficiency in distinguishing synucleinopathy cases from controls based on various kinetic parameters, the correlation of these parameters with the vast array of clinical assessments has proven to be a challenging endeavor.
Utilizing SAA kinetic parameters such as maximum Th-T fluorescence (Fmax), time to Fmax (T50), lag phase (Tlag), protein aggregation rate (PAR), and area under the curve (AUC), α-synuclein pathology have been identified in different biological samples.21,24,54–56 However, despite these advancements, the relationship between these kinetic parameters and clinical measures has remained elusive, with most studies reporting no significant correlations.
In an SAA study, despite showing high sensitivity identifying PD from controls, parameters such as Fmax and T50 did not correlate with any clinical parameter. 47 Another extensive CSF SAA study involving patients with Mild Cognitive Impairment with LBs (MCI-LBs) echoed these findings, as it did not find any substantial connections between SAA parameters and a range of demographic and clinical evaluations. 57
For clinical assays, serial dilution of biological samples isn’t a common practice, but it can be employed to determine the SD50 value, which is the seeding dose required for 50% of the replicates to exhibit positivity, calculated using the Spearman-Karber method. 28 In a couple of studies, SAA SD50 values exhibited correlation with disease duration, and higher SD50 values were associated with increased pathological α-synuclein deposits in postmortem autopsy analyses.23,47,58 Yet, these correlations have not been consistently found across other disease features, and the method itself is time-consuming, rendering it impractical for routine application.
Another recent study on the PPMI cohort reported moderate correlations of SAA parameters Fmax, Tlag, and AUC with disease duration and UPDRS-III scores. 47 Furthermore, a larger study with the same cohort did not report correlation between SAA metrics and clinical evaluations but instead focused on frequency SAA status (negative or positive) in individuals in the prodromal cases of PD (exhibiting RBD and hyposmia) as well as in symptomatic and asymptomatic carriers of LRRK2 mutations linked to the disease. 59
In the context of clinical trials, not only is diagnostic accuracy required but also the ability to track disease progression and response to therapies. 18 While SAA has shown promise in differentiating PD from controls, its application in clinical trials faces significant challenges. The lack of correlations between SAA kinetic parameters and clinical variables poses a problem for clinical trials, which often rely on these clinical variables as primary or secondary endpoints. For instance, while SAA can identify the presence of synucleinopathies, it may not effectively measure the severity or progression of motor symptoms as assessed by the MDS-UPDRS-III scale or cognitive decline as indicated by MMSE or MoCA scores. 60
Correlating SAA parameters with various clinical measures has been arduous, with mostly modest successes. While slight correlations have been observed with age and/or disease duration parameters in some studies, it becomes apparent that the development of more precise seed quantitation methodology may open the door for more accurate longitudinal assessments of synucleinopathies, fostering stronger correlations with clinical evaluations.
Longitudinal studies using SAA
Most existing SAA studies are cross-sectional, providing a single snapshot of α-synuclein pathology rather than capturing changes over time. This limits the interpretation of SAA results, as synucleinopathies like PD show considerable variability in clinical presentation across disease stages. Moreover, early PD diagnosis is challenging, with approximately 10% of initial diagnoses later reassigned to other conditions, such as essential tremor, progressive supranuclear palsy, or MSA.20,61 These conditions often mimic PD symptoms, emphasizing the need for diagnostic criteria that incorporate longitudinal clinical observations to improve diagnostic precision.
Longitudinal cohort studies are essential for understanding the progression of PD and assessing treatment effectiveness. However, SAA is underutilized in such studies, with limited data available that only captures binary outcomes and lacks the quantitative detail necessary to track disease progression accurately. This scarcity of longitudinal data restricts the potential of SAA as tools for monitoring PD and synucleinopathy progression, evaluating patient response to treatment, and better understanding the natural history of these diseases. 22 Although the FDA has encouraged scientists to utilize the α-synuclein SAA biomarker in their research and clinical trial efforts, 62 its value for routine clinical diagnostics requires further evidence from long-term studies.
Research using SAA to detect α-synuclein aggregates in REM sleep behavior disorder (RBD) and hyposmia patients that are prodromal to synucleinopathies has shown promising results, with studies demonstrating high sensitivity and specificity for α-synuclein detection in these conditions.32,63 These findings highlight SAA's potential as a valuable tool for tracking disease progression in RBD, which often precedes the onset of synucleinopathies like PD and DLB. Given these promising results, further exploration of SAA in RBD offers a productive avenue for understanding longitudinal disease progression and potential early intervention strategies.
Developing quantitative SAA methods could significantly enhance their utility in longitudinal research, offering detailed insights into how α-synuclein seed levels and characteristics change over time and correlate with disease progression and symptoms. This would mark a significant advancement, transitioning SAA from supportive diagnostic tools to central instruments for managing and studying PD and related disorders. Furthermore, longitudinal data is critical in clinical trials, where the effectiveness of interventions depends on detecting subtle changes over time—changes that current SAA methodologies may not yet be sensitive enough to identify reliably. 18
Given the progressive and variable course of PD, establishing the full diagnostic value of SAA will require longitudinal follow-up of both positive and negative assay results, ideally including postmortem verification when possible. Long-term observation of SAA changes could reveal how α-synuclein aggregation corresponds to disease progression, enhancing our ability to identify individuals at risk of developing PD or related synucleinopathies and enabling earlier, more targeted interventions.
Emerging solutions and future research directions
Addressing the limitations and operational challenges in SAA requires innovative solutions and further research into standardization, accessibility, and assay adaptability. Key areas of focus include efforts to standardize assay protocols across labs, adapt SAA for strain-specific detection, expand non-invasive sampling options, and incorporate advanced molecular detection tools.18,20,25,26 These advancements have the potential to establish α-synuclein SAA as reliable and accessible diagnostic tools for synucleinopathies.
The variability in SAA outcomes underscores the critical need for standardized protocols and reagents to ensure reliability and comparability across studies. Differences in preparation, handling, and quality of reagents, such as monomeric α-synuclein, can significantly affect assay performance. Implementing rigorous quality control measures for reagents, such as validating purification methods and establishing centralized production standards, would minimize variability.25–27 Standardized protocols for assay conditions, including buffer composition and incubation parameters, are also essential to enhance reproducibility. These steps will pave the way for greater consistency and reliability, supporting the broader clinical application of SAAs.
The advent of SAA has revolutionized the diagnostic landscape for PD and other synucleinopathies by providing a highly sensitive method for detecting misfolded α-synuclein aggregates. However, instances of SAA-negative results in clinically diagnosed PD cases have raised questions about the assay's validity. For example, the largest SAA study to date reported that ∼12% of clinically defined PD patients did not exhibit detectable seeding activity, highlighting the pathological heterogeneity of the disease. 59 Such findings suggest that not all PD cases are associated with α-syn pathology detectable by SAA, underscoring the importance of interpreting assay results alongside other diagnostic tools. Future advancements in this area should focus on enhancing diagnostic criteria to more effectively distinguish between different synucleinopathies, such as PD and MSA, 38 by combining SAA findings with additional biomarkers and imaging techniques. Exploring alternative biomarkers, such as tau and amyloid plaque pathology or neuroinflammatory markers, could enhance diagnostic specificity and provide a more comprehensive understanding of disease mechanisms.
Post-translational modifications (PTMs) of α-synuclein, such as phosphorylation, truncation, acetylation, and ubiquitination, play a critical role in modulating its aggregation, seeding activity, and toxicity, yet they have not been widely incorporated into seed SAA. The use of monomeric recombinant α-synuclein in its unmodified wild-type form may limit the ability of SAAs to fully replicate the pathological characteristics of α-synuclein observed in disease. For example, Ghanem et al. (2022) demonstrated that phosphorylated α-synuclein fibrils exhibited decreased seeding activity compared to their unmodified counterparts in RT-QuIC assays, highlighting how phosphorylation modulates seeding efficiency and assay sensitivity. 64 Similarly, Poggiolini et al. (2021) found that using truncated α-synuclein substrates in RT-QuIC assays improved the differentiation between PD and dementia with Lewy bodies, further emphasizing the importance of PTMs in mimicking disease-relevant α-synuclein species. 65 Incorporating PTM-specific α-synuclein substrates into SAAs could enhance their biological relevance by more closely reflecting the pathological forms observed in synucleinopathies. This approach could also reduce variability by standardizing the use of modified substrates, thereby improving the diagnostic and mechanistic utility of SAAs.
The expansion of sample types for SAA, including non-invasive methods like blood, olfactory mucosa, and saliva, presents promising avenues for research.21,23,66 In blood, serum SAA was developed including an immunoprecipitation step that allowed for effective detection of pathogenic α-synuclein aggregates that distinguished synucleinopathies from controls. 67 Another method also developed involved isolating neuronal extracellular vesicles derived from plasma. This method has demonstrated potential for identifying misfolded α-synuclein years before the onset of clinical symptoms and predicting PD risk.68,69 However, challenges such as the known matrix effect present in blood samples, interference from substances like albumin, and the complexity of protocols, including extracellular vesicle isolation and immunoprecipitation, limit the scalability of these methods.70,71 Addressing these technical barriers and simplifying workflows will be critical for enabling widespread application of blood-based diagnostics. Skin biopsies have shown comparable diagnostic accuracy to CSF-based methods also detecting α-synuclein pathology in prodromal cases. 63 However, variability in α-synuclein levels across different skin regions, potential sample degradation, and the need for standardized protocols pose challenges to their broader application.23,54,63,72 Additionally, large-scale validation across diverse cohorts is essential to confirm their diagnostic utility. Together, blood- and skin-based α-synuclein detection methods represent promising, less invasive diagnostic tools that could complement existing approaches. By addressing the current limitations and optimizing protocols, these methods have the potential to improve accessibility and enable earlier detection of synucleinopathies, advancing both clinical care and research.
In an attempt to improve the sensitivity and specificity of SAA, two research groups have suggested a biological staging system for PD that incorporates a combination of various biological markers. 17 The proposals for diagnosing and classifying PD include detecting α-synuclein seeds in CSF using SAA as the S marker, along with imaging diagnostics like 123I-FP-CIT PET and MRI neuromelanin as markers for dopaminergic neurodegeneration (N(D) marker). Genetic markers such as SNCA, LRRK2, and GBA are also used alongside clinical symptoms for diagnosis. In a recent personal view, Höglinger et al. introduced the SynNeurGe classification system, which combines S, G, and N markers with clinical symptoms (C marker) to categorize PD. 73 Simuni et al.74,75 proposed the neuronal α-synucleinopathy integrated staging system, which considers genetic background and integrates S markers, D markers, and clinical symptoms for staging. In this system, patients with a fully penetrant SNCA variant but no S, N, or clinical symptoms are classified as stage 0, while the presence of an S marker advances them to stage 1, with further stages determined by D markers and clinical symptoms. Additionally, individuals at genetic risk who have not developed the disease are classified into low-risk and high-risk groups based on age and genetic predisposition.
Both the SynNeurGe classification and the Neuronal α-synuclein Disease Integrated Staging System (NSD-ISS) represent significant advancements in the understanding and diagnosis of PD, albeit with distinct approaches and implications. The SynNeurGe classification offers a more comprehensive framework that integrates multiple biological components, including the use of sensitive SAA to detect α-synuclein pathology. This integration not only enhances diagnostic accuracy but also significantly improves the sensitivity of identifying early-stage disease, allowing for timely interventions. In contrast, the NSD-ISS focuses on a biological definition centered around α-synuclein pathology, aiming to streamline therapeutic interventions primarily for sporadic cases and early stages of the disease. However, its limitations in addressing the complexity of genetic forms and the broader spectrum of parkinsonian disorders may restrict its applicability. By acknowledging the heterogeneity of PD and incorporating evolving biomarkers, the SynNeurGe system holds the potential to advance both clinical practice and research, paving the way for targeted therapies and improved patient outcomes. Together, these systems underscore the importance of a biological approach in the ongoing quest to understand and effectively treat PD and other synucleinopathies.
While SAA has primarily been used qualitatively to detect misfolded α-synuclein aggregates, efforts to quantify these seeds through kinetic analysis and the EP-QuIC method have shown promise.16,48 To advance this field, researchers should prioritize the development of standard calibration curves using aggregates that closely resemble endogenous α-synuclein seeds, which would enhance the quantitative accuracy of the assay. This would allow for a more precise estimation of α-synuclein aggregate levels, facilitating better diagnosis and monitoring of synucleinopathies. This would be particularly beneficial in longitudinal studies which are essential for understanding the dynamics of α-synuclein aggregation, particularly in at-risk populations, to identify early intervention points. 18 Additionally, further innovations in assay protocols could be explored to improve the sensitivity and specificity of the quantification process. Investigating the structural properties of synthetic versus biological fibrils will also be crucial to ensure that the seeding activity of synthetic fibrils is representative of that in biological samples.
Moreover, quantifying α-synuclein levels can serve as a reliable biomarker for monitoring disease progression and treatment response. This allows for the establishment of clear endpoints in clinical trials, enabling researchers to assess the efficacy of new therapies more effectively. A quantitative approach can help in the selection of appropriate dosing regimens and treatment durations by correlating α-synuclein levels with clinical outcomes. This data-driven strategy can lead to more personalized treatment plans, optimizing therapeutic efficacy while minimizing potential side effects. Finally, the ability to quantify α-synuclein seeds can enhance the reproducibility and reliability of trial results, as it provides a standardized method for assessing treatment effects across different study sites. This consistency is vital for regulatory approval and can ultimately accelerate the translation of research findings into clinical practice. Overall, adopting a quantitative approach in SAA can significantly improve the design, execution, and outcomes of clinical trials targeting synucleinopathies. Lastly, by addressing the current limitations, SAA has the potential to significantly improve early detection and differentiation of neurodegenerative diseases, ultimately leading to better patient outcomes and targeted therapeutic strategies. It is noteworthy that fostering collaboration across disciplines—between neurologists, pathologists, and researchers—will be essential to drive innovation and breakthroughs in understanding and diagnosing synucleinopathies. By pursuing these directions, the field can significantly enhance diagnostic capabilities for synucleinopathies and improve patient outcomes through earlier intervention and tailored treatment strategies.
Conclusion
SAA represents a transformative step in the early detection of synucleinopathies, with high sensitivity for identifying α-synuclein aggregates central to PD, MSA, and DLB. However, key limitations—including standardization needs, reproducibility challenges, and difficulty differentiating among synucleinopathies—necessitate further advancements. Future directions in quantitative methods, non-invasive sampling, and multi-biomarker integration hold promise to enhance SAA's clinical applicability. Prioritizing cross-disciplinary collaboration and longitudinal studies will be critical to transitioning α-synuclein SAA from research tools to routine diagnostics, facilitating earlier diagnosis, improved disease differentiation, and tailored treatments. These improvements could significantly impact patient outcomes and advancing the management of neurodegenerative diseases.
Footnotes
Acknowledgments
The authors have no acknowledgments to report.
Ethical considerations
Not applicable
Consent to participate
Not applicable
Consent for publication
Not applicable
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: Professor El-Agnaf's laboratory is funded by Qatar Biomedical Research Institute Internal Grant (no.: VR-98) and Qatar Research, Development and Innovation Council -Academic Research Grant (no.: ARG01-0514-230148). Dr. El-Agnaf is a consultant for Roche Pharmaceuticals and the founder of the startup company QABY BIOTECH.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability
Not applicable
