Abstract
Background
Parkinson's disease is a progressive neurodegenerative disorder. Awareness and the evidence supporting the merits of palliative care (PC) approaches to people with Parkinson's disease (PwP) are increasing.
Objective
This review aimed to address four key questions related to PC for PwPs and their caregivers: i) What are the indicators for timely access to PC? ii) When should PC be introduced? iii) What are the current care models for providing PC? iv) What are the barriers and facilitators at the organizational level?
Methods
A systematic literature search was conducted in PubMed, CINAHL, Cochrane, EMBASE, and MEDLINE (2006–2024). Six reviewers independently screened abstracts and full texts, and thematic synthesis was applied to develop analytical themes. Reporting followed PRISMA guidelines.
Results
Out of 894 studies, 70 were included. PwPs were infrequently referred to PC services, and while several referral criteria were identified, no consensus emerged. Barriers to accessing PC included insufficient information, inadequate education, difficulties determining referral timing, limited home-based care options, inconsistent provider support, and disparities linked to socioeconomic and cultural factors. Facilitators included improved care coordination and education for PwPs, caregivers, and healthcare providers. Effective PC models were identified, including home-based, hospital-based, and community-based approaches, which improved quality of life and reduced healthcare costs.
Conclusions
Establishing consensus on referral timing and criteria is essential for integrating PC into Parkinson's disease care. Overcoming barriers requires enhanced education, better care coordination, and targeted interventions to address disparities, ensuring comprehensive, patient-centred care for PwPs and their caregivers.
Keywords
Introduction
The World Health Organization (WHO) defined palliative care in 2002 as a holistic approach aimed at improving the quality of life for patients and their families facing challenges associated with life-threatening illnesses. 1 However, numerous economic, social, legal, cultural, and health policy barriers hinder the early implementation of palliative care (PC) in neurology, particularly for people with Parkinson's disease (PwP) and related disorders.2–4 Despite growing awareness and an expanding body of research on PC, several factors continue to obstruct its integration into clinical practice.
First, PC remains a linguistically and conceptually misunderstood term, often equated with end-of-life or terminal care, even among healthcare professionals (HCPs). 2 Governments have been hesitant to invest in PC services due to uncertain implementation timelines, limited supporting evidence, and a lack of consensus on planning strategies. 5 Furthermore, access to educational programmes at both basic and specialist levels remains insufficient. Surveys reveal that only a small proportion of HCPs have received PC training specific to managing PwPs.6,7 This lack of training undermines the availability and quality of specialized PC for PwPs, as untrained HCPs are often ill-equipped to address these needs and may undervalue the benefits of PC. 8
Existing PC models, predominantly developed in Western healthcare systems, lack adequate racial and ethnic diversity in their design and implementation. 9 Socioeconomic status, race, and ethnicity have been associated with decreased PC engagement and a higher likelihood of aggressive end-of-life treatments, highlighting significant disparities in access and outcomes.9–12
Although PwPs experience substantial morbidity and mortality and evidence increasingly supports the benefits of early PC integration, their PC needs often remain unmet.13,14 PwPs frequently report traumatic diagnostic experiences, insufficient management of non-motor symptoms, and inadequate hospice care planning, often leading to institutionalized deaths. 15 Recognizing the critical role of PC in managing Parkinson's disease (PD) and enabling timely access to it through advance care planning (ACP) is essential. 16 ACP, a patient-centred approach, ensures care aligns with the patient's autonomy, wishes, and decision-making capacity. 17
Recent studies have explored specific aspects of PC, such as indicators for its provision, caregiver experiences, and ACP.16,18,19 However, PC is not solely about end-of-life care. Early integration of PC addresses physical, emotional, psychosocial, and spiritual needs, fostering a patient-centred care approach. Sharing care preferences effectively with relatives, partners, and HCPs ensures these preferences are respected when patients can no longer communicate their wishes.
This study aims to map the current research landscape to understand how early integration of PC for PwPs can be achieved. Four research questions guide this investigation, focusing on both PwPs and their caregivers:
This question identifies starting points for integrating PC into the care pathways of PwPs at an early stage.
This involves determining clinical criteria, such as disease status or symptom severity, that inform the timing of PC.
This question examines existing approaches to PC delivery for PwPs.
This explores organizational aspects, including intra- and inter-organizational factors and interactions with individuals, that influence access to PC for PwPs.
Methods
A systematic mixed-methods review 20 of the published literature on palliative care for PwPs was conducted, with its protocol registered in PROSPERO (PROSPERO 2021 CRD42021254848). The review adheres to the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) guidelines, and the PRISMA 2020 Checklist is provided in Supplemental Material 1.
Search strategy
Peer-reviewed articles published in English between January 1, 2006, and May 31, 2021, were retrieved from PubMed and via OVID to access CINAHL, Cochrane, EMBASE, and MEDLINE. The literature search was conducted in May 2021 and updated in December 2022 and March 2024. Additional studies were identified through manual forward and backward citation tracking. The timeframe was chosen because, in 2006, the World Health Organization issued a policy document highlighting the public health challenges and impact of neurological conditions. 21 The full search strategy is provided in Supplemental Material 2.
Eligibility criteria
The inclusion and exclusion criteria for study selection were developed using the PICO (Population, Intervention, Context, Outcome) framework. The following eligibility criteria were applied:
Only studies published in English were considered. Preclinical studies, conceptual papers, review articles, books, book chapters, book reviews, conference proceedings, editorials, national guidelines, dissertations, and all non-peer-reviewed publications were excluded. No geographical restrictions were applied.
The inclusion criteria were further refined based on the specific research questions (RQs):
Conditions under which care organizations operate and their interactions at individual and organizational levels. Structures, processes, and cultures of healthcare organizations. Interactions within and between care organizations. The impact of these factors on healthcare outcomes and organizational performance.
Selection process
Search results were imported into the review management tool Covidence (Covidence—Better Systematic Review Management, www.covidence.org), where duplicates were removed. The study selection process followed a two-stage screening procedure: the first stage focused on titles and abstracts, and the second stage involved a detailed examination of full-text articles. Two independent reviewers (V.L. and G.G.) conducted the screening of titles and abstracts and retrieved full-text articles for detailed review. Any conflicts were resolved by a third independent reviewer (P.P.).
The full texts of all included studies were independently screened by two of the six reviewers (B.W.D., C.M., C.W., D.T., M.G., P.P.), who decided on inclusion or exclusion based on the predefined criteria. In cases of disagreement, a third reviewer from the group was consulted. The selection process is summarized in a flow diagram presented in Figure 1.
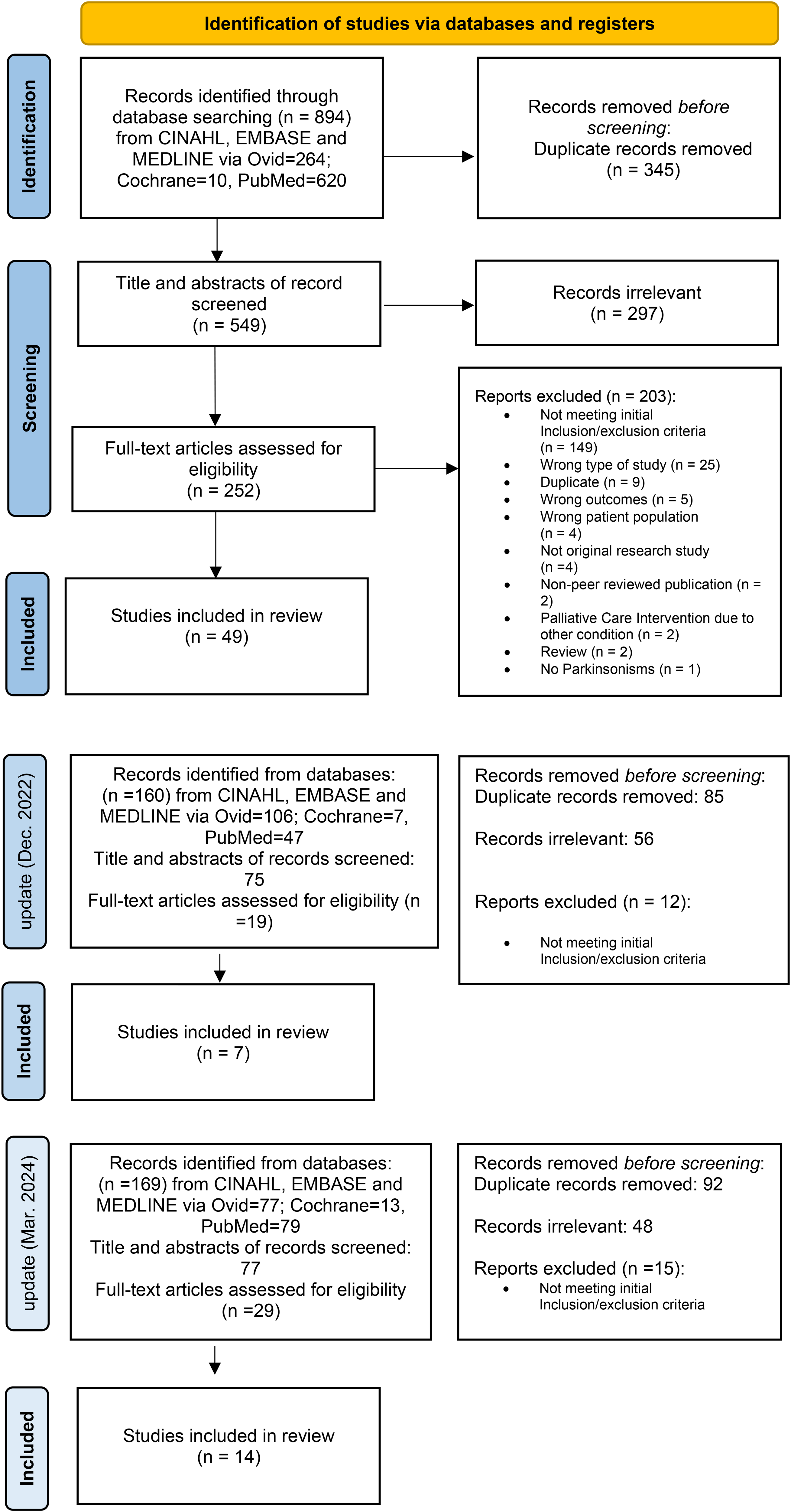
PRISMA flow diagram.
Data items
The extracted data from the included reports covered the following items: first author, year of publication, country, study design, number of participants and their clinical characteristics. In addition, specific elements were extracted according to the research question, i.e., RQ1: Indicators for timely access to palliative care RQ2: Indicators for palliative care referral (when), palliative care needs (physical, social, psychological, spiritual, informational) RQ3: Study design and/or care models, use of care, healthcare providers involved, care coordination between healthcare providers, frequency of visits and place of visits, evaluation of palliative care model RQ4: Hindering factors, facilitating factors
Risk of bias in individual studies
To evaluate the extent to which potential bias was addressed in the design, conduct, and analysis of the included qualitative and quantitative studies, the Quality Assessment Criteria for Evaluating Primary Research Papers from a Variety of Fields (QualSyst) 23 were applied. Two reviewers, selected from a group of five (M.G., B.W.D., C.M., C.W., and P.P.), independently assessed the risk of bias. Any disagreements were resolved through discussion, and in cases of irreconcilable disagreement, a third reviewer was consulted.
Thematic synthesis
The data were analyzed using thematic synthesis. 24 The process involved the following steps: Firstly, two reviewers prepared the extracted data for analysis. The data were organized into structured tables in Microsoft Word, tailored to address the four research questions. Secondly, a combined table was created for research questions 1 and 2, as there was significant overlap in the content. Separate tables were created for research questions 3 and 4 to maintain clarity and specificity. Thirdly, we identified the themes. First, descriptive themes were initially extracted from the literature, closely adhering to the body of evidence presented in the studies. Second, analytical themes were then grouped and refined into seven broader analytical themes through iterative group discussions among the reviewers. Finally, any discrepancies in theme identification or categorization were resolved through a consensus process involving all reviewers. This structured approach ensured that the thematic synthesis was thorough, transparent, and directly aligned with the research questions (Supplemental Material 3).
Results
Study identification and selection
A total of 894 records were retrieved, of which 252 were retained after excluding duplicates and non-relevant papers. Subsequently, 203 records were excluded for not meeting the inclusion criteria. Following the initial search and screening process, 49 reports were included. The first update added 7 articles, and the second update contributed an additional 14 articles, bringing the total to 70 publications comprehensively evaluated for this study.
For the research questions:
12 reports were included for research question 1, 22 for research question 2, 40 provided additional information on the palliative care needs of the population, resulting in a total of 47 included publications, 32 reports were considered for research question 3, 17 for research question 4.
It is important to note that some reports contained relevant information applicable to multiple research questions (see Figure 2).
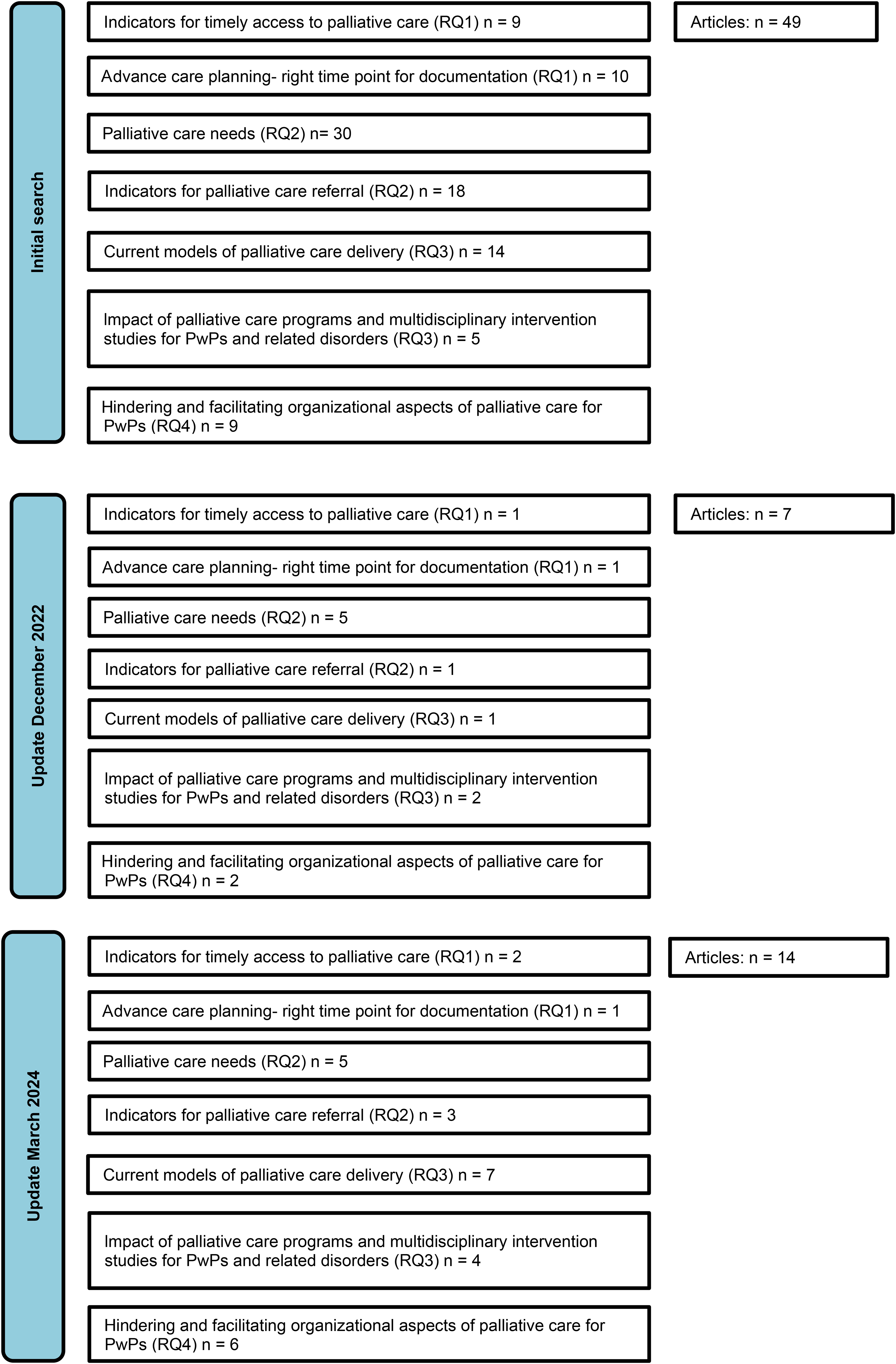
Flowchart depicting the article inclusion process for thematic analysis.
Study characteristics
The majority of the articles originated from the United States (49%), followed by the United Kingdom (13%). The studies comprised quantitative studies (47 out of 70, 67%), qualitative studies (17 out of 70, 24%), mixed-method studies (4 out of 70, 6%), others (2 out of 70, 3%). A detailed overview of the characteristics of the included studies is provided in Tables 1–4.
Studies presenting indicators for timely access to palliative care and when it should be addressed for PwP and their caregivers.
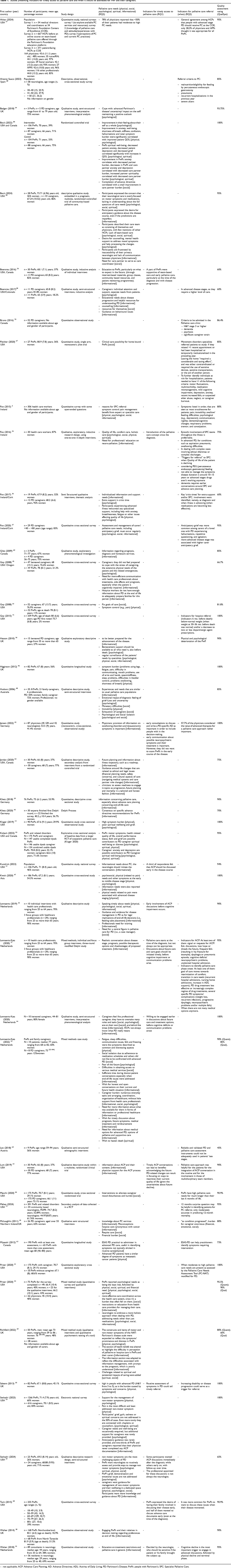
- : not applicable; ACP: Advance Care Planning; AD: Advance Directives; ADL: Activity of Daily Living; PD: Parkinson's Disease; PwPs: people with Parkinson's; SPC: Specialist Palliative Care.
Use of palliative care services.

GPs: General Practitioners; PD: Parkinson's Disease; PwPs: people with Parkinson's.
Intervention studies evaluating (new) models for providing palliative care.

PD: Parkinson's Disease; PwPs: people with Parkinson's.
Facilitating and hindering factors in healthcare organizations for providing palliative care to PwP.

- : not available; GP: general practitioner; HCPs: Health care professionals; PD: Parkinson's Disease; PwPs: people with Parkinson's.
Study results
The thematic synthesis identified the following themes:
Indicators for timely access to palliative care (RQ1), Advance care planning - right time point for documentation (RQ1), Palliative care needs (RQ2), Indicators for palliative care referral (RQ2), Current models of palliative care delivery (RQ3), Impact of palliative care programmes and multidisciplinary intervention studies for PwPs and related disorders (RQ3), Hindering and facilitating organizational aspects of palliative care for PwPs (RQ4).
Based on the WHO definition of palliative care, the needs of PwPs, caregivers, and healthcare professionals (RQ2) extracted from the records were categorized into four domains: physical, social, psychological, and spiritual needs.
1
Given the high prevalence of informational and educational needs highlighted in the publications, a fifth dimension, informational needs, was added.
Indicators for timely access to palliative care
Twelve of the 70 articles (17%) contributed to addressing RQ1 (see Table 1). Although evidence-based guidelines are lacking, a significant proportion of PwPs expressed support for early palliative care, particularly at the time of diagnosis and as the disease progresses.25–28 However, incorporating palliative care from the point of diagnosis does not represent a “one-size-fits-all” solution. Some patients and caregivers prefer to discuss palliative care approaches and ACP only when they perceive a worsening of the disease.27,29
Regular symptom assessment and increased sensitivity among neurologists to the needs of PwPs and their families are essential to ensure timely referrals and the initiation of ACP discussions.13,27,30 Despite this variability in preferences, there is growing support for starting ACP discussions earlier in the care process, 31 ideally before cognitive impairment develops, which could hinder effective communication about patients’ wishes and preferences.27,28,32,33
Advance care planning – right time point for communication and documentation
The introduction, discussion, and implications of having ACP documentation were addressed in the included studies. ACP discussions are part of a process that allows individuals to define their goals and preferences for future medical treatment and care, share these preferences with family and HCPs, and document and review them as needed. 2 These discussions covered various topics and terms, including Advance Directives, living wills, and medical orders.
Specific indicators for initiating ACP discussions were identified.
27
When at least two of the following indicators were present simultaneously, it was considered appropriate to begin these discussions:
A request or signal from PwPs to discuss end-of-life issues. Expressions of fear or uncertainty about the future. Frequent falls resulting in injuries. Episodes of dysphagia or pneumonia. Cognitive deficits or neuropsychiatric issues.
ACP was not always desired by PwPs or their families in the early stages of the disease, often due to the hope for a potential cure.33,36 Additionally, HCPs frequently postponed end-of-life discussions, fearing that patients might find such conversations overwhelming or that it could diminish their sense of hope.
35
A recent U.S. survey reported that physicians or advanced practice providers (APPs) offered ACP discussions or revisions only rarely, at rates of 4% and 8%, respectively.
37
Moreover, Advance Directives, which outline an individual's preferences for medical treatment, were often lacking.
In a UK study, none of the PwPs who died in a hospital had documented Advance Directives. 38 Although 9% of these patients or their caregivers had end-of-life discussions with a physician, 75% had “do not attempt resuscitation” instructions. Discussions on treatment restrictions typically occurred during the terminal phase, with approximately 70% of neurologists and neurology residents addressing these issues only at that stage.35,39 Triggers for initiating treatment restrictions or withdrawal in Advance Directives often included physical or clear cognitive decline. 35 The most frequently discussed topics were resuscitation, ventilation, admission to intensive care units, and tube feeding. 35
In a German study involving people with advanced PD meeting inclusion criteria for palliative care interventions, 70% of participants had Advance Directives. Similarly, in a U.S./Canada study involving PwPs or related disorders with moderate to high palliative care needs, 61% had Advance Directives.39,40 However, in the U.S., only 35% of people with PD listed as a cause of death had completed forms for life-sustaining treatment. 41
End-of-life discussions are recommended to occur when PwPs and their family members feel prepared to address these topics, considering factors such as age, stage of illness, and personality.36,42 These discussions often lack clarity for family members, underscoring the need for tailored guidance specific to PD-related scenarios. Recommended additions include advanced therapies, swallowing disorders, bladder and bowel issues, changes in personality, and neuropsychological symptoms. 43
Palliative care needs
In 40 out of 70 publications (57%), the needs of PwPs, caregivers, and HCPs were identified and categorized into five main areas: physical (n = 24), social (n = 31), psychological (n = 38), spiritual (n = 14), and informational needs (n = 48) 44 (see Table 1).
Physical needs were frequently mentioned, particularly regarding symptom burden and management, including the need for better pain relief.37,45–47 Despite this, physical needs were often inadequately addressed. Alongside pain, other disease-related motor and non-motor symptoms were reported as neglected or requiring greater support, such as swallowing difficulties, communication impairments, spasms and stiffness, sleep disturbances, fatigue, and shortness of breath.29,36,42,48 In general, it was noted that any clinical manifestation negatively impacting quality of life should be prioritized.13,36
Social needs could be further divided into two subcategories:
Socioeconomic Concerns: These reflect the financial burden of the disease and its impact on care provision. Caregiver Support Needs: Caregivers highlighted the effects of social and psychological isolation, emphasizing the need for solutions such as respite care.48,49
The need for improved care coordination was also expressed, with suggestions such as introducing case managers to address these social needs.
50
Case managers could provide advocacy, emotional support, and connections to community resources.32,51
PwPs often reported that psychological suffering was inadequately addressed, 52 especially during critical moments such as diagnosis, which frequently led to feelings of pain, uncertainty, and isolation for both PwPs and caregivers.36,48,53,54 Anxiety and depression, which significantly affect quality of life throughout the disease course, were often overlooked. 36
Spiritual well-being and bereavement support were rarely considered in clinical care provided by neurologists. This highlights the need for a more holistic approach that evaluates and addresses these needs to improve quality of life.13,19,25,37,55–57 Caregivers, in particular, reported feeling unprepared for their roles, struggling with stress, and managing the increasing and extensive needs of PwPs, often without receiving individualized attention or support.34,36,37,57,58
Informational needs were the most frequently reported, accounting for 48 studies. These needs were further divided into three subcategories:
Education about the Disease: PwPs and caregivers noted that the lack of information about disease progression impacted their ability to manage symptoms and develop coping strategies, increasing their suffering and uncertainty.25,32,47,58–60 They expressed a desire for anticipatory guidance, even when predictions about disease progression were not entirely accurate.29,50 Palliative Care Education: PwPs and caregivers emphasized the need for more information about palliative care, including correcting misconceptions that it is solely associated with oncology. They sought guidance on ACP, end-of-life care, and available PC services.26,35,38,39,49,61,62 ACP Documentation: PwPs and caregivers advocated for tailored ACP discussions and support from physicians to ensure their preferences were understood and documented appropriately.27,33,38,62
Additionally, terms such as guidance, training, and preparation were commonly used in the analyzed papers to describe informational needs. PwPs, caregivers, and HCPs highlighted the importance of timely discussions to include and address their needs effectively.25,32,34,58,60
HCPs also noted a lack of consensus on recommendations for conducting ACP conversations 43 and expressed an urgent need for evidence-based guidelines to manage the disease and address ethical and legal issues, such as feeding tube placement.32,61 Furthermore, they called for general education on neuro-palliation. 25
Indicators for palliative care referral
Most articles addressing palliative care for PwPs lacked clear clinical indications for referral (see Table 1). Only 31% (22 out of 70) provided information on indicators for referral. Validated scales, such as the Edmonton Symptom Assessment System Scale for Parkinson's Disease (ESAS-PD) and the Palliative Care Needs Assessment Tool (PC-NAT), have proven useful in helping clinicians and PwPs identify prominent symptoms, including those not typically addressed in routine care. A moderate-to-severe score on these scales has been proposed as an indicator for referral.51,63 However, while these tools show promise for use from early to advanced stages of the disease, they are less reliable in late-stage PwPs due to predominant cognitive decline.64,65
Another tool designed for HCPs to identify palliative care needs and indicators for ACP is the RADboud Indicator for Palliative Care Needs in Parkinson's Disease (RADPAC-PD). 27 This tool distinguishes between criteria for ACP discussions and the palliative care phase, a distinction critical for initiating early care planning while decision-making capacity and communication abilities are intact.
The burdensome progression of the disease, including its emotional, financial, and social impact on both PwPs and caregivers, has been identified as an indicator for referral.30,49,56 Advanced stages of PD often require higher levels of care due to increasing disability, physical and psychological deterioration, and prolonged palliative care needs, often extending well beyond the last six months of life.58,66 Care transitions, such as shifting the focus toward comfort, recurrent hospital admissions, or the need for nursing home placement, also serve as valid indicators for referral.27,67,68
Referral is further warranted when leaving the home becomes a significant challenge, requiring assistive devices, specialized transportation, or caregiver support to ensure safety.27,69 Specific disease-related symptoms and complications, such as weight loss, recurrent infections, severe ulcers, swallowing difficulties, malnutrition, psychiatric and cognitive changes, multiple falls, pain, rigidity, respiratory problems, tremor, and constipation, have also been identified as triggers for referral.6,68,69
PwPs meeting certain criteria, such as Hoehn and Yahr (H&Y) stage 3 or higher, the presence of dementia or psychosis, or significant caregiver strain, have been identified as requiring specialized and comprehensive care. 70 Studies investigating predictors of hospice eligibility have noted that symptoms appearing 6–12 months before death, such as significant weight loss (below normal BMI) or reduced use of dopaminergic agents (two or fewer prescriptions), are indicative of advanced needs. 71
The “surprise question” (SQ: “Would you be surprised if your patient died in the next year?”) has also been used to identify referral needs. Although its accuracy in predicting survival is modest, a negative answer to the SQ has been associated with factors such as older age, dementia, neuropsychiatric symptoms, increased comorbidities, having a care partner, and poor quality of life. 72
Specialist palliative care (SPC) services are beneficial from the time of diagnosis, particularly when medication management becomes complex or when patients and their families need support to cope with the uncertainty of the disease, its impact on identity, and maintaining a positive outlook.25,36 SPC involvement is especially valuable in managing complications such as aspiration pneumonia and swallowing difficulties. Episodic involvement or enhanced dialogue between PD specialists and SPC professionals has proven effective for discussions concerning ACP. 25
SPC also plays an advisory role in complex cases involving ethical dilemmas or challenging discharges. 25 Triggers for SPC referral identified by PD specialists include perceived deterioration in quality of life, consideration of advanced nutritional strategies (e.g., percutaneous endoscopic gastrostomy [PEG] feeding), unsuccessful symptom management, long disease duration (10–15 years), and advanced stages with cognitive decline. 25 The collaborative approach between PD specialists and SPC professionals enhances the overall quality of care for PwPs.
Current models for palliative care delivery
In examining the current use of palliative care services across eight countries (based on 22 out of 70 publications, or 31%, detailed in Table 2), it became evident that PwPs are infrequently referred to specialized PC services.31,37,42,73,74
In Australia, only 12% of PwPs accessed SPC services during their final year of life, typically in hospitals and occasionally at home. In Spain, neurologists reported that less than 10% of eligible patients with movement disorders received palliative care. 68 In the United States, only 4% of PwPs and 8% of caregivers reported that their PD providers discussed palliative care with them. Additionally, only 4.5% of hospitalized PwPs received inpatient PC consultations.73,75
Two studies noted an upward trend in PC referrals: in Australia, there was an 8% increase between 2000 and 2002, and in the U.S., a 3.6% increase was observed between 2007 and 2014.70,71 However, gender disparities were evident in the U.S., with women less likely to be referred to PC services. 75
In the UK, 14% of PwPs who died in hospitals were referred to SPC teams, and 42% were placed on the Liverpool Care Pathway (LCP). 38 Importantly, most PwPs on the LCP had their dopaminergic medications withdrawn, a practice linked to worsening symptoms and accelerated clinical decline in PD. 38
Hospice care, a well-established PC approach, provides comprehensive, multidisciplinary support for patients nearing end-of-life. In the UK, fewer than 1% of PwPs died in hospice facilities, while in the U.S., 54% of nursing home residents received hospice care.38,76–78 In Australia, a national survey found that only 5% of PwPs received PC services. 79 Similarly, a German national survey reported a 3% provision rate of PC for patients with advanced PD. 39
In a retrospective U.S. study of PwPs at the end of life, 46% received PC consultations. 80 Of these, 40% were discharged to hospice facilities or inpatient hospice, 37% received home hospice care, and 23% died in the hospital. Notably, PwPs who died in the hospital had higher rates of invasive procedures and ICU admissions compared to those who received hospice care (75% vs. 41%). 80
Impact of palliative care programmes and multidisciplinary interventions for PwPs and related disorders
Eleven of the 70 studies (16%) evaluated the effects of various PC programmes for PwPs and related disorders (detailed in Table 3).40,69,81–84
In a Chinese study, the outcomes of a six-month nurse-led PC intervention were compared to standard care provided by neurologists or physicians. While improvements in quality of life (QoL), caregiver burden, and patient satisfaction were observed in all groups, the nurse-led intervention demonstrated the most significant benefits. 85 Similarly, in Australia, community-based PC for deceased PwPs reduced hospital costs compared to those who did not receive such care. 83
In Germany, a combination of standard neurological care, supplemented with three-monthly home visits by a PD nurse and the development of individualized treatment plans, significantly improved QoL compared to standard neurological care alone. Additionally, both motor and non-motor symptoms decreased, as measured by disease-specific scales (UPDRS-III and PD-NMS, respectively). 81
In the U.S. and Canada, a hospital-based PC intervention involving a neurologist, nurse, social worker, chaplain, and palliative care specialist also enhanced QoL for PwPs. Patients in the intervention group reported higher QoL at six months compared to those receiving standard care, along with a decrease in caregiver burden at the 12-month follow-up.40,84 These findings align with results from studies in Australia and the U.S., where QoL indexes remained stable over 12 months despite disease progression, and there was no increase in the utilization of health services, such as long-term care facilities and hospitalizations.69,82,83,86 These programmes also reported high retention rates and satisfaction levels, with adjustments to therapy and referrals to non-pharmacological treatments occurring in over 40% of cases. 82
Caregivers also benefited from these interventions. For instance, interdisciplinary programmes and peer mentoring effectively mitigated and stabilized caregiver burden in a cohort of homebound PwPs. 87
A recent trial 88 tested the impact of community-based PC, which included palliative care training for community neurologists and coaching from senior PC specialists to address challenging cases. PwPs were referred to a multidisciplinary telemedicine PC team comprising a nurse, pharmacist, social worker, and chaplain. The intervention addressed overlooked needs such as spiritual support and facilitated discussions on advance ACP and future care preferences. 50
The programme demonstrated several benefits compared to standard care, including improved QoL, reduced depressive symptoms, and higher completion rates of Advance Directives at six months. Participants also experienced enhanced care coordination, increased referrals, and stronger connections to community resources. 50 Modest improvements in HCPs’ knowledge about PC were also observed. 88 These findings underscore the potential of palliative care programmes to improve both patient and caregiver outcomes through tailored, interdisciplinary approaches and innovative delivery models.
A U.S. study analyzed the costs and return on investment of team-based palliative care using Medicare reimbursement rates. In an academic medical setting, the neurology PC clinic demonstrated financial sustainability. For every dollar invested in expanding PC services, the hospital system recovered $1.68 for new patient visits and $0.13 for established patient visits. 89 This evidence underscores the variability in PC utilization across countries, the challenges in equitable access, and the potential for economic viability when PC services are effectively integrated into healthcare systems.
Barriers and facilitators for access to and provision of palliative care for pwPs
Barriers and facilitators to palliative care access, identified in 17 of 70 publications (24%) (detailed in Table 4), can be categorized as follows: education, challenges in determining the right time for palliative care, end-of-life care preferences and ACP, home-based palliative care services, and the influence of race, ethnicity, gender, and income.
Educational needs span PwPs, caregivers, and HCPs. PwPs often lack basic information about their condition, and even HCPs harbour misconceptions about palliative care.19,29,35,36,56 Training in palliative care for HCPs is identified as a key facilitating factor.37,90 Two studies emphasized that HCPs must deepen their understanding of the core principles of palliative care to support the development of effective PC systems for PD.32,91,92
To enable PwPs and caregivers to access palliative care, timely information about available services and emotional preparation, especially for end-of-life and bereavement, is essential.25,56 However, identifying clear indicators for appropriate and timely PC referrals remains challenging, affecting both caregivers and HCPs.32,56 HCPs often struggle in daily practice to recognize the needs of PwPs, interpret them accurately, and refer them promptly to appropriate services.34,37 For instance, family members may not realize that death is imminent, leaving them unprepared to handle sudden crises. 56
Another significant barrier is the discomfort surrounding discussions about ACP and end-of-life care. Many PwPs and caregivers find these conversations distressing, leading to delays. 35 HCPs themselves often feel unprepared for such discussions, citing a lack of personal strategies to navigate the emotional complexities involved. 37 When physician interactions are brief and lack psychological support or referrals to other services, care planning and coordination become inadequate, resulting in reactive, ad hoc access to services. 56
PwPs and caregivers also fear that documented ACP preferences may not be respected by physicians or the healthcare system, further hindering their willingness to engage in ACP discussions. 33 Additionally, the fragmented healthcare system provides inconsistent support to PwPs and caregivers, with limited and sporadic contact leaving many feeling “abandoned” in managing the disease. 25
A lack of continuity in care further exacerbates these challenges. PwPs and caregivers often encounter different health professionals with each visit to clinics or hospitals, leading to frustration and uncertainty about available support services.25,32,56 This fragmentation results in uncoordinated and inconsistent access to both clinical and palliative care. For PwPs without a caregiver, access to palliative care is particularly difficult, as caregivers often play a central role in organizing and facilitating care. 25
Many PwPs prefer to remain at home despite their deteriorating health. However, inadequate home-based palliative care services often make this option unattainable. 56 Beyond the availability of specialized PC services, a strong connection between services and multidisciplinary collaboration is critical to ensure access. 32 Establishing dedicated multidisciplinary teams for managing neurological conditions, supported by a central coordinator, is essential.56,62
Diversity factors also influence access to palliative care. While race or ethnicity did not affect PC referrals for PwPs in one U.S. study, 75 cultural factors in certain regions, such as traditional Chinese beliefs, create barriers. In these cultures, topics related to death and dying are taboo, preventing discussions about ACP and treatment preferences. 91
Additionally, socioeconomic factors strongly influence PC referrals. Female PwPs, those with lower incomes, and those without private insurance are significantly less likely to be referred to palliative care. 75 Addressing these disparities is crucial for ensuring equitable access to palliative care services.
Overcoming these barriers requires targeted education, better coordination of services, culturally sensitive care practices, and efforts to reduce socioeconomic disparities. By addressing these challenges, the provision of palliative care for PwPs can be significantly improved, enhancing both patient and caregiver outcomes.
Discussion
This systematic review synthesizes how PC is currently provided for PwPs and their caregivers, focusing on clinical indicators for referral and opportunities for early integration into traditional care management. It also offers a comprehensive overview of facilitators and barriers to PC access.
Delivering care to PwPs is inherently complex due to the diverse phenotypes, progression rates, and individual needs/preferences associated with PD.93,94 As disease duration increases, so does the likelihood of hospitalization and nursing home placement, underscoring the growing difficulty of managing care at home. 93 Despite the recognized benefits, PC provision remains sparse, inconsistent, and largely limited to high-income countries (e.g., U.S., UK, Netherlands, Australia, Germany, Canada, Sweden). Referral to PC services often occurs only in the advanced or terminal stages of the disease.38,39,56,73,75,95,96
Significant barriers hinder PC access, including inadequate information, education, and training for PwPs, caregivers, and HCPs. These gaps result in unmet needs, heightened disease burden, and inequities in care due to income, gender, and the lack of home-based services. Fragmented healthcare systems exacerbate these challenges, with poor integration of disciplines and inconsistent interactions between HCPs and patients undermining trust and continuity of care. Furthermore, HCPs often struggle to identify the optimal timing for PC referral and are insufficiently trained to address the comprehensive needs of PwPs and their caregivers.
This fragmented reality highlights the urgent need for innovative, patient-centred care delivery models. However, few studies have rigorously evaluated outpatient, multidisciplinary PC interventions.69,81–83 These trials demonstrated that multidisciplinary care—incorporating neurology, nursing, social care, physical and occupational therapy, spiritual, and psychological support—can significantly improve QoL, reduce caregiver burden, and alleviate global symptom burden for advanced PwPs.40,69,81,83 Additionally, these approaches reduced healthcare costs by mitigating preventable crises. Hence, improving access to PC for PwPs requires systemic changes, including better training for HCPs, integration of disciplines, and advocacy for evidence-based, multidisciplinary care models that address the complex and evolving needs of PwPs and their caregivers.
Despite advancements in recognizing the needs of PwPs, care delivery remains predominantly focused on motor symptom management.46,47,61 PwPs report limited interactions with HCPs, centred mainly on pharmacological treatment, while non-motor needs significantly impacting QoL are often overlooked.36,48 As the disease progresses, its impact extends beyond medical care to encompass social, spiritual, and financial challenges, as well as caregiver distress.48,49,97
Education remains a critical barrier to implementing PC. PwPs, caregivers, and HCPs frequently lack adequate information about PC services and their application.35,36,56 HCPs often fail to adopt a holistic approach to managing PD due to limited knowledge of PC principles.25,43 Evidence suggests that educating allied HCPs improves care quality, reduces complications, and lowers healthcare costs. 98 Emerging evidence-based educational programmes specifically address palliative care best practices in PD, targeting diverse audiences, including specialists, to enhance understanding of disease progression, prognosis, and care planning.90,99–101 PC specialists have proven effective in managing non-motor complications, complex discharges, and ethical dilemmas, highlighting the need for stronger collaboration between neurology and PC disciplines.25,36
This review identified multiple criteria for PC referral but no clear consensus. Criteria can be broadly categorized into:
Disease-related symptoms or complications, such as weight loss, recurrent infections, and cognitive or psychiatric changes. Indicators of burdensome disease progression and increasing needs, including escalating impacts on PwPs and caregivers.
Informational, social, psychological, and spiritual needs often drive referrals, emphasizing a patient-centred rather than disease-centred approach.18,102 Although several studies advocate for early PC referral, starting at diagnosis,13,25–28,30,35 limited evidence compares outcomes for early versus late PC initiation. Routine evaluations and open dialogues between HCPs, PwPs, and caregivers can help tailor PC interventions and determine the appropriate timing.
103
ACP is vital for addressing long-term care preferences, yet discussions often occur too late. Early initiation of ACP is recommended to mitigate the risks associated with cognitive decline, apathy, and depression, which impede communication and decision-making.28,36 Annual assessments of readiness for ACP discussions are suggested to ensure these conversations occur proactively rather than during crises like hospitalizations.33,36
Evidence shows that ACP documentation influences end-of-life decisions, increasing the likelihood of PwPs choosing comfort measures over life-sustaining treatments and dying in their preferred setting, such as hospice or home. For example, individuals with documented HCP orders for life-sustaining treatment were half as likely to die in inpatient facilities compared to those without such documentation. 28
Integrating PC early in the disease trajectory requires improving educational efforts, enhancing multidisciplinary collaboration, and tailoring care to individual needs. Addressing these gaps can support PwPs and caregivers, improve QoL, and align care delivery with patient preferences across all stages of the disease.
Limitations
This systematic review has several limitations that should be acknowledged. The primary limitation is that many of the included studies did not directly address the specific research questions posed in this review.
First, indicators for timely access to palliative care were often not explicitly mentioned in the included studies, requiring interpretation to extract relevant findings. Second, there is a notable lack of research examining organizational aspects at the meso and macro levels. Instead, the findings included in this review were identified as relevant organizational factors and integrated to provide an organization-specific perspective, raising questions about the representativeness of the results.
Third, the distinction between palliative care services and multi-professional teams specialized in the care of PwPs is not always clearly defined in the literature. While it is evident that multi-professional teams provide a holistic approach to meeting the needs of PwPs and their caregivers, these teams are not always comprised of specialists in palliative care.
Future research should focus on exploring palliative care for PwPs from an organizational perspective and on generating stronger evidence to guide clinical practice effectively.
Conclusions
In conclusion, the multifaceted and heterogeneous nature of PD underscores the need for a comprehensive, patient-centred approach to care that emphasizes the early integration of PC principles. Achieving personalized and holistic care requires the active involvement of a multidisciplinary team, including PC specialists, to address the complex physical, emotional, social, and spiritual needs of PwPs and their families.
Enhanced coordination of care and effective collaboration among the various healthcare providers involved in PD management are critical for improving care continuity and outcomes. Additionally, prioritizing education and training on PC for PwPs, caregivers, and healthcare professionals is essential to enhance the overall quality of care and QoL.
Implementing these strategies not only improves the quality of care but also empowers PwPs to actively participate in determining their care preferences, fostering a more patient-centred approach throughout the disease trajectory.
Supplemental Material
sj-docx-1-pkn-10.1177_1877718X251321110 - Supplemental material for Early thinking palliative care for people with Parkinson's disease: A thematic synthesis based on a systematic mixed-methods review
Supplemental material, sj-docx-1-pkn-10.1177_1877718X251321110 for Early thinking palliative care for people with Parkinson's disease: A thematic synthesis based on a systematic mixed-methods review by Michela Garon, Christiane Weck, Valentina Leta, Bauke W Dijkstra, Catharina Muente, Giovanni Gentile, Dhaval Trivedi, Marieke M Groot, Stefan Lorenzl, Per Odin, Spyridon Konitsiotis, David J Pedrosa, Dimitrios I Fotiadis, Marjan J Meinders, Bastiaan R Bloem, Anette E Schrag, Laura Grover, Pille Taba, K Ray Chaudhuri, Angelo Antonini, Piret Paal and in Journal of Parkinson's Disease
Supplemental Material
sj-docx-2-pkn-10.1177_1877718X251321110 - Supplemental material for Early thinking palliative care for people with Parkinson's disease: A thematic synthesis based on a systematic mixed-methods review
Supplemental material, sj-docx-2-pkn-10.1177_1877718X251321110 for Early thinking palliative care for people with Parkinson's disease: A thematic synthesis based on a systematic mixed-methods review by Michela Garon, Christiane Weck, Valentina Leta, Bauke W Dijkstra, Catharina Muente, Giovanni Gentile, Dhaval Trivedi, Marieke M Groot, Stefan Lorenzl, Per Odin, Spyridon Konitsiotis, David J Pedrosa, Dimitrios I Fotiadis, Marjan J Meinders, Bastiaan R Bloem, Anette E Schrag, Laura Grover, Pille Taba, K Ray Chaudhuri, Angelo Antonini, Piret Paal and in Journal of Parkinson's Disease
Supplemental Material
sj-docx-3-pkn-10.1177_1877718X251321110 - Supplemental material for Early thinking palliative care for people with Parkinson's disease: A thematic synthesis based on a systematic mixed-methods review
Supplemental material, sj-docx-3-pkn-10.1177_1877718X251321110 for Early thinking palliative care for people with Parkinson's disease: A thematic synthesis based on a systematic mixed-methods review by Michela Garon, Christiane Weck, Valentina Leta, Bauke W Dijkstra, Catharina Muente, Giovanni Gentile, Dhaval Trivedi, Marieke M Groot, Stefan Lorenzl, Per Odin, Spyridon Konitsiotis, David J Pedrosa, Dimitrios I Fotiadis, Marjan J Meinders, Bastiaan R Bloem, Anette E Schrag, Laura Grover, Pille Taba, K Ray Chaudhuri, Angelo Antonini, Piret Paal and in Journal of Parkinson's Disease
Footnotes
Acknowledgments
The authors have no acknowledgments to report.
ORCID iDs
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The PD_Pal project has received funding from the European Union's Horizon 2020 research and innovation programme under grant agreement No. 825785.
Declaration of conflicting interests
The authors declared the following potential conflicts of interest with respect to the research, authorship, and/or publication of this article: C.W. has received honoraria for lectures for AbbVie. V.L. declares that she has received grants and/or honoraria for speaker-related activities from Abbvie, Bial, Britannia Pharmaceuticals, Invisio, Profile and UCB unrelated to the submitted work. S.L. has received honoraria for lectures from AbbVie, BIal and STADA as well as CAnnaXan. P.O. has received honoraria for lectures and advice from AbbVie, Bial, Britannia, Nordic Infucare, Stada and Zambon. A.A. has received compensation for consultancy and speaker-related activities from UCB, Boehringer Ingelheim, Ever Pharma, General Electric, Britannia, AbbVie, Kyowa Kirin, Zambon, Bial, Theravance Biopharma, Jazz Pharmaceuticals, Roche and Medscape; has received research support from Bial, Lundbeck, Roche, Angelini Pharmaceuticals, Horizon 2020 Grants 825785 and 101016902, Ministry of Education University and Research Grant ARS01_01081, Cariparo Foundation and Movement Disorders Society for NMS Scale validation; and has served as consultant for Boehringer–Ingelheim for legal cases on pathological gambling. D.P. has received honoraria as a speaker at symposia sponsored by Boston Scientific Corp., Medtronic, AbbVie Inc., Zambon, and Esteve Pharmaceuticals GmbH. He received payments as a consultant for Boston Scientific Corp, Bial and Bayer, and he received a scientific fellowship from Boston Scientific Corp. for a project entitled: “Sensor-based optimization of Deep Brain Stimulation settings in Parkinson's disease” (COMPARE-DBS). Finally, D.P. was reimbursed for travel expenses by Esteve Pharmaceuticals GmbH and Boston Scientific Corp for attending conferences. S.K. and D.I.F. have received funding from PD Neurotechnology LTD. P.T. received support from the Estonian Research Foundation (Grant No. PRG957). A.E.S. reported receiving a salary from the National Institute for Health and Care Research (NIHR) Biomedical Research Council during the conduct of the study; grants from the NIHR for investigator-led trials, Movement Disorders Society Development of the Movement Disorder Society Nonmotor Rating Scale, and the European Commission for studies on anxiety in Parkinson disease and care aspects in Parkinson disease; advisory and speaker fees from AbbVie; salary from the University College London; and book royalties from Oxford University Press outside the submitted work. B.R.B. serves as the co-Editor in Chief for the Journal of Parkinson's disease, serves on the editorial board of Practical Neurology and Digital Biomarkers, has received fees from serving on the scientific advisory board for the Critical Path Institute, Gyenno Science, MedRhythms, UCB, Kyowa Kirin and Zambon (paid to the Institute), has received fees for speaking at conferences from AbbVie, Bial, Biogen, GE Healthcare, Oruen, Roche, UCB and Zambon (paid to the Institute), and has received research support from Biogen, Cure Parkinson's, Davis Phinney Foundation, Edmond J. Safra Foundation, Gatsby Foundation, Hersenstichting Nederland, Horizon 2020, IRLAB Therapeutics, Maag Lever Darm Stichting, Michael J Fox Foundation, Ministry of Agriculture, Ministry of Economic Affairs & Climate Policy, Ministry of Health, Welfare and Sport, Netherlands Organization for Scientific Research (ZonMw), Not Impossible, Parkinson Vereniging, Parkinson's Foundation, Parkinson's UK, Stichting Alkemade-Keuls, Stichting Parkinson NL, Stichting Woelse Waard, Topsector Life Sciences and Health, UCB, Verily Life Sciences, Roche and Zambon. BRB does not hold any stocks or stock options with any companies that are connected to Parkinson's disease or to any of his clinical or research activities. B.R.B. is co-Editor in Chief for the Journal of Parkinson's Disease but was not involved in the peer-review process nor had access to any information regarding its peer-review. K.R.C. declares that he has received honoraria for advisory board membership from 4D Pharma, AbbVie, Acadia, Britannia, Global kinetics (GKC), Lobsor, Profile Pharma, Roche, Scion, Stada, Synovion, Therevance, UCB and Zambon; speaker's honoraria for lectures from AbbVie, Bial, Boeringer Ingelheim, Britannia, Kyowa Kirin, Novartis, SK Pharma, UCB, and Zambon; investigator initiated research grants from AbbVie, Bial; Britannia, UCB, Global Kinetics; and academic grant funding from EU Horizon 2020, Parkinson's UK, Wellcome Trust, Kirby Laing Foundation and the Medical Research Council; royalty payments or license fees from Oxford University Press, and Cambridge University Press, MAPI institute (King's Parkinson Disease Pain Scale (KPPS) and Parkinson Disease Sleep Scale 2 (PDSS-2)); and payment for expert testimony from the General Medical Council unrelated to the submitted work. K.R.C. is an Editorial Board Member of this journal but was not involved in the peer-review process nor had access to any information regarding its peer-review.
Data availability
The data supporting the findings of this study are available within the article and/or its Supplemental Material.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
