Abstract
Background
Parkinson's disease (PD) is a progressive degenerative disease affecting people at any age; however, it is generally more prevalent in older age groups. Younger individuals, especially women, are facing significant challenges that pertain to pregnancy. Unfortunately, the available information is limited and supported mostly by anecdotical experience.
Objective
To gain insight into the interaction between early-onset PD and pregnancy.
Methods
We report on a case series of women, diagnosed with PD, that underwent pregnancy to explore the disease catachrestic, natural history, and different challenges associated with these unique group of people.
Results
We identified a total of 9 participants who met our inclusion criteria, who had gone through a total of 12 pregnancies after onset of PD motor symptoms. In 10 out of 12 pregnancies, participants did not use any PD medication; throughout 1 pregnancy, a participant used pramipexole and throughout 1 pregnancy, a participant used levodopa/carbidopa.
Conclusions
Our study supports the importance of further multicentric studies on pregnant women with PD to prospectively collect data on complications, birth outcomes, lactation, and PD severity.
Introduction
Parkinson's disease (PD) is a chronic, progressive disease that mainly occurs in people in their mid- to late 60 s. 1 However, ∼5 to 10% of all people with PD have early-onset Parkinson's disease (EOPD) when first symptoms occur under age 50.2,3 EOPD differs significantly from late-onset PD in clinical features and underlying pathophysiology. 2 Moreover, people with EOPD often experience different social and societal issues that are specific to a younger stage-of-life compared to people who get diagnosed at a later age. 2 Additionally, due to the relatively young age at disease onset, people with EOPD may still consider having children after diagnosis.4,5 In women, this provides additional challenges, due to the largely unknown interaction between EOPD, pregnancy, and medication. 4
The exact number of women with PD that are undergoing pregnancy remains unknown, 6 although it is increasing due to the average increment of maternal age at pregnancy 5 and also for the progressive increment of people living with PD is in every age group. The decision to have children has a huge impact. Therefore, it is important to make a well-informed, scientifically substantiated decision on this matter. However, if it comes to PD and pregnancy, there is a lack of knowledge on the safety of PD medication in pregnancy, the impact of pregnancy on PD, potential fetal risks, risks during birth, safety of breastfeeding and long-term birth outcomes. The available literature is limited and consists of case reports and case series and does not provide any definite conclusions. 7
Because of the lack of knowledge about this topic, it is currently impossible to provide women PD with a proper preconceptional advice on the risks of pregnancy when diagnosed with PD. Thus, the desire to have children becomes uncertain and quite difficult for women and support them during pregnancy, potentially hindering the decision-making process. There are no guidelines available to properly advise, treat, and support women with PD during pregnancy.
Therefore, we aimed to gain insight into the complex interaction between EOPD and pregnancy reviewing our cohort of women with EOPD and reviewing the outcomes of women with EOPD who had been pregnant after the onset of PD motor symptoms.
Methods
We used a retrospective approach by collecting data through medical records of women with EOPD. All women who were diagnosed with EOPD at the Mayo Clinic (USA) with an onset of motor symptoms between 1989 and 2021 were identified using the Mayo Clinic Data Management tool. A movement disorder specialist reviewed all medical records to confirm a clinical EOPD diagnosis. EOPD was defined as the onset of motor symptoms between the ages of 21 and 50.
We reviewed medical records of patients to identify demographic characteristics, as well as data on pregnancies and deliveries that occurred after the onset of motor symptoms. We collected data on the use of PD medication during pregnancy, anomalies on prenatal ultrasounds and in prenatal genetic testing, complications during pregnancy, birth outcomes, maternal age at delivery, number of weeks gestation at birth, delivery type, complications during delivery, birth weight, complications after delivery and severity of PD symptoms throughout pregnancy. Women were eligible for inclusion in the study if medical records at least provided information on birth outcomes and maternal age at delivery.
Ethical approval and informed consent
The study received approval from the Institutional Review Board of the Mayo Clinic, and all study participants provided written informed consent for the passive use of their medical information in research.
Data availability
All relevant data appear in this article; data regarding case ascertainment of parkinsonism and methodology on case identification have been previously published. Collected data are summarized in Table 1.
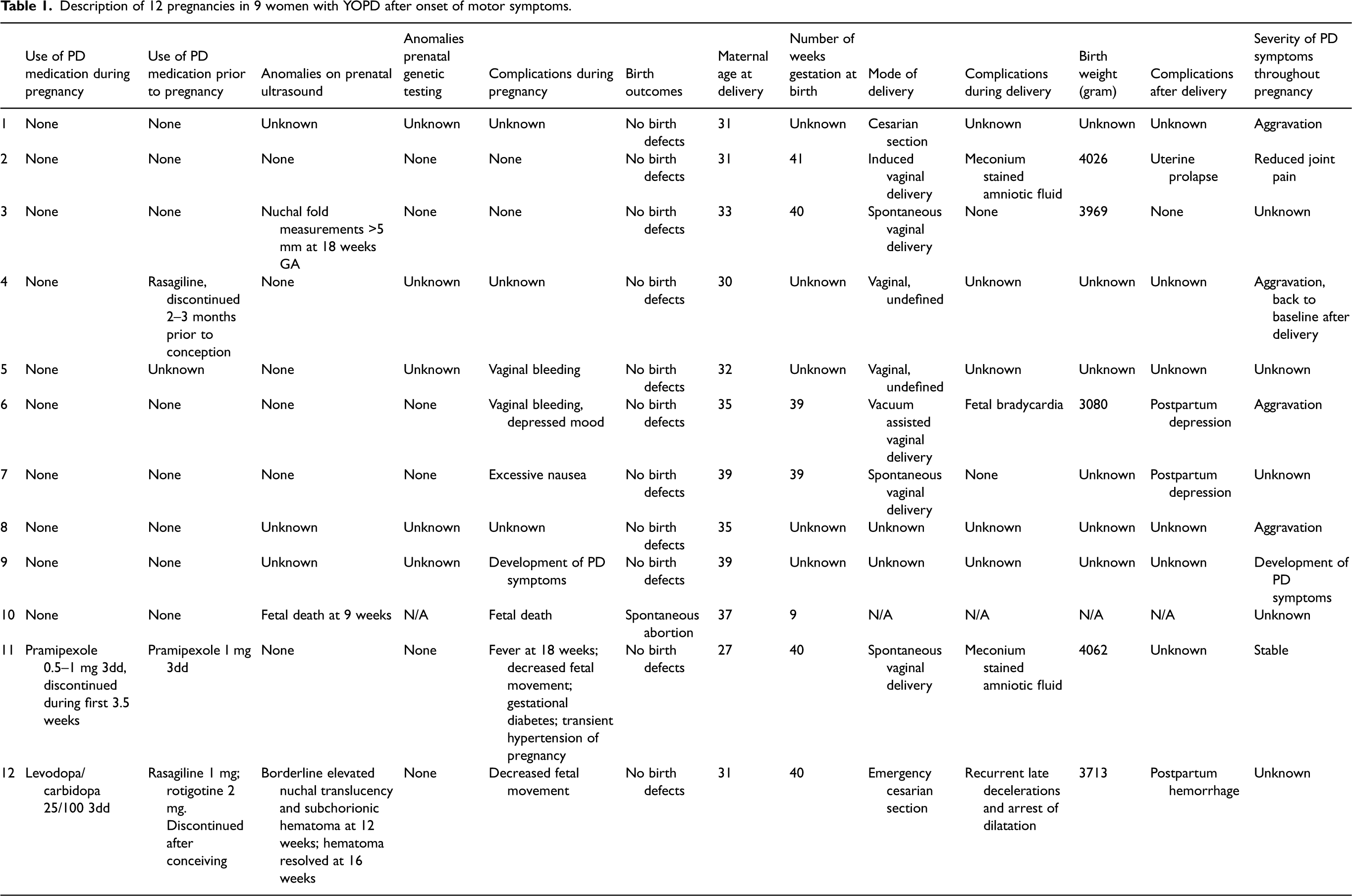
Description of 12 pregnancies in 9 women with YOPD after onset of motor symptoms.
Results
We identified a total of 9 participants who met our inclusion criteria, who had gone through a total of 12 pregnancies after onset of PD motor symptoms. During 10 out of 12 pregnancies, participants did not use any PD medication; throughout 1 pregnancy, the participant used pramipexole and throughout 1 pregnancy, the participant used levodopa/carbidopa.
Pregnancies without use of PD medication
Of the 10 pregnancies that were carried out without the use of PD medication, 9 resulted in live births. One pregnancy ended in a spontaneous abortion after fetal death was established during the prenatal ultrasound at 9 weeks gestation. In one pregnancy, the prenatal ultrasound showed nuchal fold measurements of more than 5 mm at 18 weeks gestation. This pregnancy resulted in the birth of a healthy infant. In the other pregnancies, no anomalies were reported on prenatal ultrasounds or in genetic testing. Complications during pregnancy were reported in 7 cases; in 2 pregnancies, vaginal bleeding occurred during the first trimester; depressed mood and excessive nausea were each present in one pregnancy and one participant reported the onset of their first PD motor symptoms during pregnancy. Maternal age at delivery was between 30 and 39 years (mean = 34 years; SD ± 3.4 years). The number of weeks gestation at birth was reported in 5 pregnancies; 4 of these pregnancies were carried to term, with the number of weeks gestation at birth between 39 and 41 weeks. The mode of delivery was reported in 7 pregnancies and consisted of 2 spontaneous vaginal deliveries, one induced vaginal delivery, one cesarian section, two vaginal deliveries without further clarification and one vacuum assisted delivery. The latter was provoked by fetal bradycardia during birth. One birth was complicated by meconium in the amniotic fluid, which did not result in any further complications. Birth weight was reported in only three cases and was between 3080 and 4026 grams. Three births eventually resulted in complications after delivery. One participant developed a postpartum depression after her second birth and one participant developed a uterine prolapse. In 5 pregnancies, aggravation of PD symptoms was reported; in one case, symptoms returned to baseline after delivery. No data were available on the other pregnancies.
Pregnancy and pramipexole
One pregnancy was carried out while the participant was on pramipexole to treat her PD symptoms. Although her medication use was initially halted after conception, she restarted pramipexole 3.5 weeks after conception due to the severity of her motor symptoms. Throughout the rest of pregnancy, she used 0.5–1 mg pramipexole 3dd. No anomalies were found on prenatal ultrasounds or in genetic testing. Several factors complicated this pregnancy: an episode of maternal fever in the second trimester, decreased fetal movement perceived by the mother, gestational diabetes and transient hypertension of pregnancy. Nevertheless, the pregnancy resulted in a spontaneous vaginal delivery with live birth at 40 weeks of gestation. The maternal age at delivery was 27 years and the newborn's birth weight was 4062 grams. The birth was complicated by meconium-stained amniotic fluid, which did not result in any further complications. There was no additional information about complications after delivery, but PD symptoms remained stable throughout the pregnancy.
Pregnancy and levodopa/carbidopa
One pregnancy was carried out while the participant was on levodopa/carbidopa. The 31-year-old participant originally used rotigotine and a MAO-B inhibitor but switched to levodopa/carbidopa 25/100 3dd after conception. The prenatal ultrasound scan at 12 weeks of gestation showed a borderline elevated nuchal translucency and a subchorionic hematoma. The latter was resolved at 16 weeks gestation. No anomalies were found in prenatal genetic testing. The pregnancy was complicated by maternal perception of decreased fetal movement, for which labor was induced at 40 weeks of gestation. During birth, fetal bradycardia occurred as well as an arrest of dilatation, which eventually resulted in an emergency cesarian section. The birth weight of her newborn was 3713 grams. The birth was further complicated by a postpartum hemorrhage of 1.4 liters at the surgical site. There was no additional information about progression of PD symptoms throughout pregnancy.
Discussion
We described 12 pregnancies in 9 women who went through pregnancy after onset of PD motor symptoms. 11 pregnancies resulted in a live birth, while 1 pregnancy resulted in a spontaneous abortion at 9 weeks gestation. Although some complications occurred in pregnancy, during delivery and after delivery in the 11 remaining pregnancies, this did not result in any long-term complications. Aggravation of PD symptoms was present after 5 births, although data are limited.
Pregnancy in Parkinson's disease
Our study revealed an aggravation of PD symptoms throughout pregnancy in 5 pregnancies. Unfortunately, no information about severity of PD symptoms was available in 4 pregnancies; in 1 pregnancy, symptoms remained stable and in 1 pregnancy, symptoms improved. Most women in our study did not use any PD medication during pregnancy, either because they did not receive a diagnosis yet or had not started taking medication yet. In only 3 pregnancies, medication was used prior to conception. Our findings are in line with other studies on pregnancy and PD, where some studies show an aggravation of PD symptoms,8–13 while others show stability13–16 or improvement of PD symptoms. 17 The pathophysiology behind aggravation of PD symptoms during pregnancy remains poorly understood. It could be the result of discontinuation or a change in PD medication around pregnancy,11,13 since literature suggests that symptoms typically do not aggravate as much in women who continue their PD treatment during pregnancy compared to women who discontinue their medication use. 18 Optimizing physical well-being is especially important in preparation of childbirth by the end of the third trimester. 18 Changes in PD symptoms during pregnancy could also be explained by changes in hormonal exposure during pregnancy (e.g., exposure to estrogen) or be the result of increased physical stress and physiological changes that occur during pregnancy.18,19
Within our case series, one participant used pramipexole throughout the pregnancy and one participant used levodopa/carbidopa. Complications occurred in both pregnancies, of which the most significant was an emergency cesarian section that was performed because of signs of fetal distress in a situation of failure to progress and fetal bradycardia. This occurred in the levodopa/carbidopa pregnancy. In both cases of medication use, no birth defects were reported. Levodopa has been described in literature to be the safest option for PD medication use during pregnancy.6,20 It is important to note that any complication due to levodopa/carbidopa use would be caused solely by levodopa, since carbidopa does not pass the placental barrier. 21 Previous studies show no indications of possible complications due to levodopa use.9,11,14,15,16,17 Since our study only describes one case, our findings could be coincidental. Information on the use of pramipexole during pregnancy is limited, with case reports suggesting possible safety of its use during pregnancy.8,12,13
One out of 12 pregnancies resulted in a miscarriage at 9 weeks gestation. During this pregnancy, no PD medication was used. Previous research does not show a clear link between PD and an increased risk of miscarriages or fetal anomalies.18,20
Implications for healthcare professionals
The decision to have children after being diagnosed with a chronic progressive disease has a vast impact on an individual's life. Healthcare professionals should be aware of this issue and proactively inquire if women with PD are still considering pregnancy. Pregnant women with PD should be monitored by both a neurologist and a gynecologist to adjust treatment if necessary and intervene in case of complications.
Strengths and limitations
Our study provides additional information on a topic that so far has been largely undiscovered. Data of this study could be used as an example on pregnancy in women with PD can be approached and are an important step in building towards a guideline on treatment of pregnant women with PD.
However, our study was not without limitations. First, our study sample was small and not all relevant information was available int the 13 identified cases. Second, in 10 out of 12 pregnancies, women did not use PD medication during pregnancy, making it difficult to assess the actual risk of use of PD medication during pregnancy. Third, in most cases, there was no information about long term birth outcomes of these pregnancies. Fourth, evaluation of motor symptoms during pregnancy was scarce and only described as ‘aggravated’ or ‘stable’, without any objective description of these symptoms. Most records did not describe active consultation of a neurologist throughout the pregnancy, so these data were based on information provided through gynecological consultations.
Implications for future research
Although our study adds to our current understanding of pregnancy in women with PD, does not yield any conclusive, clinically applicable results. Our study supports the importance of further multicentric studies on pregnant women with PD to prospectively collect data on complications, birth outcomes, lactation and PD severity. The relatively low frequency of pregnant women with PD calls for an international collaboration on this important topic.
Footnotes
Acknowledgments
The authors have no acknowledgments to report.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability
The data supporting the findings of this study are available on request from the corresponding author. The data are not publicly available due to privacy or ethical restrictions.
