Abstract
Background
Autopsy and in vivo molecular imaging studies suggest altered binding of the α4β2-nicotinic cholinergic receptor (α4β2-nAChR) with cognitive dysfunction in Parkinson's disease (PD).
Objective
To determine the relationship between cortical and hippocampal binding of the α4β2-nAChR with [18F]XTRA PET, a high-affinity radiotracer that enables quantification of α4β2-nAChR in these regions, and cognitive function in individuals with PD.
Methods
Individuals with PD (N = 32) and age-similar, controls without PD or dementia (N = 10) completed a cognitive assessment and one 90-min, [18F]XTRA PET scan. Metabolite-corrected arterial input function radioactivity time-activity curves were generated to obtain total distribution volume (VT) across 12 regions of interest (ROIs). [18F]XTRA binding was compared 1) between controls and people with PD and 2) between controls, persons with PD with normal cognition (PD-NC), and persons with PD with MCI (PD-MCI).
Results
[18F]XTRA binding was higher in the occipital cortex of the combined group of PD participants compared to age-similar controls. No regions showed lower binding in PD. VT with, but not without, partial volume correction was different between controls, PD-NC, and PD-MCI groups, and this was driven by higher binding in PD-MCI compared to controls. Regression of regional VT on cognitive domain T-scores, adjusting for age, showed that worse performance in visual-spatial memory tasks was associated with higher VT in the precuneus and the entire parietal cortex.
Conclusions
Higher α4β2-nAChR binding in posterior cortical regions is found in PD and associated with worse visual perception and memory, possibly due to lower receptor occupancy by endogenous acetylcholine.
Plain Language Summary
Cognitive impairment, such as problems with decision-making, memory, or other “thinking” issues, is a large source of disability in people with Parkinson's disease (PD) but there are few treatments available to address this issue. To better understand the cause of cognitive impairment—the first step in being able to design new treatments for it—we used a type of brain imaging called PET to look at the binding of a specific receptor in the brain and explored how this was associated with cognitive function in a group of people with PD. Earlier, similar studies used a type of PET scan that shows deeper parts of the brain but we used a PET scan that can better show the outer surface of the brain, called the cortex, where these receptors are more sparse but important in normal brain function. We found that binding of this receptor was higher in people with PD compared to people of a similar age without neurologic issues and that those with PD who had more cognitive impairment, showed higher binding than those with PD who did not have cognitive impairment. Also, in those with PD, higher binding was associated with more cognitive impairment on tests of visual memory, such as learning and remembering an array of pictures. This knowledge may inform the development of medications or other treatments that specifically target this receptor to treat cognitive issues in PD.
Introduction
While dementia eventually occurs in up to 80% of people with Parkinson's disease (PD) by 20 years of disease duration, 1 even mild cognitive impairment (MCI) often begins early 2 in the course of PD and contributes to poorer quality of life. 3 Given the impact of cognitive deficits on quality of life in PD, understanding the neurobiological mechanisms underlying PD-MCI and developing more effective treatment targets are research priorities. A relationship between decreased cortical cholinergic innervation and cognitive impairment in PD has been established. 4 However, the involvement of specific cholinergic receptor subtypes has not been as extensively studied, especially in PD patients with cognitive deficits. Understanding the specific cholinergic receptors involved in cognitive deficits is crucial to inform the development of targeted therapies beyond the currently available options of boosting overall cholinergic tone with cholinesterase inhibitors, which are effective in dementia 5 but have off-target side effects due to increased cholinergic signaling through many muscarinic and nicotinic receptors in and outside of the CNS. Because the tolerated doses of cholinesterase inhibitors only provide low occupancy of CNS cholinesterase, 6 targeting receptors may be more effective for modulating cholinergic pathways. For instance, is α4β2-nAChR agonism, antagonism, or partial agonism might be most appropriate depending on the stage or severity of cognitive impairment in PD as it relates to the directionality of α4β2-nAChR density at that time.
Mild cognitive impairment in PD (PD-MCI) presents differently than in other conditions like Alzheimer's disease. Executive dysfunction is nearly universally present in PD-MCI, while language comprehension or memory deficits may only occur in 20 or 45%, respectively. 2 Deficits in visual-spatial function are more prominent than in Alzheimer's disease and may be associated with development of dementia in PD. 7 This cognitive profile is explained by unique patterns of brain network dysfunction, owing to multiple, underlying pathoetiologies, including abnormal protein deposition and neurotransmitter deficits. While α-synuclein pathology in neocortical and limbic regions correlates with dementia in PD, 8 cognitive deficits occurring in earlier stages of disease correlate with neurotransmitter deficits, including decreased dopaminergic 9 and cholinergic activity. 10 In fact, disrupted cholinergic signaling has been proposed to account for more rapid cognitive decline. 11 Thus, the heterogeneity of cognitive syndromes in PD 11 may be explained by the variable involvement of neurotransmitter systems 12 and differences in regional pathology/neurodegeneration. 13
Neuropathological 14 and molecular imaging10,15 studies have shown that cholinergic innervation and nicotinic (more so than muscarinic 16 ) receptor availability in cortical regions are abnormal in PD patients with MCI and dementia. The α4β2 subtype of the nicotinic cholinergic receptor (α4β2-nAChR) is broadly distributed through the neocortex, limbic and subcortical regions and is involved in multiple cognitive processes. 17 Most notably, attention and working memory have been associated with α4β2-nAChR agonism in the dorsolateral prefrontal cortex, 18 and visual-spatial learning and memory has been associated with α4β2-nAChR agonism. 19 In extra-striatal regions, α4β2-nAChRs are mostly found pre-synaptically, but also post-synaptically in the subiculum and entorhinal cortex and in neocortical areas such as the outer and upper middle layers of the motor cortex, middle layers of striate cortex, and outer and middle layers of the sensory cortex. 20 α4β2-nAChRs localize to a large number of neurons throughout the cortex 21 rather than being restricted to large pyramidal neurons like α7β*-nAChR receptors. The cortical and hippocampal innervation by cholinergic neurons is thought to be important for multiple cognitive processes such as attention, working memory, and spatial learning/memory, but given the lower concentration of α4β2 in these regions relative to the thalamus, higher affinity α4β2-nAChR radioligands than those that were previously available are needed to image α4β2 receptors in these regions. 22 For example, PET studies of 2-[18F]-FA showed that reduced subcortical (midbrain) α4β2-nAChR binding was associated with worse global cognition, but cortical α4β2-nAChR imaging was limited by low signal-to-noise ratio related to a ligand affinity that could reliably measure α4β2-nAChR only in high-binding regions like the thalamus and midbrain. 15
To address the need to image cortical and hippocampal α4β2-nAChR, a more recently developed α4β2-nAChR radioligand, 23 [18F]XTRA ((-)-7-methyl-2-exo-[3′-(2-[18F]fluoropyridin-3-yl)-5′-pyridinyl]-7-azabicyclo[2.2.1]heptane, [18F](-)-6b), with high binding affinity (Ki = 0.06 nM) and lipophilicity (LogD7.4 = 0.67) was evaluated and showed more rapid and favorable pharmacokinetics compared to the previously developed radioligands (e.g., 2-[18F]-FA) for human imaging of α4β2-nAChRs. 24 [18F]XTRA has demonstrated enhanced extra-thalamic affinity and has sufficiently fast kinetics for derivation of distribution volume (VT) with 90-min emission scans, as compared to up to 7 h with a prior α4β2-nAChR PET radioligand. 15 The regional distribution of [18F]XTRA binding was consistent with the known distribution of α4β2-nAChRs in humans.25,26
Using this high-affinity radioligand, we measured the differences in [18F]XTRA binding between persons with PD and controls and focused on cortical and limbic regions relevant to cognitive dysfunction in PD. Because reduced presynaptic cholinergic innervation has been associated with worse cognitive performance,27,28 we also tested whether lower α4β2-nAChR density, imaged with [18F]XTRA binding, in specific regions would be associated with worse performance in various cognitive domains. These hypothesized cognition-regional associations include: attention – prefrontal cortex 29 ; executive – prefrontal cortex, anterior cingulate, posterior cingulate 30 ; verbal memory – mesial temporal cortex (MTC), hippocampus 31 ; visual-spatial perception/construction – parietal cortex, occipital cortex 32 ; visual-spatial memory – parietal cortex, precuneus. 33 Additionally, we investigated whether binding in these regions would be lower in people with PD who had MCI (PD-MCI) than in people with PD who were cognitively normal (PD-NC).
Methods
This was a cross-sectional, observational study with data collected from two visits: a neuropsychological evaluation and a PET scan acquired after bolus administration of [18F]XTRA.
Subjects
The study was approved by the Johns Hopkins Institutional Review Board and complied with the declaration of Helsinki, including informed consent. The target sample size of 30 participants with PD was calculated based on the anticipated strength of correlation (r = 0.6) between midbrain 2-[18F]-FA binding and performance on a cognitive measure BPND and cognitive performance, 15 with alpha at 0.001 and power at 80%. Participants with idiopathic Parkinson's disease based on UK Brain Bank Criteria, age 50 and above were recruited serially from the Parkinson's and Movement Disorders Center through direct clinic recruitment (by K.M.) or screening patients of other collaborating providers under a HIPAA waiver. Potential participants were excluded if they had secondary parkinsonism or parkinsonism as a symptom of a neurodegenerative condition other than PD. Potential participants were excluded for claustrophobia and contraindications to MRI (metal implant), evidence of stroke or tumor on the MRI scan, meeting criteria for dementia based on Clinical Dementia Rating (CDR) of 1 or greater and use of anticholinergic or CNS-acting cholinesterase inhibitor agents or use of nicotine. They were also excluded if, in the judgment of the investigator, cognitive deficits prevented them from providing informed consent, as outlined by institutional standards. PD participants were allowed to be taking a stable dose of a selective serotonin reuptake inhibitor or serotonin-norepinephrine reuptake inhibitor. Age similar controls, included in a previously published study, 25 were used for comparison. The controls had no diagnosis of a psychiatric or neurological disorder and were determined to be cognitively normal as assessed by the Clinical Dementia Rating Scale (CDR = 0). 34 The combination of a MoCA score less than 26 35 and participant or informant interview indicating subjective cognitive impairment was used to establish the presence or absence of level I PD-MCI in participants with PD. 36
Cognitive assessments
In the PD participants, cognitive assessments were performed 30–60 days prior to the [18F]XTRA PET scan by a licensed psychometrician overseen by a neuropsychologist. The duration of the battery was 90–120 min. The PD participants were tested in the optimally medicated state, as defined by the participant reporting they feel in the medication “on” state. If the patient encountered “wearing-off” of dopamine replacement medication effect during testing, they were given a break to re-dose. Hopkins Verbal Learning Test – Revised 37 and Brief Visuospatial Memory Test – Revised 38 were performed to assess verbal and visual-spatial learning and recall, respectively. The Trail-Making test (Parts A and B) 39 assessed processing speed and set-shifting. The Speed and Capacity of Language Processing (SCOLP) 40 was used to evaluate word knowledge and comprehension, while the discrepancy score (word knowledge minus comprehension speed) was used as a surrogate for decline from pre-morbid knowledge. The Boston Naming Test 41 was used to evaluate semantic language processing. Letter (F, A, S) and category (animals, supermarket) fluency 42 were used to assess verbal fluency, which combines working memory, attention, and semantic knowledge. The D-KEFS Tower Test 43 was used to evaluate sequential planning as part of executive function. Visual-spatial perception and construction were tested with cube copy (from Montreal Cognitive Assessment (MoCA) 44 ) and the Clock Draw Task. 45 Cognitive performance measures were calibrated and T-scored based on sex, age, and education (years) using the Calibrated Neuropsychological Normative System (PAR, https://paa.com.au/product/cnns/), which assigns a T-score based on normative data from the Johns Hopkins Aging, Brain Imaging, and Cognition Study incorporating demographic covariates in regression modeling. Composite cognitive domain scores were generated as the mean T-score of domain-specific tests, except for visual-spatial perception/construction, which did not have calibration data (Supplemental Table 2). The MoCA was performed at baseline in the PD participants.
[18F-XTRA] PET scan
PET scans were performed after obtaining a structural MRI scan (3 T Philips dStream Ingenia Elition, Best, Netherlands). As is typically done in PET neurochemical imaging studies, participants were instructed to withhold antiparkinsonian medications after 10 PM on the night prior to the PET scan, which began at 10:45 AM. The methods for synthesis of [18F]XTRA, PET acquisition and analysis have previously been published. 46 PET scans were obtained on the High-Resolution Research Tomograph (Siemens Healthcare, Knoxville, TN). Each subject was fitted with a thermoplastic mask modeled to their face to reduce head motion during the PET scan. Radial arterial lines were placed by an anesthesiologist for serial arterial blood sampling to determine the input function corrected for the amount of unchanged radiotracer in arterial plasma. An intravenous line was placed in the opposite arm to inject the radioligand. A 6-min transmission scan was acquired using a 137Cs point source prior to the emission scan. Then, the radioligand [18F]XTRA was injected and the emission scan began immediately. The emission scan data were collected in list mode for at least 90 min. After the first 7 PD participants were scanned for 180 min, it was determined that 90 min of data were sufficient for quantification. The serial arterial sampling protocol was 0.4 mL samples drawn every 5–6 s for the first 1.5 min, every 30 s until minute 3, then every 5 min until 90 min. Additional samples were obtained in a subset of arterial blood sample time points to determine the amount of remaining parent radioligand fraction via high performance liquid chromatography (HPLC). Emission PET data were reconstructed using the iterative ordinary Poisson ordered-subset expectation-maximization algorithm, with correction for radioactive decay, dead time, attenuation, scatter, and randoms, and binned into 30 (90-min scans) or 48 (180-min scans) frames (four 15 s, four 30 s, three 1 min, two 2 min, five 4 min, and twelve or thirty 5 min). The reconstructed image volume spanned 31 cm x 31 cm transaxially and 25 cm axially, with an image matrix of 256 × 256 × 207 voxels (voxel size of 1.22 × 1.22 × 1.22 mm3).
PET region of interest and voxel-wise analyses
Region of interest (ROI) parcellation was performed on a T1 volumetric MRI sequences using the FreeSurfer software (version 7.1, Boston, MA) according to standard anatomical boundaries. 47 The twelve ROIs were chosen based on 1) proposed localization of cognitive functions of interest in PD: prefrontal cortex – attention 29 /executive function 30 ; anterior and posterior cingulate – working memory 48 ; hippocampus and mesial temporal cortex – verbal learning 31 ; and occipital / parietal cortex and precuneus – visual-spatial perception and memory,32,33 respectively, 2) regions with high levels of cholinergic innervation (midbrain, striatum), and 3) regions hypothesized to have low nicotinic receptor binding as a possible reference region to measure non-specific binding (corpus callosum and cerebellum). The ROIs for the midbrain, mostly reflecting binding in the ventral tegmentum (VTA) and lateral geniculate nucleus, were defined on averaged PET images (of 40–90 min frames) using the threshold mode of a locally developed ROI definition tool (IDAE, version 2, https://github.com/IDAETeam/The-Integrative-Data-Analysis-Environment-for-Brain-PET). The final midbrain ROIs (volumes = 0.26 mL) were projected on the matching MRI to visually confirm the spatial agreement of the ROI with published locations of VTA and lateral geniculate nucleus.
Co-registration of the PET frames for post-hoc head motion correction and MRI to PET co-registration was performed in Statistical Parametric Mapping, version 12 (SPM12; Institute of Neurology, London). The ROIs were transferred from MRI to PET space using the co-registration parameters to generate time-activity curves (TACs) of the regions. The metabolite-corrected plasma TACs were obtained as the products of the total plasma TACs and the metabolite fractions that were fitted by a first-order Hill's equation. The regional TACs were obtained for the 12 ROIs (summed across both hemispheres within ROI). Total distribution volume (VT) of regions were obtained using Logan plasma-reference graphical analysis method 49 with metabolite-corrected arterial input function since prior work showed a high correlation between VT values derived from plasma-reference graphical analysis and two tissue compartment models. 25 The starting point for the graphical analysis was set at the asymptote of 20 min. 23 The primary outcome measure was VT due to the lack of a human brain region known to be completely devoid of the α4β2-nAChR. Even the corpus callosum has been shown to have displaceable α4β2 nACHRs. 50 Since there is not a clear brain reference region for measurement of non-displaceable binding with [18F]XTRA 51 and displacement by nicotine in the corpus callosum has been shown with another α4β2-nAChR radioligand, 50 we were unable to use a reference tissue method such as non-displaceable binding potential (BPND). Distribution volume ratio (DVR) also could not be used because we could not assume uniform VND (non-specific binding) across gray matter regions. We noted that TACs of the thalamus and midbrain continued to increase up to 180 min in participants who had 180 min scans, suggesting the lack of dissociation of the tracer in these regions. Therefore, we excluded these regions from further analyses This emphasized [18F]XTRA's utility as a high affinity ligand for imaging of cortical and limbic regions in time frames tolerated by most patients with PD in the unmedicated state. Other ligands such as 2-[18F]-FA are more optimal for imaging regions of very high binding.
We report the VT values both with and without partial volume correction (PVC) as the primary outcome measures. While we applied one of the most widely used PVC methods available, there is no consensus or standardization of the use of correction for partial volume effects. 52 While PVC can mitigate some of the bias introduced by differential atrophy between groups in neurodegenerative research, some studies have shown that some PVC methods can introduce noise and reduce precision.53,54 The Müller-Gärtner method 55 was applied, given its frequent use in brain PET and SPECT to adjust for partial volume effects. Because the cholinergic-relevant midbrain ROI could not be determined with MRI segmentation (the ROIs were identified on the PET scan), we were unable to apply partial volume correction here. Parametric VT images of [18F]XTRA were transferred to the MNI space smoothed by a Gaussian kernel of 8 mm, full-width at half-maximum 56 to perform voxel-wise between-group and correlational analyses. The spatial normalization was achieved by applying the parameters of the PET-to-MRI co-registration and spatial normalization of the MRI images in one step in Statistical Parametric Mapping version 12 (SPM12).
Statistical analysis
Mean values with standard deviations or proportions were calculated for demographic variables and were compared and modeled using STATA 14 (StataCorp. 2023. Stata Statistical Software: Release 18. College Station, TX: StataCorp LLC). A two sample Student's t-test or test of proportions was performed to test whether mean demographic variables were different between controls and participants with PD. Our primary outcome was ROI VT values given the justification for ROIs provided above. Wilcoxon sign-rank tests were used to test whether [18F]XTRA VT was different for each ROI for the combined PD compared to the control group. Participants with PD were then separated by MCI status and the distribution of ROI [18F]XTRA VT between these three groups (Controls, PD-NC, PD-MCI) was tested for normality with the Shapiro-Wilk test. All VTs within the Controls and PD-MCI group were normally distributed, but within the PD-NC group, they were non-normally distributed in the cerebellum, striatum, or prefrontal cortex, so Kruskall-Wallis tests were used to test whether variance of the demographic variables and [18F]XTRA VT (with and without PVC) was different across the three groups: Controls, PD-NC, and PD-MCI. Leven's test showed that the assumption of heterogeneity was violated in that the variance for the PD-MCI group was larger than that of the controls. However, we proceeded with use of means testing because a) with this sample size, heterogeneous variances should bias us toward type II statistical error (false negative) and b) the difference was below a factor of 4-times the smallest variance. We also performed a between-group, voxel-wise analysis of variance in VT between Controls, PD-NC, and PD-MCI, with a pre-specified cluster-level, family-wise error-corrected threshold of p < 0.05, a peak voxel uncorrected threshold of p ≤ 0.001 and extent threshold (k) > 50 voxels. We performed these analyses with and without adjusting for age, then also repeated them using PVC VT.
To evaluate the relationship between regional α4β2-nAChR binding and cognitive function, we assessed the correlation between a global measure of cognitive function (MoCA) and each of the 12 a priori ROls VT. Using the entire cohort of participants with PD, we then performed a multiple linear regression for each cognitive domain using the domain composite T-score as the dependent variable and the hypothesized ROI VT as the independent variable, adjusting for age. To address whether this relationship could be confounded by overall disease severity, we repeated the analyses also including disease duration as a covariate. As an exploratory, secondary analysis, we performed voxel-wise regression analyses using the multiple regression design of SPM12, of [18F]XTRA VT on cognitive domain T-scores, adjusting for age, given the previously described relationship between age and [18F]XTRA VT. 25 The significance criteria consisted of a cluster-level, family-wise error (FWE)-corrected threshold of p ≤ 0.05 and a peak voxel uncorrected threshold of P ≤ 0.001 and extent threshold (k) > 50 voxels.
Results
We enrolled 32 participants with PD (54 to 76 years) with moderately severe motor disease overall (Table 1), 10 of whom met Level I criteria for PD-MCI. We compared the PET data for the PD patients to 10 age-similar controls (51–87 years) 25 who were cognitively normal (Clinical Dementia Rating Scale = 0). The controls were slightly, but not statistically significantly, older than the PD group (Table 1). Years of education did not differ significantly between those PD, with or without PD-MCI, nor did levodopa dose or treatment with antidepressants. As expected, PD participants with PD-MCI had a higher MDS-UPDRS Part III ON and OFF medications and, by definition, MoCA was worse in this group relative to those without PD-MCI. On the cognitive battery, performance across all cognitive domains was significantly worse in patients with PD-MCI compared to those PD patients without PD-MCI (Table 1). Means and standard deviations for the mass, specific activity, and radiotracer dose for each group can be found in Supplemental Table 1. There were no between-group differences in the radiotracer characteristics.
Demographics and clinical characteristics of study participants.
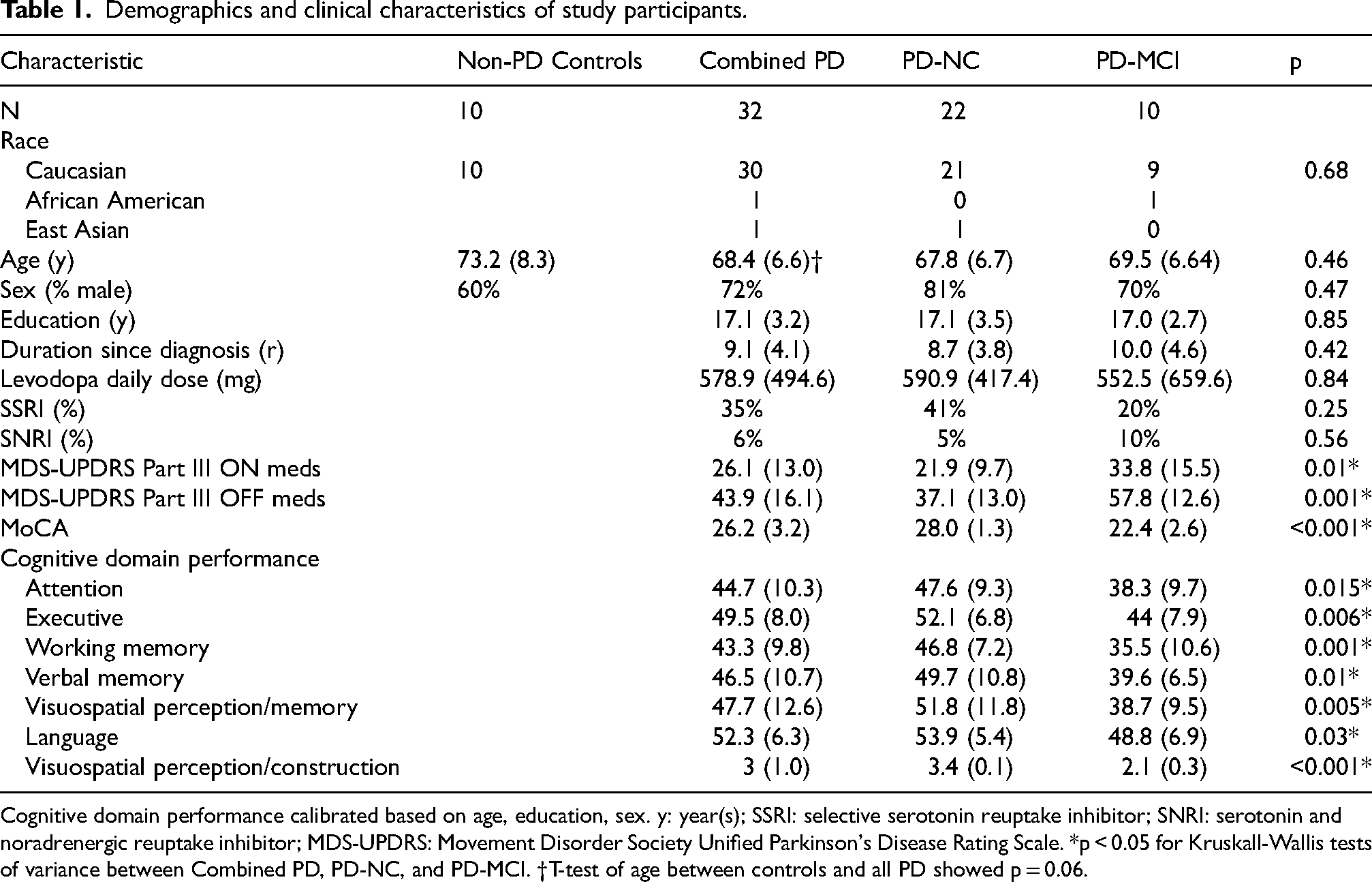
Cognitive domain performance calibrated based on age, education, sex. y: year(s); SSRI: selective serotonin reuptake inhibitor; SNRI: serotonin and noradrenergic reuptake inhibitor; MDS-UPDRS: Movement Disorder Society Unified Parkinson's Disease Rating Scale. *p < 0.05 for Kruskall-Wallis tests of variance between Combined PD, PD-NC, and PD-MCI. †T-test of age between controls and all PD showed p = 0.06.
We first analyzed group-wise comparisons of VT from ROIs between PD versus non-PD controls and then, between controls, PD-NC, and PD-MCI. As seen in Table 2, total volume of distribution (VT) measured by [18F]XTRA was higher in the occipital cortex in the combined PD participants compared to non-PD controls (13.1 ± 2.2 vs. 11.4 ± 2.2, p = 0.047), but not in any of the other regions examined without PVC. With PVC, VT was higher in PD than controls in the mesial temporal cortex, occipital cortex, and precuneus. In comparing ROI VTs without PVC, one-way ANOVA did not show a statistically significant difference in VT variance between controls, PD-NC, and PD-MCI in any region (Table 3). With PVC, there were differences in the occipital cortex and precuneus (Table 3, Figure 1), where post-hoc means testing showed a statistically significant difference between controls and people with PD-MCI. In each case, higher [18F]XTRA VT was seen in PD-MCI relative to controls.
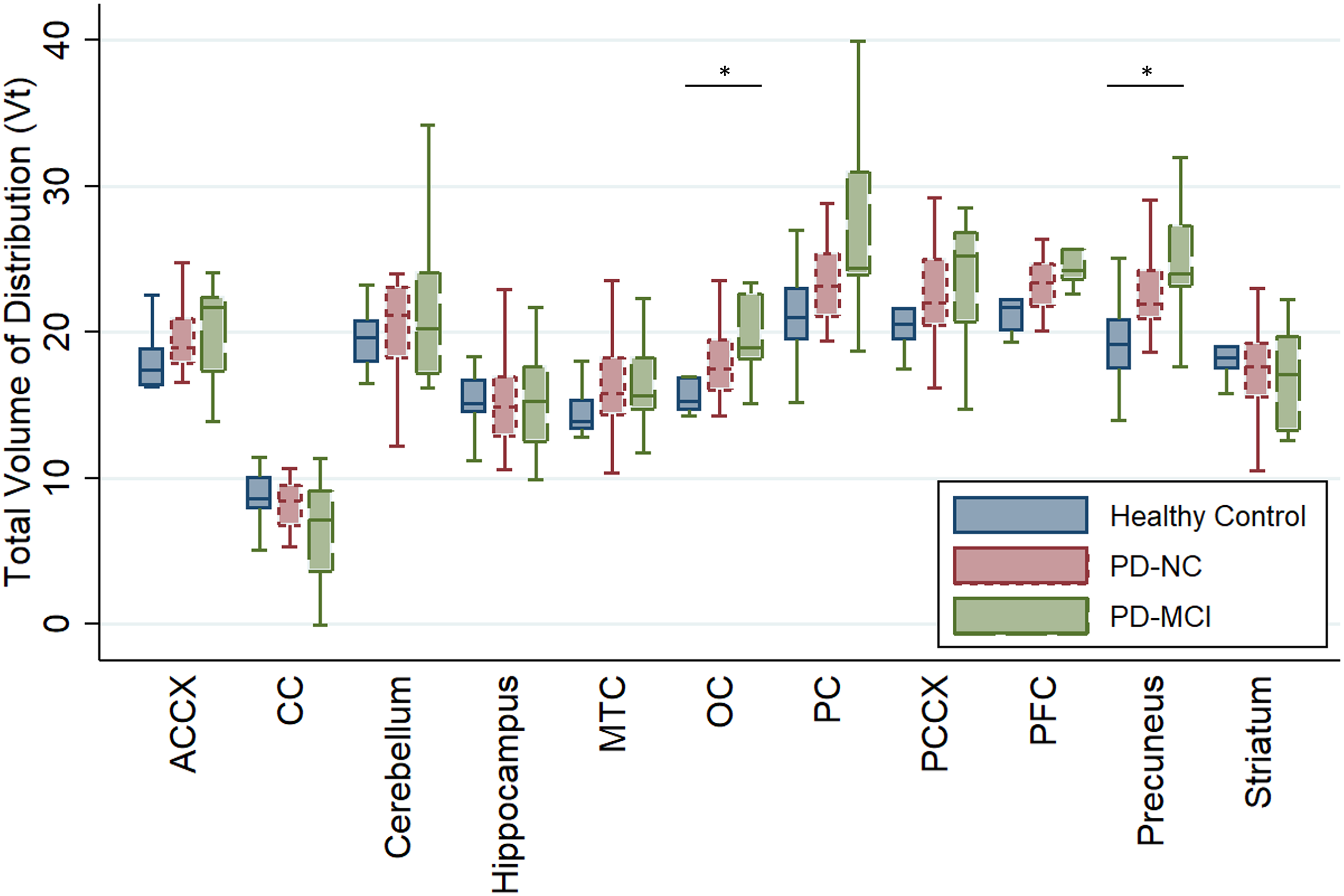
[18F]XTRA VT with PVC in healthy elderly controls, Parkinson’s disease with normal cognition (PD-NC), and Parkinson’s disease mild cognitive impairment (PD-MCI) across regions of interest pertinent to cognitive function in PD. Box shows 75th-25th quartile range, line shows median, and upper and lower whiskers show 75th or 25th quartile plus/minus 1.5 times the interquartile range, respectively. *p < 0.05 for Kruskall-Wallis test of variance in in VT variance between Controls, PD-NC, and PD-MCI. Of note, combined PD group of PD-MCI + PD-NC showed higher VT in MTC, OC, and Precuneus (p < 0.05).
Mean [18F]XTRA VT in controls and Parkinson's disease participants with and without partial volume correction.
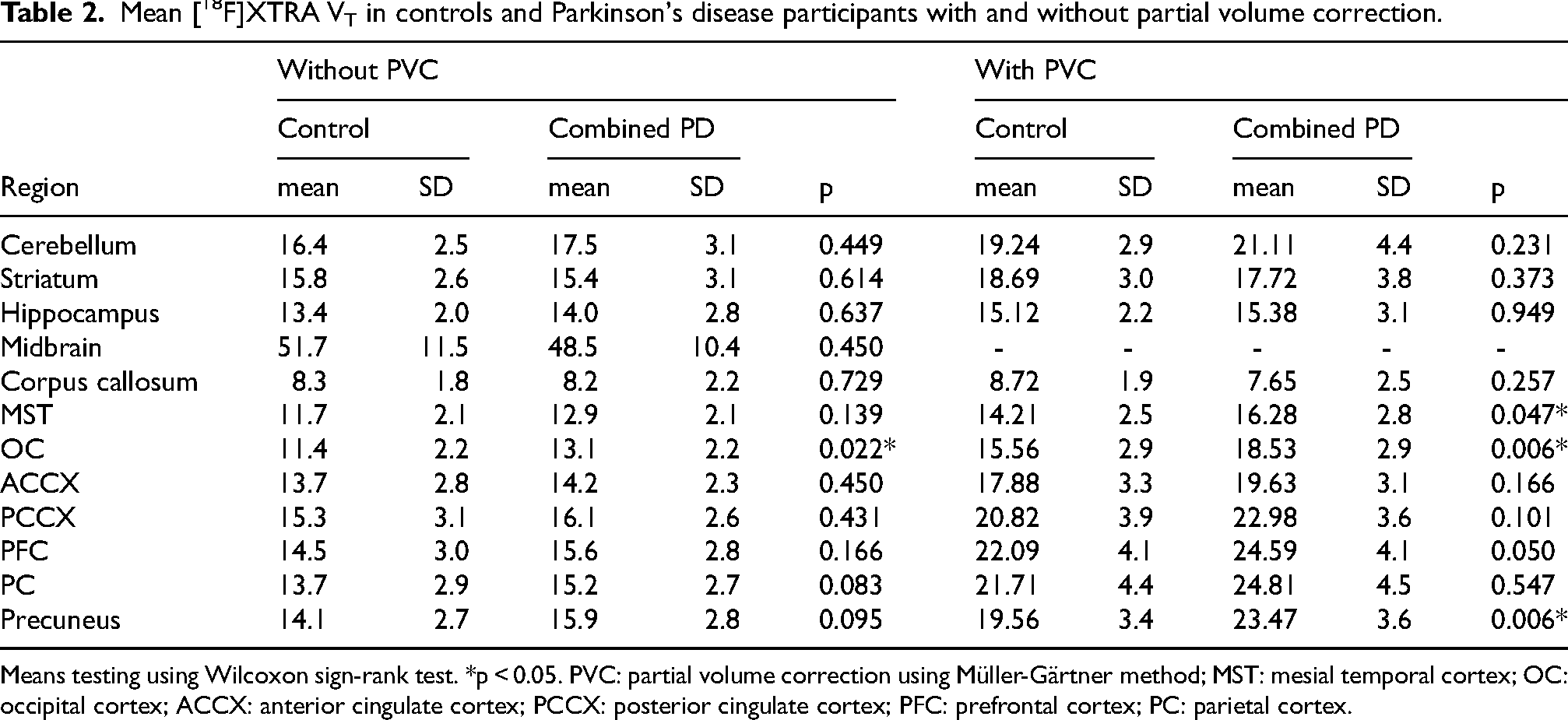
Means testing using Wilcoxon sign-rank test. *p < 0.05. PVC: partial volume correction using Müller-Gärtner method; MST: mesial temporal cortex; OC: occipital cortex; ACCX: anterior cingulate cortex; PCCX: posterior cingulate cortex; PFC: prefrontal cortex; PC: parietal cortex.
Results of Kruskall-Wallis tests comparing [18F]XTRA VT across controls, PD normal cognition (PD-NC), and PD with mild cognitive impairment (PD-MCI), with and without partial volume correction.
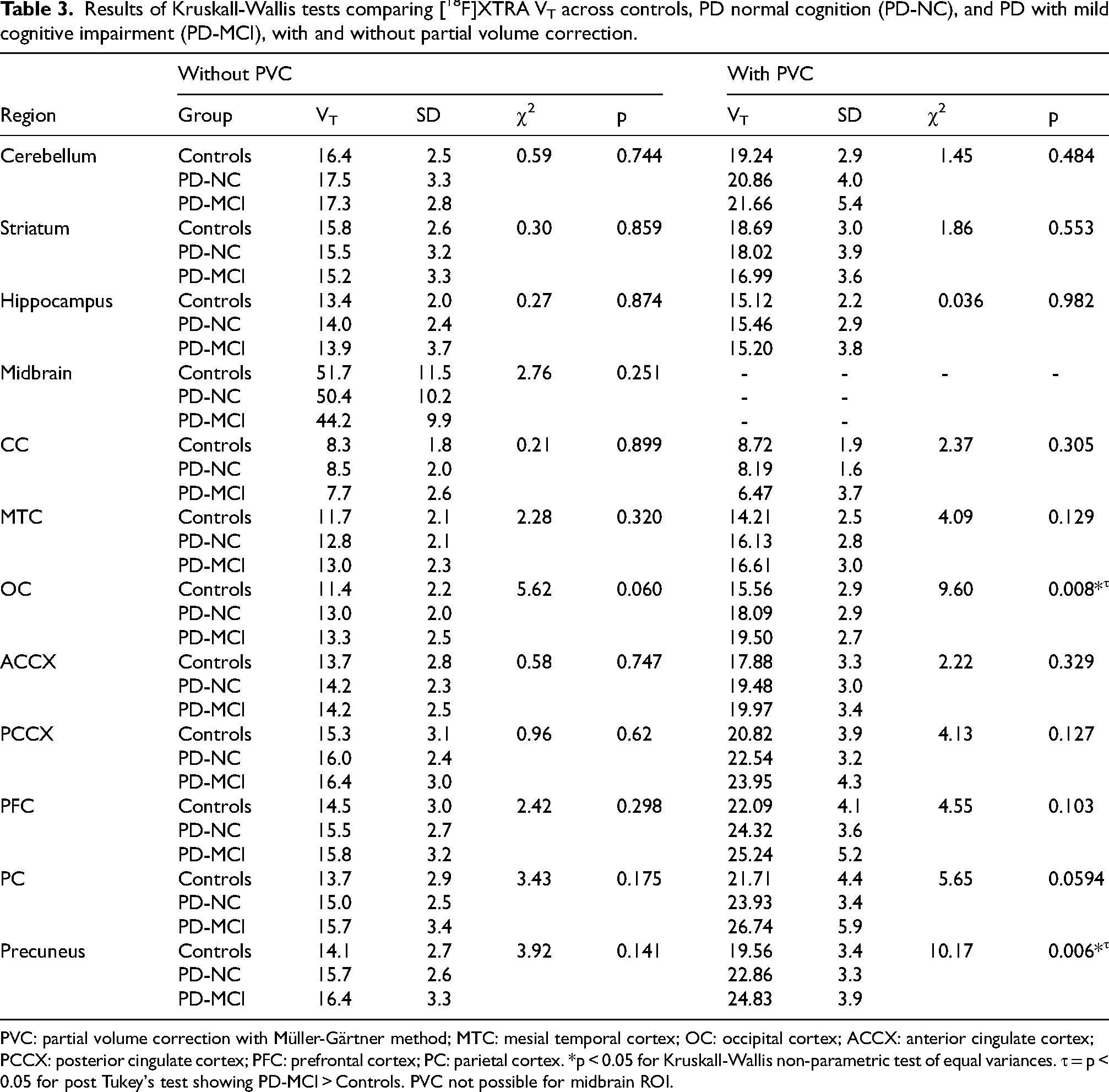
PVC: partial volume correction with Müller-Gärtner method; MTC: mesial temporal cortex; OC: occipital cortex; ACCX: anterior cingulate cortex; PCCX: posterior cingulate cortex; PFC: prefrontal cortex; PC: parietal cortex. *p < 0.05 for Kruskall-Wallis non-parametric test of equal variances. τ = p < 0.05 for post Tukey's test showing PD-MCI > Controls. PVC not possible for midbrain ROI.
We performed exploratory, voxel-wise comparisons of VT or VT after PVC between controls, PD-NC, and PD-MCI as well as between controls and the combined PD group. These analyses did not reveal statistically significant clusters. These voxel-wise analyses were also then modeled adjusting for age. Again, no statistically significant clusters were found where VT differed between groups.
Correlation between [18F]XTRA and cognitive function
Restricting our analysis to all PD participants (with and without MCI), we next assessed the relationship between ROI VT values of [18F]XTRA and cognitive function. When evaluating region-specific associations between [18F]XTRA VT and composite T-scores from five cognitive domains calibrated for age, sex, and education, multiple linear regression analyses revealed an association between visual-spatial memory domain score and VT in the parietal cortex (β=-1.78, p = 0.025) and the precuneus (β=-1.69, p = 0.033), indicating higher [18F]XTRA VT was associated with worse cognitive performance (Table 4). With PVC, these associations remained similar in strength. Though our cognitive domain T-scores were already adjusted for age, to determine whether the relationship between age and [18F]XTRA VT affected the results, we also then repeated these analyses using age as a covariate and found that it did not alter the direction or magnitude of association (Supplemental Table 3). In a sensitivity analysis to test for confounding by disease duration, we re-ran these models adding disease duration as a covariate and the association between visual-spatial memory and parietal VT (β=-1.713, p = 0.037) and precuneus VT (β=-1.62, p = 0.046) persisted. The coefficients for disease duration or age were not significant in any model. Voxel-wise analysis between VT and cognitive domain composite T-scores, with family-wise error (FWE) correction, did not show statistically significant associations.
Association between regional [18F]XTRA VT and cognitive performance with and without partial volume correction.
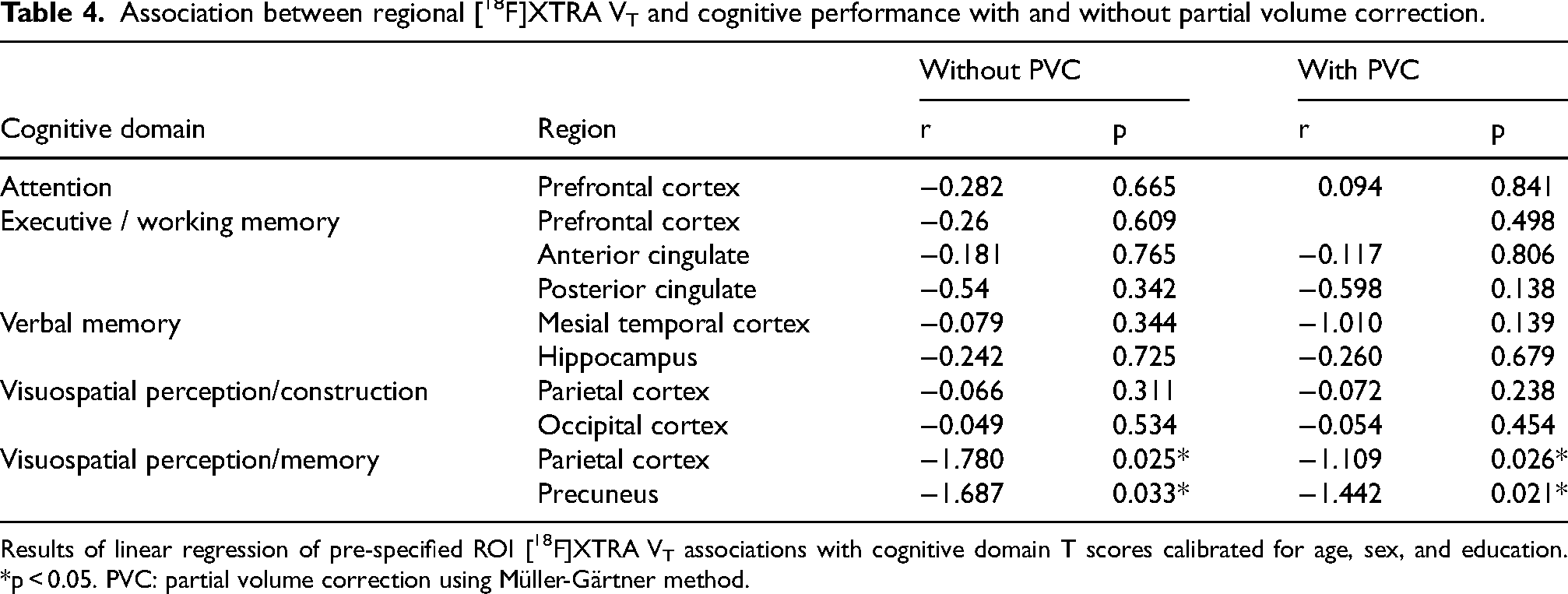
Results of linear regression of pre-specified ROI [18F]XTRA VT associations with cognitive domain T scores calibrated for age, sex, and education. *p < 0.05. PVC: partial volume correction using Müller-Gärtner method.
Discussion
It has been acknowledged for some time that cholinergic deficits may be even greater in PD with dementia than in Alzheimer's disease, 57 but the relationship between cognitive dysfunction and specific, potentially targetable, nicotinic cholinergic receptor subtypes remains less clear. The availability of nicotinic receptor radiotracers with improved imaging properties has made it possible to study the relationship of specific cholinergic receptor subtypes with cognitive domains in vivo, at early stages of cognitive impairment such as MCI. Our study adds to that growing literature15,58 by showing that when using a high-affinity α4β2-nAChR radioligand that can measure a greater dynamic range of binding in cortical regions than radioligands previously used, occipital cortex, mesial temporal cortex, and precuneus binding is higher in PD relative to controls when adjusting for the partial volume effects to correct for the effects of cerebral atrophy. Within the PD cohort, there is higher binding in participants with more cognitive impairment in the parietal cortex and precuneus, cortical regions where cholinergic denervation is hypothesized to be clinically relevant to cognitive deficits in PD. 59 The high variance of binding within our cognitive groups (PD-NC, PD-MCI) may be explained by our findings demonstrating an inverse association between cognitive performance on tasks known to be affected in PD, including visual-spatial perception/memory, and α4β2-nAChR binding in regions known to be involved in successful performance of these tasks.
The finding of an association between cognitive performance and α4β2-nAChR binding has been suggested previously, but the direction of this association has varied across studies. Previous studies have reported cortical cholinergic deficits in people with PD using radiotracers that measure cholinergic turnover (cholinesterase)10,60 or cholinergic terminal density (vesicular acetylcholine transporter), 28 but more recent preliminary results may suggest a sequence of cholinergic upregulation in earlier stages of PD.61,62 If nicotinic receptors follow these bidirectional changes, there may be a complicated relationship between α4β2-nAChR binding and cognition. Differences in the imaging properties between the α4β2-nAChR radiotracers used in the initial PD studies (e.g., 2-[18F]FA-85380) and more recent studies may contribute to the variability in results, as discussed below. Specifically, Meyer et al. 15 showed that 2-[18F]FA-85380 was lower in patients with PD-MCI than in controls in several cortical regions where we found an increase: the occipital lobe and in the precuneus region of the parietal cortex. They found correlations between midbrain binding and the mini-mental status examination, as well as between midbrain, temporal lobe, and cerebellum and the DemTect cognitive battery (which focuses mostly on executive function), such that higher binding was associated with better performance. 15 We suspect that these findings are due to differences in the radiotracers used; 2-[18F]FA-85380 has a lower BPND in cortical regions 23 and the cortical measurements may be more susceptible to noise. For example, the greatest differences between groups and the regions with strongest correlations with a cognitive measure were the midbrain and cerebellum, likely due to the 2-[18F]FA-85380 radiotracer having a greater dynamic range of sensitivity for high-binding regions like the midbrain, thalamus, and cerebellum. Another study, using 5-[123I]-IA85380 SPECT, also demonstrated positive correlations between cognitive performance on word list intrusions and the Boston Naming Test (a test of word retrieval in response to visual stimuli) and α4β2-nAChR binding in non-demented people with PD. 63 Again, most of these correlations were in subcortical regions and thalamus, likely due to lower binding potential of the 5-[123I]-IA85380 radiotracer to cortical receptors. Another 5-[123I]-IA85380 SPECT study showed reduced binding in the thalamus, but non-statistically significant reductions of 3% and 5% in the occipital and frontal cortices. 64 Yet another 5-[123I]-IA85380 SPECT study in cognitively normal people with PD showed reduced binding in the brainstem and frontal cortex, but not the parietal or occipital cortex, relative to controls. 65 The lower spatial resolution of SPECT imaging, relative to PET on the HRRT and differences in radiotracer properties and cognitive outcomes, could explain correlations between cortical α4β2-nAChR binding and cognitive performance that are lacking or different from our results. Contradicting the idea that disease progression is associated with lower α4β2-nAChR binding, another study showed that greater α4β2-nAChR binding was positively associated with disease duration in “cognitively intact” people with PD and that binding was higher in early PD than in controls, 66 whereas the Meyer et al., 22 Oishi, 65 Fujita, 64 and Lorenz et al. 63 studies suggested lower α4β2-nAChR binding with cognitive manifestations of advancing disease. In addition, increased occipital cortical binding of 5-[123I]-IA85380 SPECT was observed in patients with dementia with Lewy bodies compared to controls, and this increase was even greater in those with recent visual hallucinations. 67 It is quite possible that the lower spatial resolution for cortical regions with SPECT, causing increased noise in detecting differences between groups in radioligand binding, and the lower affinity of other PET radiotracers (2-[18F]FA-85380) explain the differences between some of these studies and our results. The affinity and binding potential of [18F]XTRA is optimized for examining α4β2-nAChR density in cortical regions and thus our findings in these regions are important to compare with prior studies using lower affinity tracers or lower resolution techniques.
The inverse association between α4β2-nAChR binding by [18F]XTRA and visual-spatial performance in PD in the current study is in the opposite direction of association found in other studies mentioned above, and could be explained by at least two mechanisms: 1) compensatory up-regulation of pre-synaptic α4β2-nAChR expression to offset cognitive dysfunction caused by spreading synucleinopathy or other mechanisms, or 2) lower levels of endogenous acetylcholine in those with worse cognition that would result in less competition for binding to the α4β2-nAChR and higher [18F]XTRA binding. Regarding the first explanation, recent unpublished data in abstract form have shown a subgroup of PD patients in which [18F]FEOBV, a ligand specific for presynaptic cholinergic terminals, is reduced in the posterior cortex, relative to controls, but increases were seen in the cerebellum, cholinergic forebrain, and subcortical grey matter. 61 A prior publication also using [18F]FEOBV to image cholinergic terminals also showed areas of both increased and decreased binding in early stage PD patients. 62 Thus, an “attempted compensation” that is failing to sustain normal cognitive function could explain our findings of higher α4β2-nAChR binding in posterior cortical regions. On the other hand, some of these findings are only in abstract form and the posterior cortex still shows reduced cholinergic terminal density, which may be more supportive of possibility #2: Regional cholinergic denervation with lower synaptic concentrations of acetylcholine could allow an increased number of receptors available for [18F]XTRA binding, explaining higher binding in PD versus controls and the association of higher α4β2-nAChR binding with worse cognitive function in PD. Supportive of this relationship, another radioligand with high affinity to α4β2-nAChR, (-)-[18F]flubatine, was displaced by a potent partial agonist, varenicline (Ki = 0.11–0.17 nM) 68 and was displaced by a physostigmine-induced increase in acetylcholine in human studies. 69 In fact, [18F]XTRA binding was decreased by administration of a α4β2-nAChR partial agonist in non-human primates, 23 but human studies are needed to confirm the sensitivity of [18F]XTRA binding to endogenous acetylcholine concentrations as a possible mechanism.
The relevance of the higher α4β2-nAChR to cognitive deficits in PD is supported by our observation that the visual-spatial perception/memory domain score showed a statistically significant association with α4β2-nAChR binding in the parietal lobe and precuneus, known to have important roles in spatial function. 33 In addition to attentional deficits, visual-spatial deficits are specifically seen more in Lewy body disorders than in Alzheimer's disease and are specifically associated with cholinergic denervation as well. 12 Accordingly, the brain region and cognitive domain specificity of our finding is consistent with current hypotheses regarding the role of cortical cholinergic denervation in cognitive deficits observed in PD, as reviewed in Bohnen et al. 70 While we observed a correlation between higher α4β2-nAChR binding and cognitive deficits, the opposite result (lower binding) was seen with a first-generation, lower-affinity α4β2-nAChR PET radiotracer that has less sensitivity for imaging extra-thalamic structures, including the cortex. 22
There are some limitations to consider when interpreting these results. The PD participants in the present study ranged from having normal cognitive function to PD-MCI, but did not meet criteria for dementia. By not directly studying patients with PD dementia or dementia with Lewy bodies, we do not know whether the directionality of this association persists as cognitive impairment continues, or whether the positive association between higher [18F]XTRA binding and visuo-spatial perception/memory in our cohort would reverse with more severe impairment. Also, the uneven sample sizes between controls, PD-NC, and PD-MCI could affect the assumption of equal variances and the PD-MCI group did violate Levin's test of homogeneity, but we proceeded with means testing because heterogenous variances should bias us toward a type II statistical error (false negative) and the difference was below a factor of 4 times the smallest variance. One limitation of the radioligand used in our study, [18F]XTRA, is that it has three active radiolabeled metabolites in humans. 25 One slightly lipophilic metabolite, unlike the polar metabolites, was detected in plasma and brain at similar fractions (6.1% and 7.2%, respectively at 30 min post-injection) in mice. 24 However, we assumed that the fraction of the radioactive metabolite was negligible in human and non-human brains since the plasma fractions were negligible even at 90 min post-injection (less than 1%; i.e., the radioactive metabolites of [18F]XTRA remained outside the brain). 25 To address this, we corrected the arterial input function for the metabolite fraction. The challenging study design also has potential limitations in that PD participants were studied in the “off” medication state for the PET scans to eliminate the variable effect of dopamine replacement dosage across patients, and because levodopa-induced dyskinesia would cause significantly more motion within and between acquisition frames in a cohort where motion is already an issue. By doing so, we do not know whether α4β2-nAChR binding would be different in the dopamine replete state and thus show different associations with cognitive tasks performed in the medication “on” state. However, we believe that the added distraction and discomfort from motor symptoms while performing cognitive tasks in the medication “off” state would negatively impact the validity of those results, and that this impact would be larger than the small effect of dopamine replacement 71 on cognitive performance. Further, cognitive testing in the med “on” state should reflect deficits related to mechanisms other than dopaminergic depletion (such as cholinergic changes) and thus would potentially isolate the cholinergic effect we aimed to study. Lastly, we tested hypotheses regarding [18F]XTRA VT differences between groups and associations with cognitive domains and ROIs that were proposed a priori without adjusting for multiple comparisons in the primary analysis. However, these early findings in a patient sample including individuals with PD-MCI and using a second-generation α4β2-nAChR ligand do not agree with previous imaging studies of the α4β2-nAChR in individuals with PD dementia using older α4β2-nAChR radioligands 15 and are important to explore even with a higher risk of type I statistical error. As the comparisons were planned a priori based on hypothesized relationships between region and cognitive function in PD, we reported ROI-based analyses that did not correct for multiple comparisons. However, we did utilize an FWE correction for the voxel-based correlation analyses because those analyses were exploratory. The findings should be replicated in a larger sample size of patients, including PD patients across a broad spectrum of cognitive impairment.
In conclusion, this study demonstrated that higher [18F]XTRA binding is associated with worse cognitive performance in patients with PD that is specific to the parietal cortex and its subregion of the precuneus. The direction of this association, in light of other studies showing higher or lower binding with cognitive impairment, highlights the complexity of imaging the α4β2-nAChR in PD given variations in study design, outcome variables, and binding affinity across brain regions of radioligands. Further studies are warranted to understand better the temporal association between α4β2-nAChR binding and progression of cognitive impairment longitudinally, and how this and similar studies 67 can inform the development of targeted therapy for cognitive deficits in parkinsonian disorders.
Supplemental Material
sj-docx-1-pkn-10.1177_1877718X241313373 - Supplemental material for Cortical α4β2-nicotinic acetylcholine receptors and cognitive decline in Parkinson's disease
Supplemental material, sj-docx-1-pkn-10.1177_1877718X241313373 for Cortical α4β2-nicotinic acetylcholine receptors and cognitive decline in Parkinson's disease by Kelly A Mills, Hiroto Kuwabara, Yong Du, Gabriela Gomez, Chelsie S Motley, Yana Skorobogatova, Ergi Spiro, Jennifer M Coughlin, Wojciech Lesniak, Jason Brandt, Vidya Kamath, Martin G Pomper and Gwenn S Smith in Journal of Parkinson's Disease
Footnotes
Acknowledgments
The authors would like to thank the participants of this study and the PET Center staff including but limited to Dr Robert F. Dannals and Karen Edwards.
Funding
This work was supported by NINDS / NIH 1K23NS101096-01A1, NCATS / NIH KL2TR001077 (Parent UL1TR001079), and Johns Hopkins Nexus Award (Parent UL1TR001079) to KM.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability
The data supporting the findings of this study are available on request from the corresponding author. The data are not publicly available due to privacy or ethical restrictions.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
