Abstract
Isolated rapid eye movement (REM) sleep behavior disorder (iRBD) highlights an early α-synucleinopathy. This study compared health-related quality of life (hrQoL) in 62 individuals with iRBD with 19 healthy controls (HC) and 29 individuals with Parkinson's disease (PD) and identified factors linked to poorer hrQoL. HrQoL was significantly lower in individuals with iRBD (83.33 ± 16.96) compared to HC (92.29 ± 5.49, p = 0.027). Poorer hrQoL in individuals with iRBD was linked to severity of multiple non-motor symptoms, most prominently fatigue and depressive symptoms being significant predictors (p < 0.001, adjusted R 2 = 0.81). This study highlights the importance of non-motor symptoms for hrQoL in prodromal PD.
Plain language summary
This study looked at health-related quality of life (hrQoL) in people with isolated REM sleep behavior disorder (iRBD), a sleep condition that can be an early sign of Parkinson's disease (PD). We compared 62 people with iRBD to 19 healthy individuals and 29 people with PD. Our findings showed that people with iRBD had lower hrQoL than healthy individuals. This reduced hrQoL was mostly due to non-motor symptoms like fatigue and depression, which had the strongest impact. These results underline the importance of addressing non-motor symptoms early to improve quality of life in people at risk of PD.
Introduction
As an incipient α-synucleinopathy,1,2 isolated rapid eye movement (REM) sleep behavior disorder (iRBD) is the most specific prodromal marker of Parkinson's disease (PD).3,4 Although individuals with iRBD exhibit merely motor symptoms,5,6 non-motor symptoms are frequent.7,8
Health-related quality of life (hrQoL) is essential in evaluating the impact of a disease on a patient's life. In PD, a significantly lower hrQoL compared to HC has been reported, with a stronger link to non-motor symptoms severity compared to motor symptoms.9,10 Only one study has assessed hrQoL in iRBD, highlighting reduced hrQoL in iRBD individuals compared to HC. 11 Identifying factors associated with reduced hrQoL is vital, particularly in prodromal PD, as it may help guidance of therapeutic interventions even in face of missing disease-modifying agents.
In the present study, we studied hrQoL in individuals with iRBD compared to HC and manifest PD, and its relation to non-motor symptoms and subtle motor symptoms. We hypothesized that hrQoL is diminished in individuals with iRBD compared to HC but still better than in PD. Comparable to PD, non-motor symptoms may contribute to poorer hrQoL in individuals with iRBD beyond emerging motor symptoms.
Methods
Study design and participants
This case-control study included participants recruited from July 2020 to November 2021 at the University Hospital of Cologne. 12 Inclusion criteria for individuals with iRBD were age 50 to 80 years, the ability to give informed consent, and polysomnography-confirmed iRBD according to the International Classification of Sleep Disorders III criteria.12,13 Exclusion criterion was a relevant cognitive impairment (Montreal Cognitive Assessment, MoCA; cutoff <22). 14
We also included individuals with PD diagnosed according to the current consensus criteria 15 with corresponding in- and exclusion criteria. For HC, depressive symptoms operationalized by the Beck's Depression Inventory II (BDI-II; cutoff ≥9) 16 and symptoms of RBD or any movement disorder were additional exclusion criteria.
Standard protocol approvals, registrations, and patient consents
The local ethics committee of the Medical Faculty of the University of Cologne approved the study. All participants gave written informed consent prior to participation.
Assessment of health-related quality of life
The 36-Item Short Form Health Survey (SF-36) 17 was used to measure hrQoL, with eight domains: physical function, physical role, bodily pain, general health, vitality, social functioning, emotional role, and psychological well-being. Scores range from 0 to 100, with higher scores indicating better hrQoL.
Clinical assessment
Subjects completed questionnaires on various non-motor symptoms. The Pittsburgh Sleep Quality Index (PSQI) 18 assessed sleep quality, the Regensburg Insomnia Scale (RIS) 19 examined insomnia, the Epworth Sleepiness Scale (ESS) 20 interrogated daytime sleepiness. Anxiety and depressive symptoms were determined using Beck's Anxiety Inventory (BAI), 21 and the BDI-II. 16 Additional assessments included the Apathy Evaluation Scale (AES), 22 as well as the Fatigue Scale for Motor and Cognitive Functions (FSMC). 23 Subjective cognitive impairment (SCI) was measured with Subjective Memory Impairment Questionnaire (SMI-Q).24,25 The SCOPA-AUT 26 questionnaire and the non-motor symptom questionnaire (NMSQ) 27 assessed autonomic symptoms and non-motor symptoms in general. Higher total scores indicate a higher symptom burden.
All participants underwent a comprehensive, non-blinded clinical work-up including the 12-item Sniffin’ Sticks test (Burkhardt®, Wedel, Germany), MoCA, 4 and MDS-UPDRS Part III 28 (except for HC participants).
Statistical analyses
Statistical analyses were conducted using IBM SPSS Statistics version 28.0 (IBM Corp, Armonk, NY, USA) and R Studio. Normality was tested, and group differences were assessed with Kruskall-Wallis and Mann-Whitney-U tests. Chi-Square tests interrogated categorical variables. The relationship between hrQoL and recorded data was analyzed with Spearman partial correlations, controlling for age and sex. Based on a significant correlation in the bivariate partial correlation analyses (p < 0.050), non-motor symptom scales were selected for multiple linear regression analyses in iRBD. The significance level was set at p < 0.050 uncorrected.
Results
The analyses included 62 individuals with iRBD, 19 HC participants, and 29 PD individuals. Demographic and clinical characteristics are shown in Supplemental Tables 1 and 2.
A Kruskal-Wallis test revealed significant group differences in the SF-36 total score (H(2) = 35.32, p < 0.001). HC participants had a significantly higher SF-36 total score than individuals with iRBD (U = 390.00, Z = −2.218, p = 0.027, r = 0.246), but individuals with PD had a significantly lower SF-36 total score compared to iRBD (U = 323.50, Z = −4.902, p < 0.001, r = 0.514) and HC (U = 30.00, Z = −5.176, p < 0.001, r = 0.747). Furthermore, there were significant differences between individuals with iRBD and HC in the SF-36 domains general health (U = 346.00, Z = −2.720, p = 0.007, r = 0.302), vitality (U = 402.00, Z = −2.096, p = .036, r = 0.233), and psychological well-being (U = 375.50, Z = −2.414, p = 0.016, r = 0.268, see Figure 1, Supplemental Table 3). HrQoL in PD was significantly lower than in individuals with iRBD and HC in all SF-36 domains.
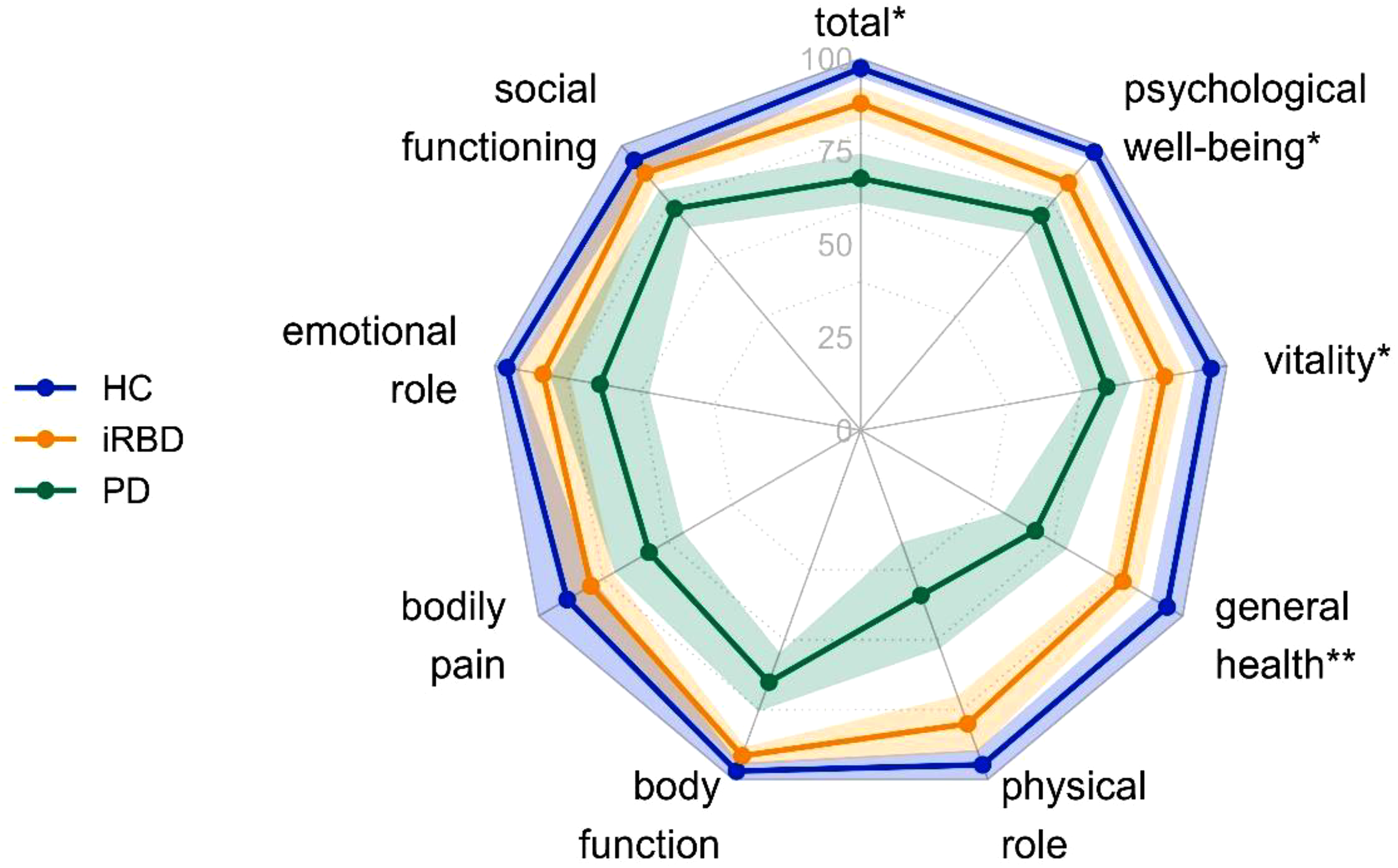
Hrqol comparisons of all SF-36 domains as well as the total score. N = 110. Dots express mean values, and colored areas show 95% confidence intervals. HC: Healthy control participants (n = 19); iRBD: iRBD individuals (n = 62); PD: Parkinson's disease individuals (n = 29). SF-36 scores range from 0 to 100, with higher scores indicating better hrQoL. *p < 0.05, **p < 0.01. Asterisks indicate significant group differences between HC and iRBD in the post-hoc Mann-Whitney-U tests. All group differences between iRBD and PD were significant (all p < 0.01).
We identified a link between various non-motor symptoms and reduced hrQoL in iRBD: PSQI, RIS, ESS, BAI, BDI-II, AES, FMSC, SCI, SCOPA-AUT, and NMSQ scores were negatively correlated with hrQoL (all r = −0.25 to −0.76, all p < 0.05, see Supplemental Figure 1). MoCA was positively correlated with vitality in individuals with iRBD (r = 0.25, p = 0.039). Incipient motor symptoms were not associated with hrQoL in individuals with iRBD (all r = −0.02 to −0.12).
Based on the significant bivariate Spearman correlations, we selected the candidates for the multiple linear regression analyses. PSQI, RIS, AES, and NMSS were each a predictor of singular SF-36 domains: total score, body role, vitality, and emotional role, respectively. However, fatigue and depressive symptoms were significant determinants of a large set of hrQoL subdomains of the SF-36 with substantial importance in terms of partitioned R2 (Table 1, Supplemental Tables 4–12).
Multiple regression analyses (corrected for age and sex) of all SF-36 domains and the total score in iRBD individuals.
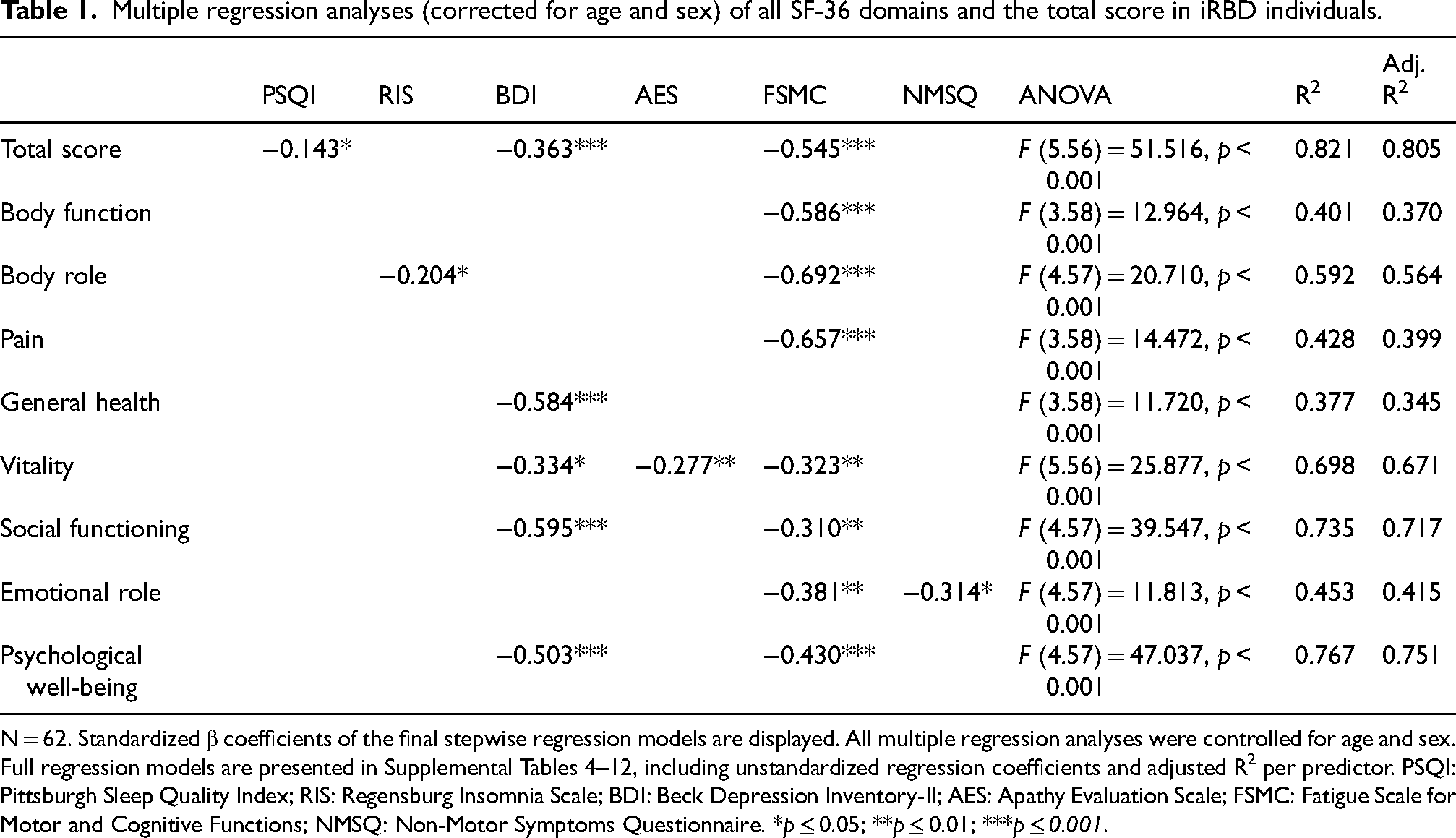
N = 62. Standardized β coefficients of the final stepwise regression models are displayed. All multiple regression analyses were controlled for age and sex. Full regression models are presented in Supplemental Tables 4–12, including unstandardized regression coefficients and adjusted R2 per predictor. PSQI: Pittsburgh Sleep Quality Index; RIS: Regensburg Insomnia Scale; BDI: Beck Depression Inventory-II; AES: Apathy Evaluation Scale; FSMC: Fatigue Scale for Motor and Cognitive Functions; NMSQ: Non-Motor Symptoms Questionnaire. *p ≤ 0.05; **p ≤ 0.01; ***p ≤ 0.001.
Discussion
This study identified reduced hrQoL in individuals with iRBD, independent of emerging PD motor symptoms. In particular, hrQoL was lower in individuals with iRBD than HC in the SF-36 total score and the domains general health, vitality, and psychological well-being. Compared to individuals with PD, hrQoL was still higher in individuals with iRBD. In line with the affected hrQoL domains, diminished hrQoL in individuals with iRBD was primarily correlated with more self-reported neuropsychiatric symptoms, sleep-wake disturbances, and autonomic symptoms. Regression analyses revealed more severe fatigue and depressive symptoms as drivers of poorer hrQoL in individuals with iRBD.
In individuals with PD, sleep-wake disorders as well as depression and fatigue are prevalent comorbidities closely related to poorer hrQoL.8,29–33 That underscores the importance of clinician awareness when caring for individuals with early PD, as addressing these potentially treatable symptoms could substantially impact individuals’ well-being. Depression may alter hrQoL ratings directly and exacerbate other non-motor symptoms, indirectly influencing hrQoL. 34
Previous studies in PD individuals support our results, showing non-motor symptoms to more significantly impact hrQoL than motor symptoms.9,10,30,35 Strikingly, non-motor symptoms of individuals with iRBD and PD are often overlooked and rarely treated in the clinical routine.36,37 However, non-motor symptoms may precede motor symptoms in emerging PD.7,36 In a recent survey, individuals with PD expressed a desire for improved non-motor symptom management. 38 Additionally, untreated individuals with recently diagnosed PD show hrQoL decline over time, whereas those receiving treatment showed a trend towards improved hrQoL. 39 Consequently, adequate diagnosis and treatment of non-motor symptoms is crucial, already in the prodromal phase of PD, due to the close link to increased functional disability, cognitive impairment, caregiver burden, and poor subjective hrQoL.38,40,41
Limitations of this study include the lack of longitudinal data. Small and heterogenous samples sizes in the HC and PD groups limited the statistical power of group comparisons and hence generalizability. Additionally, the study was conducted in recently diagnosed individuals with iRBD and self-reported depressive symptoms may have been aggravated due to the recent diagnosis.12,42 However, the study's strengths include the well-characterized sample due to the comprehensive clinical assessments as well as the possibility to compare hrQoL across healthy aging, prodromal and manifest PD.
In summary, careful screening for non-motor symptoms in individuals with iRBD is crucial. Depressive symptoms and fatigue are the most relevant therapeutic targets in the prodromal stage of PD to enhance individuals’ quality of life.
Supplemental Material
sj-docx-1-pkn-10.1177_1877718X241310726 - Supplemental material for Non-motor symptoms in prodromal Parkinson's disease are linked to reduced quality of life
Supplemental material, sj-docx-1-pkn-10.1177_1877718X241310726 for Non-motor symptoms in prodromal Parkinson's disease are linked to reduced quality of life by Sinah Röttgen, Marie-Sophie Lindner, Aline Seger, Johanna Kickartz and Kim-Lara Weiß, Christopher EJ Doppler, Gereon R Fink, Anja Ophey, Michael Sommerauer in Journal of Parkinson's Disease
Footnotes
Acknowledgments
We thank all study participants for their participation. ChatGPT, version GPT-4o mini, a language model developed by OpenAI (![]() ), was used for language refinement of the Plain Language Summary. Open access publication was funded by the Deutsche Forschungsgemeinschaft (DFG, German Research Foundation) – 491111487.
), was used for language refinement of the Plain Language Summary. Open access publication was funded by the Deutsche Forschungsgemeinschaft (DFG, German Research Foundation) – 491111487.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: C.E.J.D. is supported by the Clinician Scientist Program (CCSP), funded by the Deutsche Forschungsgemeinschaft (DFG, German Research Foundation, FI 773/15-1). G.R.F. receives grants from Deutsche Forschungsgemeinschaft (DFG, German Research Foundation), Project-ID 431549029, SFB 1451. A.O. received a grant from the Koeln Fortune Program (grant-no. 329/2021), Faculty of Medicine, University of Cologne, and the “Novartis-Stiftung für therapeutische Forschung”, both outside the submitted work. M.S. received grants from the Else Kröner-Fresenius-Stiftung (grant number 2019_EKES.02), and the Koeln Fortune Program, Faculty of Medicine, University of Cologne. M.S. receives additional funding from the program “Netzwerke 2021”, an initiative of the Ministry of Culture and Science of the State of Northrhine Westphalia. The Federal Ministry of Education and Research (BMBF) is funding the project within the framework of the funding programme ACCENT (funding code 01EO2107). Open access publication was funded by the Deutsche Forschungsgemeinschaft (DFG, German Research Foundation) – 491111487.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Data availability
The data included in this study are available on reasonable request to the corresponding author. The data are not publicly available due to privacy or ethical restrictions.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
