Abstract
Background
The availability of deep brain stimulation (DBS), a highly efficacious treatment for several movement disorders, remains low in developing countries, with scarce data available on utilization and outcomes.
Objective
We characterized the DBS cohort and outcomes at a Malaysian quaternary medical center.
Methods
A retrospective chart review was done on DBS-related surgery at the University of Malaya, including clinico-demographic, genetics, and outcomes data focusing on post-operative medication reduction and complications.
Results
149 Parkinson's disease (PD) patients underwent DBS targeting the subthalamic nucleus. Six had globus pallidus internus DBS (primarily for dystonia). Only 16.1% of patients were government-funded. Of the 133 PD patients operated in the past decade (2013–2022), 25 (18.8%) had disease duration <5 years. At 6–12 months post-DBS, median levodopa-equivalent daily dose (LEDD) reduction was 440.5 [418.9] mg/day, corresponding to a reduction of ≥50% and ≥30% in 42.2% and 69.8% of patients, respectively. LEDD reductions were larger in the early-onset and short-duration subgroups. Three patients (1.9% of 155) had symptomatic intracranial hemorrhage, resulting in stroke in two. Pathogenic monogenic or GBA1 variants were detected in 12/76 (16%) of patients tested, mostly comprising the “severe” GBA1 variant p.L483P (12%).
Conclusions
This is the largest report on DBS from Southeast Asia. The procedures were effective, and complication rates on par with international norms. Our study found a high frequency of GBA1-PD; and included a substantial number of patients with short-duration PD, who had good outcomes. It also highlights regional inequities in access to device-aided therapy.
Plain language summary
Deep brain stimulation (DBS) is a highly effective treatment for several movement disorders, including Parkinson's disease (PD). However, there is very limited published information about DBS in developing countries, and on “real-life” outcomes when DBS is performed earlier in the course of PD. Here, we describe a relatively large cohort of patients who underwent DBS at a major hospital in Kuala Lumpur, Malaysia. DBS was effective with low complication rates, and at 6–12 months after surgery, the PD medication dosage was reduced by at least one-half in 4 out of 10 patients and by at least one-third in 7 out of 10 patients. Patients with PD who underwent DBS early in their disease course (within 5 years from diagnosis, comprising one-fifth of the patients), and those diagnosed at age 50 years or younger (comprising one-half of the patients), appeared to have larger reductions in their PD medications. A significant proportion of patients with PD (16%) who had genetic testing done were found to harbor monogenic (causative) gene variants or GBA1 risk gene variants, which have been associated with worse disease progression, and these patients had lesser medication reduction compared to GBA1 variant-negative patients. Less than two out of 10 patients (16%) had their DBS funded via the public health system. To our knowledge, this is the largest report on DBS from Southeast Asia and documents good outcomes overall, including in PD patients operated on early in their disease course, but highlights limited government funding for this potentially life-changing therapy as a significant barrier in developing countries.
Keywords
Introduction
Deep brain stimulation (DBS) is an established treatment for Parkinson's disease (PD) and other movement disorders, often dramatically improving motor functions with sustained benefits for ≥10 years.1,2 Since its introduction in the 1990s, advances in hardware, software, and targeting have further improved treatment efficacy and device functionality. 3 DBS therapy has become more inclusive with expanded disease indications, and a trend towards offering the procedure earlier in the disease course.2–4 The EARLYSTIM trial 4 published a decade ago showed that DBS performed earlier in PD (participants were aged ≤60 years, with disease duration ≥4 years and ≤3 years of motor complications) was effective and safe.4–6 This seminal study, together with the advent of longer-life implantable pulse generators (IPGs; lasting ≥15 years) have likely changed the DBS landscape worldwide,7,8 but few studies have documented this paradigmatic shift in real-world practice.9,10 Concerns also continue to be put forth in opposition to this approach, including, understandably, the possible misdiagnosis of early-stage atypical parkinsonism for PD.11,12
Because DBS availability in developing countries remains low, due to its high cost, and limitations in government healthcare expenditure, facilities, and expertise,13,14 there is a paucity of data on DBS utilization and outcomes, e.g., from Southeast Asia with a population of >650 million (total n = 384 patients across 13 studies, each with n = 1–95 - see Supplemental Material 1). Other device-aided therapies (infusions of dopaminergic agents) are even costlier in this region, making them even less available/accessible to patients.14,15 The lack of access to these potentially life-changing treatments in low- and middle-income countries, where most PD patients in the world reside, has been highlighted by the World Health Organization as an area of health disparity in need of global action. 16 (Fittingly, in calling “health inequality in PD … a global phenomenon which needs to be addressed”, the Editors of the Journal of Parkinson's Disease have recently launched a new Section dedicated to highlighting the diverse challenges and unique features of PD in different geographical regions). 17
There has also been increasing interest in exploring genotype-phenotype correlations, including the influence of genetic variants on the disease course of PD, dystonias, and other movement disorders, and the responsivity of variant carriers to DBS.18–22 In the era of personalized precision medicine, greater understanding of the role of genetics can help to refine the selection of treatments for optimized outcomes.21–24
We aimed to characterize the clinico-demographic features of our DBS cohort and their outcomes, at a quaternary medical center in Malaysia. The results of genetic testing were also reviewed.
Methods
Patients, DBS procedure, and data collection
A retrospective chart review was done on consecutive patients undergoing DBS surgeries at the University of Malaya (UM), from inception in September 2004 to December 2022. We included patients who underwent new/primary DBS implantations, as well as those having IPG replacement or revision surgeries. Our DBS protocol is detailed in Supplemental Material 2. The vast majority (>90%) of patients were managed by two movement disorder neurologists (SYL, AHT) and one DBS neurosurgeon (KAM).
Clinico-demographic and outcomes data focusing on post-operative PD medication reduction and complications were collected. Patients were managed pragmatically, and motor improvement post-operatively was usually assessed qualitatively, in a variety of ways, including reduction in OFF-period severity/duration, dyskinesias and/or tremors, and improvements in functional abilities or quality of life. PD medications were not withdrawn after completion of DBS programming to formally measure OFF-medication, ON-DBS status unless there was uncertainty about a lack of stimulation efficacy. Thus, post-operative reduction in PD medications (calculated as levodopa-equivalent daily dose [LEDD]25,26) was used as a surrogate measure for motor improvement after subthalamic nucleus (STN) DBS. LEDDs were calculated at: pre-DBS (T1); 6–12 months post-surgery (T2); and the most recent hospital visit (>12 months post-surgery) (T3). In the literature, LEDD reductions post-STN DBS have typically been ∼30–50%,2,27,28 with an increasing trend (∼10%) on prolonged follow-up.2,28 Rating scales were not systematically administered in the small dystonia cohort, and the benefit from DBS was rated qualitatively (small, medium, or large).
We examined the proportion and outcomes of patients undergoing DBS with short duration or early onset of PD. Since guidelines commonly recommend ≥5 years disease duration before performing DBS (referred to as “standard-duration” PD), to avoid inclusion of atypical parkinsonian disorders,7,11,12,29 we defined “short-duration” PD patients as those undergoing DBS <5 years after diagnosis. Early-onset PD (EOPD) was diagnosed aged ≤50, and late-onset PD (LOPD) > 50 years.
The results of genetic tests were reviewed. These were done on a research basis, and in almost all patients, the results only became available post-surgery.
The study received ethical approval from the Medical Ethics Committee, UM.
Statistical analyses
Data were analyzed using SPSS Version 29.0 (SPSS, Inc., Chicago, IL, USA). Shapiro-Wilk test was used for normality testing. Between-group differences in clinico-demographics and LEDD reduction were analyzed using Chi-square and Mann-Whitney U tests. Wilcoxon signed-rank test was used to analyze LEDD trends at the different timepoints. Two-tailed p < 0.05 was the significance threshold.
Results
DBS procedures
161 patients underwent DBS-related surgeries (Figure 1, Table 1). Of the 155 patients undergoing the primary bilateral DBS surgery at UM, 147 (94.8%) had synchronous bilateral STN DBS; two (1.3%) had staged bilateral STN DBS (one because of intra-operative subdural hematoma; and one, a patient with severe left-sided PD features, initially underwent right-sided STN DBS in 2005 using a Medtronic Soletra® IPG); and six (3.9%) had synchronous bilateral globus pallidus internus (GPi) DBS. Of the 149 PD patients whose first DBS surgery was done at UM, only one (with severe dyskinesias on low-dose dopamine agonist) underwent GPi DBS. The remaining five patients who underwent GPi DBS had dystonic disorders. There were no patients of thalamic DBS for tremor disorders.
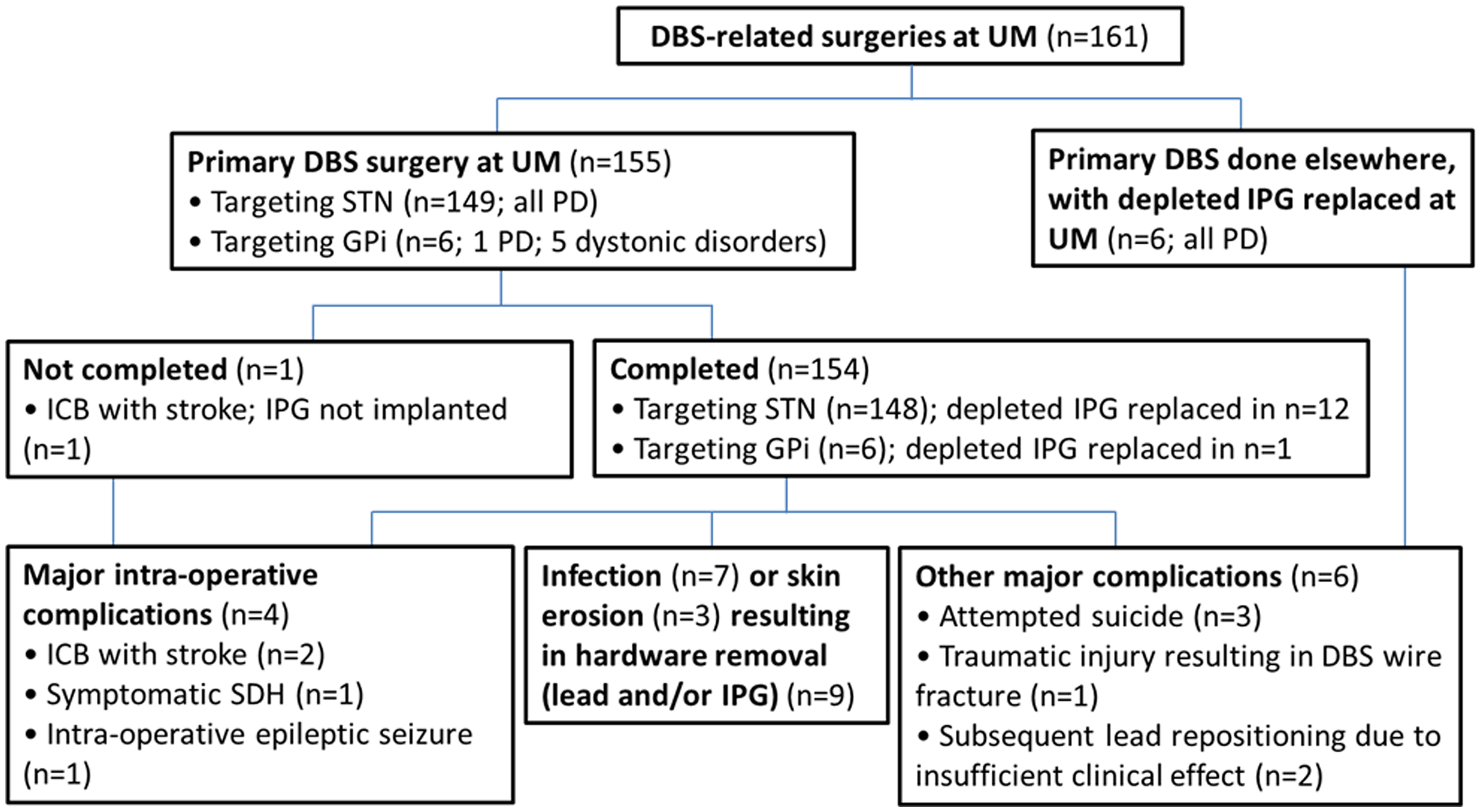
Patient flow diagram.
Clinico-demographic data of patients undergoing deep brain stimulation-related surgeries at the University of Malaya.
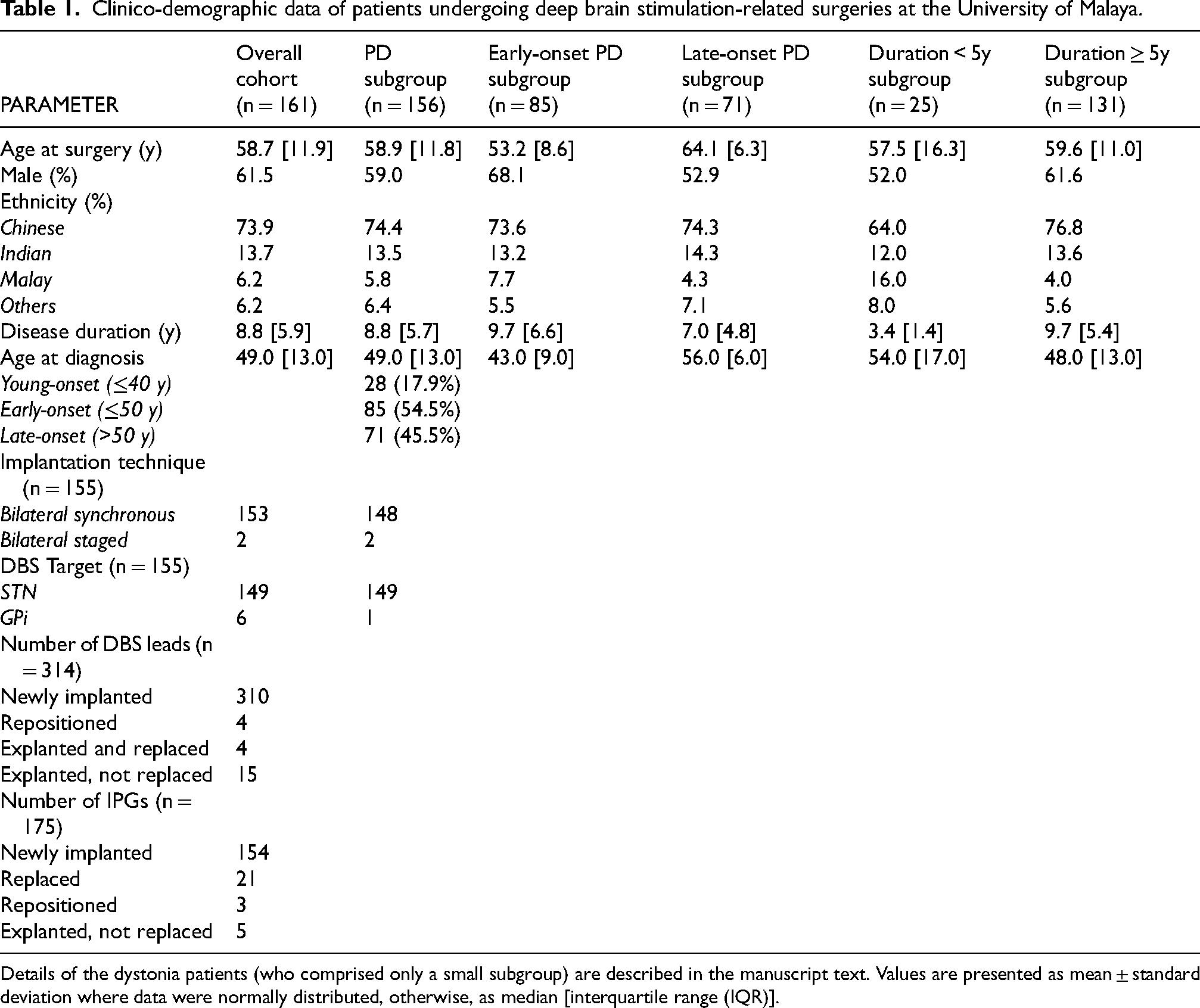
Details of the dystonia patients (who comprised only a small subgroup) are described in the manuscript text. Values are presented as mean ± standard deviation where data were normally distributed, otherwise, as median [interquartile range (IQR)].
The number of primary DBS surgeries performed increased over time, except for the period 2020–2021, due to the COVID-19 pandemic (Figure 2). On average, 19 primary DBS surgeries were performed annually from 2016–2022.
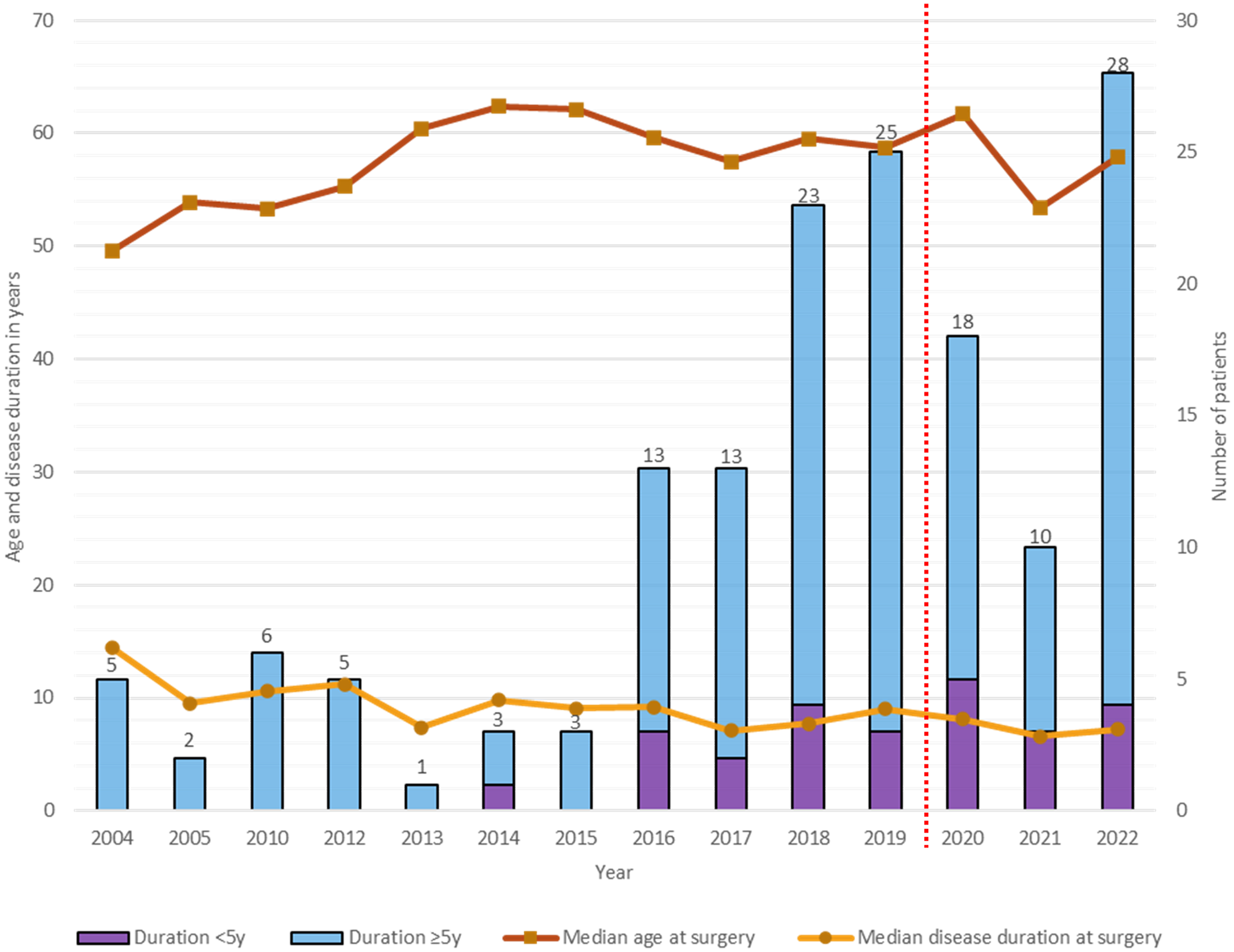
Disease duration and age at first/primary deep brain stimulation (DBS) surgery for Parkinson's disease (PD), and the number of patients with short-duration and standard-duration PD from 2004–2022.
The vast majority of the DBS surgeries were self-paid or reimbursed by private health insurance, costing ∼RM150,000 (∼USD35,000) using the (rechargeable) Medtronic Activa RC® device (utilized in almost all patients from 2015 onwards) (in comparison, the median monthly household income in Malaysia in 2022 was ∼RM6,300 [mean RM8,500 30 ]). The amounts reimbursed by private health insurance typically covered ≥2/3 of this cost. Only 26 patients (16.1%) received government funding (which covered the cost of the entire procedure), under a quota system (∼3 slots annually) reserved for government-employed patients.
The duration of post-operative follow-up was 2.9 [interquartile range: 3.5] years (range: 0–17.4 years). The majority of the cohort were still under follow-up; 24 (14.9%) were lost to follow-up and another 16 (9.9%) were deceased at T3 (age and disease duration at death 70 [8.6] and 17.5 [9.7] years, respectively). One patient died within the first post-operative year (41 weeks post-DBS), from hypokalemia-related cardiac arrhythmia.
Surgery and hardware-related complications
Among the 155 patients who had their primary DBS surgery at UM, two (1.3%) developed large intracerebral (frontal lobe) bleeds intra-operatively causing stroke, with persisting neurological disability (Figure 1 and Supplemental Table 1). Another patient with intra-operative subdural hematoma causing severe headache underwent immediate hematoma evacuation without neurological deficits. One patient (0.6%) with pre-existing occipital lobe infarction had intra-operative seizure.
In the overall cohort, 7/161 (4.3%) and 3/161 patients (1.9%) developed surgical site infection (SSI) and skin erosion, respectively (Supplemental Table 1). Despite antibiotic treatment and surgical debridement, explantation of the DBS leads and/or IPG was needed in nine patients. Dermatological conditions led to SSI in two of them (one with drug-induced Stevens-Johnson syndrome, another with bullous pemphigoid and diabetes mellitus).
Two patients had bilateral lead repositioning for suboptimal stimulation effect; another patient with suspected suboptimal lead positioning declined revision surgery. Additionally, three patients underwent re-implantation of DBS leads: two for infection (bilateral STN leads in one, and unilateral STN lead in another who initially had DBS in another institution), and one for lead fracture after a major fall (six years after the initial DBS surgery).
PD patients
There were 156 patients with PD, the majority male and Chinese (Table 1), similar to the overall PD population at UM. 31 The median ages at diagnosis and at first DBS surgery were 49.0 [13.0] (range: 25.0–68.0) years and 58.9 [11.8] (35.8–77.3) years, with disease duration from diagnosis of 8.8 [5.7] (range: 2.3–24.3) years. EOPD (n = 85) comprised 54.5% of patients.
Over time, there was a trend for reducing PD duration at the first DBS surgery (Figure 2), with increasing numbers, since around 2014, of patients with short-duration PD. Disease duration from diagnosis was <5 years in 25 patients (of whom 16 had disease duration <4 years) (age at surgery: 59.6 [11.0] years; disease duration: 3.4 [1.4] years), comprising 18.8% of the 133 PD patients operated in the past decade (2013–2022). As for the overall PD group, troublesome motor response complications were the usual primary indication for DBS in this subgroup (Supplemental Table 2), with only a few patients having troublesome medication-unresponsive tremor. None had diagnostic reassignment to a Parkinson-plus syndrome over follow-up (3.9 [2.8], range: 0.1–7.2 years).
In the overall PD cohort, median LEDD reduction at T2 (vs. T1) was 440.5 [418.9] mg/day (p < 0.001; n = 116), with 42.2% (n = 49) and 69.8% (n = 81) of patients having ≥50% and ≥30% reductions, respectively (Figure 3A-C). The median LEDD reduction at T3 (vs. T1) was 297.6 [494.3] mg/day (p < 0.001; n = 110), with 30.0% (n = 33) and 58.2% (n = 64) having ≥50% and ≥30% reductions (Figure 3A, B, and D). Median LEDD was significantly higher at T3 vs. T2 (p = 0.007).
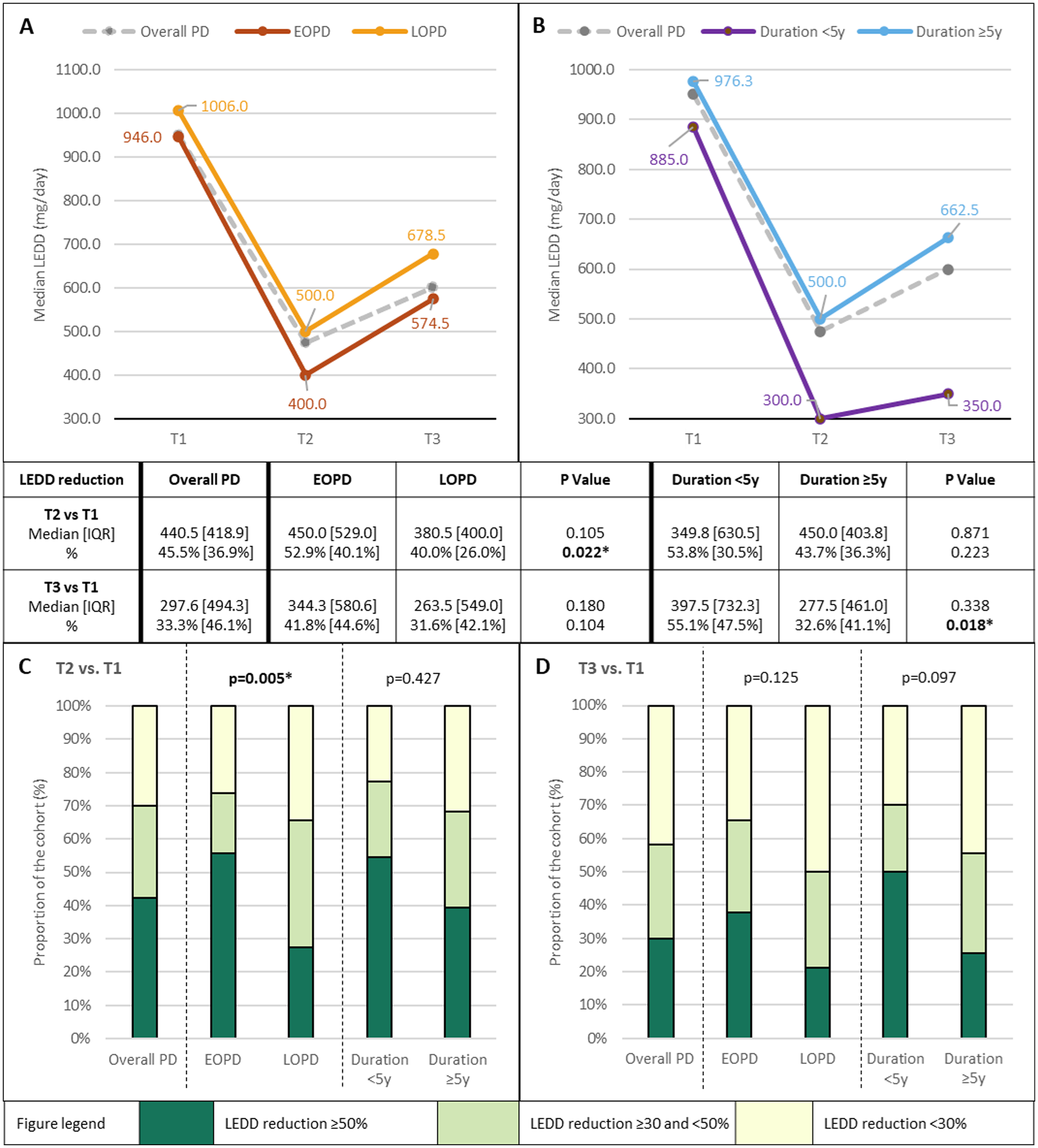
Changes in levodopa-equivalent daily dose (LEDD) after deep brain stimulation (DBS) surgery.
In the subgroup analyses of EOPD and short-duration patients: (i) the T2-vs.-T1 median % LEDD reduction was significantly greater in the EOPD vs. LOPD subgroup (52.9% [40.1%] vs. 40.0% [26.0%], p = 0.022) with a significantly higher proportion of EOPD patients with ≥50% LEDD reduction (55.7% vs. 27.3%; p = 0.005) (Figure 3A and C); and (ii) the T3-vs.-T1 median % LEDD reduction was significantly greater in the short- vs. standard-duration PD subgroup (55.1% [47.5%] vs. 32.6% [41.1%], p = 0.018), with a trend for a higher proportion of short-duration patients with ≥50% LEDD reduction (50.0% vs 25.6%, p = 0.097) (Figure 3B and D). Follow-up duration was not significantly different between the short- vs. standard-duration PD groups (2.5 [2.4] years vs. 2.9 [3.5] years, p = 0.567).
Regarding neuropsychiatric problems post-DBS, suicide was attempted in three patients (1.9%), one of whom succumbed to aspiration pneumonia after one week of hospitalization related to sedative drug overdose (this patient had a long history of depression and anxiety predating DBS, and her mother who had depression died from suicide). Four patients displayed impulsive-compulsive behaviors (ICBs): one had dopamine dysregulation syndrome, which existed pre-DBS but was concealed by the patient/family; one patient each had continued problematic gambling and shopping behaviors that predated DBS (the latter patient also had a prior history of opioid addiction; both had continued taking pramipexole against medical advice); and the fourth patient had new-onset problematic gambling two years post-operatively and impulsively attempted suicide because of the financial losses plus isolation during the COVID-19 pandemic; this patient had a gambling “habit” prior to DBS, involving small amounts of money.
Four patients had new-onset psychosis within the first post-operative year, one of whom required urgent psychiatry referral. Another patient with pre-existing psychosis had worsening psychosis post-DBS that also required urgent psychiatry care. Twelve patients (7.5%) developed dementia a median of 4.6 years post-DBS. Age at the last visit (67.5 [38.1] vs. 62.5 [52.8] years, p = 0.036) and disease duration (15.3 [27.8] vs. 11.3 [31.5] years, p = 0.031) were significantly higher/longer in those with dementia vs. those without. Only one patient of 12 who developed dementia came from the short-duration PD subgroup.
Results of testing for monogenic and GBA1-related PD (prioritizing familial and/or EOPD patients) were available for 76 patients: whole genome sequencing under the Global Parkinson's Genetics Program (GP2) (n = 61), 20 next generation sequencing-based PD gene panel (n = 22), 18 and/or multiplex ligation-dependent probe amplification (n = 13). 32 Twelve (15.8%) were found to have pathogenic or likely pathogenic variants in the risk factor GBA1 gene (n = 10)18,33 and in the monogenic LRRK2 (n = 1) 34 or PRKN genes (n = 1) 32 (Supplemental Table 3). Among GBA1 variant-positive patients, only two could have their LEDD substantially reduced (by ≥30% or ≥50%) post-operatively. Although the sample size of GBA1-PD patients was relatively small, median % LEDD reductions were significantly less in these patients vs. GBA1 variant-negative patients at both T2 (by 19.0% [31.5%] vs. 50.9% [36.6%], p = 0.004) and T3 (20.0% [50.5%] vs. 45.0% [45.0%], p = 0.045), compared to T1 (with no significant between-group difference in baseline LEDD). GBA1 variant carriers accounted for two of the patients with dementia (PD-1414 33 and PD-0203 18 ; Supplemental Table 3), and one of the patients (PD-2045) having problematic gambling and attempting suicide (described above). A good DBS outcome was obtained in the PARK-PRKN case 32 whereas the benefit was small-to-medium only in the patient with LRRK2 p.R1441C. 34
The “Asian” LRRK2 risk variants p.R1628P and p.G2385R were detected in 10.0% (15/150) and 7.3% (10/137) of patients, respectively. The median LEDD at baseline and the median % LEDD reductions at T2 and T3 (vs. T1) among these patients did not differ significantly vs. those without the LRRK2 variants (data not shown).
Dystonia subgroup
There were five patients, with: tardive dystonia (n = 2; one generalized and another segmental); TOR1A-negative familial pure generalized dystonia (n = 1); whole exome-negative familial generalized dystonia-parkinsonism (n = 1); and idiopathic Meige syndrome (n = 1). Outcomes post-GPi DBS were mixed, with benefits rated as medium-to-large; small; medium; small; and small-to-medium, respectively.
Discussion
DBS can dramatically improve the motor function and quality of life of patients with movement disorders that are not responding satisfactorily to medical therapy.1,2 However, largely due to its high cost, 13 the vast majority of patients worldwide are unable to benefit from a technological advance that has been “available” for >20 years. 14 Although our study represents the largest published on DBS from Southeast Asia, the number of patients operated is dwarfed by high-volume centers in advanced economies. For example, Canada and Australia which have comparable population sizes to Malaysia (∼30–40 million persons) reported DBS volumes35,36 that were at least one order of magnitude greater than in Malaysia (totaling ∼350 so far in Malaysia since the first case in February 2003 at Sunway Medical Centre, of which slightly >60% were done at UM - personal communication, Brian Kah, Medtronic, on 29 July 2024). In our study, less than one-fifth of patients were government-funded, reflecting the fact that in developing countries, healthcare costs are often largely borne by patients and families.13,24,37–39
Another factor limiting more widespread application of DBS is that outcomes are highly dependent on the skills of the DBS team in terms of which patients are offered to undergo this treatment, neurosurgical targeting, and post-operative management. These take time to develop, as does the trust and confidence among the patient and medical communities in the safety and efficacy of the procedure done in the local setting. 13 In our particular instance, the creation, around 2016, of a local “DBS support group” where operated patients provided personal testimonials and encouragement, gave many others the assurance to undergo DBS. We believe this peer support helped to bridge the gaps that are known to exist between the knowledge, perceptions, and priorities of patients/families, and those of the treating doctors. 40 Over time, and in parallel with rechargeable IPGs providing an improved benefit-to-cost ratio (∼RM10,000 per year when dividing the RM150,000 cost over 15 years), our center has been able to provide DBS treatment to a growing number of patients, with overall good outcomes as evidenced by complication and clinical efficacy metrics that are on par with those reported internationally. For example, the rates of symptomatic intracranial hemorrhage were 1.9% (vs. 0.6–6.0%); SSI 4.3% (vs. 0–15.2%); and wire fracture 0.6% (vs. 0.7–4.4%);41–43 with LEDD reductions ≥50% and ≥30% in 42.2% and 69.8% of PD patients, respectively, 6–12 months post-DBS.
Despite the safety and efficacy of DBS, however, outcomes can sometimes be less than satisfactory, for patients and families, as well as the treating clinicians. This may arise because of unrealistic expectations; 44 surgical complications; ongoing difficulties with motor response complications (e.g., dyskinesias that fail to abate despite LEDD reduction); 8 or cognitive-behavioral, gait and balance problems caused by lesioning from the brain surgery, off-target stimulation effects, disease progression, and/or comorbidities.
Thus, one strategy may be to offer DBS at earlier stages in younger patients with: (i) lower complication risks; (ii) still-intact functional (e.g., occupational) capacities; and (iii) more prolonged benefit (before this becomes overshadowed by disease progression or comorbidities).4,7 Experts have highlighted that “discussing device-aided treatments is a stepwise process, which may occur over several years”. 45 This, combined with the experience of patients “missing the boat” for DBS because of late referrals, motivated us to introduce the concept of DBS treatment to patients earlier in their disease course. Sometimes, this has been broached with younger patients who may not yet have developed (but are anticipated to be at heightened risk for) motor complications. Thus, in our cohort, there has been a trend of decreasing disease duration at the time of DBS surgery, corresponding to an increasing number of patients with short-duration PD (however, we would like to emphasize that all patients selected to undergo DBS experienced troublesome motor response complications and/or tremor not responding satisfactorily to medical treatment). Additionally, the fact that most patients are self-paying may also have tilted the scales towards earlier onset/shorter duration patients, as these patients are more likely to have an active income. Finally, younger patients who on the whole are more digitally savvy were more likely to be linked with the DBS support group mentioned earlier, as many of the group's activities are conducted via WhatsApp, videoconferencing, and emails.
The validity of this strategy seems to be supported by our safety and efficacy data – besides having acceptable complication rates, our short-duration patients had on average greater and more sustained LEDD reductions compared to standard-duration patients. Importantly, while misdiagnosis of atypical parkinsonism for PD is cited as a rationale to defer early DBS,11,12,29 none of our short-duration patients had their diagnosis reassigned during follow-up. We acknowledge that longer-term follow-up and continued vigilance are needed. It is known that these disorders may sometimes not declare themselves until much later, e.g., in a series from the Mayo Clinic, several patients undergoing DBS for PD “converted” to multiple system atrophy 15–17 years after onset. 46
Publications regarding the relationship between PD genetic variants and presentation for, and response to, DBS are still limited. Our rate of monogenic and GBA1-related PD in the subset of patients tested (12/76 = 15.8%) was overall lower compared to DBS cohorts in the United States 47 (26.5% of 100 EOPD patients) and the United Kingdom 48 (28.7% of 94 unselected patients) (to our knowledge, similar studies have not yet been performed in Asia). However, the frequencies of GBA1 variants (13.2%, vs. 12.1–20.0%) were comparable.47–49 GBA1-PD is reported to be associated with less favorable outcomes post-DBS,19,50 including worse cognitive decline, axial motor features, function, and quality of life, and less LEDD reduction, consistent with our observations. Indeed, the 19.0–20.0% LEDD reduction in our patients was remarkably similar to the meta-analyzed figure of 22%, involving 30 GBA1-PD patients from white populations. 50 The “severe” p.L483P variant in particular has been associated with worse disease progression in whites, 51 and the findings in our DBS cohort (where p.L483P accounted for 90.0% of the pathogenic/likely pathogenic GBA1 variants, vs. 18.8–25.0% in previous reports47,48) suggest that this may also be the case in Asians. 14
Our rates of LRRK2 Asian risk variants were comparable to overall (non-DBS) Asian PD populations (each variant being present in ∼5–10% of patients). 14 Only one study, involving Han Chinese PD, systematically investigated p.G2385R frequency in DBS patients and found this to be relatively high (8/57 = 14.0%), 52 suggesting that in some populations its presence could be associated with motor complications 53 warranting DBS. Asian LRRK2 variant positivity did not seem to adversely affect treatment response in our patients, similar to the findings of Chen et al. 52 Higher-powered, systematic studies will allow more definitive conclusions regarding the roles of LRRK2, and other risk variants/polygenic risk scores, in the context of DBS.21,22,54
The sample size for dystonia in this study was very small, allowing only tentative observations. DBS efficacy for tardive syndromes remains to be demonstrated in larger randomized studies; 55 in line with this, our two patients with tardive dystonia had mixed outcomes and continued to have significant disability and require botulinum toxin injections. Certain monogenic dystonias (e.g., DYT-TOR1A and DYT-KMT2B) respond favorably to DBS, 56 however, in our clinics, we have detected DYT-KMT2B in only two patients 57 and DYT-TOR1A in none, possibly explaining the limited benefit of DBS in our dystonia patients.
Interestingly, although DBS is an established treatment for essential tremor (ET), no patient in our cohort underwent DBS for ET. While ET is commonly diagnosed, it seems that this is rarely severe enough for our patients to be considered for (or for them to agree to undergo) DBS, again suggesting potential ethno-geographic differences in movement disorder phenotypes.33,58 An informal survey of high-volume DBS programs in Asia revealed similarly low numbers of ET patients undergoing DBS: n = 1 (Bangalore/NIMHANS, personal communication, Prof. Pramod Pal); n = 1 (Kerala/Sree Chitra Tirunal Institute, Prof. Asha Kishore); n = 2 (Tokyo/Juntendo, Dr Genko Oyama); and n = 0 (Bangkok/Chulalongkorn, Dr Onanong Phokaewvarangkul).
Our study has several major limitations and strengths. It was single-center, and the sample size is relatively small compared to reports from developed nations. On the flip side, patients were managed consistently by a small team comprising one functional neurosurgeon and two movement disorder neurologists, which could increase the reliability of between-group comparisons (e.g., short- vs. standard-duration PD). The study also represents the largest published on DBS from Southeast Asia and provides a valuable picture of DBS practice in a globally underserved region. Another important limitation was the lack of quantitative documentation of a range of disease-related variables (motor and non-motor function, functional abilities, quality of life, etc.) before and after DBS. The gold standard to confirm motor improvement after DBS is to evaluate patients after overnight withdrawal of PD medications; however, we lacked the human resources to routinely do this. Furthermore, since the evidence base for DBS efficacy in terms of motor function is well established, 27 these assessments were deemed to pose unnecessary physical and financial hardship to patients.14,24 That said, it would still be valuable for future studies to carefully document changes in various motor and non-motor features, particularly in subgroups of patients (e.g., those with specific genetic findings). On the positive side, the study provides data that are reflective of real-world clinical practice, and included all operated patients, not just those willing or able to participate in more demanding research protocols. Although conveying only a partial picture of a patient's condition post-DBS, dopaminergic medication reduction is considered a good surrogate indicator of STN DBS efficacy in improving PD symptoms, and attenuates medication-related adverse effects including dyskinesias and hyperdopaminergic behaviors.2,6,21 Indeed, the rates of overt medication-related neuropsychiatric complications in our patients were low. However, we acknowledge that patients did not routinely receive neuropsychiatric evaluations post-operatively, due in part to resource limitations and a reluctance among many patients to be referred for psychiatric care. Clinical neuropsychologists (e.g., to help detect and manage incipient cognitive decline) are unfortunately very scarce in Malaysia and this is another important gap in our current DBS setup. Finally, only one PD patient underwent GPi-DBS in our center. Although large-scale studies did not report major differences in outcomes comparing STN vs. GPi DBS in the treatment of PD,59,60 many centers around the world currently still favor STN DBS, in part due to their experience and familiarity with this target. To our knowledge, the only double-blind sham-controlled study of DBS in PD used bilateral STN as the target. 61 Moreover, in our local setting, dopaminergic medication reduction is often an important outcome for patients (in part due to cost as medications are often paid for out-of-pocket, as well as medication-related adverse effects), 14 and this is usually only achieved with STN and not GPi DBS. The authors would like to emphasize however that dopaminergic medication post-DBS is not per se the main objective of the procedure, rather the primary aims are improvements in symptoms, function, and quality of life for the patient and family.
In conclusion, we described real-world experience of DBS in a developing country and showed the procedures to be safe and effective. A notable aspect of our cohort was the inclusion of a substantial number of patients with a short duration of PD, in whom good clinical outcomes were seen, with larger and more sustained reductions in PD medication requirement, and none developing atypical parkinsonism. Our findings also provide Asian-relevant genotype-phenotype insights, particularly regarding GBA1 and LRRK2 variants that are commonly encountered. Genetics play an increasing role in the delivery of personalized precision medicine, and enhanced PD genetics knowledge that is also inclusive of underrepresented non-European patient cohorts will provide clinicians, patients, and families worldwide with better predictive ability to enable informed decision-making regarding the benefits and risks of DBS.62,63
Importantly, the study highlights inequity of access to a potentially life-changing treatment. A relatively small proportion of DBS cases in our cohort was government-funded, reflecting, we believe, a more general situation in developing countries where healthcare costs, especially those involving more expensive procedures, often have to be borne by patients and families.13–16,64 This makes many treatments which are taken for granted as “standard of care” in advanced economies inaccessible to the vast majority of patients worldwide,13–16,64–66 and even within certain segments of developed countries.64,67 There is thus an urgent need for the clinical-scientific and patient-support communities, the device industry, and governments, to work together towards closing this gap across different groups and regions.13,16,68 These efforts could include performing research to comprehensively understand the circumstances under which health disparities arise;64,67 employing larger-scale and modernized production techniques to reduce the cost of devices;1,13 increasing investment to strengthen public healthcare capacity (including trained workforce and equipment/device procurement) while ensuring the efficient and equitable use of available resources; and other innovative solutions.13–16,64,69
Supplemental Material
sj-docx-1-pkn-10.1177_1877718X241297715 - Supplemental material for New insights from a Malaysian real-world deep brain stimulation cohort
Supplemental material, sj-docx-1-pkn-10.1177_1877718X241297715 for New insights from a Malaysian real-world deep brain stimulation cohort by Alfand Marl F. Dy Closas, Ai Huey Tan, Yi Wen Tay, Jia Wei Hor, Tzi Shin Toh, Jia Lun Lim, Choey Yee Lew, Chun Yoong Cham, Carolyn Chue Wai Yim, Kok Yoon Chee, Chong Guan Ng, Lei Cheng Lit, Anis Nadhirah Khairul Anuar, Lara M. Lange, Zih-Hua Fang, Sara Bandres Ciga, Katja Lohmann, Christine Klein, Azlina Ahmad-Annuar, Kalai Arasu Muthusamy and Shen-Yang Lim in Journal of Parkinson's Disease
Footnotes
Acknowledgments
The authors gratefully acknowledge funding from The Ministry of Higher Education Malaysia Fundamental Research Grant Scheme (FRGS/1/2020/SKK0/UM/01/2) and University of Malaya Parkinson's Disease and Movement Disorders Research Program (PV035-2017). This project was supported by the Global Parkinson's Genetics Program (GP2). GP2 is funded by the Aligning Science Across Parkinson's (ASAP) initiative and implemented by The Michael J. Fox Foundation for Parkinson's Research (![]() ). For a complete list of GP2 members see https://gp2.org. Open access funding was provided by a generous donation from Mr. Shahabudin bin Maideen. The authors also gratefully acknowledge Medtronic staff Mr Brian Kah Chun Hor, Mr Lybron Lee Kian Pooi, and Ms. Amira Aina Hasbullah for providing DBS-related technical support to patients and their families.
). For a complete list of GP2 members see https://gp2.org. Open access funding was provided by a generous donation from Mr. Shahabudin bin Maideen. The authors also gratefully acknowledge Medtronic staff Mr Brian Kah Chun Hor, Mr Lybron Lee Kian Pooi, and Ms. Amira Aina Hasbullah for providing DBS-related technical support to patients and their families.
This research was supported in part by the Intramural Research Program of the NIH, National Institute on Aging (NIA), National Institutes of Health, Department of Health and Human Services; project number ZIAAG000534, as well as the National Institute of Neurological Disorders and Stroke.
ORCID iDs
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by a grant awarded to SYL from the Ministry of Higher Education Malaysia (FRGS/1/2020/SKK0/UM/01/2) and the University of Malaya Parkinson's Disease and Movement Disorders Research Program (PV035-2017) awarded to SYL and AHT. AMD was supported by a grant from the Global Parkinson's Genetics Program (GP2) funded by the Michael J. Fox Foundation.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
SYL receives grants from the Michael J. Fox Foundation (MJFF) and the Ministry of Education Malaysia Fundamental Research Grant Scheme. He serves on the Editorial Board for Neurotorium. He has received honoraria for talks sponsored by Eisai, Lundbeck, Medtronic, and the International Parkinson & Movement Disorder Society (MDS). AHT receives grants from the MJFF, Ministry of Education Malaysia Fundamental Research Grant Scheme, and Toray Science Foundation Science & Technology Grant. She has received honoraria for talks sponsored by Sanofi, and the MDS. AMFDC has received travel grant from MDS. JWH has received research grant from Malaysia - TORAY Science Foundation (Science & Technology Research Grant). LCL receives grants from the MJFF. ZHF has a contract with MJFF for Parkinson's Research. LML has received fellowship grant from the Bachmann-Strauss Dystonia and Parkinson's Foundation. KL receives grants from German Research Foundation (DFG). CK has received honoraria from Bial and Desitin, and receives royalties from Oxford University Press and grants from DFG, MJFF, Federal Ministry for Education and Research (BMBF) – Federal Republic of Germany. She serves on the advisory board of Retromer Therapeutics and does consultancy for Centogene. AAA has received honoraria from Global Parkinson's Genetics Program for travel, and research grants from Malaysian Ministry of Education Fundamental Research Grant Scheme and University of Malaya Specialist Centre Research Grant. KAM has received honoraria from Medtronic for proctorship and lectures.
Data availability
The data supporting the findings of this study are available on request from the corresponding author. The data are not publicly available due to privacy or ethical restrictions.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
