Abstract
Children with severe traumatic brain injury (TBI) experience medical complications including paroxysmal sympathetic hyperactivity, disorders of consciousness, mood disruption, agitation, and upper motor neuron syndrome. This review article examines the pharmacologic agents used in management of complications of pediatric TBI and the evidence supporting the use of these medications. Much of the literature to support medications used in pediatric TBI comes from adult literature; therefore, more research is needed to examine the impact of these agents in pediatric patients.
Introduction
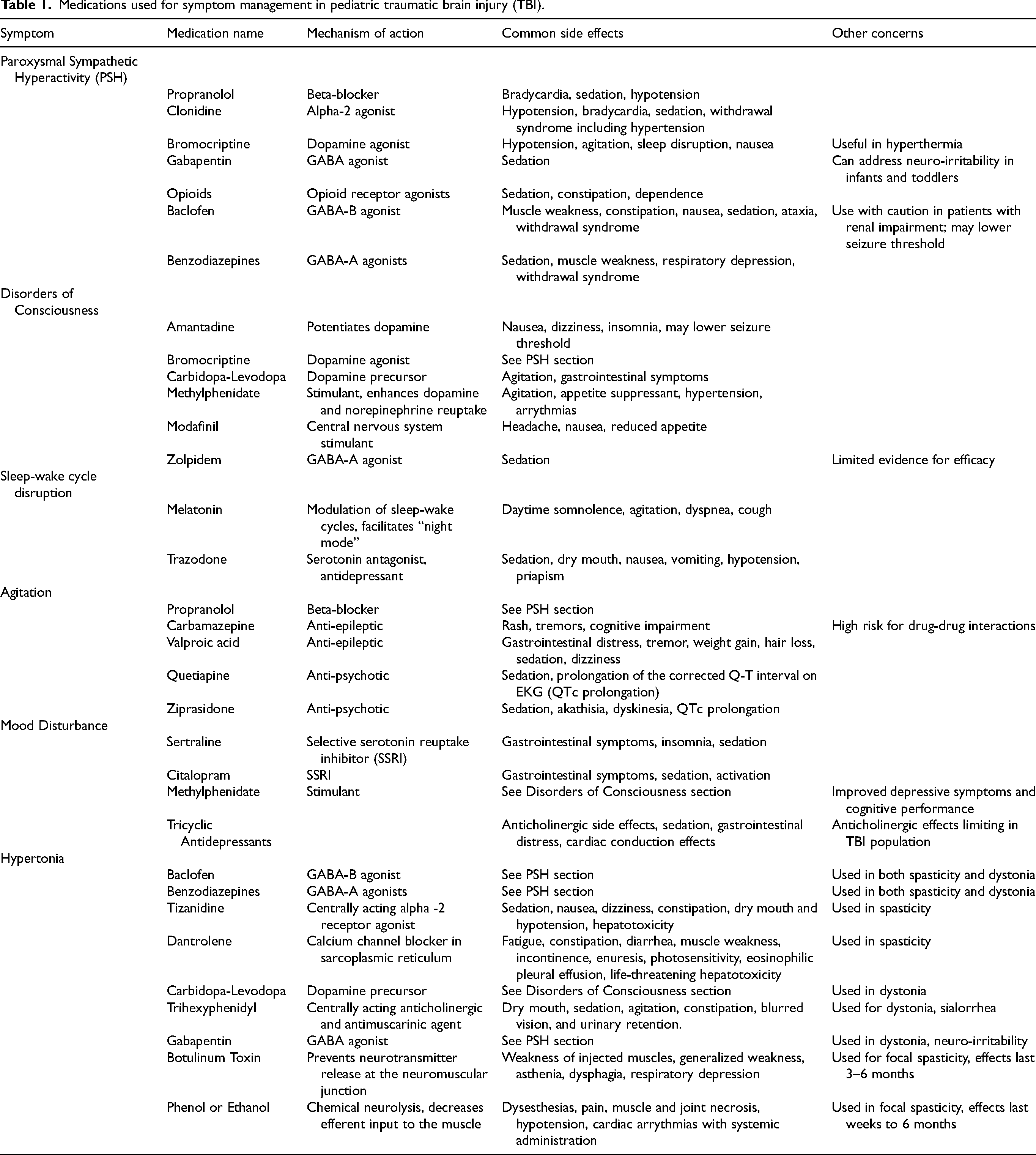
The incidence of pediatric traumatic brain injury (TBI) in the United States is approximately 697,000 cases per year, with the highest incidence in children ages 0–4 and adolescents aged 15–19. Most TBIs in children are mild; however, 5–10% of cases are moderate to severe and result in significant morbidity and functional impairment. Children with moderate to severe TBI can have an altered level of consciousness, sympathetic nervous system dysregulation, hypertonia, mood disturbances, and agitation.1,2 The primary goal of pharmacologic intervention in pediatric TBI is to support recovery and prevent secondary complications. Evidence for pharmacologic management of the sequelae of pediatric TBI is limited and is often extrapolated from adult studies. This article will review commonly used medications in the management of medical complications of TBI in children. Please see Table 1 for a chart of all medications and their indications discussed in this manuscript.
Medications used for symptom management in pediatric traumatic brain injury (TBI).
Paroxysmal sympathetic hyperactivity
Paroxysmal sympathetic hyperactivity (PSH), also known as storming or dysautonomia, refers to disinhibition of the sympathetic nervous system caused by damage to cortical, thalamic, and subcortical areas of the brain.2–4 Noxious stimuli, such as infection, pain, medication withdrawal, seizures, constipation, and urinary retention, trigger an increase in sympathetic nervous system activity. The resulting sympathetic overdrive is manifested by vital sign abnormalities, such as hypertension, hyperthermia, tachycardia, and tachypnea, as well as diaphoresis and dystonic posturing. 2 PSH is associated with longer hospital stays, higher likelihood of discharge to acute inpatient rehabilitation, and worse neurologic outcomes.2,4 PSH is often underrecognized and undertreated. 4
Treatment of PSH begins with elimination of triggers, followed by pharmacologic interventions. Environmental modifications can be made to reduce triggers for PSH, including limiting overstimulation (e.g., television, bright lights, loud noises) and optimizing sleep-wake cycles. 2 Pharmacologic management is directed at the specific symptoms of PSH for each patient. Propranolol, a lipophilic beta-blocker, is a useful agent for PSH with vital sign abnormalities, particularly tachycardia. Propranolol should be avoided in patients with bradycardia. 4 Because it crosses the blood-brain barrier, propranolol modulates sympathetic hyperactivity inside the brain and directly on peripheral targets, such as the heart, lungs, skeletal muscle, and arterioles. 5 A randomized controlled trial of propranolol in adults with TBI showed a decrease in circulating catecholamines, improvement in Glasgow Coma Scale scores, and improvement in heart rate, respiratory rate, and temperature at seven days post injury. 5 Propranolol has a neuroprotective effect post-injury by reducing cerebral metabolism and hypoxia and has been shown to improve cerebral perfusion in animal models. 4 There is case-report level evidence that propranolol can reduce the frequency of PSH events and reduce heart rate in anoxic brain injury. 6
Clonidine is a centrally acting alpha-2 adrenergic agonist that also can provide a sympathetic blockade in PSH. It has been shown to reduce circulating catecholamines in humans and rats with brain injury, and in humans, it can decrease cerebral vasoconstriction. Clonidine and propranolol have been shown to decrease PSH in adults in a randomized controlled clinical trial. 7 Clonidine has more side effects than propranolol, including sedation and rebound hypertension after discontinuation. 4 A case series of two children with TBI showed improvement in PSH after initiation of clonidine. 8 Bromocriptine, a synthetic dopamine agonist, has been used with success in patients with temperature instability and diaphoresis. While its mechanism in mitigating temperature instability in PSH is unclear,4,9 dopamine depletion has also been implicated in hyperthermia caused by neuroleptic malignant syndrome. 10 The dopaminergic properties of bromocriptine can be stimulating, potentially worsen agitation, and impact sleep, as well as cause nausea and hypotension.9,10
Medications used to treat neuropathic and nociceptive pain can control PSH triggered by pain. Gabapentin is a gamma-aminobutyric acid (GABA)-agonist that can be useful in treating PSH, particularly in infants and very young children.4,11,12 Gabapentin can address neuro-irritability and hyperalgesia, which are often noted in this age group. 12 A case series of adolescents and young adults with TBI implicated gabapentin in improvement of PSH. 11 Opioid medications may also be helpful for short-term management of PSH that may be mediated by pain.4,8 Gabapentin and opioids can cause sedation, though opioids have multiple other side effects, including constipation and dependence. Opioids inhibit catecholamine release and modulate sympathetic afferents both peripherally and centrally, which may explain their utility in PSH. 3 Case series have shown improvement with use of morphine in children with PSH. 8
Medications that mediate hypertonia are used to manage posturing that can be seen in PSH; these agents will be described in more detail in the Spasticity and Dystonia section. Baclofen can target PSH by modulating sympathetic activity and controlling associated hypertonia. 4 Benzodiazepines, such as diazepam, are used for management of PSH with hypertonia, though they should be used cautiously as they may delay cognitive recovery in adults. 4 More studies are needed to determine optimal management of PSH in pediatric patients. 2
Disorders of consciousness
Children with severe TBI can exhibit decreased wakefulness and awareness of their environment.1,13 Catecholamines, such as dopamine, modulate wakefulness through the reticular activating system, which is vulnerable to TBI.14–16 The acute post-injury phase is associated with increased dopamine release; however, chronic brain injury is associated with low dopamine levels. 17 Medications used in TBI to increase arousal are referred to as neurostimulants, and they often increase dopaminergic transmission. 18 Amantadine is the most frequently prescribed neurostimulant according to a study of pediatric patients with TBI admitted to the intensive care unit. 19 In adult patients, it is the only neurostimulant that has been shown to improve functional recovery when given between four and 16 weeks post injury. 20 It increases synthesis and synaptic release of dopamine and decreases its reuptake. The most common side effects of amantadine are nausea, dizziness, and insomnia. Amantadine may also decrease the seizure threshold, though in some cases, it can be used to treat certain types of seizures.21,22 Bromocriptine is a dopamine agonist similar to amantadine. A small retrospective case series of five patients (ages 16–27) showed functional improvement with bromocriptine, 23 and a second study showed improvement in cognition in adults with TBI after six months.24,25 Carbidopa-levodopa combines carbidopa, a dopamine precursor, and levodopa, which enables carbidopa to enter the central nervous system (CNS). 26 This medication is useful for patients with dystonia and disorders of consciousness. 1 A small retrospective study showed that carbidopa-levodopa improved alertness in children with brain injury. 27
This small study also found that methylphenidate, a CNS stimulant, improved alertness in children with brain injury. 27 Methylphenidate enhances dopamine and norepinephrine reuptake. 28 In adults, this medication may improve mental processing speed, attention, and working memory, and it is often used in patients who have emerged from disorders of consciousness to target these higher-level cognitive goals. A randomized controlled trial showed that methylphenidate improved attention-deficit/hyperactivity disorder symptoms in children five years after TBI. 29 This medication has a fast onset of action, which can show clinical improvement quickly. 28 Modafinil, used in narcolepsy, is another CNS stimulant used to target daytime sleepiness in adults with brain injury. 30 Modafinil modulates dopamine reuptake, increasing extracellular levels of dopamine in the striatum and prefrontal cortex. 30 There is a case report of improvement in daytime sleepiness with use of modafinil in an adolescent with secondary narcolepsy due to TBI. 31
Prior studies with zolpidem in adults, a GABA-A agonist, show an improvement in social and environmental interaction in approximately 5% of patients. 32 A small placebo-controlled study of three pediatric patients treated with zolpidem showed decreased responsiveness compared to placebo.13,33 There is limited evidence to recommend zolpidem for neurostimulation in pediatric TBI.
Agitation
Agitation in pediatric TBI can appear as explosive anger, physical and verbal aggression, increased psychomotor activity, disorganized thinking, impulsivity, and/or perseveration. 34 During inpatient rehabilitation, up to 70% of adults with TBI experience agitation, 35 and agitation is often encountered in pediatric patients with brain injury. Most pediatric rehabilitation specialists manage agitation using evidence from adult literature as there is a dearth of evidence in pediatric populations.
Management of agitation starts with environmental modifications and optimizing sleep/wake cycles. Wakefulness during the day can be promoted by keeping window shades open and lights on, and turning off lights and minimizing nighttime interruptions can promote consolidated sleep overnight. For children over six months of age, melatonin, a hormone secreted by the pineal gland that regulates circadian rhythm, 36 can be considered to help with sleep initiation. Trazodone, an atypical antidepressant, can be used for sleep maintenance for children over 18 months. 1
Evidence to guide pharmacologic management of agitation in pediatric TBI remains sparse. A recently published case series showed improvement in agitation in four children with TBI 48 h after the initiation of amantadine. 37 There are a few small studies supporting the use of propranolol for agitation in adults with brain injury. 38 Anti-seizure medications (ASMs), such as valproic acid and carbamazepine, have been used to manage TBI-related agitation in adults.39,40 A retrospective chart review showed improvement in agitation with initiation of valproic acid in patients with TBI as young as age 13. 39
Levetiracetam is a commonly used ASM in children with TBI, and it can cause agitation and other behavioral side effects. Supplementation with vitamin B6 (pyridoxine) can be an option to decrease irritability. One retrospective study showed that levetiracetam was less likely to be discontinued in children with epilepsy while on vitamin B6. 41 While a systematic review looking at studies between 2019 and 2022 found seven studies that suggest vitamin B6 may be effective in treatment of behavioral adverse events, placebo-controlled trials in the pediatric population are still warranted to establish efficacy. 42 If the addition of vitamin B6 is not effective in decreasing agitation, a trial of another ASM may be warranted.
Atypical antipsychotics, such as ziprasidone and quetiapine, are preferred over first-generation antipsychotics for management of agitation in patients with brain injury due to their lower side effect profile. An adult study of post-TBI agitation showed a significant reduction in aggression and irritability in seven patients who were treated for six weeks with quetiapine. 43 In another study, ziprasidone was used in twenty children ages nine months to 17 years for 3–8 days and showed a reduction in agitation. 44 Sedation was noted with the use of atypical antipsychotics in many patients, however.13,44
If mood disturbance is contributing to agitation, antidepressants may be considered, 45 though time to efficacy is a concern in the acute phase. Small studies with sertraline and amitriptyline have shown improvement in adults with TBI-related agitation, and more research is required to guide recommendations, especially in the pediatric population.46,47
Mood
TBI in pediatric patients can lead to significant neuropsychological sequelae and mood disorders. 48 In addition to alteration of dopamine pathways, TBI can disrupt serotonin and norepinephrine levels. 49 These changes can predispose patients to depression, anxiety, and other mood disturbances. The neuropharmacological landscape for treating mood-related issues in this demographic is complex and requires careful consideration of developmental factors, medication efficacy, and safety. As with management of other medical sequelae of TBI, clinical treatment of mood disturbance in children with TBI is supported by literature in the adult population.
Medications typically utilized for depressive disorders can also be effective in treating depression after TBI. Selective serotonin reuptake inhibitors (SSRIs) and tricyclic antidepressants (TCAs) have been found to alleviate depressive symptoms following TBI. Treatment with SSRIs not only helps improve depression but may also reduce associated irritability and aggression. 45 Due to concerns regarding both the tolerability and efficacy of TCAs in this population, SSRIs are typically preferred as the first-line treatment for post-TBI depression. Among SSRIs, sertraline and citalopram are often preferred due to their positive effects and comparatively fewer side effects, including fewer drug-drug interactions. 50
A small double-blind study compared methylphenidate, sertraline, and placebo. Both medications were effective in improving depressive symptoms; however, methylphenidate also enhanced neuropsychological performance. 51 Methylphenidate may be beneficial in inpatient settings or when a quick response is needed. Methylphenidate and other stimulants, like dextroamphetamine, can be used to boost responses to SSRIs, especially when cognitive issues or fatigue persist. 50
Other antidepressant classes, such as serotonin-norepinephrine reuptake inhibitors, bupropion, and monoamine oxidase inhibitors (MAOIs) have not been well-studied in patients with TBI. Bupropion can lower the seizure threshold, and the dietary cross-reactions of MAOIs can limit their use in people with cognitive impairment. 50
Spasticity and dystonia
Severe TBI can impair both the pyramidal and extrapyramidal tracts resulting in hypertonia from the upper motor neuron syndrome. Hypertonia can occur as early as the first week after brain injury and can become more prevalent during the first year. 52 The upper motor neuron syndrome may manifest as spasticity when there is injury to the corticospinal and corticobulbar tracts. Spasticity is defined as resistance to passive range of motion that varies with the speed of stretch. Injury to the basal ganglia and other subcortical structures may result in movement disorders, such as dystonia and rigidity. Dystonia denotes involuntary sustained or intermittent muscle contractions that cause abnormal posturing, twisting, or repetitive movements. Dystonia becomes more apparent with active movement. Rigidity is described as resistance of passive movement in both agonist and antagonist muscles around a joint caused by simultaneous co-contraction. 53
Treatment of the upper motor neuron syndrome in brain injury should focus on the functional goals of each individual patient. Therapies and/or bracing focused on maintaining range of motion are considered first line and should accompany any pharmacologic intervention. The severity and distribution of hypertonia, either focal or global, must be considered when selecting a medication.
Enteral medications for spasticity and dystonia should be considered for global hypertonia. Baclofen is an agonist at the beta subunit of the GABA-B receptor at the level of the spinal cord and is a commonly used medication for treatment of spasticity and dystonia. Baclofen is available in both enteral and intrathecal forms. 54 Baclofen can cause constipation, nausea, sedation, confusion, and ataxia, and it can potentially lower the seizure threshold. 55 Caution should be used in patients with renal impairment, who may require a reduction in dosing. 56
The intrathecal baclofen (ITB) pump is a surgically implantable device that can deliver medication directly to the intrathecal space, requiring a lower dose of medication than enteral administration. In the immediate post-injury period, there is concern for impairment in neurologic recovery due to central inhibition from baclofen with early ITB pump implantation; however, a small observational study of adults with acquired brain injury showed improvement in spasticity, as well as improvement in cognitive outcomes. 57 The need for a lower dose of ITB may result in fewer systemic side effects than enteral baclofen. 55 Placement of the baclofen pump catheter has typically been in the thoracic or lumbar spine, resulting in medication effect in the bilateral lower extremities, though catheter placement in the cervical spine can also treat spasticity in the upper extremities. 58 ITB can also be used to manage dystonia, though the evidence for efficacy is limited. 59 Sudden discontinuation of baclofen can lead to withdrawal, characterized by worsened spasticity, hallucinations, seizure, and fever. Withdrawal from baclofen can be life-threatening, and treatment should be initiated immediately. 53
Benzodiazepines, such as diazepam, can be used short-term for the management of hypertonia. Diazepam is an agonist at the alpha subunit of the GABA receptor in the brain and spinal cord. Systemic side effects include sedation, impairment of oral secretion control, ataxia, and risk of developing physical dependence. As with baclofen, there is also a risk of potentially life-threatening withdrawal with sudden discontinuation. Benzodiazepines can also worsen agitation and confusion in the acute brain injury period. 60 However, diazepam can be useful to manage severe hypertonia with associated neuro-irritability in infants and toddlers. 12
Tizanidine, an alpha-2 receptor agonist in the spinal cord and brain, is a medication used in adults with spinal cord injury and multiple sclerosis, though use in the pediatric population is less frequent. There are two small studies in children with cerebral palsy that suggest improvement in spasticity and motor function. Side effects of tizanidine include sedation, nausea, dizziness, constipation, dry mouth, and hypotension. Frequent laboratory monitoring is needed due to the potential for hepatotoxicity. 53
Dantrolene is a calcium channel blocker that inhibits muscle contraction in ryanodine receptors in the sarcoplasmic reticulum of skeletal muscle. Its use is limited by irreversible and potentially life-threatening hepatotoxicity, though this side effect appears to be more frequent in adults over age 35. 61 Evaluation of liver enzymes is indicated prior to initiation and periodically during its continued use. 53 It is common practice to obtain baseline liver enzyme levels and subsequent laboratory monitoring every three months, though there have been instances of hepatic dysfunction occurring as early as one month after starting this medication.62,63 Dantrolene use can also lead to muscle weakness, fatigue, constipation or diarrhea, incontinence, enuresis, photosensitivity, and eosinophilic pleural effusion.53,63
Enteral medications used primarily for dystonia include carbidopa-levodopa and trihexyphenidyl. As previously mentioned, carbidopa-levodopa is a dopaminergic agent that can also be used for neurostimulation. Injury to the basal ganglia leads to disinhibition of extraneous movements, which is implicated in dystonia. 64 Side effects include hypotension, nausea, vomiting, hallucinations, dyskinesias, and bradykinesia. 53
Trihexyphenidyl is a centrally acting anticholinergic and antimuscarinic medication used for dystonia, though its mechanism in treatment is unclear. Trihexyphenidyl is more commonly used in cerebral palsy, 65 as anticholinergics may increase confusion and agitation in acute brain injury. 60 There is a case report of a child with spontaneous basal ganglia hemorrhages who started trihexyphenidyl seven months after injury and showed improvement in dystonia without adverse effect. 66 In larger studies, data for efficacy of trihexyphenidyl is conflicting; however, the medication was initiated at low doses to avoid side effects in these studies.66As the anticholinergic side effects can be helpful in controlling sialorrhea, 67 it is reasonable to consider a trial of trihexyphenidyl in a patient with chronic TBI, dystonia, and sialorrhea while monitoring for anticholinergic side effects.
Medications used to manage spasticity and PSH can also be used in dystonia. For patients with cerebral palsy, the American Academy of Cerebral Palsy and Developmental Medicine (AACPDM) recommends a trial of enteral baclofen as a first-line agent for dystonia, and gabapentin and clonidine as second-line agents for severe dystonia. AACPDM does not recommend levodopa or trihexyphenidyl for management of dystonia in cerebral palsy. 68
Focal hypertonia can be managed using intramuscular botulinum toxins. Botulinum toxins prevent fusion of synaptic vesicles to the synaptic cleft at the neuromuscular junction, preventing neurotransmitter release and inhibiting muscle contraction. 69 Generally, the effects of botulinum toxins last between three and six months;53,70 therefore, it is important to consider the potential for improvement in hypertonia during the acute brain injury period when using botulinum toxins. Stretching and/or serial casting can prolong the effects of botulinum toxins. Botulinum toxins are relatively safe, and many studies have shown efficacy in improving hypertonia and function. However, the risk of infection or bleeding after injection must be discussed with patients, as well as the risk of distant spread of toxin, which can lead to asthenia, generalized weakness, dysphagia, or respiratory depression. 71
Proteolytic alcohols, including phenol and ethanol, are also used in focal hypertonia. When injected at the motor end plate using electromyographic or electrical stimulation guidance, proteolytic alcohols cause chemical neurolysis, decreasing efferent input to the muscle, and resulting in a decrease in spasticity in the muscles innervated by the injected nerve. 69 The onset is almost immediate, and the effects last a few weeks to approximately six months.72,73 When compared to botulinum toxin injections, phenol nerve blocks have an increased side effect profile, including dysesthesias, pain, muscle and joint necrosis, and hypotension.72,73 While phenol is more cost-effective than botulinum toxin, it is used less frequently in the United States.72,73
Medications to avoid
Children with TBI are often exposed to a multitude of sedating medications in the acute care setting, and it is important to wean these medications to facilitate arousal once they are medically stable. 1 Classes of medications that are prioritized in this weaning process include benzodiazepines, opioids, and centrally acting antihypertensives, such as clonidine, if not needed for management of PSH or hypertonia. Adult studies show that these medications can contribute to cognitive side effects including confusion, sedation, and memory impairment, and thus can interfere with brain injury recovery.60,74,75 Regardless, weaning these medications also incurs risks such as uncovering PSH, agitation, and withdrawal. To monitor tolerance of weaning opioids, the Withdrawal Assessment Tool is used in pediatric inpatient settings. 76 To minimize risk, clinicians may choose to wean one medication at a time or alternate days of weaning. The rate of weaning is tailored to each child's tolerance, which can be influenced by duration of treatment with the medication and concurrent medical complications.
Other standing medications to consider weaning or avoiding during the acute brain injury phase are anticholinergics, such as scopolamine and cyproheptadine, due to the risk of increasing confusion and sedation.60,77 Anticholinergic medications may be used to manage sialorrhea. Alternatives for sialorrhea include ophthalmic atropine drops given sublingually or botulinum toxin injections to salivary glands, as systemic side effects are limited due to the mode of delivery.78–81
The use of neuroleptic agents, especially haloperidol and risperidone, for brain injury-related agitation in adults is not a common practice for expert practitioners. 34 These medications also interfere with dopamine transmission, 82 which can interfere with cognition after brain injury. 83 Multiple studies in animals, adults, and children show motor and/or cognitive impairments following chronic administration of haloperidol or risperidone in brain injury.60,84 Haloperidol has been shown to prolong post-traumatic amnesia in adults with TBI. 60
Given concern for worsening medical complications such as agitation and sedation, limiting the above medications in children with TBI is recommended, though more studies are needed to determine the short- and long-term impacts.
Conclusion
Currently, the evidence behind pharmacologic management of sequelae of TBI in children is guided by studies in adults with TBI, with some case series and small studies in children with TBI. It is important to consider that pediatric TBI occurs in the context of rapid developmental changes in childhood 85 and that the pharmacologic agents discussed in this review may impact pediatric patients differently than adults. Therefore, more research is needed on the efficacy of these medications in the pediatric population. 86
Footnotes
Acknowledgements
The Authors have no acknowledgments.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
