Abstract
Purpose:
This study aimed to highlight the structural and functional musculoskeletal changes and associated treatment strategies in paediatric patients with spinal cord disorders (SCD).
Methods:
A systematic scoping review was conducted whereby PubMed, Excerpta Medica Database and MEDLINE Ovid databases, and grey literature were searched for articles published between January 2000 and June 2024. Study criteria included at least 50% of participant cohort being children aged under 18 years at time of SCD diagnosis, investigating the musculoskeletal effects of SCD in children, and related management or interventional strategies. Reports in which paediatric or SCD participants were less than 50% of the cohort were included if results were appropriately stratified. Included reports underwent descriptive analysis.
Results:
Forty-five reports were eligible for inclusion. Physiological Parameters, Musculoskeletal Complications, and Interventions were the main themes identified. It was observed that musculoskeletal changes following SCD can differ in children compared to their adult-injured counterparts, but the mechanisms underlying these differences are not known. Intervention studies that have been performed are important, but underpowered, and cannot yet be translated into routine care.
Conclusion:
Monitoring and treatment guidelines for musculoskeletal health in this population are scarce. Comprehensive research efforts are required to facilitate guideline development, which will be imperative in improving musculoskeletal outcomes following paediatric SCD.
Introduction
Paediatric spinal cord disorders (SCD) are rare, but the impact of SCD in childhood is significant. However, perhaps due to the rarity of this population, studies investigating the effects of SCD on the musculoskeletal system in children are lacking. The term SCD includes both traumatic spinal cord injuries (SCI) and non-traumatic spinal cord dysfunction aetiologies, of which 3.8 and 6.5 per million children younger than 15 years are diagnosed per year in Victoria, Australia, respectively. 1 This aligns with the global incidence of traumatic SCI in children, with 4.3 per million children per year under 15 in developed countries, 2 and between 3.3 to 13.2 per million throughout global regions. 3
Following a SCD, abnormal sensory and motor function below the level of injury results in the loss of the typical load on the bones from weight-bearing and muscle activity. As a result, there is a change in bone structure and disruption of bone growth below the injury site.4–6 The musculoskeletal system is still developing in children, so the effects of SCD on their bones can vary from the effects seen in adults who suffer injury to the spinal cord. 7 Bone mineral density (BMD), as measured using dual X-ray absorptiometry (DXA) and peripheral quantitative computed tomography (pQCT), is heavily reduced in both children and adults with SCD.8,9 In addition, children have an increased risk of developing musculoskeletal complications such as scoliosis, hip dysplasia, and fractures.10–12 Scoliosis and hip dysplasia are the two most common orthopaedic complications that arise following a SCD.13,14 Fracture risk is high in paediatric-onset SCD, as these patients live with this condition for the majority of their lifespan. Their compromised bone health, combined with significant injury duration, often results in fragility fractures which contribute to increased pain and reduced quality of life. 13 Furthermore, children with SCD may never attain peak bone mass levels. Peak bone mass is determined by nutritional, endocrine, and environmental factors such as physical activity, 15 which can be heavily disrupted following a SCD diagnosis. Despite these differences, paediatric SCD literature is scarce. The statistics surrounding paediatric SCD are not as well documented as for their adult counterparts, and there is currently no standard reporting procedure or data registry that exists for the paediatric SCD population in Australia.
A recent systematic review of the literature revealed that between 1974 and 2020, 176 reports were published that concerned paediatric SCD, of which only 36 looked at neuromusculoskeletal and movement-related structure and function. 16 Therefore, many aspects of care for this population may not be receiving adequate attention. While rehabilitation research and clinical practice guidelines (CPGs) are established in adult SCD, there are still substantial gaps in literature,17,18 which is even more evident in the paediatric SCD population.19,20 CPGs for the management of musculoskeletal complications that arise from SCD in children are needed. The International Perspectives on Spinal Cord Injury report provides excellent recommendations for persons living with an SCI; 21 however, these recommendations are not specific to children, and the concepts are not explored in original paediatric research elsewhere. Developing CPGs to support musculoskeletal outcomes will be key to improving quality of life and reducing the risk of pathological fracture and bone deformities that are often associated with paediatric SCD, given that these patients will live with the consequences of their injury for their lifetime.
The aim of this review was to broadly map the musculoskeletal changes following SCD in children. Therefore a scoping rather than systematic methodology was used.22–24 A scoping review of the available evidence was performed because, unlike systematic reviews, scoping reviews attempt to provide an overview of a research topic and can include evidence from a variety of sources.22,23 Systematic reviews typically provide evidence regarding the effectiveness of interventions. Thus the specificity of the research question means they are of limited scope. 24 Conversely, scoping reviews provide the opportunity to review many factors that contribute to a research question, while being produced in a systematic manner. Therefore, a scoping methodology was adopted for this review. This review was conducted according to the methodology outlined by Arksey and O’Malley 22 and refined by Peters et al. 24 The purpose of this scoping review was to explore how SCD diagnoses have been shown to impact the musculoskeletal system of children in the current literature. Therefore, the question driving the basis of this scoping review was: “What are the structural and functional musculoskeletal changes and associated treatment strategies in paediatric patients with spinal cord disorders?”
Methods
Methodology and reporting
This review is reported according to the Preferred Reporting Items for Systematic Reviews and Meta-Analyses Extension for Scoping Reviews (PRISMA-ScR) 23 guideline. The protocol for this review was pre-registered on the Open Science Framework (OSF) Registry, registration DOI: https://doi.org/10.17605/OSF.IO/YFX89
Eligibility criteria
Types of evidence sources included original research reports, systematic reviews, narrative reviews, guidelines, and grey literature websites. Following removal of duplicates, screening of the sources of evidence to be included was performed by two or more reviewers according to the following inclusion criteria:
The diagnosis of SCD occurred in children aged ≤18 years of age. Reports that included at least 50% of participants with a SCD diagnosis. If representation of paediatrics or SCD diagnoses was fewer than 50%, but results were stratified, records could be included for review. Reports investigating the musculoskeletal effects of SCD in children, and related management or interventional strategies. Reports published between January 2000 and June 2024, inclusive.
The SCD diagnoses were eligible for inclusion as outlined by the MeSH term ‘Spinal Cord Diseases’, such as traumatic injuries and non-traumatic causes like myelitis or tumour. Therefore, reports that focused on conditions that do not qualify as an acquired SCD, e.g., spinal dysraphism, were not included. Furthermore, conference abstracts that had insufficient data to extract, reports discussing acute management of the SCI, and reports discussing the methodology of surgical management procedures were excluded. To minimize potential bias, no language restrictions were applied.
Information sources
The record selection process involved searching MEDLINE OVID, Excerpta Medica Database (EMBASE), and PubMed. These were selected due to their broad coverage of material pertaining to medicine and life sciences, whilst the grey literature information sources included government or institutional documents and pamphlet-style guidelines. Reference lists of similar reports were cross-checked to find records in addition to the OSF-registered protocol. Grey literature websites included were Google Scholar, System for Information on Grey Literature in Europe, the website of the American Spinal Injury Association, and the website of the International Spinal Cord Society.
Search
A scientific librarian-assisted search was designed, and JE built and performed the search in the included databases. An example of the electronic search for PubMed database, which was also adapted for EMBASE and MEDLINE OVID code, is available in Appendix A. The search was originally conducted on July 1, 2024, and a subsequent search including all languages was conducted on March 5, 2025.
Selection of sources of evidence
Three reviewers (JE, MG and AS) performed the screening – at both ‘Title and Abstract’ and ‘Full Text’ screening stages, JE screened every record, while MG and AS shared the records as reviewer two. Each reviewer independently screened records, and if any conflicts arose, these were resolved by consensus.
Data charting process and extraction
Covidence, which is an online software tool designed to support the systematic or scoping review process, was used for the entire screening and extraction process. It facilitates the separate screening of titles and abstracts from full texts, enables independent data extraction, and provides a process to reach consensus, which minimizes the risk of bias. 25
Data extracted from sources of evidence were charted using a data extraction form built in Covidence. The form was tested before its use. Data extraction was conducted independently: data from each included report was extracted by two independent reviewers as per the screening protocol, whereby JE extracted data from every report, while MG and AS each extracted data from half of the reports as reviewer two. The data obtained from both reviewers was then compared within Covidence by a single reviewer, and the confirmed data was exported to Microsoft Excel for descriptive analysis.
Data items
The variables gathered were General Information, Methods, Participants, Outcomes and Key Findings. General Information included sub-variables: Title, Year of Publication, Author Contact Details, and Country. Methods included sub-variables: Aim, Study Design, Start and End Dates, Funding Sources, and Conflicts of Interest. Participants included sub variables: Population Description, Inclusion and Exclusion Criteria, Recruitment Method, and Total Number of Participants.
Synthesis of results
Data extracted from each source of evidence were analysed descriptively and summarised into tabular format. The reports included were organised into separate categories based on the type of outcomes discussed. To achieve this, the key findings from sources of evidence were summarised and coded for descriptive qualitative content analysis. The main outcomes or keywords were used to identify whether the findings were reporting primarily biological, functional, or interventional conclusions. Sources of evidence were then grouped into the categories Physiological Parameters, Musculoskeletal Complications or Interventions based on what the main outcomes were exploring.
Results
Search
The combined searches resulted in a total of 6042 records, with the original search returning 6015, and the subsequent search returning 27. Embase retrieved 4488 records, Medline returned 1314 records, PubMed yielded 202 and a search of grey literature revealed 38 additional records, from reference lists, Google Scholar, the American Spinal Injury Association website, and the International Spinal Cord Society website. Following removal of duplicates and screening of ineligible records, assessment of 249 full text articles resulted in 45 reports eligible for inclusion. The sources of evidence were acquired, screened and extracted as summarised in Figure 1, including all relevant reasons for exclusion.
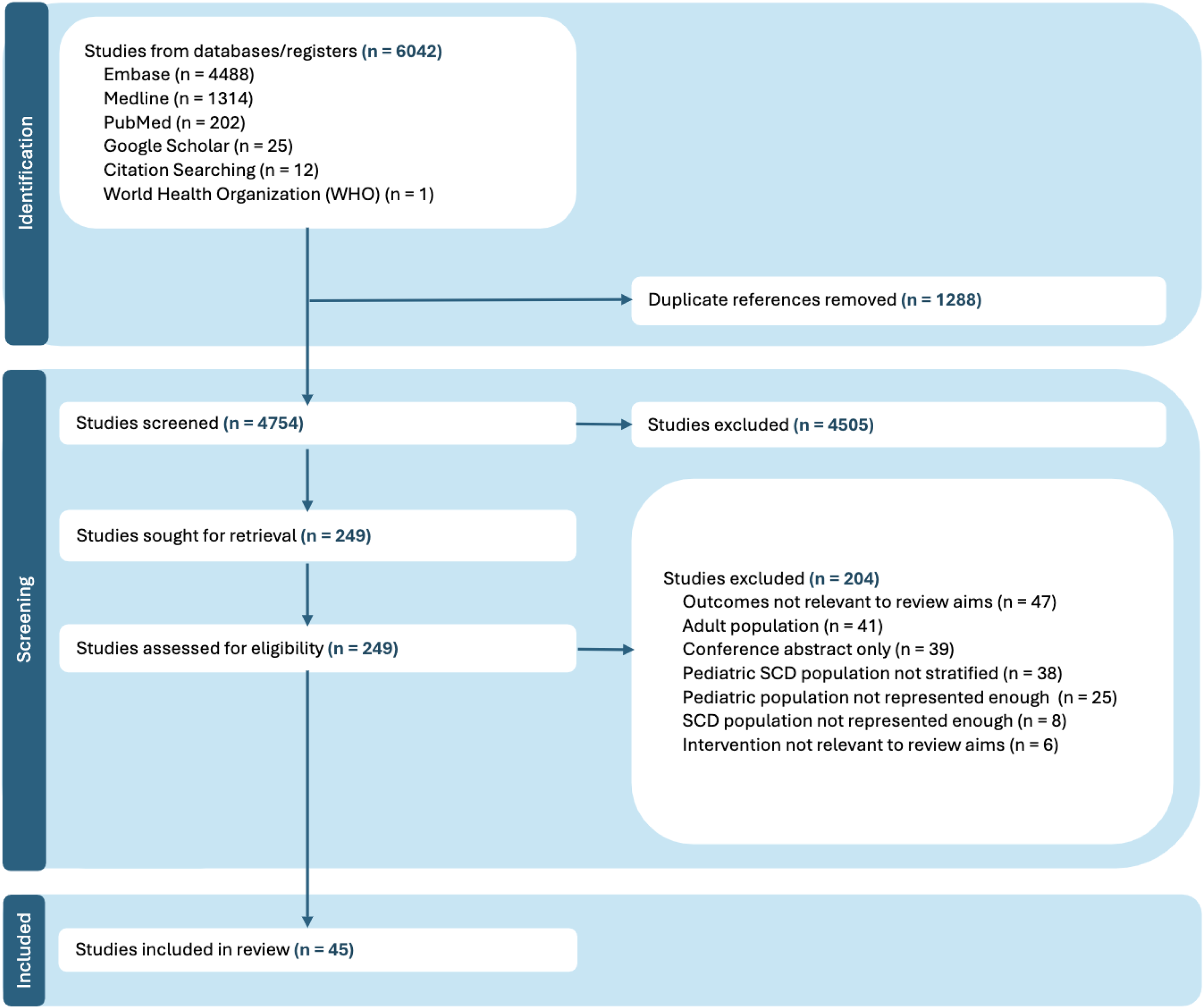
PRISMA for scoping reviews flowchart of records obtained from multiple information sources, generated in covidence.
There were 10 cross-sectional studies, nine retrospective reviews of medical records, six case series, four narrative reviews, four randomised controlled trials, three non-randomised experimental studies, two systematic reviews, two case reports, and one each of a case-control study, cohort study, book chapter, conference task force, and guideline document. Seventeen studies received no funding, 16 were supported by grants from Shriners Hospitals for Children, 10 were funded by independent foundations and charities, and two studies were supported by both independent grants and a Shriners Hospitals for Children Grant, as indicated in Tables 1, 2, and 3.
Theme: Physiological parameters.
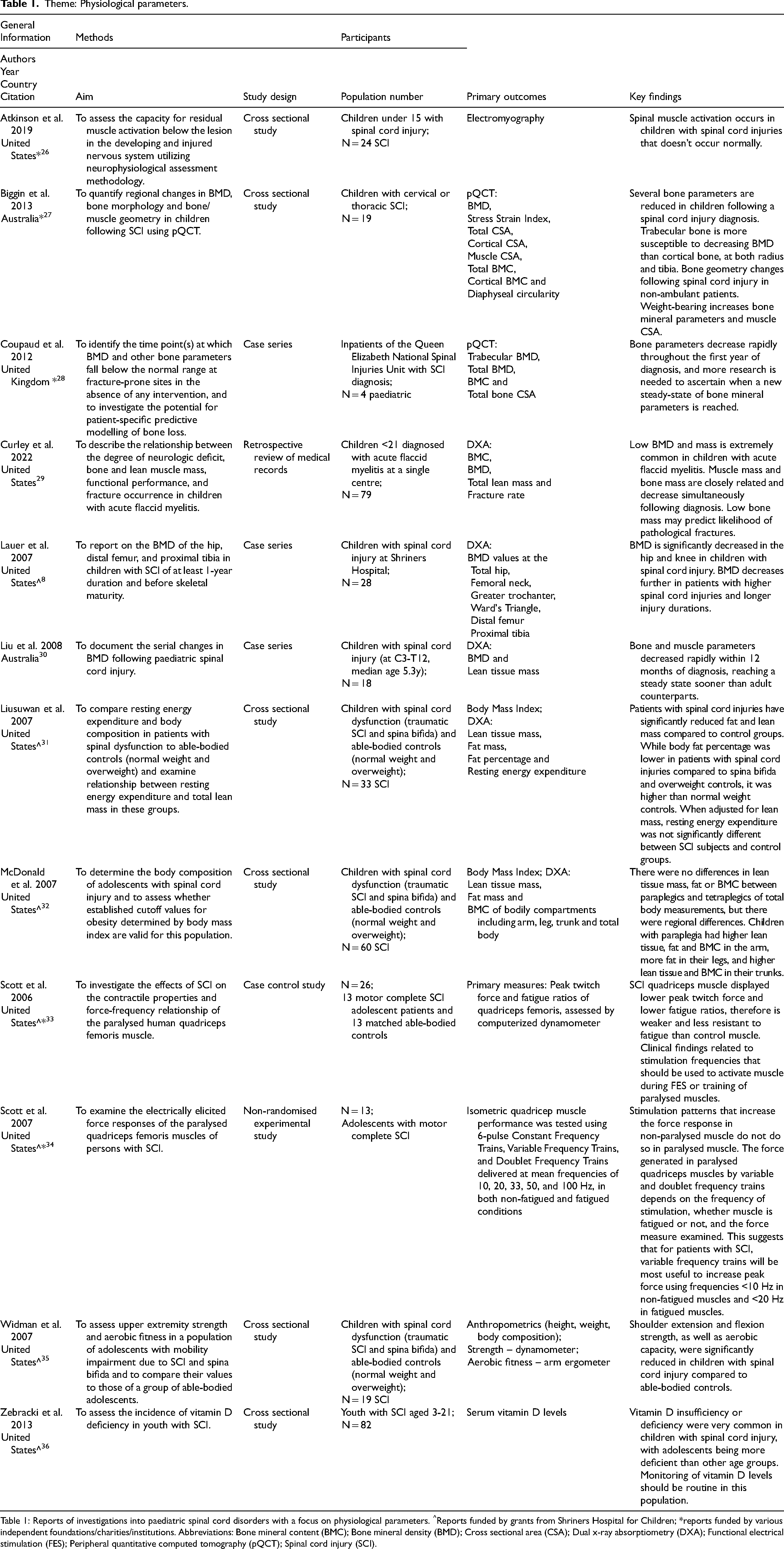
Table 1: Reports of investigations into paediatric spinal cord disorders with a focus on physiological parameters. ^Reports funded by grants from Shriners Hospital for Children; *reports funded by various independent foundations/charities/institutions. Abbreviations: Bone mineral content (BMC); Bone mineral density (BMD); Cross sectional area (CSA); Dual x-ray absorptiometry (DXA); Functional electrical stimulation (FES); Peripheral quantitative computed tomography (pQCT); Spinal cord injury (SCI).
Theme: Musculoskeletal complications.

Table 2: Reports of investigations into paediatric spinal cord disorders with a focus on Musculoskeletal Complications. ^Reports funded by grants from Shriners Hospital for Children; *reports funded by various independent foundations/charities/institutions. Abbreviations: Spinal cord injury (SCI).
Theme: Interventions.
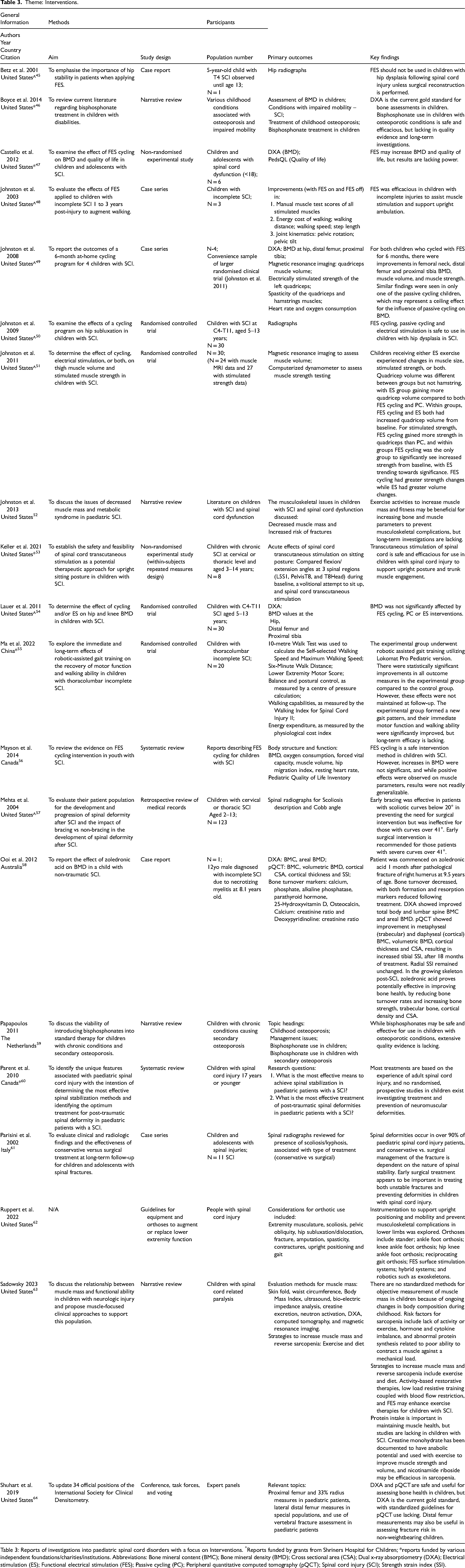
Table 3: Reports of investigations into paediatric spinal cord disorders with a focus on Interventions. ^Reports funded by grants from Shriners Hospital for Children; *reports funded by various independent foundations/charities/institutions. Abbreviations: Bone mineral content (BMC); Bone mineral density (BMD); Cross sectional area (CSA); Dual x-ray absorptiometry (DXA); Electrical stimulation (ES); Functional electrical stimulation (FES); Passive cycling (PC); Peripheral quantitative computed tomography (pQCT); Spinal cord injury (SCI); Strength strain index (SSI).
The majority of studies were conducted in North America (United States 29/45; Canada 2/45), and one was a multi-site study conducted in both countries (1/45). The remaining studies were conducted in the United Kingdom (4/45), Australia (4/45), China (2/45), Italy (1/45), Switzerland (1/45) and the Netherlands (1/45).
Examination of the included reports revealed three general types of conceptual approach. These included studies that investigated the Physiological Parameters, Musculoskeletal Complications or possible Interventions related to paediatric SCDs.
Physiological parameters
Twelve included reports were of investigations into various Physiological Parameters relating to the paediatric SCD experience (Table 1). These sources of evidence highlight that patients with SCD have significantly reduced bone mineral content (BMC), BMD, lean tissue mass and cross-sectional area (CSA),8,27,32 abnormal muscle activation, 26 and reduced muscle strength,33–35 compared to typically developing children. Cortical bone appears to be less likely to reduce in density than trabecular bone, and bone circularity changes appear to occur within the early months of diagnosis. 27 Two studies observed that these parameters decreased most rapidly throughout the first 12 months of diagnosis.28,30 It was also observed that vitamin D insufficiency and deficiency are common within this population. 36 These losses of bone and muscle physiology parameters appear to be correlated with loss of weight-bearing ability and decrease further over time.29,30
Musculoskeletal complications
Thirteen included reports were of investigations into various Musculoskeletal Complications associated with paediatric SCD (Table 2). The most common musculoskeletal complications associated with paediatric SCD included scoliosis, spasticity, hip dysplasia, hypercalcemia, fractures, contractures7,11–14,37–41,43,44 and, to a much lesser extent, heterotopic ossification. 42 These effects are more profound in patients diagnosed during childhood, compared to those injured later in life.13,14,38–40,44 A key issue may be the lack of appropriate stress upon bones during critical periods of bone growth, on the journey to peak bone mass, as ambulant children have fewer complications associated with their condition. Ambulatory status, defined as those who are community ambulators, positively correlated with BMD in the femur, based on DXA assessment of the distal femur. 29 Furthermore, load-bearing in a standing frame (or better) attenuated the losses in trabecular density, cortical area, and muscle CSA. Mobile patients able to stand without a standing frame exhibited better tibial circularity scores than patients who could not, based on pQCT assessment of the tibia. 27 Independently ambulatory patients who walked with an assistive walking device or better were also less likely to have scoliosis. 39 Age at injury was highlighted as a risk factor for scoliosis and hip dysplasia, with children diagnosed pre-puberty being at greater risk.11,12,14,40,41 Complete injuries and flaccid paralysis were also noted to correlate with the development of hip subluxation.14,41 Spasticity was associated with tetraplegia, and rates increase with injury duration.7,14 It was also highlighted that scoliosis may arise from changes to motor control of the spine following injury, rather than the fracture of the spinal column itself. 37
Interventions
Twenty included reports were of investigations into various potential Interventions to assist in the treatment of paediatric SCD (Table 3). Several evidence sources explored the use of functional electrical stimulation (FES) in paediatric SCD and found it to be safe for use in children, but these studies often had small sample sizes and therefore lacked power.45,47–51,53,54,56 Most of these studies reported trends towards increases in BMD with FES.
It was also proposed that exercise interventions52,63 to increase weight-bearing as well as dietary strategies to support muscle mass would also be of benefit for muscle and bone parameters. Some studies explored the use of robot-assisted exercise programs to facilitate muscle mass and walking ability, but long-term efficacy is unknown.55,62
Pharmacological interventions included mostly bisphosphonate treatment,46,58,59 which was reported as safe for use in children and used following fragility fractures on a case-by-case basis. 59
Two studies explored the utility of surgery or bracing to prevent paralytic scoliosis,57,61 in which bracing was only effective in cases where scoliosis was less severe, 57 and early surgical intervention was recommended to prevent scoliosis development.57,61
Discussion
Summary of evidence
This scoping review revealed several gaps in the literature regarding the musculoskeletal health and management in children with SCD. While it is well understood that musculoskeletal changes following SCD can differ in children compared to their adult-injured counterparts, the mechanisms underlying these differences are not known. The timeline of bone and muscle deterioration is not comprehensively understood, and the development of musculoskeletal complications such as spinal deformities should be investigated further. The prevalence of musculoskeletal complications following paediatric SCD have been documented, yet CPGs supporting musculoskeletal health are lacking and complications are generally treated on a case-by-case basis.
While the existing sources of evidence are important in the grand scheme of paediatric SCD research, there is an overwhelming dearth of high-quality evidence that could support the development of evidence-based CPGs. However, a recent report on CPGs for the management of autonomic dysfunction in SCD highlighted that lower-level evidence can be used to develop CPGs if there is consensus, and that is the only evidence available. 65 For example, based on the findings from this review, the first point to discuss for future guidelines for managing musculoskeletal health in paediatric SCD would be to alert clinicians to the high prevalence of these complications in these children compared to adults with SCD. This is a health issue that is often not considered during acute management and can be ignored in these early stages when other health complications are more pressing, yet the deterioration of musculoskeletal health can result in significant complications such as spinal deformities or fragility fractures. By the time these occur, they are too late to prevent and they negatively impact quality of life. Therefore, the findings of this scoping review could provide the basis for a similar undertaking to develop CPGs regarding musculoskeletal health for children with SCD. Implementation of preventative monitoring strategies may reduce the incidence of these complications, which in turn may reduce associated pain and increase quality of life.
The current standard for bone health assessment in paediatric SCD is DXA.64,66 But while DXA is used to assess bone mineral parameters, often following a fragility fracture, it does not provide the best data by which to judge outcomes.27,66 pQCT has superior resolution and assesses several meaningful bone health parameters that DXA does not, such as volumetric BMD, skeletal geometry, and bone strength, and distinguishes between trabecular and cortical bone. 67 However, while it has been utilised in some prospective studies, it is not used in routine clinical care for children with SCD due to insufficient paediatric reference data and technical issues pertaining to the positioning of the 4% scan. 68 The 4% site measures the trabecular bone at the metaphyseal region of the radius and tibia, which is proximal to the growth plate. This makes obtaining measurements at the same reference point within this growing population rather difficult. 68 Further studies are required to ascertain the utility of each imaging modality for the purposes of bone health monitoring in paediatric SCD. However, this concept of including musculoskeletal imaging assessments before pathological complications arise could help improve the care and management of bone health in this population.
In terms of potential interventions to promote musculoskeletal health in paediatric SCD, very few controlled studies have been performed. Of the 20 reports that discussed potential interventions, only eight involved prospective study designs, of which there were only five distinct cohorts – four of these reported multiple outcomes from the same cohort,49–51,54 with the remaining four reporting on independent cohorts.47,48,53,55 This highlights a significant lack of studies aimed at developing interventions to support musculoskeletal health in paediatric SCD. These studies, while often suggesting promising effects, lacked power due to small sample sizes.47–49,53 Given the overall low prevalence of paediatric SCD, this may be a persistent issue; therefore, national and international collaborative research efforts may require consideration.
Bisphosphonates were the primary focus for pharmacological intervention.46,58,59 They are used in adults and children to increase BMD and treat bone fragility conditions such as osteoporosis, as they increase bone mass and prevent resorption. 69 However, prospective studies to support routine clinical use are lacking. 59 Overall, interventions for musculoskeletal health are lacking, but quality intervention studies cannot be performed until more research is conducted within this population. For example, longitudinal studies exploring the timeline and mechanisms of bone deterioration 28 would help contribute to the foundation of knowledge of bone health in paediatric SCD that this scoping review highlights.
Limitations
A key observation from this scoping review revealed that nearly half of the evidence sources were supported by grants from one centre in the USA. Eighteen of the 45 reports included arose from research efforts by the Shriners Hospital for Children. However, this condition is not a phenomenon exclusive to the USA, and this large contribution by one centre to the research field may introduce bias. Corroborating data from other treatment centres outside of the USA, in both hospital and community centres, is very much needed due to potential effects of differing healthcare systems and to avoid potential bias in reporting.
Due to the breadth of available sources of evidence, it was challenging to standardise the information extracted from each reference. The varying methodologies and limited number of publications on this topic meant that uniform analysis of available literature was difficult. For example, while the recommendations from some of the studies included in this review may be useful and relevant for paediatric SCD, many of the techniques discussed were more related to the methods used for assessment of bone health in children in general, not specifically to children with SCD. The overall lack of studies performed on this topic meant that the published reports are difficult to compare and generalise to the entire population of paediatric SCD.
This is further emphasised by the aetiology focus of most of the included reports. While this scoping review has revealed the status of the literature landscape for musculoskeletal health in paediatric SCD, it appears that many studies include traumatic aetiologies only. Given that nearly two-thirds of the paediatric SCD population are represented by non-traumatic causes,1,3,70 the lack of discussions of non-traumatic aetiologies in the literature means that the experience of over half of the paediatric population with SCD is not being adequately captured. Non-traumatic diagnoses result in heterogenous SCD experiences; the injuries to the spinal cord are often incomplete yet extensive, with damage commonly occurring across several spinal levels. 71 While this may make it difficult to study this population, it should not be ignored.
Conclusions
This scoping review aimed to ascertain from the literature the structural and functional musculoskeletal changes and associated treatment strategies in paediatric patients with spinal cord disorders. Gaps highlighted included a lack of underlying mechanistic knowledge for the timeline of musculoskeletal deterioration and development of complications. This underscores the lack of available CPGs to support the management of musculoskeletal health in these children.
Implications of the findings for research, practice, and policy
Investigations into potential interventions to support musculoskeletal outcomes are imperative. What has been achieved to date has shown several potentially positive outcomes, but these studies are underpowered, thus their findings have not yet been translated into routine clinical care. Future studies further exploring the effects of standing and weightbearing interventions, electrical stimulation, and pharmacological strategies to manage musculoskeletal complications following paediatric SCD would be beneficial, however, it would be possible for CPGs to be produced if consensus can be reached. Therefore it is also recommended that paediatric SCD clinicians and researchers engage in discussions of the findings from this scoping review, to reach consensus on a set of guidelines based on current knowledge and evidence to date, thus improving the management of musculoskeletal complications for these children.
Footnotes
Acknowledgements
We thank Poh Chua from The Royal Children's Hospital library for her assistance building the database searches used in this study.
Funding
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Appendix A: Sample search strategy (PubMed)

| #1. Title/abstract |
| “sci” OR “scd” OR “spinal-cord-injur*” OR “spinal-cord-disorder*” OR “spinal-cord-disease*” |
| #2. Title/abstract |
| “newborn*” OR “new-born*” OR “baby” OR “babies” OR “neonat*” OR “neo-nat*” OR “infan*” OR “toddler*” OR “pre-schooler*” OR “preschooler*” OR “kinder” OR “kinders” OR “kindergarten*” OR “kinder-aged” OR “boy” OR “boys” OR “girl” OR “girls” OR “child” OR “children” OR “childhood” OR “pediatric*” OR “paediatric*” OR “school-age*” OR “schoolage*” OR “schoolchild*” OR “schoolgirl*” OR “schoolboy*” OR “adolescen*” OR “youth” OR “youths” OR “teen” OR “teens” OR “teenage*” |
| #3. Title/abstract |
| “skeletal” OR “musculoskeletal” OR “fracture*” OR “scoliosis” OR “dislocation*” OR “contracture*” OR “bone*” OR “tibia” OR “radius” OR “femur” OR “ulna*” OR “hip” OR “pelvis” OR “paraplegi*” OR “quadriplegi*” OR “DEXA” OR “DXA” OR “dual energy x-ray absorptiometry” OR “pQCT” OR “peripheral quantitative computed tomography” OR “bisphosphonate*” OR “vitamin D” OR “bone mineral density” OR “muscle” |
| #4. Title/abstract |
| “longterm” OR “long-term” OR “repeat*” OR “serial” OR “longitudinal*” OR “follow-up” OR “followup” OR “cohort*” OR “retrospective*” OR “prospective*” OR “clinical-stud*” OR “clinical-articl*” OR “clinical-trial*” |
| #5. NOTNLM OR publisher[sb] OR inprocess[sb] OR pubmednotmedline[sb] OR indatareview[sb] OR pubstatusaheadofprint |
| #6. #1 AND #2 AND #3 AND #4 AND #5 |
| Limit from year 2000 onwards |
