Abstract
Background
The CD155-TIGIT axis, a breast cancer progression biomarker, underscored neoadjuvant chemotherapy (NAC) response variability in triple-negative breast cancer (TNBC), urging biomarker-based patient stratification for timely therapy.
Methods
Thirty-nine TNBC patients who received NAC were recruited. The expression of TIGIT, CD155, CD226, and CD96 on tumoral and stromal cells in the tumor microenvironment was detected by immunohistochemistry, and their relationships with NAC response were explored.
Results
10.3% patients exhibited grade 1 (G1) response to NAC, and 20.5% achieved a complete pathological response. Notably, CD155 and CD96 were predominantly detected on tumor cells, whereas CD226 and TIGIT were predominantly detected on stromal cells. The expression of these markers did not significantly correlate with response to NAC (p > 0.05), and each individual marker lacked predictive power for determining NAC therapeutic efficacy (p > 0.05). However, a specific combination of tumoral cells expression of CD226(≥4%), CD155(≥40%), and CD96(≥35%), coupled with TIGIT expression on tumoral (<35%) and stromal cells (<12.5%), was able to identify patients with G1 response to NAC.
Conclusion
Expression levels of TIGIT/CD155/CD226/CD96 on tumoral and stromal cells might collectively serve as predictive biomarkers for NAC response in TNBC. This implied that CD155-TIGIT axis could be prospectively applied clinically to identify NAC-resistant TNBC patients.
Keywords
Introduction
In 2020, breast cancer (BC) was identified as the most prevalent malignant neoplasm among women, with 2.26 million new cases reported worldwide. 1 Triple-negative breast cancer (TNBC), which comprised approximately 10–20% of BC cases, was noted for its unfavorable prognosis.2,3 Despite the continuous development of innovative therapeutics, TNBC was associated with a higher risk of recurrence and metastasis.4–7
Characterized by high immunogenicity, high mutation rates, and extensive lymphocytic infiltration, TNBC was recognized as a “hot tumor” suitable for immunotherapy.8–12 Neoadjuvant chemotherapy (NAC) had been a commonly used therapeutic approach for TNBC, however, a subset of patients exhibited reduced sensitivity, increased progression risk, and potential loss of the optimal window for surgical intervention. Consequently, there was a pressing need for biomarkers or diagnostic tools to identify non-responsive patients and provide effective treatment.
The tumor microenvironment (TME), a complex system consisting of neoplastic cells, immune constituents, extracellular matrix, fibroblasts, cytokines, and metabolic byproducts, was dynamically interacting to modulate the immunological phenotype of the tumor. 13 These immunological elements were demonstrated to dynamically influence tumor development and prognosis.14–16
TIGIT, a transmembrane inhibitory protein expressed on the surface of various immune cells, had ligands including CD155, CD112, and CD113.17,18 Co-stimulatory and co-inhibitory receptors such as CD226 and CD96 were also expressed on TME immune cells. 19 These glycoproteins interacted to form the CD155-TIGIT immune checkpoint axis, which influenced antitumor immune responses through multiple pathways. The interaction between TIGIT and CD155 was shown to suppress the cytotoxic activity of natural killer (NK) cells, weaken T cell functions, and inhibit inflammatory responses. 20 Moreover, TIGIT exhibited stronger binding affinity to CD155 compared with CD226 and CD96, thereby antagonizing the antitumor benefits derived from CD226-CD155 interaction. 18 Therefore, this study was designed to investigate the collective predictive effects of TIGIT, CD155, CD226, and CD96 on NAC efficacy in the pre-surgical setting. The overarching goal was to facilitate the identification of vulnerable patients with low likelihood of benefiting from NAC and enable personalized therapeutic planning.
Materials and methods
Ethical approval
All procedures involving human participants complied with the ethical standards of the Institutional Review Board of Beijing Shijitan Hospital, Capital Medical University (sjtkyll-1x-2021(108)) and adhered to the 1964 Helsinki Declaration and its subsequent amendments. The requirement for informed consent was waived by the Institutional Review Board due to the retrospective, de-identified nature of the data and minimal risk to participants, in accordance with applicable ethical guidelines.
Study setting and design
This retrospective study was conducted using tumor biopsy samples obtained from adult female patients diagnosed with TNBC at Beijing Shijitan Hospital, Capital Medical University between 2010 and 2020. The diagnosis of TNBC based on the preoperative biopsy and pathological detection. Inclusion criteria: (1) Patients were pathologically confirmed with primary TNBC. (2) Patients had not received any form of BC-directed therapy. (3) ECOG performance status was ≤ 2. (4) There was no radiologically confirmed distant metastases. (5) Patients had available paired tumor specimens (pre-NAC core needle biopsies and post-NAC surgical resection samples). Exclusion criteria: (1) Lactating or pregnant females. (2) Severe dysfunction of vital organs. (3) History of autoimmune disorders or previous use of immunomodulatory therapies. (4) Concurrent or past diagnosis of other malignant neoplasms. Subsequently, the patients received preoperative NAC and followed surgical resection or palliative chemotherapy. The efficacy of NAC was assessed by pathological detection on the surgical tissues.
Data collection and definition
The study collected clinical and pathological data, including age, pathological diagnosis, tumor histological grade, tumor stage, Ki-67 status and NAC regimen.
TNBC was identified using immunohistochemistry (IHC) and fluorescence in situ hybridization (FISH) techniques. For IHC testing of negative estrogen receptor (ER) and progesterone receptor (PR) status, the nuclear staining of tumor cells was less than 1%. The human epidermal growth factor receptor 2 (HER-2) negative status was assessed as 0 or 1 + by IHC. When a 2+ IHC result was obtained, FISH testing was required to confirm negativity.
Tumor histological grading was defined according to the Nottingham grading system, which integrated the proportion of gland formation, nuclear pleomorphism, and mitotic count to determine the overall tumor grade. The score ranges for grade I, II and III was 3–5, 6–7 and 8–9, respectively. Tumor staging was performed according to the tumor node metastasis (TNM) classification system, in accordance with the guidelines of the 8th edition of the American Joint Committee on Cancer. TNM stage was classified as I (T1N0M0), II (T0∼1N1M0, T2N0∼1M0, T3N0M0), III (T0∼2N2M0, T3N1∼2M0, T4N0∼2M0, T0∼4N3M0), and IV (T0~4N0~3M1). Ki-67 expression was defined as brown nucleus in BC cells by IHC on 4 μm-thick formalin fixed paraffin-embedded sections, and Ki-67 index was measured as the proportion of Ki-67 expression among BC cells in the hot-spot area.
The NAC regimens in the study participants consisted of the following: EC(A)-T, EC(A), TE(A), T, TX-EC(A), TP-EC(A), TX, TX-EC(A), and EC(A). Definitions of regimen components were as follows: (1) T (Taxanes): docetaxel, paclitaxel, albumin-bound paclitaxel, or liposomal paclitaxel. (2) E(A) (Anthracyclines): epirubicin, doxorubicin, or pirarubicin. (3) C: cyclophosphamide. (4) X: capecitabine. (5) P: carboplatin. All regimens were administered in 14- or 21-day cycles. For monotherapy or combination regimens, treatment duration was 4–6 cycles. Sequential regimens comprised 4 cycles of the initial protocol followed by 4 cycles of the subsequent regimen. Dose modifications were permitted based on toxicity management or disease progression, with protocol adjustments overseen by board-certified oncologists.
NAC efficacy
The Miller & Payne assessment system was utilized to evaluate the NAC chemotherapeutic efficacy on the primary lesion. G1 response indicated the invalid NAC treatment and responses of G2 to G5 indicated effective treatment.
IHC detection and scoring of TIGIT, CD155, CD226 and CD96
The expression of TIGIT, CD155, CD226, and CD96 on the membrane of tumor cells and stromal immune cells was detected via IHC using the EnVision two-step method. Two pathologists estimated stromal tumor-infiltrating lymphocytes (TILs) located within the borders of the invasive tumor areas, excluding regions with crush artifacts, necrosis, regressive hyalinization, and biopsy sites. All mononuclear cells (including lymphocytes and plasma cells) were scored, whereas polymorphonuclear leukocytes were excluded. In cases of scoring discrepancies between the two pathologists, a third senior pathologist reviewed the IHC staining.
Monoclonal antibody against CD155 (rabbit anti-human, #81254S) was purchased from Cell Signaling Technology Co. Ltd Monoclonal antibodies against CD226 (rabbit anti-human, #ab2120772) and TIGIT (rabbit anti-human, #ab243903) were purchased from Abcam Co. Ltd Polyclonal antibody against CD96 (rabbit anti-human, #PA5-97568) was purchased from Invitrogen Co. Ltd Secondary antibodies were purchased from Beijing Zhongshanjinqiao biotechnology Co. Ltd.
Positive expression was defined as brown membrane staining in cells. The percentage of positive tumor cells was calculated as the proportion of tumor cells with molecule expression across the entire section, while the percentage of stromal TILs was defined as the proportion of stromal immune cells with positive molecular expression in the whole section.
Statistical analysis
All analyses were performed using R software (v4.4.2, https://www.R-project.org/). Continuous data were described using the median and interquartile range (IQR). The Mann-Whitney U test was used to assess differences in the expression of CD155/CD226/CD96/TIGIT on tumor cells and TILs between NAC-effective and NAC-non-effective groups. Spearman's rank correlation test was employed to evaluate associations between biomarker expression and response subgroups (G1-G5). A decision tree model was constructed using the Classification and Regression Tree algorithm with Gini impurity as the splitting criterion to assess the diagnostic utility of molecular biomarkers in predicting NAC non-response. The receiver operating characteristic (ROC) curve was employed to evaluate the model's diagnostic accuracy. Ten-fold cross-validation was performed to estimate the model's generalizability. All analyses were two-tailed with the significant level of 0.05.
Results
Clinical information of TNBC patients
Totally 39 patients were enrolled in this study, with a median age of 52 years (IQR = 15) and a median Ki-67 index of 60% (IQR = 50%, Table 1). 27 patients were diagnosed as histological grade Ⅱ and 12 patients were in grade Ⅲ (Table 1). Most patients (80%) were in stage Ⅱ and Ⅲ (Table 1). Almost half of TNBC patients chose E(A)C-T regimen (49%), and other NAC regimens accounted for 10%, respectively (Table 1). 90% patients obtained an effective NAC efficacy but 10% patients had an invalid efficacy (Table 1). Among the patients with ineffective treatment, 3 of them received the EC-T regimen, and the remaining one received the AC regimen for NAC.
Basic characteristics of TNBC patients receiving neoadjuvant therapy.
Expression of TIGIT, CD155, CD226 and CD96
The median percentage of CD155 was 60% (IQR = 40%) in TME tumor cells, but the TILs did not show any CD155 expression (Table 2). The median percentage of CD226 expression was 1% and 40% on tumor cells and TILs, respectively (Table 2). The median percentage of CD96 expression was 60% and 20% on tumor cells and TILs, respectively (Table 2). The median percentage of TIGIT expression was 0% and 10% on tumor cells and TILs, respectively (Table 2).
Expression of TIGIT, CD155, CD226 and CD96 in tumor microenvironment.

Association between NAC efficacy and individual molecule
The expression of TIGIT, CD155 and CD96 on TME tumor cells and TILs did not present any association with pathological responses of NAC (Table 3). TNBC patients with invalid NAC treatment had slightly higher median percentage of CD226 expression on tumor cells than those with effective NAC treatment (15% vs. 0%, p = 0.092, Table 3).
Expression differences of immune check-point molecules between pathological responses of NAC patients.
Prediction of TIGIT, CD155, CD226, and CD96 expression in tumor cells and stromal TILs for NAC efficacy
A decision tree model was constructed to predict pathological responses to NAC. Patients with tumoral CD226 expression (<4%) uniformly exhibited NAC-effective (Figure 1A). Among the tumoral CD226 high expression (≥4%) subgroup, the probability of NAC non-response increased from 10.3% to 26.7% (Figure 1B). Within this tumoral CD226 high expression (≥4%) subgroup, further stratification revealed that patients with stromal TIGIT expression at least 12.5% maintained NAC-effective (Figure 1A), whereas those with stromal TIGIT expression less than 12.5% showed a 50.0% non-response rate (Figure 1B). Among these lower stromal TIGIT expression (<12.5%) patients, those with low tumoral CD155 expression (<40%) all responded to NAC (Figure 1A), whereas those with higher tumoral CD155 expression (≥40%) showed a 66.7% non-response rate (Figure 1B). Within the higher tumoral CD155 expression (≥40%) subset, only one patient with low tumoral CD96 expression (<35%) responded to NAC (Figure 1A), while those with higher tumoral CD96 expression (≥35%) exhibited an 80.0% non-response rate (Figure 1B). In the final stratification, one patient with higher tumoral TIGIT expression (≥35%) responded to NAC (Figure 1A), whereas all four patients with lower tumoral TIGIT expression (<35%) were non-responders (Figure 1A), resulting in a 100.0% non-response rate (Figure 1B). The decision tree model demonstrated excellent diagnostic performance for predicting non-response to NAC, with an area under the ROC of 1.0 (Figure 2). Following 10-fold cross-validation, the mean misclassification rate for the model was 8.1%.
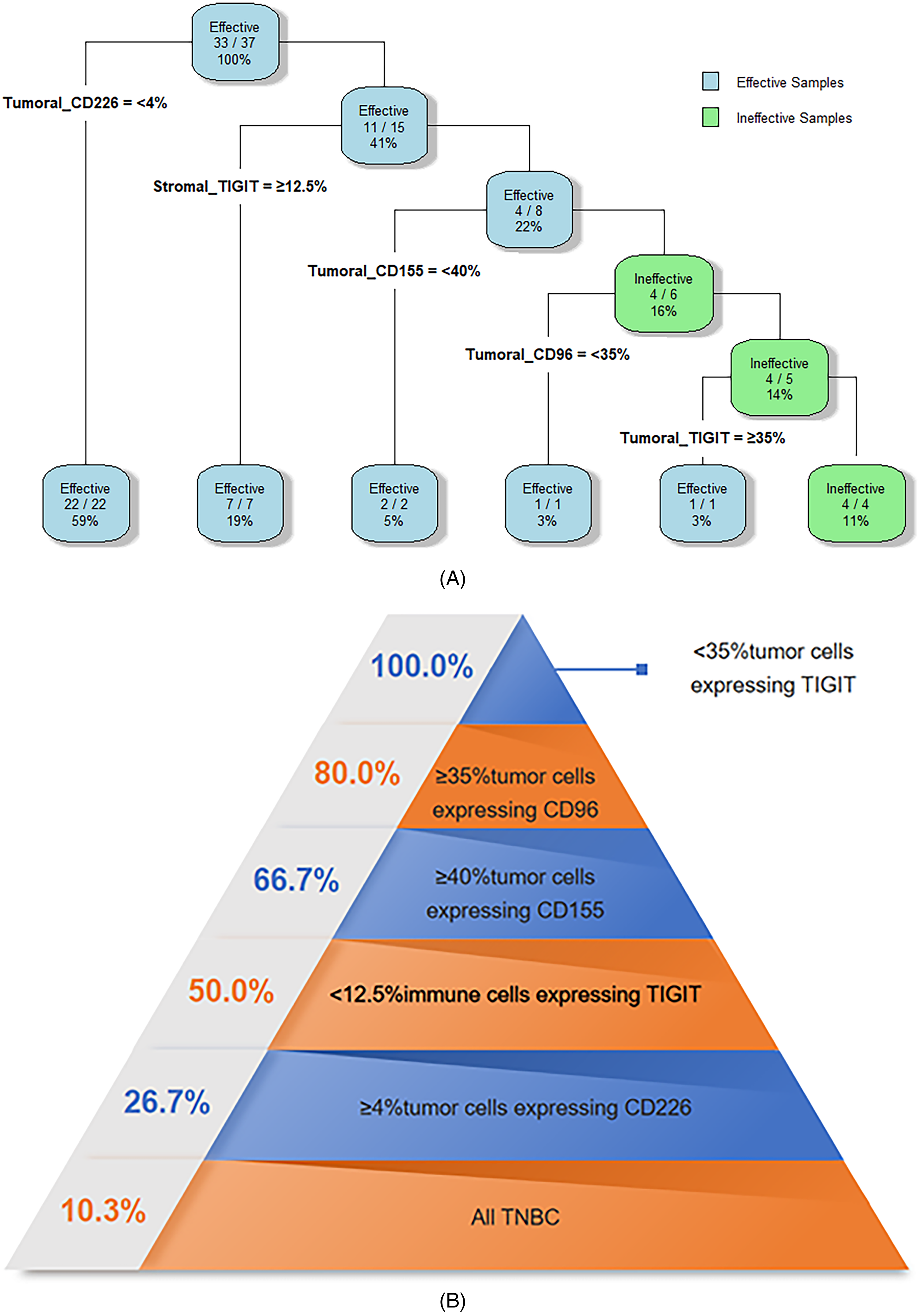
Prediction process of NAC treatment efficacy based on sequential assessment of TME CD226/TIGIT/CD155/CD96 in TNBC, A. Novel decision tree model for predicting NAC efficacy, B. Probability of invalid NAC with reference to TME CD226/TIGIT/CD155/CD96 expression.
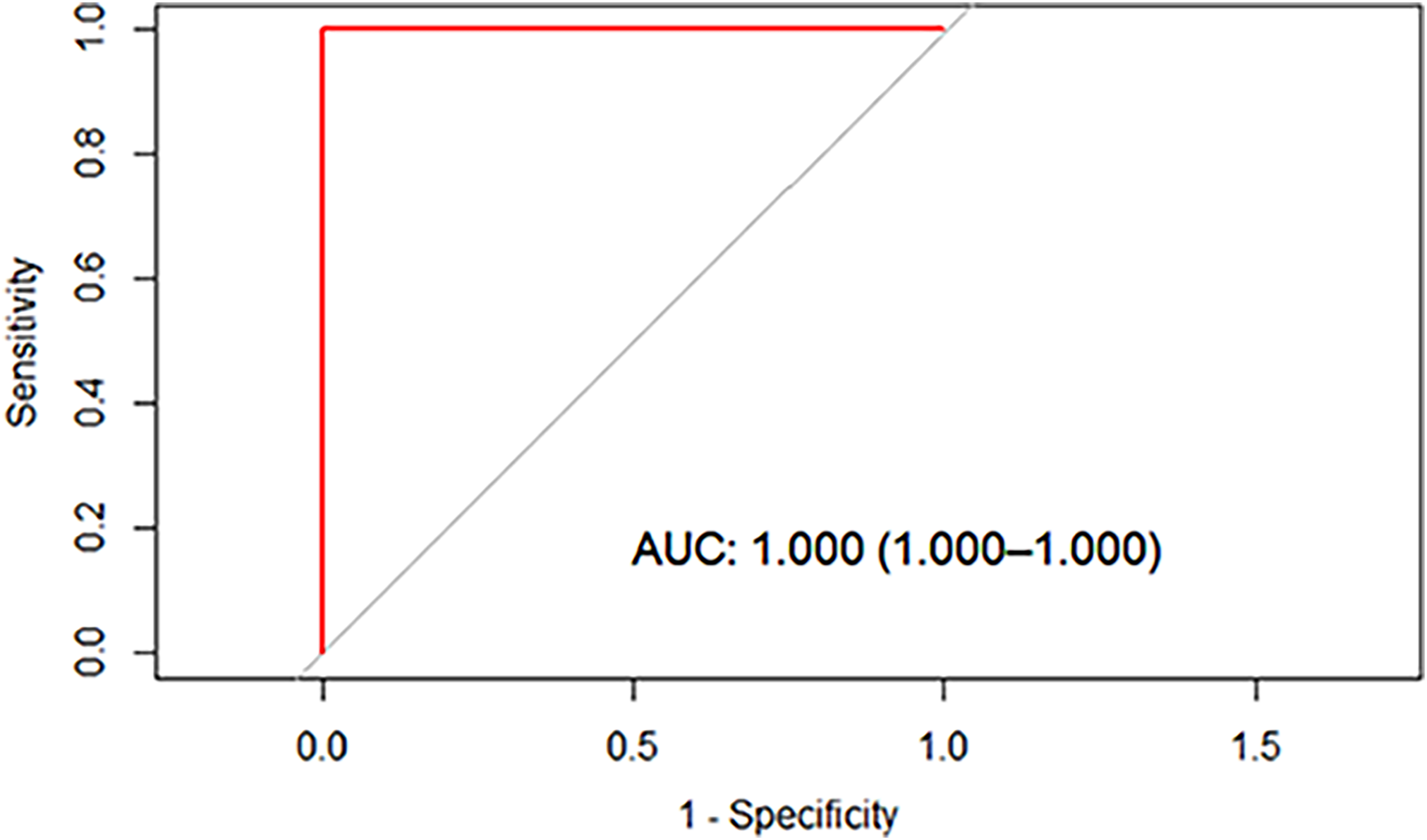
Receiver operating characteristic curve of the decision tree model.
Discussion
This study innovatively investigated the checkpoint complex expression of CD155-TIGIT/CD226/CD96 and found CD155 was exclusively expressed in tumor cells, whereas TIGIT/CD226/CD96 exhibited both tumoral and stromal expression within the TME. In addition, sequential detection of tumoral CD226, stromal TIGIT, tumoral CD155, tumoral CD96 and tumoral TIGIT expression was found to predict the ineffective NAC efficacy and identify vulnerable patients who may benefit from alternative therapies.
Characterized by its complex composition and functional diversity, the TME was dynamically influenced by clinical therapies.9,13,21–25 The components of the TME were closely correlated with the clinical outcomes of malignancies.26,27 TILs was reported to modulate chemotherapy efficacy and contribute to chemoresistance.24,26 PD-L1 and TILs were previously documented to possess the potential to predict NAC efficacy in BC.28,29 Concurrently, studies conducted by Zeng et al., 30 Abbasov et al., 31 and Cabioglu et al. 32 observed that CD155 and TIGIT immune checkpoint in the TME of TNBC exhibited predictive potential for NAC treatment efficacy.
Similar to PD-L1, TIGIT expression was higher in TNBC patients compared to other molecular subtypes.33,34 TIGIT was found to suppress T cell function, and its expression was increased when co-expressed with immune suppressive receptors such as PD-1, LAG-3, and TIM-3, while concurrently downregulating CD226. 35 Additionally, TIGIT mRNA levels were correlated with primary tumor size, hormone receptor status, and other clinical variables of BC. 19 Although TIGIT expression tended to be higher in later tumor stages, increased TIGIT expression was associated with longer overall survival(OS), 33 and higher expression of TIGIT on T and NK cells was linked to better responses to NAC treatment among BC patients. 31
CD155, also known as Necl-5, was involved in cell-to-cell adhesion and highly expressed on tumor cells. 36 Binding to its receptors CD96 or TIGIT induced suppression of NK cell function, thereby facilitating immune evasion. 20 In contrast, engagement of CD155 with CD226 triggered the activation of NK cells, leading to tumor cell lysis.20,36,37 Elevated CD155 expression was identified in various solid tumors and was associated with poor prognosis. 20 Patients with high CD155 expression tended to exhibit poorer responses to chemotherapy and higher tumor residue. 32 Even in the context of immunotherapy, high CD155 expression was correlated with reduced responses to anti-PD-1/PD-L1 therapy.32,37
CD96 served as an indirect indicator of the abundance of T cells and NK cells, 38 and was identified as a molecule activating T cells function.38,39 However, CD96 expressed in tumor cells was reported to correlate with poorer prognosis. 40 Elevated CD96 was indicated as an unfavorable factor for NAC efficacy in BC, and blockade of CD96 was shown to enhance response to chemotherapy. 40 Moreover, through its interaction with CD155, CD96 was demonstrated to stimulate chemoresistance in BC tumor stem cells. 40 The CD155-TIGIT immune checkpoint network was suggested to have potential for predicting chemotherapy responsiveness. 40
CD226, also known as DNAM-1, acted as a costimulatory molecule by enhancing the function of T cells and NK cells, and was primarily involved in tumor recognition and immune surveillance. 12 An elevated TIGIT/CD226 ratio was associated with a predisposition to metastasis and poor outcomes with immunosuppressive therapy.12,35 Bioinformatics analysis showed that upregulation of the CD226 gene in BC TME was associated with favorable OS. 12 Guillerey et al. reported that CD226 expression enhanced the therapeutic efficacy of cyclophosphamide and bortezomib in multiple myeloma treatment. 41
TIGIT, CD96, CD155, and other molecules within this immune checkpoint axis were observed to co-regulate with PD-1 and exhibited similar correlations with clinical and pathological parameters. 27 With the involvement of CD226, immunotherapies targeting TIGIT, CD96, CD155, and PD-1 demonstrated synergistic clinical effects in malignant tumors, even achieving 100% complete remission rates.20,35,42 Furthermore, these molecules were suggested to be utilized for evaluating the anticipated benefits of treatment strategies prior to their implementation.28,43 This study determined the potential feasibility of the CD155-TIGIT immune checkpoint axis complex as a predictive tool for NAC drug sensitivity and treatment efficacy. It highlighted the necessity of collectively applying these markers to identify the vulnerable patients with G1 responses to NAC.
The major limitation of this study was the limited sample size, which confined multivariate and subgroup analyses. The lack of interaction analysis with NAC regimens constituted the second limitation, and further studies should combine investigations of chemotherapy regimens with these molecular markers. Thirdly, this study focused solely on primary tumor sites, without considering metastatic lesions, which might exhibit distinct immunological profiles. 44 Moreover, the reliance on a hospital-based sample introduced Berkson bias, which could have limited the generalizability of findings to broader populations. Therefore, future studies should further explore and validate our findings in larger, multicenter TNBC cohorts that include standard immunotherapy regimens.
Conclusion
In this study, the CD155-TIGIT immune checkpoint axis complex was demonstrated to possess potential as a collective panel for predicting TNBC patients vulnerable to non-responsiveness to NAC treatment. Further studies were warranted with a larger sample size to verify the predictive efficacy of the entire immune checkpoint complex. Such identification could facilitate the timely implementation of alternative treatment strategies, thereby potentially reducing the risk of disease progression.
Supplemental Material
sj-xls-1-cbm-10.1177_18758592251390248 - Supplemental material for The predictive effect of the CD155-TIGIT immune checkpoint axis complex on neoadjuvant chemotherapy efficacy in triple-negative breast cancer: A preliminary study
Supplemental material, sj-xls-1-cbm-10.1177_18758592251390248 for The predictive effect of the CD155-TIGIT immune checkpoint axis complex on neoadjuvant chemotherapy efficacy in triple-negative breast cancer: A preliminary study by Keyu Yuan, Xin Ou, Shuzhen Lyu, Feng Shi, Quan Zhou, Yanjie Zhao, Jiangping Wu, Yu Cao, Yanping Li and Qingkun Song in Cancer Biomarkers
Footnotes
Author contributions
KY, XO, JW conducted the detection of molecule, and were major contributors in writing the manuscript. YZ, YC and SL were responsible for data collection and analysis. QZ and FS performed the detection of molecule in tumor microenvironment. YL and QS designed, supervised and optimized the entire study. All authors read and approved the final manuscript.
Funding
This research was funded by Beijing Municipal Committee of Science and Technology grant number Z19110006619041, High Level Public Health Technical Talents Construction Project from Beijing Municipal Health Commission (2022-2-025) and Beijing Hospital's Authority (XMLX202114). The supporting organizations had no role in study design, data collection, analysis, and interpretation.
Conflict of interest
The authors declare that they have no competing interests.
Availability of data and materials
The datasets generated and analysed during the current study are not publicly available due to privacy and ethical but are available from the corresponding author on reasonable request.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
