Abstract
Background
Hypoxia and leptin receptors (also called obesity receptors, OB-R) are evident markers of tumor progression and have been demonstrated to be essential oncogenes in a variety of cancers. However, the specific role of OB-R in lung cancer, especially non-small cell lung cancer (NSCLC) and its correlation with HIF1α remains unclear. Present study aims to explore the potential functions and mechanisms of OB-R in NSCLC.
Methods
The RNA levels of HIF1α and OB-R in NSCLC cells were detected by quantitative real-time PCR (qRT-PCR) and western blotting. The HIF-1α, OB-R, and Ki67 levels in tumor tissues were detected by immunohistochemistry. CCK8 assays for proliferation, transwell assays for migration were performed to determine the role of HIF-1α and OB-R in vitro, while subcutaneous tumors in nude mice were used for in vivo functional studies. Mechanically, chromatin immunoprecipitation and luciferase reporter gene analyses were executed to determine the relationship between HIF-1α and OB-R.
Results
qRT-PCR and western blotting revealed that HIF-1α and OB-R was highly expressed in NSCLC cells. Moreover, hypoxia up-regulated OB-R expression in NSCLC cells via HIF-1α. Hence, down-regulating HIF-1α significantly reduced the mRNA level of OB-R. In addition, HIF-1α silencing reduced cell proliferation and migration in vitro. Xenograft mouse models indicated that decrease of HIF-1α led to tumor growth by decreasing OB-R in vivo. Mechanically, we unveiled that HIF-1α bound to the promoter region (−831 to −824) and positively regulated OB-R expression by activating its transcription. Additionally, by immunohistochemical staining, we observed that high levels of HIF-1α and OB-R were positively associated with tumor size and lymph node metastasis.
Conclusion
In conclusion, our present results demonstrated that HIF-1α positively regulates the expression of OB-R, which acts as an oncogene in NSCLC. HIF-1α and OB-R are potential therapeutic targets in NSCLC.
Introduction
Worldwide, lung cancer, which has the highest morbidity and mortality among all malignant tumors, ranks the first cause of cancer-related deaths in China. 1 Among lung cancer, approximately 80% to 85% of cases are histologically classified as non-small-cell lung cancer (NSCLC). 2 Hypoxia of tissues is a fundamental characteristic of lots of solid tumors that promotes cancer development in different ways. 3 Lack of oxygen is a key factor that makes most cancers difficult to cure, and also leads to increased drug resistance in cancer cells. Hypoxia-inducible factor-1 (HIF-1) is a master regulator of the response to tumor hypoxia, and it has a heterodimer, which includes an alpha subunit (HIF-1α) and a beta subunit (HIF-1β).4,5 When cells are under normoxic conditions, HIF-1α is oxygen-sensitive and is quickly degraded through the ubiquitin-proteasome pathway, with a half-life of less than 1 min. However, cells faces to hypoxic stress, the level of HIF-1α is significantly increased. 6 This happens due to decreased ubiquitination levels, which causes HIF-1α and HIF-1β to quickly polymerize, thus a dynamic dimer is formed and regulating downstream genes expressions. 7 HIF-1α is highly expressed and has been widely proposed to be a significant prognostic factor in lung cancers.8,9 Many studies have found that HIF-1α not only directly promotes obesity gene(OB) transcription in breast cancer cells, but also in pancreatic, colon and endometrial cancer cells, showing that HIF-1α could be involved in OB-R regulation in kinds of cancers. 10
Leptin is a peptide hormone synthesized by adipocytes, which is the product of the OB gene and is the ligand of the leptin receptor (OB-R) to regulate energy expenditure.11,12 In humans, OB-R has several alternatively spliced forms, including OB-Ra, OB-Rb, OB-Rc, OB-Rd, OB-Re, and OB-Rf. 13 However, only OB-Rb is the functional receptor of leptin because of its full-length isoform possessing an extended intracellular domain, which contains several motifs. These motifs interact with other proteins and activate subsequent signaling pathway. Emerging studies have found that both leptin and OB-R are highly expressed in a various cancers, such as breast, colon, prostate, and pancreatic cancers.14–16 The leptin and OB-R could activate the JAK/STAT, MAPK, and PI3K signaling pathways to facilitate tumor proliferation, metastasis, angiogenesis, and drug resistance.12,17 Previous studies showed that lung cancer patients of China had higher leptin levels than normal in serum and lung tissue. Furthermore, the expression of leptin is an independent and unfavorable prognostic factor for NSCLC.18,19 However, rare studies have investigated the regulatory mechanisms of OB-R in NSCLC.
In our study, both HIF-1α and OB-R were highly expressed and positively correlated with the tumor size and lymph node metastasis in NSCLC. Moreover, HIF-1α facilitated OB-R expression by activating transcription. Targeting HIF-1α and OB-R could be a novel therapeutic strategy for NSCLC.
Materials and methods
Cell culture
Human lung cancer cell lines H1299, H1975, A549, H446 were purchased from the Chinese Academy of Sciences (Shanghai, China) and cultured in Dulbecco's modified eagle medium (DMEM) supplemented with 10% fetal bovine serum (FBS, Gibco, USA) adding 1% penicillin-streptomycin (Gibco). H520 and normal human bronchial epithelial cells (HBE) was purchased from the Fuheng BioLogy(Shanghai, China) and cultured in RPMI 1640 with 10% FBS. All cells were cultured in a humidified atmosphere containing 95% air and 5% CO2 at 37°C. For hypoxia treatment, the cells were cultured in a modulator-incubator in an atmosphere consisting of 94% N2, 5% CO2, and 1% O2.
Cell transfection with siRNA
To knockdown HIF-1α, three small interfering RNAs (siRNAs) targeting human HIF-1α were obtained from GenePharma (Shanghai, China). The si1 HIF-1α and si3 HIF-1α had higher silencing efficiency, and were selected for subsequent research. The target sequences were si1 HIF1α: 5’-GCACAGTTACAGTATTCCA-3’ and si3 HIF1α: 5’-AGTTCACCTGAGCCTAATA-3’. The lung cancer cells were transiently transfected with the siRNA kit (GenePharma), following the manufacturer's instructions. All of these experiments were performed either in 6-well plates or in 96-well plates when 50%-80% density was reached on the day of transfection. And cells were cultured in mediated culture medium for 48 h to 72 h after transfection before protein and RNA extraction.
Quantitative real-time PCR
Total RNA was extracted using TRIzol reagent (Invitrogen, San Diego, CA, USA). cDNA was synthesized using a GoldenstarTM RT6 cDNA Synthesis Kit (TsingKe Biotech, Beijing, China) and quantitative real-time PCR (qRT-PCR) was performed by 2xT5 Fast qPCR Mix (TsingKe Biotech). PCR were performed in 95°C for 15 min, 40 cycles of 95°C for 15 s, 57°C for 30 s, and 72°C for 45 s before melting curve program. The experiment was performed on a StepOnePlusTM Real-Time PCR System. GAPDH was detected as a loading reference control. The relative sequences of the primers pairs used were as follows: OB-R-F: ACCTCTGGTTCCCCAAAAAGG; OB-R-R:TTGGCACAGGCACAAGACAT; HIF-1α-F:GAACGTCGAAAAGAAAAGTCTCG; HIF-1α-R:CCTTATCAAGATGCGAACTCACA; GAPDH-F:AGGTCGGTGTGAACGGATTTG; GAPDH-R:GGGGTCGTTGATGGCAACA.
Western blot
Total protein was lysed using SDS lysis buffer(Beyotime, Nantong, China), which is supplemented with a protease inhibitor cocktail (Sigma). The BCA protein assay kit (Thermo Scientific) was used to detect concentrations of proteins and equal amounts of protein were detected on an 8–10% SDS-polyacry lamidegel (Beyotime), and the cell proteins were transferred onto nitrocellulose filter membranes. Then, the membranes were probed with the appropriate antibodies at 4°C overnight, followed by secondary anti-mouse or anti-rabbit antibodies at room temperature for 2 h. Protein expression levels were determined using an enhanced chemiluminescence detection system (Bio-Rad). Image J was used to quantify protein expression levels and antibodies specific for OB-R (Abcam, MA, USA), HIF-1α (Abcam), and Ki-67(Abcam), following the manufacturer's recommendations. An antibody specific for GAPDH (Proteintech) was used as an internal control. Recombinant human leptin was purchased from R&D Systems (Minneapolis, MN, USA).
Cell viability assay
Cell counting kit-8 (CCK-8; Beyotime) was used to measure cell proliferation. Transfected lung cancer cells were seeded into 96-well plates (3000 cells/well) and incubated for 24 h, 48 h, and 72 h to detect viability. After transfection with siRNA for 48 h, the lung cancer cell lines were treated with leptin (50, 100, 150, 200, and 250 ng/mL) for 48 h, and 10 ul CCK-8 reagent was added into each well incubated at 37°C for 1.5 h. Then, 450 nm absorbance was measured. All experimental points were set up in five duplicate wells and all experiments were independently performed more than three times.
Transwell migration assay
The 8.0 um pores inserts of 24-well were obtained from Millipore. Lung cancer cells were re-suspended in DMEM and counted by cell counter. A total of 200 uL of DMEM containing 2 × 105 cells was added to the upper chamber. The lower chambers were added with 1 mL DMEM containing 10% FBS. After 48 h, the cells were fixed with 4% paraformaldehyde and stained with 0.25% crystal violet. Cells on the upper surface were wiped away by a cotton swab. Migrated cells were randomly counted in three fields under microscope.
Animal studies
Female BALB/c nude mice(four-week-old) were purchased from the Department of Laboratory Animal Center of Nanjing Medical University. The experimental animals were performed following the Institutional Animal Care and Use Committee of Nanjing Medical University. Cells were washed with PBS three times and then collected in 1.5 mL EP tube. Cells were re-suspended with cold PBS at 107 cells/mL, and 0.1 mL(106 cells) were injected subcutaneously into the flank of nude mice. Mice were intraperitoneally injected with digoxin (2 mg/kg) three days after tumor cell implantation. Tumor volume and weight was determined every three days based on caliper measurements. The tumors were obtained on day 28 and fixed in formalin. Then, tumor tissues were embedded in paraffin, and then immuno-stained using the corresponding antibodies.
Immunohistochemistry
For one hundred PFA-fixed tumor tissues, the embedding section was performed first. After blocking endogenous peroxides and proteins, 4 um-thick slice were incubated with primary antibodies for OB-R, HIF-1α, and Ki67 at 4°C overnight and then incubated with MaxVisionTM 2 followed by DAB chromogen and hematoxylin counterstaining. The results were scored and the intensity of staining was defined as (0 = negative, 1 = low, 2 = medium, 3 = high). Extent of staining was scored as 0 = 0% stained, 1 = 1–25% stained, 2 = 26–50% stained, and 3 = 51–100% stained. Three random fields were selected under a light microscope. The final score was calculated by multiplying the intensity scores by the extent of staining, ranging from 0 to 9. Final scores were defined as less than 1 as negative (−); 1–2 as low (+); 3–4 as medium(++), and 6–9 as high(+++).
Statistical analysis
All data were obtained from at least three independent assays. And the results were presented as mean ± standard deviation (SD). The statistical analyses were performed using Student's t-test. P < 0.05 was considered to indicate statistical significance. The Spearman rank-correlation coefficient test was used to analyze the association between the differences. * p < 0.05; ** p < 0.01; *** p < 0.001; **** p < 0.0001.
Results
HIF-1α and Ob-R are highly expressed in NSCLC cell lines
Lung adenocarcinoma cell lines (H1975, H1299, and A549), lung squamous cell line (H520), small-cell lung cancer line (H446), and normal human bronchial epithelial cell line (HBE) were used in our experiments. All lung cancer cell lines expressed higher OB-R and HIF-1α mRNA levels than the HBE cell line. Moreover, H520 and H1299 cells had the highest HIF-1α and OB-R RNA levels, whereas H446 cells had the lowest expression (Figure 1A, B). Consistent with this, western blotting also indicated that the protein expression levels of HIF-1α and OB-R were significantly higher in lung squamous cell and adenocarcinoma cell lines than in HBE, while H446 cells had the lowest HIF-1α and OB-R expression without statistical significance (Figure 1C, D, E). Therefore, H520 and H1299 were used for HIF-1α knockdown, while A549 and H446 were used for hypoxia experiments.
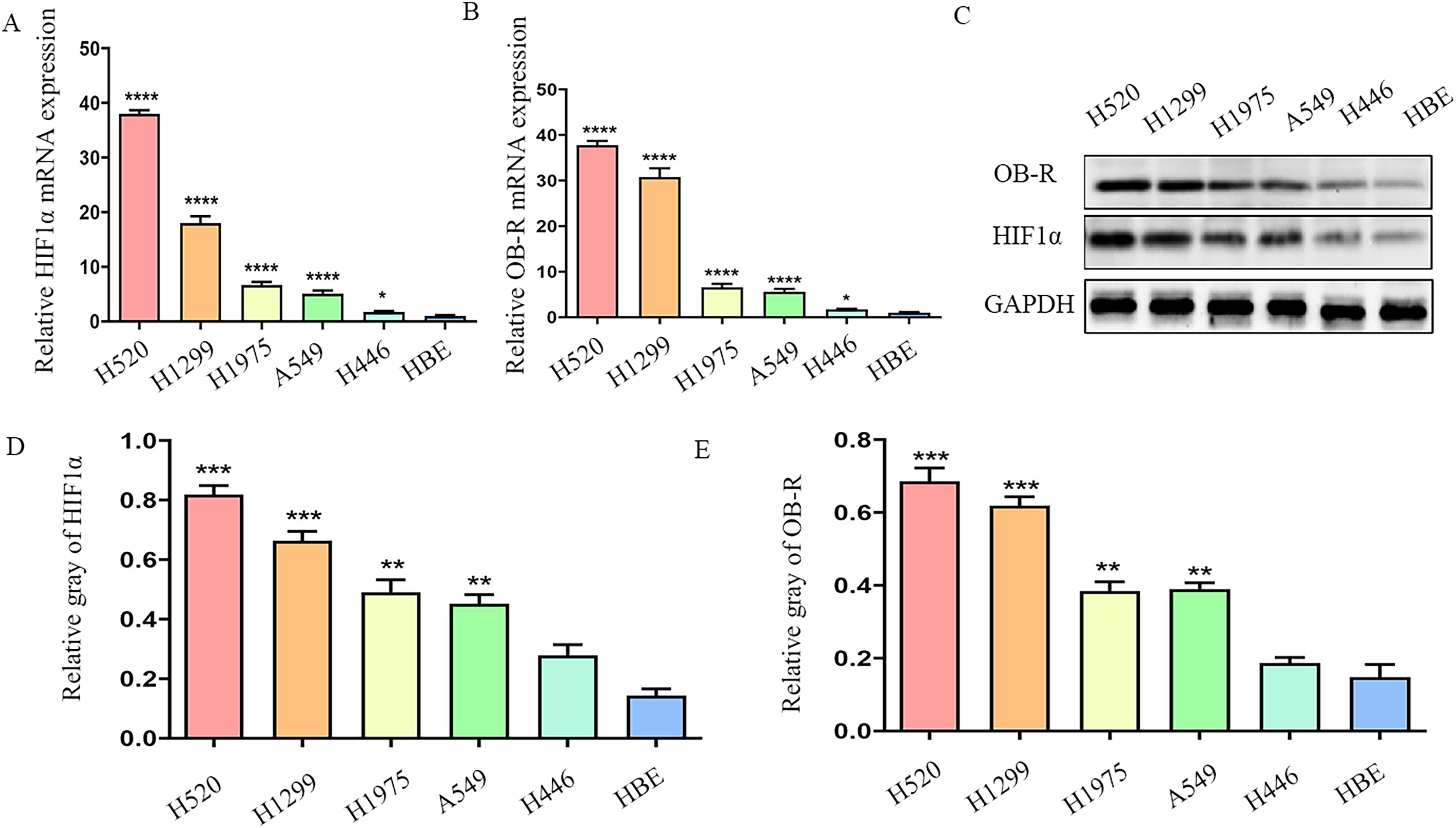
The expression of hypoxia-inducible factor-1α (HIF-1α) and OB-R in NSCLC is significantly increased. A, B, Human bronchial epithelial cells (HBE), lung squamous cell line (H520), and lung adenocarcinoma cell lines (H1299, H1975, A549, and H446) were cultured in vitro. mRNA of the cells was extracted. The mRNA expression levels of HIF-1α and OB-R were detected by RT-PCR. C-E, Cell protein was extracted, and the expression levels of HIF-1α and OB-R protein were detected using western blot. *P < 0.05, **P < 0.01, ***P < 0.001, ****P < 0.0001.
Hypoxia induces HIF-1α and Ob-R expression
To explore the effect of hypoxia on OB-R, two lower HIF-1α expression cell lines (A549 and H446) were chosen for the study. The mRNA and protein were extracted, and the expression levels of HIF-1α and OB-R were verified by RT-PCR and western blotting. The expression levels of HIF-1α and OB-R mRNA in A549 and H446 cells were significantly increased under hypoxia (1% oxygen) compared with that in normal oxygen culture (Figure 2A, B). At the same time, a similar trend was observed in the protein level, and the protein expression levels of HIF-1α and OB-R in A549 and H446 cells were significantly increased (Figure 2C, D, E).
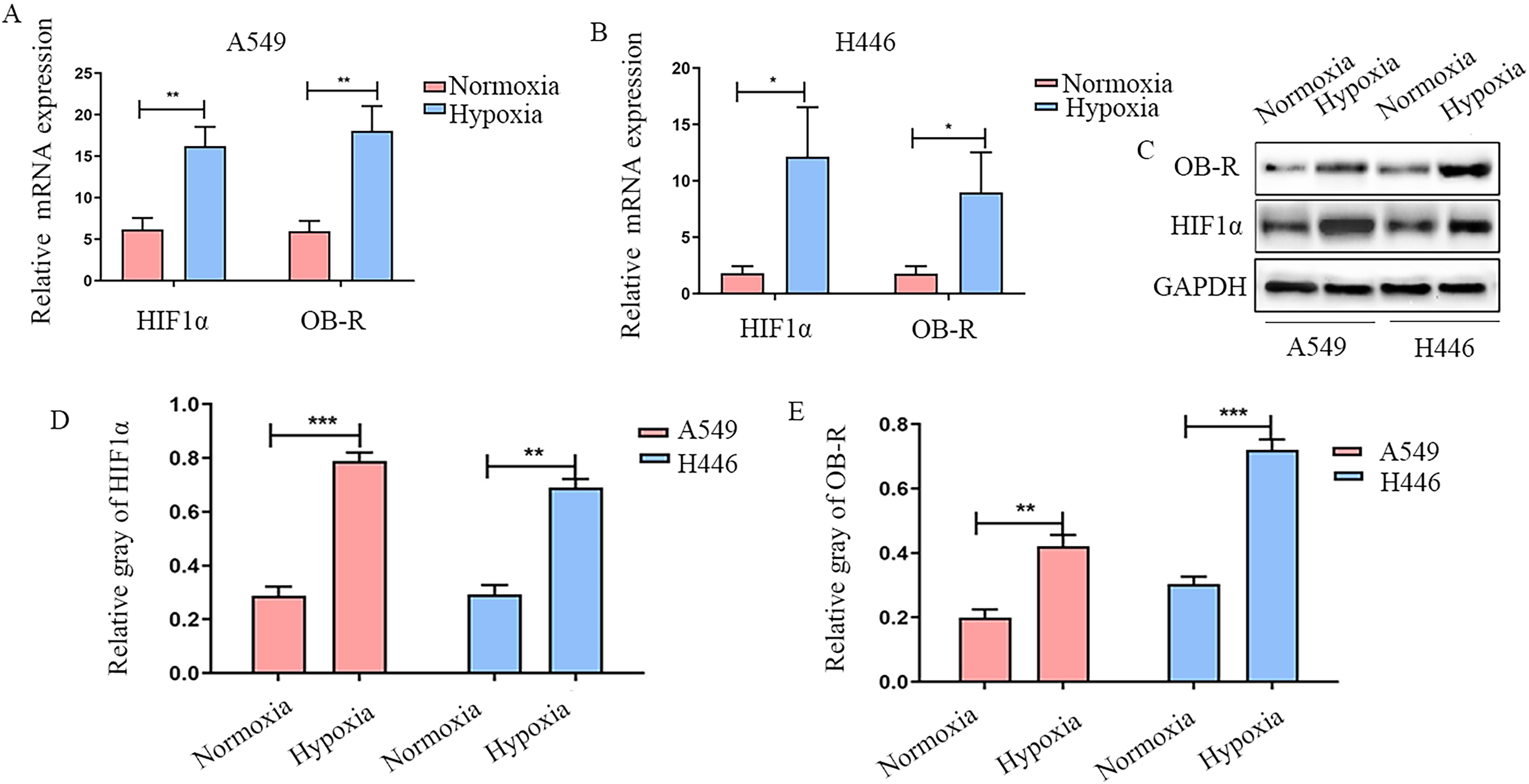
The expressions of HIF-1α and OB-R in lung cancer cell lines were significantly increased under hypoxic condition. A, B, Lung adenocarcinoma A549 and small cell carcinoma H446 cell lines were cultured in vitro under 2% oxygen conditions. mRNA of the cells was extracted and then RT-PCR was used to detect HIF-1α and OB-R mRNA expression. C-E, Cell protein was extracted, and the expression levels of HIF-1α and OB-R were detected using western blot. *P < 0.05, **P < 0.01, ***P < 0.001.
Down-regulation of HIF-1α significantly inhibits Ob-R expression
To investigate whether HIF-1α is involved in the genesis and development of NSCLC through OB-R, we constructed three siRNA targets of HIF-1α (si1 HIF-1α, si2 HIF-1α, and si3 HIF-1α) acting on H520 and H1299 cells and extracted cell mRNA and protein. According to the RT-PCR results, the silencing effects of si1 HIF-1α and si3 HIF-1α were particularly obvious, with inhibition rates of nearly 99%. Moreover, silencing HIF-1α reduced OB-R mRNA levels (Figure 3A, B, F, G). Similarly, western blotting showed that HIF-1α was significantly inhibited by si1 HIF-1α and si3 HIF-1α, confirming that the interference was effective. Furthermore, inhibition of HIF-1α decreased OB-R protein levels (Figure 3C, D, E, H, 1, G). Overall, these results demonstrate that HIF-1α is involved in the regulation of NSCLC through the regulation of OB-R.
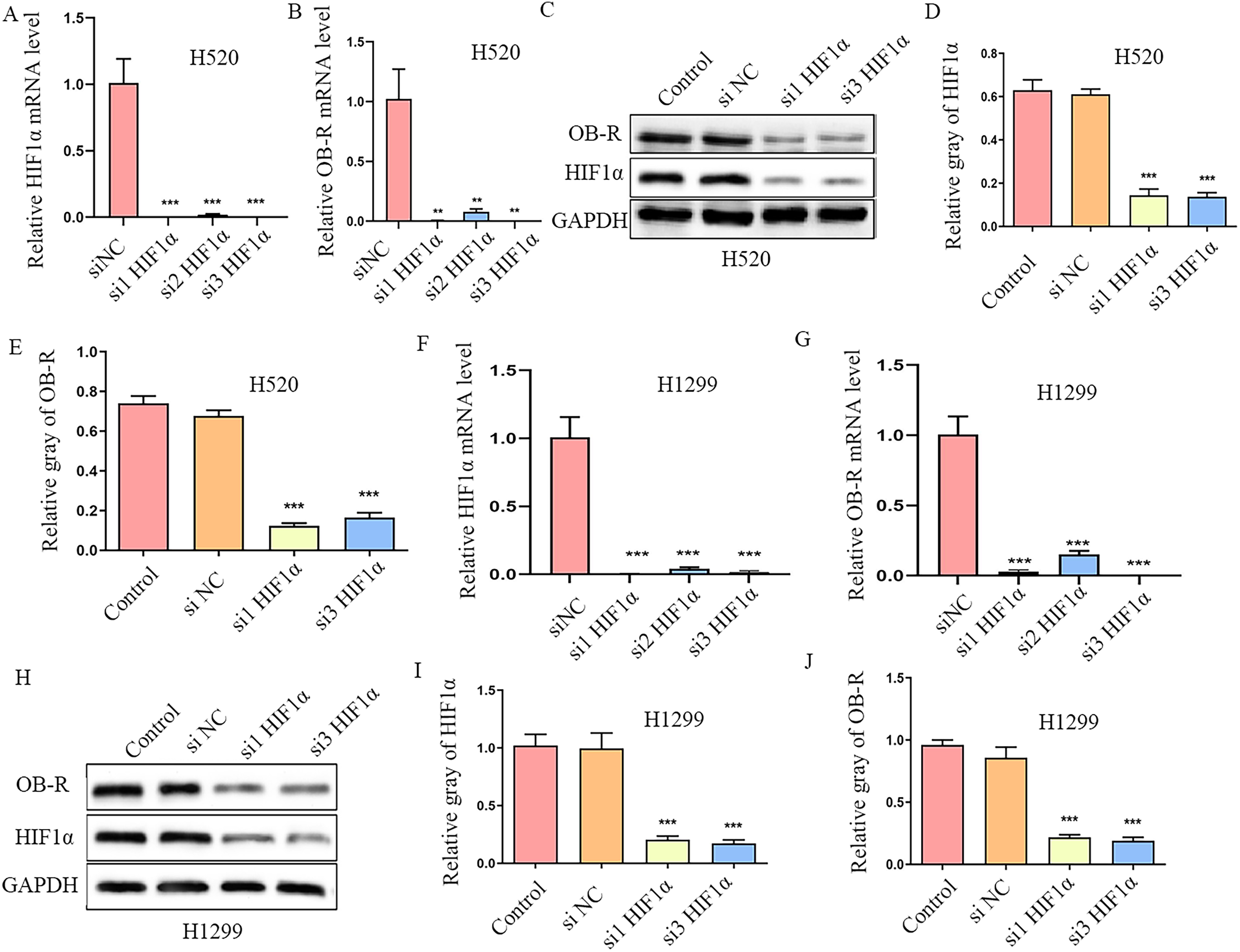
Silencing HIF-1α significantly inhibits OB-R expression. A, B, mRNA expression levels of HIF-1α and OB-R were detected using RT-PCR in H520 cells after transfection with HIF-1α siRNA and the control blank. C, D, E, Cell protein was extracted, and the expression levels of HIF-1α and OB-R were detected using western blot in H520 cells after transfection with HIF-1α siRNA and the control blank. F, G, mRNA expression levels of HIF-1α and OB-R were detected using RT-PCR in H1299 cells after transfection with HIF-1α siRNA and the control blank. H, I, J, Cell protein was extracted, and the expression levels of HIF-1α and OB-R protein were detected using western blot in H1299 cells after transfection with HIF-1α siRNA and the control blank. **P < 0.01, ***P < 0.001.
Silencing HIF-1α expression represses cell proliferation and migration of NSCLC induced by leptin
Leptin signals mainly through its interaction with OB-R to promote tumor angiogenesis, invasion, and metastasis. To verify the function of leptin in NSCLC cell proliferation, different concentrations of leptin (0, 50, 100, 150, and 200 ng/mL) was used to treat H520 and H1299 cells. And results showed that leptin promoted the proliferation of H520 and H1299 cells in a dose-dependent manner. To further verify whether HIF-1α participated in the development of NSCLC through leptin/OB-R, we treated H520 and H1299 with 200 ng/mL leptin, and selected si1 HIF-1α and si3 HIF-1α to further treat the cells. Cell proliferation was detected and it showed that silencing HIF-1α expression inhibited leptin-induced proliferation of NSCLC cells (Figure 4A, B, C, D). Transwell assays also showed that, compared with the no-load transfection group, inhibition of HIF-1α expression significantly repressed the migration-promoting ability of leptin (Figure 4E, F, G). Overall, above results suggest that HIF-1α accelerates cell proliferation and migration of NSCLC via leptin.
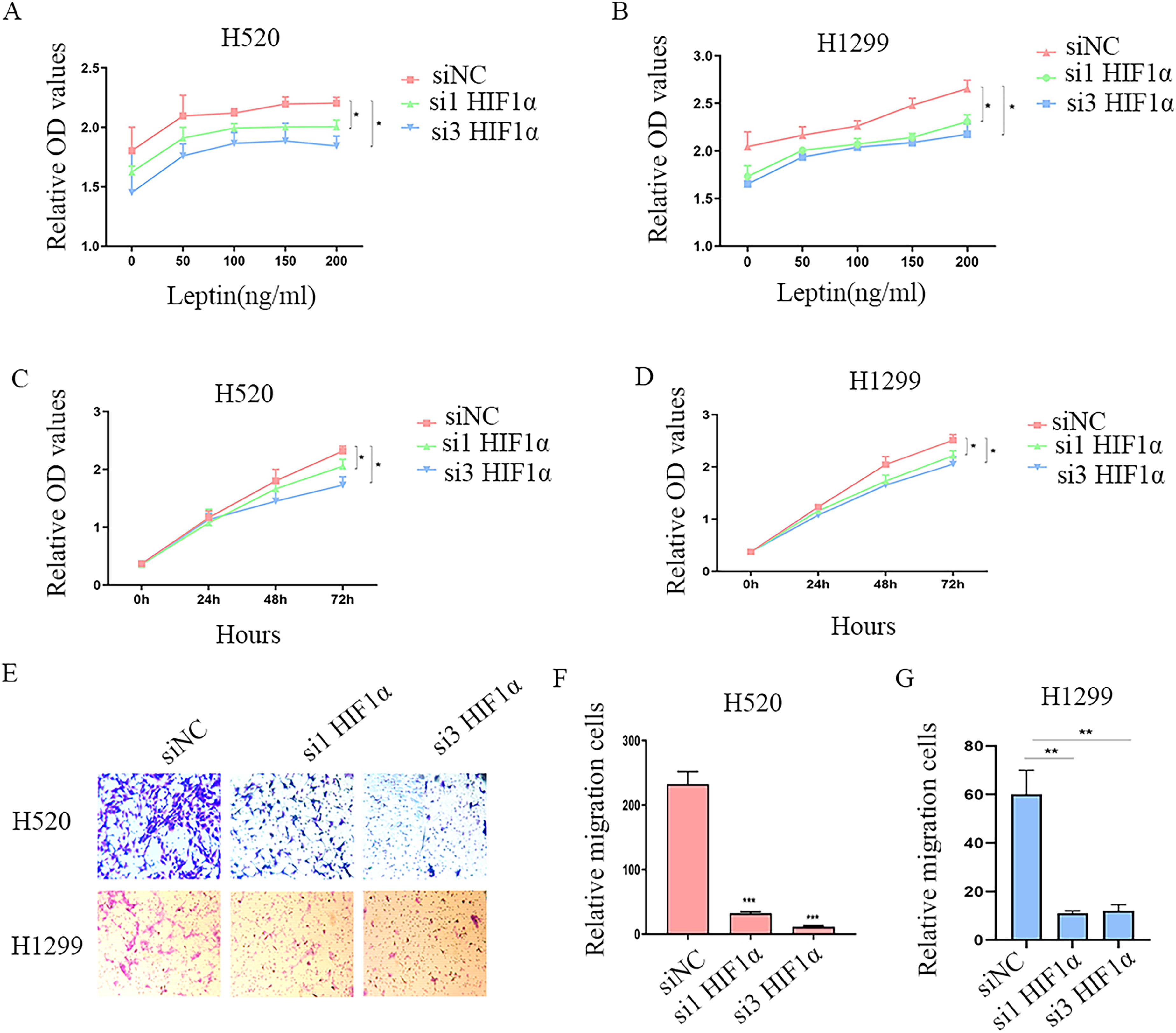
Down-regulation of HIF-1α inhibits leptin-induced proliferation and migration. A, B, H520 and H1299 cells were treated with different concentrations of leptin (0, 50, 100, 150, and 200 ng/mL) and HIF-1α siRNA was transfected. C, D, Knockdown of HIF-1α decreased cell proliferation in both H520 and H1299 cells treated by 200 ng/mL leptin, the cell proliferation was detected at different time points (0, 24 h, 48 h, and 72 h) and absorbance of 450 nm was detected 48 h later. E, F, G, H520 and H1299 were treated with 200 ng/ml leptin, and si1 HIF-1α and si3 HIF-1α had significant interference effect and were selected for further treatment. The migration ability of NSCLC cells was detected by transwell assay 72 h later. *P < 0.05, ***P < 0.001.
Digoxin suppresses tumor growth of NSCLC in vivo
Digoxin, an HIF-1α inhibitor, was injected three days before tumor cell transplantation in nude mice. Tumor size was measured four times one week, and the results showed that the tumor gradually grew larger with the extension of time, which indicates that the animal model was successfully built and that tumor growth could be inhibited after the intraperitoneal injection of digoxin (Figure 5A, B, C). Immunohistochemical staining of tumors tissues showed that HIF-1α, OB-R, and the cell proliferation marker Ki67 were significantly decreased in the digoxin injection group (Figure 5D, E). These results suggested that digoxin may inhibit NSCLC growth by inhibiting OB-R expression.
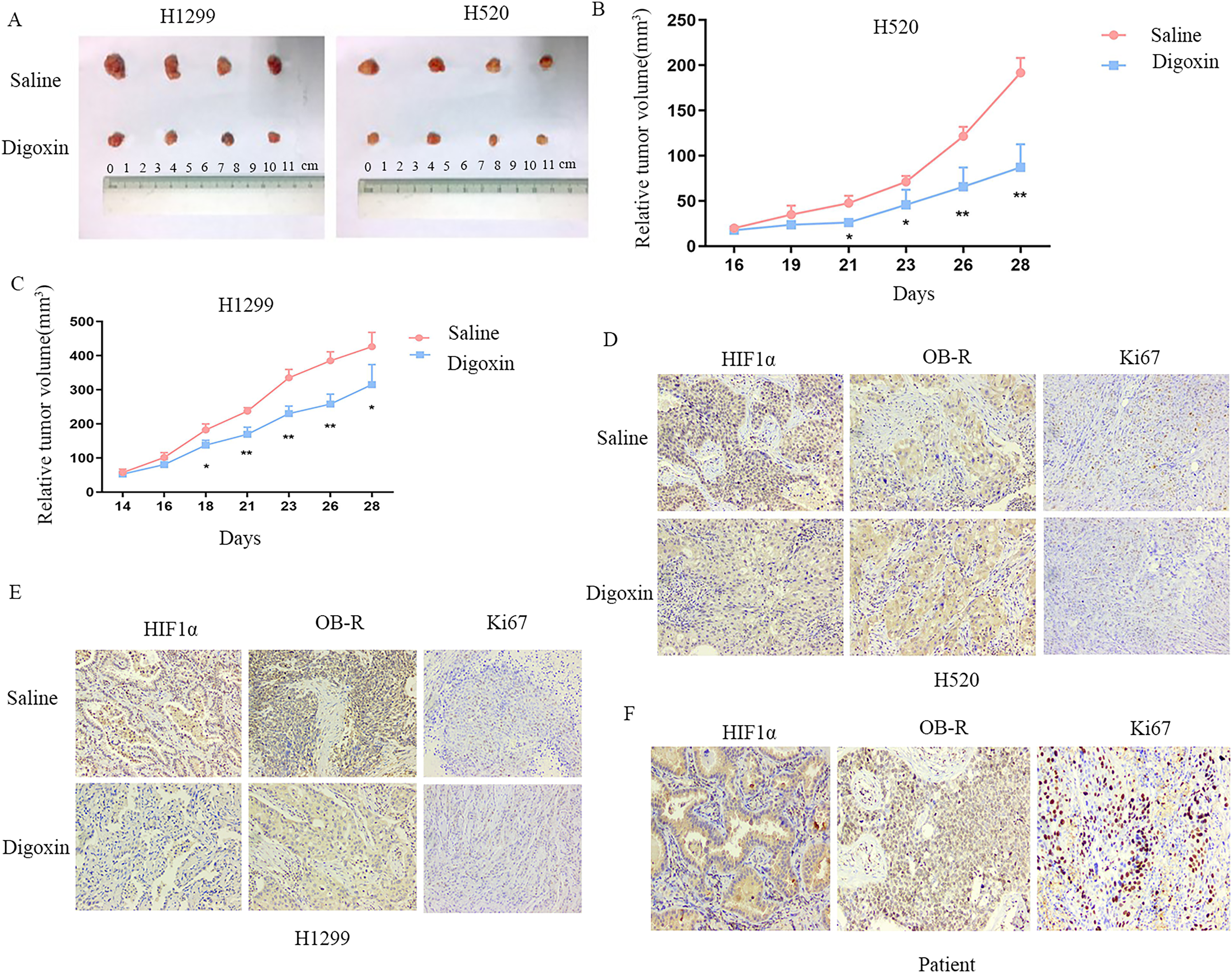
Digoxin inhibits the growth of NSCLC in vivo. A, Tumor formation was observed and nude mice were randomly divided into groups, digoxin or control group. H520 and H1299 cells were injected subcutaneously in nude mice, and the HIF-1α inhibitor digoxin (2 mg/kg/d) was injected intraperitoneally in nude mice starting 3 days before tumor cell transplantation. B, C, Tumor volume was followed for four weeks in H520 and H1299 cells. D, E, Tumor tissues derived from H520 and H1299 cells were stained with HIF-1α, OB-R, and Ki67 using immunohistochemical assays. F, Tumor tissues of NSCLC patients were stained with HIF-1α, OB-R, and Ki67 by immunohistochemical assays. *P < 0.05, **P < 0.01.
HIF-1α and Ob-R are significantly increased in NSCLC tissues
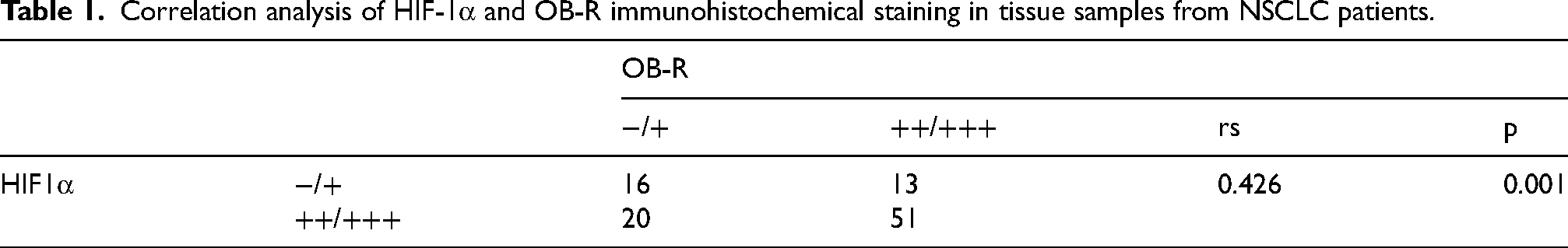
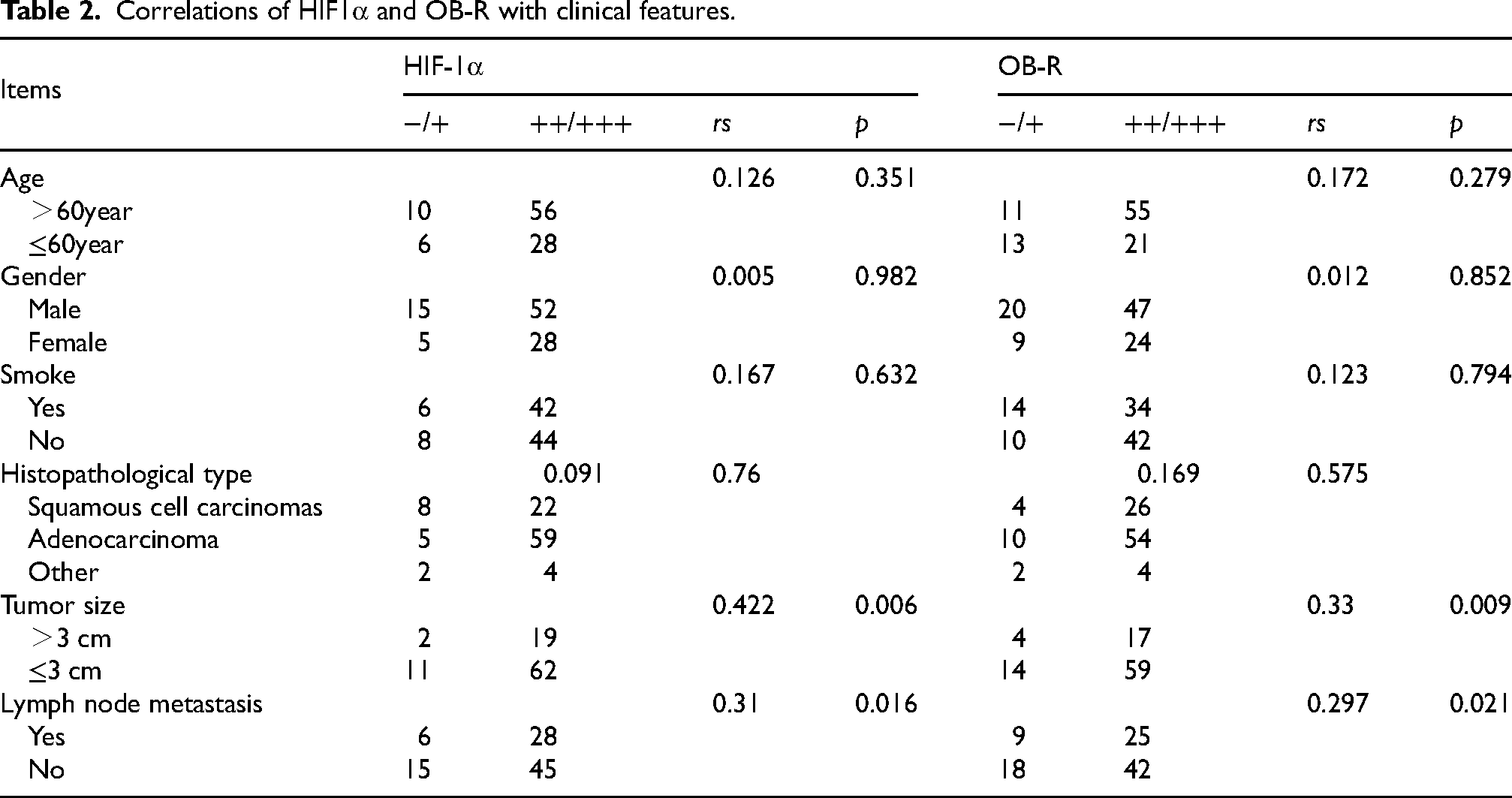
To evaluate HIF-1α and OB-R in NSCLC tissues, lung cancer tissue samples surgically resection from 94 patients and percutaneous needle lung biopsy from 6 patients with NSCLC were collected for immunohistochemical analysis. HIF-1α, OB-R, and Ki67 in patients with NSCLC were significantly increased (Figure 5F). In addition, OB-R levels were positively correlated with HIF-1α levels in NSCLC (rs = 0.426, p = 0.001) (Table 1). In addition, we analyzed the correlation between HIF-1α and OB-R and it showed that HIF-1α and OB-R was positively correlated with tumor size and lymphatic metastasis (Table 2).
Correlation analysis of HIF-1α and OB-R immunohistochemical staining in tissue samples from NSCLC patients.
Correlations of HIF1α and OB-R with clinical features.
HIF-1α directly activates Ob-R transcription
We analyzed the interaction between HIF-1α and OB-R using the String (https://string-db.org/) and UniProt databases (https://www.uniprot.org/). It was found that HIF-1α and OB-R did not interact directly, but indirectly regulated the leptin via STAT3 pathways (Figure 6A). In contrast, we used the Jaspar database (Version 2022, Jaspar.uIO.no) to predict the binding site of HIF-1α in the promoter region of OB-R. We identified two potential hypoxia response elements (HRE) located 675 and 824 bases upstream of the coding initiation sequence, with predicted binding sites of “CTGCGTGC” and “CACGCACT"(Figure 6B). Chromatin immunoprecipitation assays were performed on H520 and H1299 cell lines under hypoxic and normoxic conditions to study the binding of HIF-1α to promoter of OB-R. In the chromatin components pulled down by anti-HIF-1α antibodies, PCR fragments of the OB-R promoter (−831/-824) were detected under normoxic conditions and enhanced under hypoxia. In contrast, the PCR fragment was not detected in the control IgG antibody samples. Another potential HRE (−682/-675) failed to pull down by anti-HIF-1α (Figure 6C). Moreover, a luciferase promoter vector for OB-R was constructed and co-transfected with HIF-1α plasmid. Results showed that HIF-1α increased OB-R transcription. Furthermore, OB-R promoter mutant in HRE (−831/-824) was constructed and co-transfected with HIF-1α plasmid. Interestingly, we found that the luciferase activity of the mutant vector was significantly lower than that of the wild-type (Figure 6D–F). In summary, HIF-1α directly activated OB-R transcription by binding to the OB-R promoter region HRE (−831/-824).
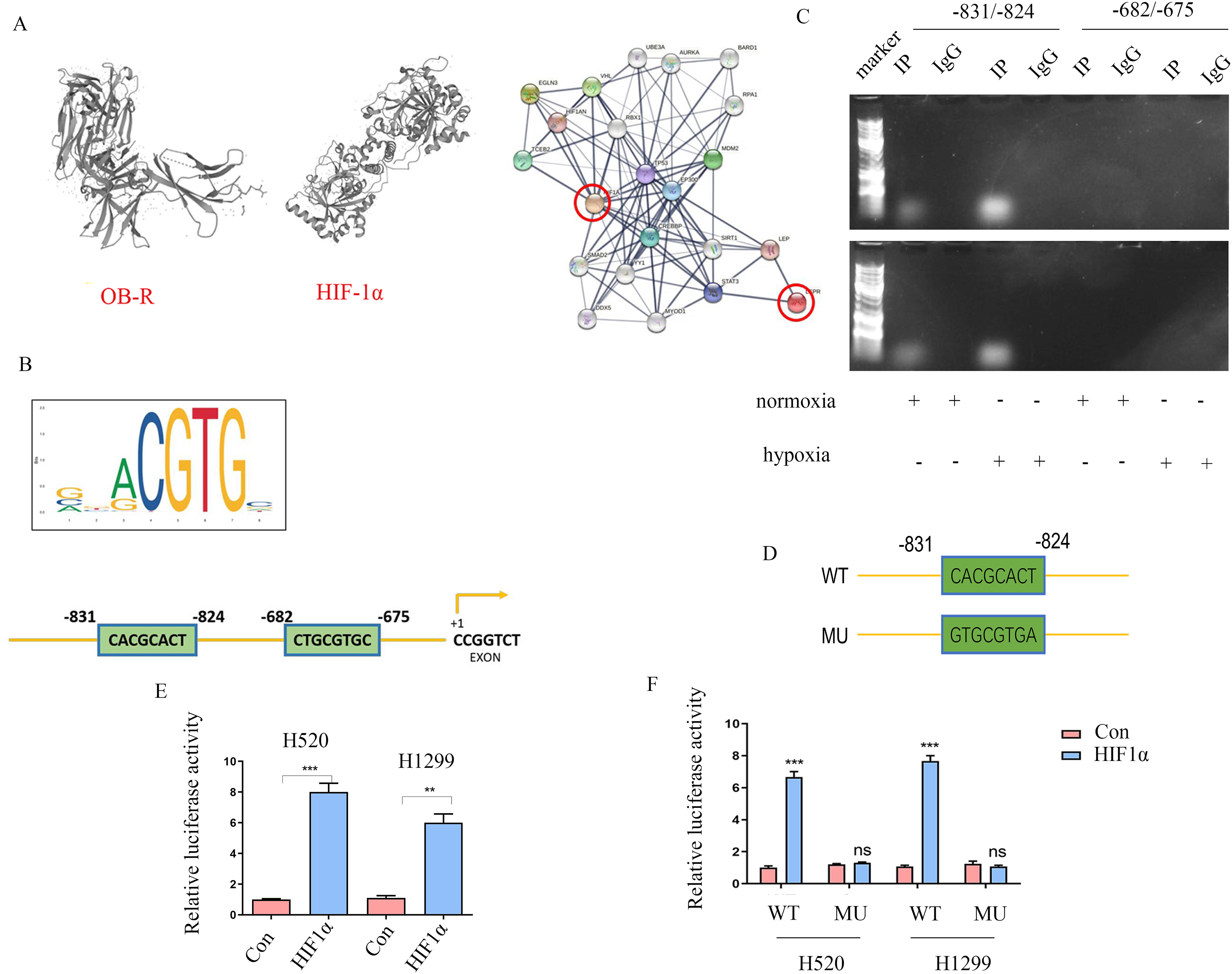
HIF-1α directly activates OB-R transcription. A, Three-dimensional structure and protein interaction analysis of HIF-1α and OB-R. B, Prediction of the HRE upstream promoter of OB-R. C, The ChIP-PCR assay was conducted under normal oxygen and hypoxic conditions, IP represents HIF-1α co-immunoprecipitation, and IgG was used as a negative control. D, Model diagram of the HIF-1α binding region mutant of the OB-R promoter region −831/-824. E, Luciferase reporter gene assay showed that HIF-1α binds to the OB-R promoter (2000 bp upstream of the start codon). F, In the luciferase reporter gene experiment, HIF-1α could not activate OB-R transcription after binding site mutations. **P < 0.01, ***P < 0.001.
Discussion
Lung cancer has the highest morbidity and mortality in China and worldwide.20,21 Although some achievements have been made in understanding the mechanism and developing a treatment for NSCLC, the overall survival rate is extremely low.22,23 Inhibiting leptin expression can inhibit lung cancer. 24 In this study, HIF-1α and OB-R was found highly expressed in NSCLC cells, and the increase was more significant under hypoxia. Inhibition of HIF-1α decreased OB-R expression, indicating that HIF-1α enhanced the malignant behaviors of NSCLC cells by activating OB-R transcription. Furthermore, digoxin, an inhibitor of HIF-1α, inhibited tumor growth in vivo.
Leptin is secreted by the adipose tissue and involved in the regulation of energy metabolism. 25 It is mainly involved in angiogenesis and can stimulate cell proliferation through OB-R on tumor cells surface and participate in the progression of tumors. 24
Hypoxia not only promotes the metabolism of tumor cells to enhance survival, but also promotes tumor angiogenesis, metastasis and drug resistance. HIF-1α is now widely recognized as a major regulator of hypoxia response. HIF-1α is widely associated with the biological functions of the body, including pathophysiological processes, such as cell differentiation, migration, metabolism, apoptosis, and cell cycle regulation.26,27 Recent studies demonstrated HIF-1α was closely related to NSCLC development, playing an important role in this process. HIF-1α is highly expressed in NSCLC cells under hypoxia and plays a leading role in regulating hypoxia-related genes in NSCLC.28–30
HIF-1α and OB-R has been extensively studied in various tumors and has been reported promotes metastasis of pancreatic cancer by activating OB-R.31,32 The relationship between these two genes in NSCLC has not yet been reported. In present study, HIF-1α and OB-R was highly expressed in NSCLC cells. Subsequent studies focused on exploring the regulatory relationship between HIF-1α and OB-R in NSCLC. Chromatin immunoprecipitation and luciferase reporter gene assays showed HIF-1α could bind to the upstream hypoxia reactive element (−831/-824) of the OB-R promoter region to activate its transcription, thereby increasing OB-R expression. Above results indicated that HIF-1α promoted NSCLC by regulating OB-R.
In conclusion, hypoxia significantly increased HIF-1α and thus activated OB-R transcription, which enhanced the proliferation and migration of NSCLC cells through OB-R. HIF-1α/OB-R facilitated NSCLC and may be new targets and strategies for the future treatment of NSCLC.
Footnotes
Acknowledgements
Not applicable.
Ethics approval and consent to participate
Animal study was approved by Institutional Animal Care and Use Committee (IACUC)of Nanjing Medical University.
Consent for publication
Not applicable.
Authors’ contributions
CONCEPTION: Yan Li
INTERPRETATION OR ANALYSIS OF DATA: Bo Chen, Shuangshuang Wu,
PREPARATION OF THE MANUSCRIPT: Yan Li, Yijue Zhong
REVISION FOR IMPORTANT INTELLECTUAL CONTENT: Yan Li, Yuxing Zhang
SUPERVISION: Jianqing Wu
Funding
This work was supported by the National Key R&D Program of China (grant numbers 2018YFC2002100, 2018YFC2002102), the National Natural Science Foundation of China (grant numbers 82171576, 81871115, 81572259).
Conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Competing interests
The authors have declared that no competing interest exists.
Availability of data and material
All of the data of this study are available from the corresponding author.
