Abstract
Background
It is unclear what proportion of the population cancer burden is covered by current implementation of USPSTF A/B screening recommendations.
Objective
We estimated the proportion of all US cancer deaths caused by cancer types not covered by screening recommendations or cancer types covered but unaddressed by current implementation.
Methods
We used 2018–2019 National Center for Health Statistics mortality data, Surveillance, Epidemiology, and End Results registries incidence-based mortality data, and published estimates of screening eligibility and receipt.
Results
Of approximately 600,000 annual cancer deaths in the US, 31.4% were from screenable cancer types, including colorectal, female breast, cervical, and smoking-associated lung cancers. Further accounting for the low receipt of lung cancer screening reduced the proportion to 17.4%; accounting for receipt of other screening reduced it to 12.8%. Thus, we estimated that current implementation of recommended screening may not address as much as 87.2% of cancer deaths—including 30.4% from individually uncommon cancer types unlikely ever to be covered by dedicated screening.
Conclusions
The large proportion of cancer deaths unaddressed by current screening represents a major opportunity for improved implementation of current approaches, as well as new multi-cancer screening technologies.
Keywords
Introduction
Although cancer mortality rates in the US have been decreasing, 1 the absolute number of cancer deaths continues to increase, making cancer the leading cause of death in US adults aged ≤80 years. 2 Declines in cancer mortality rates are partially attributable to standard-of-care, guideline-based population screening programs for early detection of lung, colorectal, female breast, and cervical cancers.3,4 However, these four cancer types account for only approximately 40% of the 600,000 cancer deaths that occur annually in the US. 5 To date, what has remained uncertain is the actual proportion of these deaths that occurs in individuals eligible for or receiving recommended screening based on age and, for lung cancer, based on smoking history.3,4 These numbers can quantify the unmet need of cancer deaths that could be avoided by improving the existing standard of care and/or adding new cancer screening approaches.
As an addition to standard-of-care screening, novel blood-based tests that detect circulating cell-free DNA (cfDNA) have the potential to enable earlier detection of cancer types not addressed by existing screening guidelines, as well as cancers missed by current implementation of recommended screening strategies due to incomplete receipt or access. Some of these new tests are formulated as single-cancer early detection (SCED) tests, which target individually common cancer types (e.g., lung, colorectal, breast), typically with relatively high sensitivity for the cancer type, but at the cost of a relatively high false-positive rate per test. 6 Other cfDNA-based tests are formulated as multi-cancer early detection (MCED) tests, which simultaneously screen for multiple cancer types with sensitivity that varies by cancer type and is generally lower than that from targeted approaches, but have the benefit of a far lower overall false-positive rate. 6 Additions of novel SCED and MCED tests to existing standard-of-care, guideline-based screening are now being contemplated, and it is important to understand the relative contributions of these approaches to the public health goal of reducing the overall cancer death rate. Therefore, to evaluate the unmet burden of cancer mortality that is not currently being addressed by extant efforts, and that might be addressed by novel screening modalities, we estimated the proportion of cancer deaths in the US unaddressed by current implementation of cancer screening with A- or B-grade recommendations from the US Preventive Services Task Force (USPSTF). 4
Materials and methods
To obtain recent, pre-COVID-19 pandemic counts of US cancer deaths by type, we used 2018–2019 age- and sex-specific mortality data from the National Center for Health Statistics (NCHS), accessed via the National Cancer Institute's Surveillance, Epidemiology, and End Results (SEER) program. 7 Our primary analysis included all age groups. We conducted secondary analyses limited to age groups (classified by age at death) that are more likely to be eligible for population-based cancer screening, including lung, colorectal, breast, and cervical cancer screenings currently recommended by USPSTF, 4 i.e., adults aged ≥50 years (with no upper age limit because cancer deaths may occur several years after screening and diagnosis); and those generally eligible for US federal health insurance through Medicare, i.e., adults aged ≥65 years.
Next we sought to further stratify cancer deaths by stage at diagnosis. Because stage at cancer diagnosis is not typically recorded on death certificates, this information is not available for the entire US population. Therefore, we used 2018–2019 incidence-based mortality data from 17 SEER geographic regions, 8 with stage classified according to SEER Summary Stage. 9 We accessed the most recent SEER data using SEER*Stat software version 8.4.1. 10
To account for the proportion of US lung cancer cases meeting current USPSTF eligibility criteria for lung cancer screening (i.e., ages 50–80 years with a 20 + pack-year history of smoking and either currently smoking or having quit within the past 15 years 4 ), we used a recent published estimate based on modeling of data from two large clinical trials of lung cancer screening, combined with 2015–2018 National Health Interview Survey data on smoking prevalence. 11 We further applied rates of lung cancer screening receipt (assuming either full adherence or full non-adherence to recommendations) based on 2019 reports from the American College of Radiology's Lung Cancer Screening Registry.12,13 These reports were required by Medicare to be submitted by radiology facilities for reimbursement of all lung cancer screening scans. To acknowledge that current receipt of standard-of-care colorectal, breast, and cervical cancer screenings is reportedly near (albeit below) national targets,14,15 and that any further gains in screening receipt from implementation of new tests are likely to be incremental, we did not further account for receipt of guideline-based screening for these cancer types in our primary analysis. In a secondary analysis, however, we applied 2019 self-reported screening rates from the National Health Interview Survey 14 (again assuming either full adherence or full non-adherence to recommendations) to estimate cancer deaths not addressed by current implementation of screening recommendations for these three cancer types.
We did not consider prostate cancer to be addressed by current screening recommendations because prostate cancer screening is not recommended by USPSTF as part of the usual standard of care. 4 Nevertheless, in 2021, 37.1% of US men aged 55–69 years reported having a prostate-specific antigen test in the past year; 16 therefore, we separately estimated the proportion of cancer deaths due to prostate cancer and other relatively common cancer types that lack USPSTF-recommended screening.
To estimate the proportion of cancer deaths due to cancer types that are individually uncommon and therefore unlikely to be targeted by SCED tests, we used the National Cancer Institute's definition of rare cancers (types affecting <40,000 persons per year in the US). 17
Due to the secondary use of de-identified data, this study was not subject to institutional review board approval and informed consent was not required.
Results
A total of 1,198,854 persons died of cancer in the US in the two-year period of 2018–2019, including 281,681 deaths from lung cancer (23.5% of cancer deaths), 104,059 from colorectal cancer (8.7%), 84,745 from female breast cancer (7.1%), and 8290 from cervical cancer (0.7%) (Figure 1). Thus, of all cancer deaths, 39.9% were attributable to cancer types covered and 60.1% to cancer types not covered by USPSTF screening recommendations. Among cancer types without a USPSTF A- or B-grade screening recommendation, the leading causes of cancer death were pancreatic cancer (7.6% of cancer deaths), prostate cancer (5.3%), liver/intrahepatic bile duct cancer (4.6%), leukemia (3.9%), and lymphoma (3.6%). Cancer types that are individually uncommon in the US—including brain cancer (2.9% of cancer deaths), esophageal cancer (2.6%), ovarian cancer (2.3%), myeloma (2.1%), stomach cancer (1.9%), and other types that individually account for <2% of cancer deaths—cumulatively amounted to 30.4% of cancer deaths.
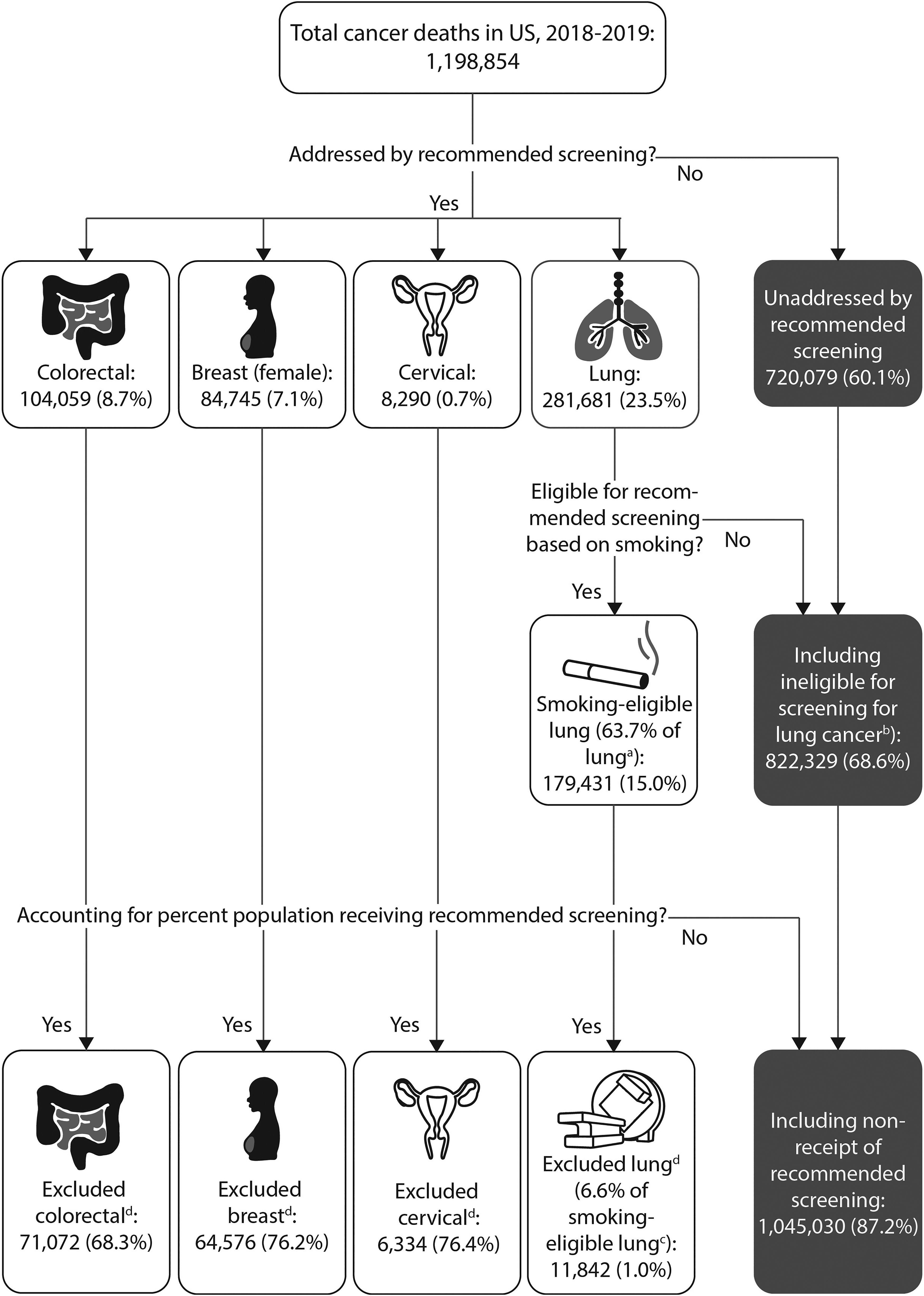
Deaths from cancer by primary type in 2018–2019, US National Center for Health Statistics, 7 with eligibility for and receipt of USPSTF guideline-based cancer screening.
After accounting for the percentage of US lung cancer cases meeting current USPSTF smoking-based eligibility criteria for lung cancer screening (63.7% 11 ), the proportion of cancer deaths addressed by screening recommendations decreased to 31.4% (Figure 1). Further accounting for receipt of lung cancer screening in the eligible population (6.6% based on radiology facility reports12,13), the estimated proportion of addressed cancer deaths was reduced to 17.4%, leaving 82.6% of cancer deaths (approximately 500,000 per year in the US) unaccounted for by current implementation of USPSTF cancer screening guidelines. When we additionally applied rates of 76.2% for breast cancer screening, 76.4% for cervical cancer screening, and 68.3% for colorectal cancer screening based on self-reported national survey data, 14 the estimated proportion of addressed cancer deaths further declined to 12.8%, leaving 87.2% of cancer deaths unaddressed by current implementation of standard-of-care screening guidelines.
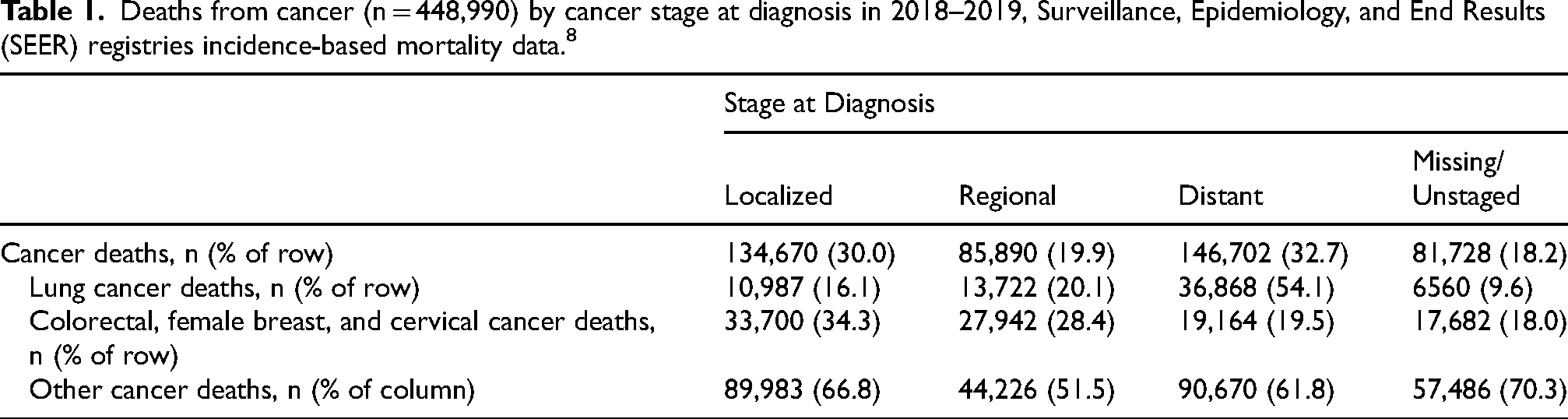
According to incidence-based mortality data from 17 SEER geographic regions in 2018–2019 for deaths from all cancer types, 146,702 (39.9%) of 367,262 deaths with known stage at diagnosis were from cancers diagnosed at a distant stage, and 85,890 (23.4%) were diagnosed at a regional stage (Table 1). Of the cancer deaths diagnosed at a distant stage, 90,670 (61.8%; 24.7% of all stageable cancer deaths) were from cancer types other than lung, colorectal, female breast, and cervical, i.e., those unaddressed by screening guidelines.
Deaths from cancer (n = 448,990) by cancer stage at diagnosis in 2018–2019, Surveillance, Epidemiology, and End Results (SEER) registries incidence-based mortality data. 8
In secondary analyses, we found similar proportions of deaths addressed by screening after restriction to deaths among adults aged ≥50 years, who are more likely than younger groups to be eligible for population cancer screening. Of the 1,139,712 cancer deaths among US adults in this age group in 2018–2019 (95.1% of all cancer deaths), 60.0% were from cancer types that are not addressed by USPSTF screening guidelines, including 29.7% from individually uncommon cancer types. Applying the same percentage of lung cancer cases eligible for USPSTF-recommended screening based on smoking history, the estimated proportion of cancer deaths among adults aged ≥50 years that are not covered by screening guidelines rose to 69.8%; and applying the same rate of receipt further increased the estimate to 84.3% of cancer deaths unaddressed by current implementation of USPSTF screening guidelines in adults aged ≥50 years. Results were also similar for adults aged ≥65 years, who are generally eligible for Medicare health insurance. This age group experienced 866,564 cancer deaths in 2018–2019 (72.3% of all cancer deaths), with estimates of 60.9% of deaths from cancer types not addressed by USPSTF guidelines (29.4% from individually uncommon cancer types), 69.8% after accounting for lung cancer screening eligibility, and 84.3% after accounting for receipt of lung cancer screening.
Incidence-based mortality results were also similar after restriction to ages ≥50 years or ≥65 years. Among adults aged ≥50 years, 39.4% of 350,437 stageable cancer deaths were diagnosed at a distant stage, and 61.7% of these were from cancer types without USPSTF-recommended screening. Among adults aged ≥65 years, there were 278,663 stageable cancer deaths, of which 36.5% were diagnosed at a distant stage, and 64.0% of distant-stage deaths were from cancer types lacking USPSTF-recommended screening.
Discussion
A large proportion of cancer deaths in the US is unaddressed by current screening efforts. Specifically, 68.6% of all cancer deaths are from cancer types that are overtly left unaddressed by current screening guidelines, and when factoring in the low current receipt of lung cancer screening, this percentage climbs to 82.6%, representing more than 500,000 cancer deaths per year. Within the existing standard of care, a profound increase in receipt of recommended low-dose computed tomography screening for lung cancer would be required to move from the latter proportion (82.6%) toward the former (68.6%). Additionally accounting for incomplete receipt of recommended colorectal, breast, and cervical cancer screening further increases to 87.2% the proportion of cancer deaths potentially not addressed by current implementation of guideline-based cancer screening. The impact of these gaps on the overall cancer death burden underscores the importance of developing and implementing new cancer screening technologies, as an addition to current standard-of-care screening, that are not only effective and safe, but also broadly accessible and affordable, for example, blood-based testing as opposed to invasive or inaccessible procedures such as endoscopy or targeted imaging.18,19
The major benefit of an MCED approach is its ability to simultaneously screen for multiple cancer types that are cumulatively responsible for a substantial proportion of the overall cancer death burden. This coverage, coupled with a single, very low false-positive rate, portends a lower number needed to screen and lower total cost of screening than a system based on multiple SCED tests, as an addition to standard-of-care screening. 20 In contrast, adding new blood tests to the existing recommended colorectal cancer screening paradigm might increase overall receipt of screening, but would not be expected to materially reduce the overall cancer death burden. 21 Given that hundreds of cancer types exist, it is impractical to add many more SCED tests to the existing paradigm, as each SCED test has its own number needed to screen and false-positive rate, leading to high cumulative false-positive rates, related unnecessary diagnostic workups, and an inflated overall cost of screening. 20
A significant opportunity exists for new screening technologies to reduce the population burden of cancer, especially through earlier detection of the one-quarter of cancer deaths from distant-stage tumors that have no recommended population-level screening. Individually uncommon cancers, which collectively comprise nearly one-third of cancer deaths, particularly represent an opportunity for MCED tests to address cancer types that are unlikely to be targeted by SCED tests in the general population due to the challenges of screening for low-prevalence conditions, 22 many of which would be missed if screening were restricted to high-risk subgroups. Given that chronologic age is by far the most important risk factor for all cancers combined, 23 restriction of any cancer screening to a selected subpopulation through risk stratification could exclude the preponderance of cancers that arise in the general population. Moreover, the proportion of lung cancers covered by smoking-based screening eligibility criteria is expected to decrease over time with the declining prevalence of smoking. 24
A strength of our study is the use of nationwide mortality data and population-based incidence-based mortality to estimate the proportion of cancer deaths in the US not addressed by current screening practice. On the other hand, our analysis is limited by the lack of population-based data on the proportion of cancer patients eligible for or detected by current standard-of-care cancer screening. The latter information is available, for example, in England from the National Health Service, which routinely collects data on routes to diagnosis among cancer patients, including whether they were diagnosed through routine screening programs (6.2% of cancer diagnoses in England in 2019). 25 In the absence of observed data, we relied on an estimate of the proportion of lung cancer patients who are eligible for USPSTF-recommended screening. 11 This estimate of 63.7% may be an overestimate, since it is based largely on data from lung cancer cases diagnosed in 1993–2009; 11 however, the estimate is consistent with observational results from other US patient populations.26–31
In conclusion, the vast majority of cancer deaths in the US occur among individuals who are ineligible for recommended guideline-based cancer screening or, if eligible, do not follow recommended screening guidelines. Results were similar for adults aged ≥50 years (i.e., generally eligible for recommended cancer screenings based on age) or ≥65 years (i.e., generally eligible for Medicare coverage), among whom the absolute risk of cancer is highest. 23 The large burden of unscreened cancer mortality, combined with the substantial proportion of cancer death from distant-stage cancers, especially those lacking recommended population-based screening strategies, underpins the need for new MCED tests that are safe, effective, accessible, and affordable. If added to existing evidence-based strategies for cancer control, including tobacco control and standard-of-care single-cancer screening programs, MCED tests could enable earlier detection and effective treatment for the full spectrum of cancer types contributing to the overall cancer burden.
Footnotes
Acknowledgments
Project management assistance was provided by Prescott Medical Communications Group (Chicago, IL). Editorial assistance was provided by Erin Spohr (ENGAGE Labs, LLC, Greenwood Lake, NY). All support and study funding were provided by GRAIL, Inc.
Author contributions
Conception: JJO, ETC, CAC, EH
Interpretation or analysis of data: ETC, ARK
Preparation of the manuscript: JJO, ETC, CAC, EH, ARK
Revision for important intellectual content: AJ, WD, AWK, GAC, AVP
Supervision: JJO
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Declaration of conflicting interests
JJO, ETC, CAC, ARK, and EH are current or former employees of GRAIL, Inc., report stock or other support from Illumina, and report other support from GRAIL, Inc., during the conduct of the study. In addition, EH has multiple patents in the field of cancer detection pending to GRAIL, Inc. GAC reports other support from NIH outside of the submitted work. AWK reports a past grant from Myriad Genetics outside of the submitted work.
