Abstract
Purpose:
A significant percentage of patients develop complications related to the use of implants made of stainless steel or titanium. The purpose of our study is to retrospectively analyze the clinical and radiological outcomes of allograft screws implanted in the Pediatric Orthopedic unit of our center.
Methods:
A retrospective analysis was conducted, including 30 patients (16 females and 14 males) with lower limb pathologies who received 50 human cortical bone allograft screws. The study period covered 48 months, from 1 January 2020, to 1 January 2023. The average age was 12 years 7 months (range 3, 15 years). The mean follow-up was 44 months (range 24–66).
Results:
After an average follow-up of 44 months, no patients developed minor or major complications, delayed union, or infections. Three patients reported discomfort due to implant prominence, and two of them (7%) required reoperation for screw head remodeling. The average resorption time was 9 months (range 5, 15 months). The postoperative American Orthopaedic Foot & Ankle Society score increased by 38.28 points compared to the preoperative score.
Conclusions:
This is the first reported series assessing cortical bone allograft screws in the pediatric population. Our findings suggest that these implants may reduce the need for hardware removal and associated morbidity. It is important to avoid implant prominences that may cause discomfort to the patient and potentially require reoperation.
Level of evidence:
IV, Therapeutic.
Keywords
Introduction
The traditional surgical method for performing osteosynthesis has involved the use of screws, plates, pins, or nails made primarily of stainless steel or titanium. These materials undergo an inevitable process of corrosion, which releases free metal ions that can activate the host’s immune system through various mechanisms.
Previous reports suggest that sensitization to metal ions, as detected by patch testing, may be present in approximately 10%–15% of the general population. 1 However, clinically significant hypersensitivity reactions directly attributable to orthopedic implants appear to be considerably less common, and robust data specifically in pediatric populations are limited. Extrapolation of these adult results to pediatric patients should be approached with caution. The clinical relevance of metal hypersensitivity in children remains uncertain, and caution is warranted when extrapolating from adult studies.
A significant proportion of complications associated with orthopedic implants are related to the presence of permanent foreign materials, particularly metallic devices.1–7 While metal hypersensitivity and the release of free metal ions are well-documented phenomena, other complications, such as infection or local soft tissue irritation, are not exclusive to metallic implants and may occur with any form of internal fixation. In some cases, the implant removal surgery presents a challenge to the surgeon, with a risk of implant material breakage. 8 These complications have also been reported in dental implants or pacemakers.9–11
In pediatric orthopedic surgery, the need for implants that can provide adequate mechanical stability while minimizing long-term foreign-body presence is especially relevant due to skeletal growth, higher bone remodeling rates, and the desire to avoid secondary procedures for hardware removal.
In 2013, Pastl and colleagues 12 developed an alternative by introducing screws made from cortical bone allograft (Shark Screw®, SurgeBright) to optimize osteosynthesis methods. Once implanted, these screws undergo cellular colonization and progressive remodeling, ultimately transforming into host bone. Since 2021, the first case series have been published on hand and foot surgeries, including osteosynthesis and arthrodesis.13–16 In 2022, the first biomechanical study was published, examining rotator cuff repair in cadavers using cortical bone allograft sutures. 17 All these published studies exclusively review patients of adult age.
The purpose of our study is to retrospectively analyze the clinical and radiological outcomes of allograft screws implanted in the Pediatric Orthopedics Unit of our center between January 2020 and January 2023 and to compare our findings with those previously reported in adult series. We hypothesize that allograft screws offer comparable stability with fewer implant-related complications. A review of the literature shows that no studies have been published analyzing the use of human cortical bone allograft screws in pediatric patients, despite the unique biological and mechanical considerations of the growing skeleton, which may significantly influence implant behavior and clinical outcomes.
Materials and methods
This single-center retrospective study included 30 patients (16 females and 14 males) with lower limb pathologies, in whom a total of 50 human cortical bone allograft screws were implanted. The study period spanned 48 months, from 1 January 2020 to 1 January 2023, and the surgeries were performed in the Pediatric Orthopedics Unit of our center. The average age was 12 years 7 months (range 3–5). The mean follow-up was 44 months (range 24–66).
Among the variables studied were the incidence of minor and major complications, reoperation, and the AOFAS scale. The absorption time and structural integrity of the screws, including the rate of fracture, were evaluated using follow-up radiographs every 1–2 months and periodically until the end of the follow-up period. Pain was also assessed using the visual analog scale (VAS), and postoperative satisfaction was measured using a five-point Likert scale.
Informed consent was obtained from the legal guardians of the patients. The study was approved by the Ethics and Research Committee of our center, and the Strengthening the Reporting of Observational Studies in Epidemiology guidelines were followed for its preparation.
The study population included a heterogeneous group of pathologies and surgical indications. Due to the limited sample size, subgroup analyses were not performed, as they would lack sufficient statistical power. This heterogeneity limits direct comparison between cases and should be considered when interpreting the reported outcomes. All procedures included in this series were elective surgeries. The use of cortical bone allograft screws for acute fracture fixation or trauma-related indications was not evaluated in this study.
Statistical analysis
Continuous variables were expressed as mean and standard deviation. The Wilcoxon signed-rank test (to compare non-parametric data between two related groups) and the Mann–Whitney U test (to compare non-parametric data between two independent groups) were used to assess the statistical significance of the differences. A p-value < 0.05 was considered statistically significant.
Due to the limited sample size, the study was not powered to detect differences between subgroups. Results are therefore primarily descriptive. Confidence intervals (CIs) were calculated for the AOFAS outcome to aid clinical interpretation.
Inclusion and exclusion criteria
Patients included in the study were aged between 3 and 15 years with lower limb pathologies requiring osteosynthesis or arthrodesis, in which cortical bone allograft screws (Shark Screw, SurgeBright®) were used. Patients were excluded if their legal guardians declined participation in the study, had prior open fractures or contaminated surgeries in the area of interest, systemic immunodeficiency, prior graft-versus-host reaction, or if insufficient clinical data were recorded in their medical history. A flow diagram illustrating patient selection and exclusions is provided in Figure 1.
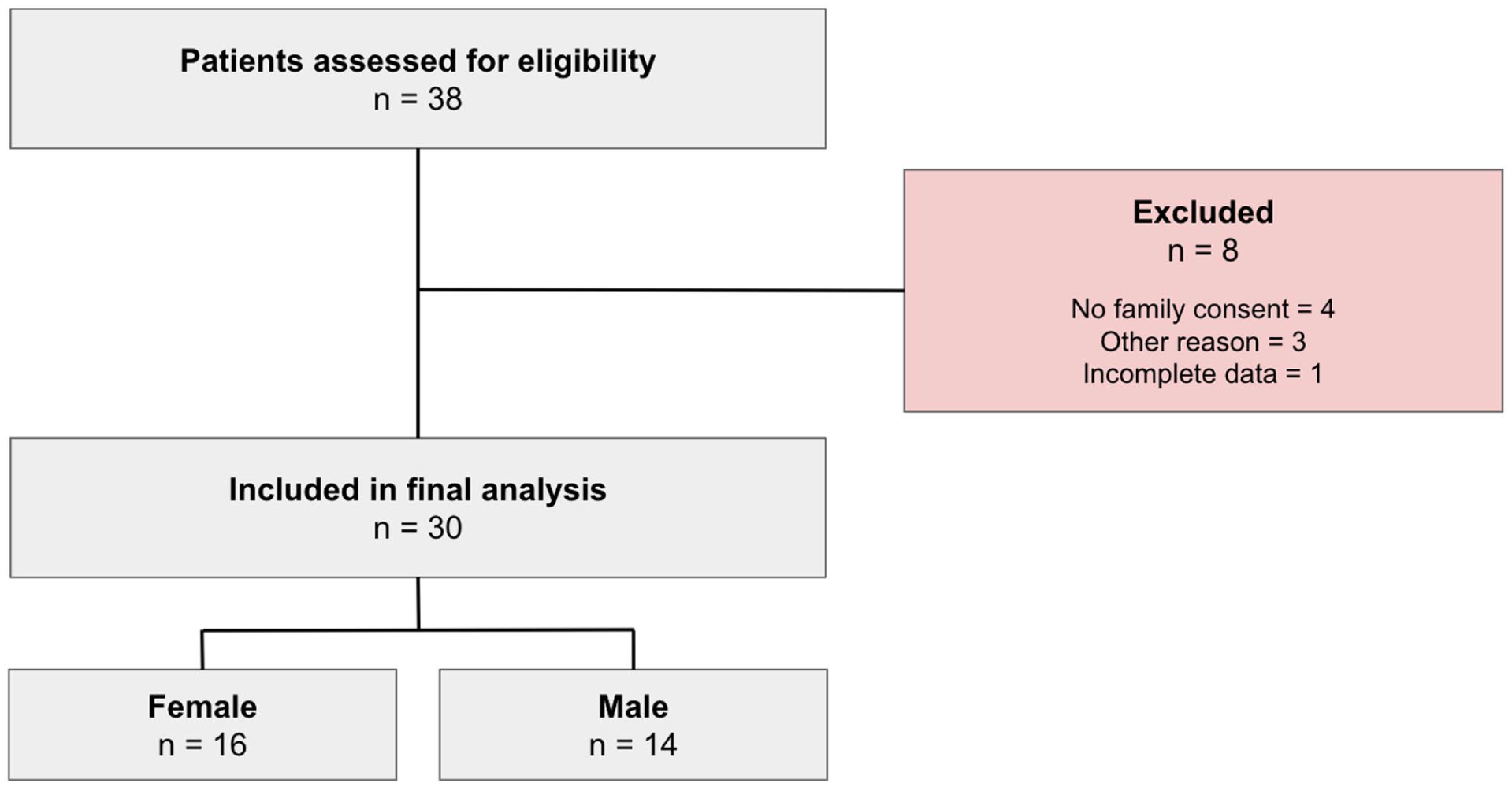
Flow diagram of the study.
Cortical bone allograft screws
A total of 50 human cortical bone screws were used, with 3 possible lengths (15 mm, 35 mm, and 45 mm), 4 possible diameters (3.5 Ø, 4.0 Ø, 4.5 Ø, and 5.0 Ø), and 4 available morphologies (Cut, Diver, Tendon, and Suture), depending on the needs of each surgical intervention (Figure 2). The total cost of each screw is approximately 1000 euros per implant, according to institutional procurement data.

Cortical bone allograft screws showing the four available morphologies: (a) Cut, (b) Diver, (c) Suture, and (d) Tendon.
The surgical technique followed a standardized protocol across all cases. After exposure of the osteotomy or arthrodesis site, the bone was drilled according to the manufacturer’s specifications to accommodate the Shark Screw® allograft implant. The screws were inserted manually under fluoroscopic guidance to ensure optimal positioning and stability, taking advantage of the initial radiopacity of the allograft screw relative to the surrounding bone (Figure 3). No additional graft material other than the allograft screws themselves was used. Intraoperative fluoroscopy was systematically employed in all procedures to verify correct screw orientation, depth, and compression, minimizing the risk of malposition or cortical breach.
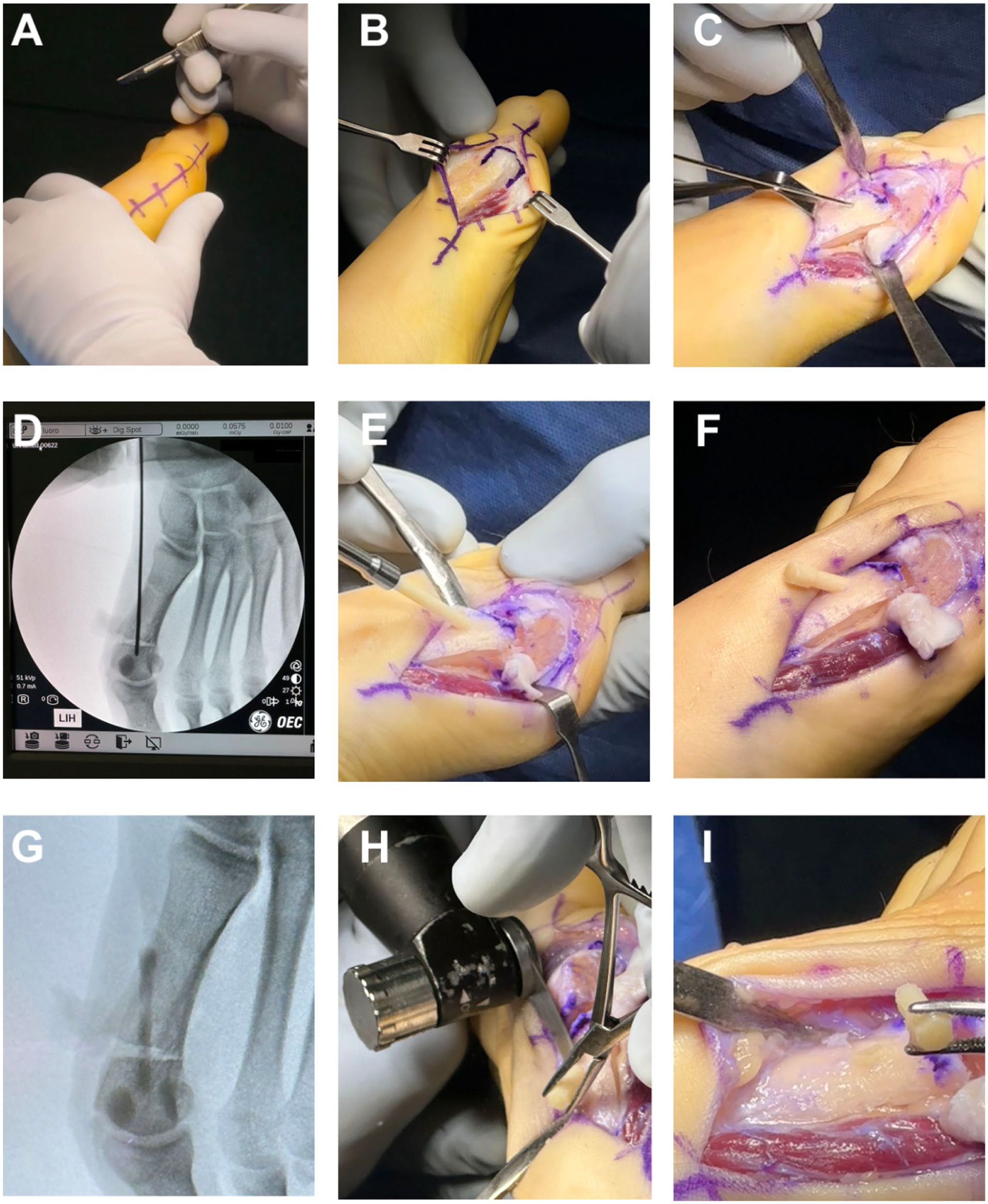
Intraoperative images of case 4 demonstrating the surgical technique for Hallux Valgus correction using a human cortical bone allograft screw.
Ethical and regulatory considerations
All procedures involving human cortical bone allograft screws (Shark Screw®, SurgeBright, Austria) were conducted in accordance with current ethical and regulatory standards for the use of human-derived tissues. The implants were provided by certified tissue banks operating under the European Union Directives 2004/23/EC and 2006/17/EC, as well as ISO-13485 quality management requirements for medical devices. Each allograft screw was fully traceable from donor screening to implantation, following strict protocols for donor selection, serological testing, and sterilization to ensure biocompatibility and safety.
Informed consent specifically included discussion of the novel use of cortical bone allograft screws in pediatric patients. Families were informed of alternative conventional fixation options, including metallic implants, and the decision to proceed was made after shared decision-making with the patient’s caregivers.
Results
The surgeries performed included 9 Chevron-Akin procedures (15 screws; Figure 4), 7 metatarsophalangeal or interphalangeal arthrodesis (10 screws), 6 posterior or anterior tibial tendon reinsertion procedures (six screws; Figure 5), 4 Dwyer osteotomies (12 screws; Figure 6), 2 Super Knee procedures using Paley’s technique (three screws), 1 Elmslie-Trillat osteotomy for recurrent patellar dislocation (two screws; Figure 7), 1 neglected hip dislocation in cerebral palsy using McHale Castle’s technique (one screw), and 1 Evans osteotomy of the cuboid (one screw; Figure 8; Table 1). Procedures were categorized by anatomical region, and the average number of screws per region is detailed in Supplemental Table 1.
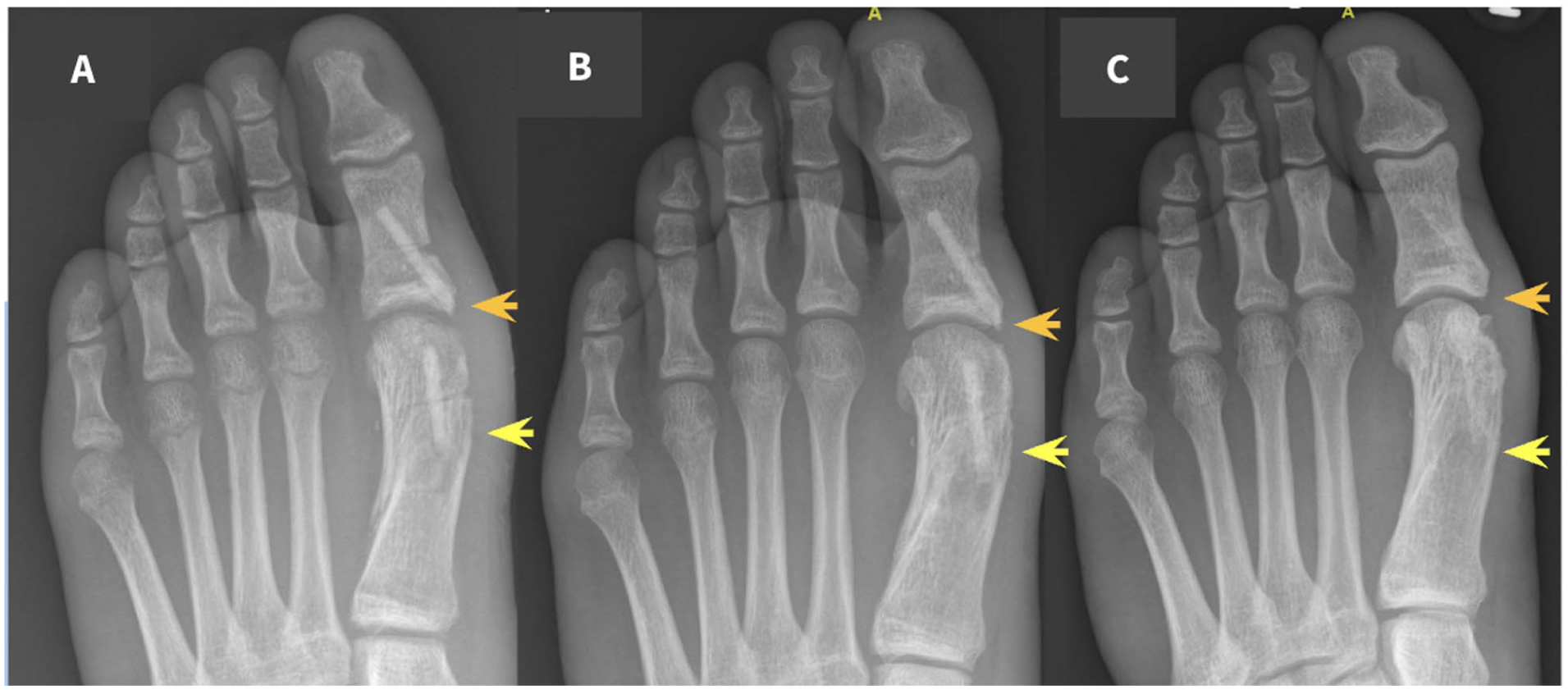
Chevron-Akin osteotomies in a 14-year-old male with Juvenile Hallux Valgus.
Reinsertion of the posterior tibialis following resection of an accessory navicular bone in a 12-year-old female.
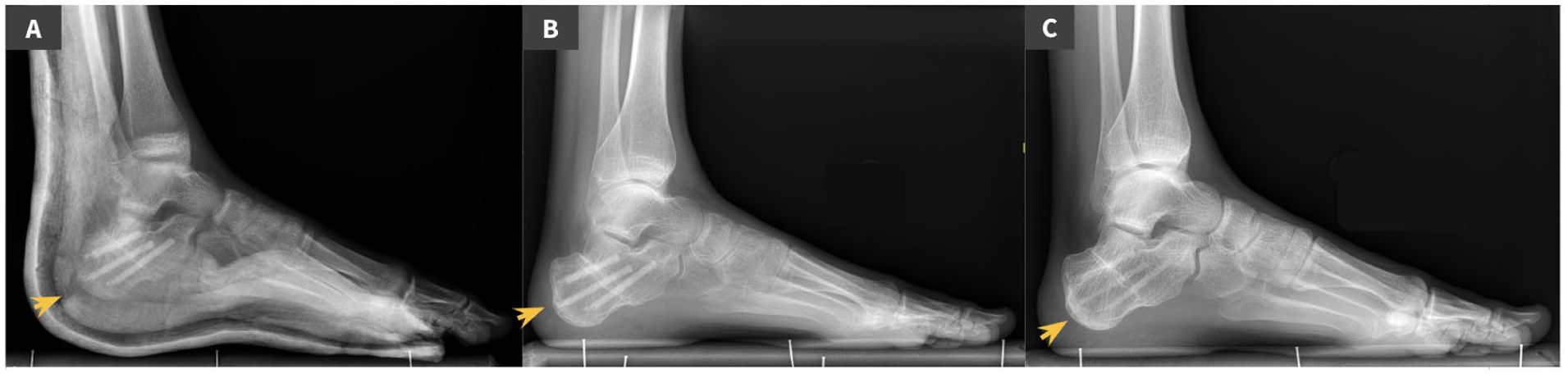
Dwyer osteotomy in a 13-year-old male.
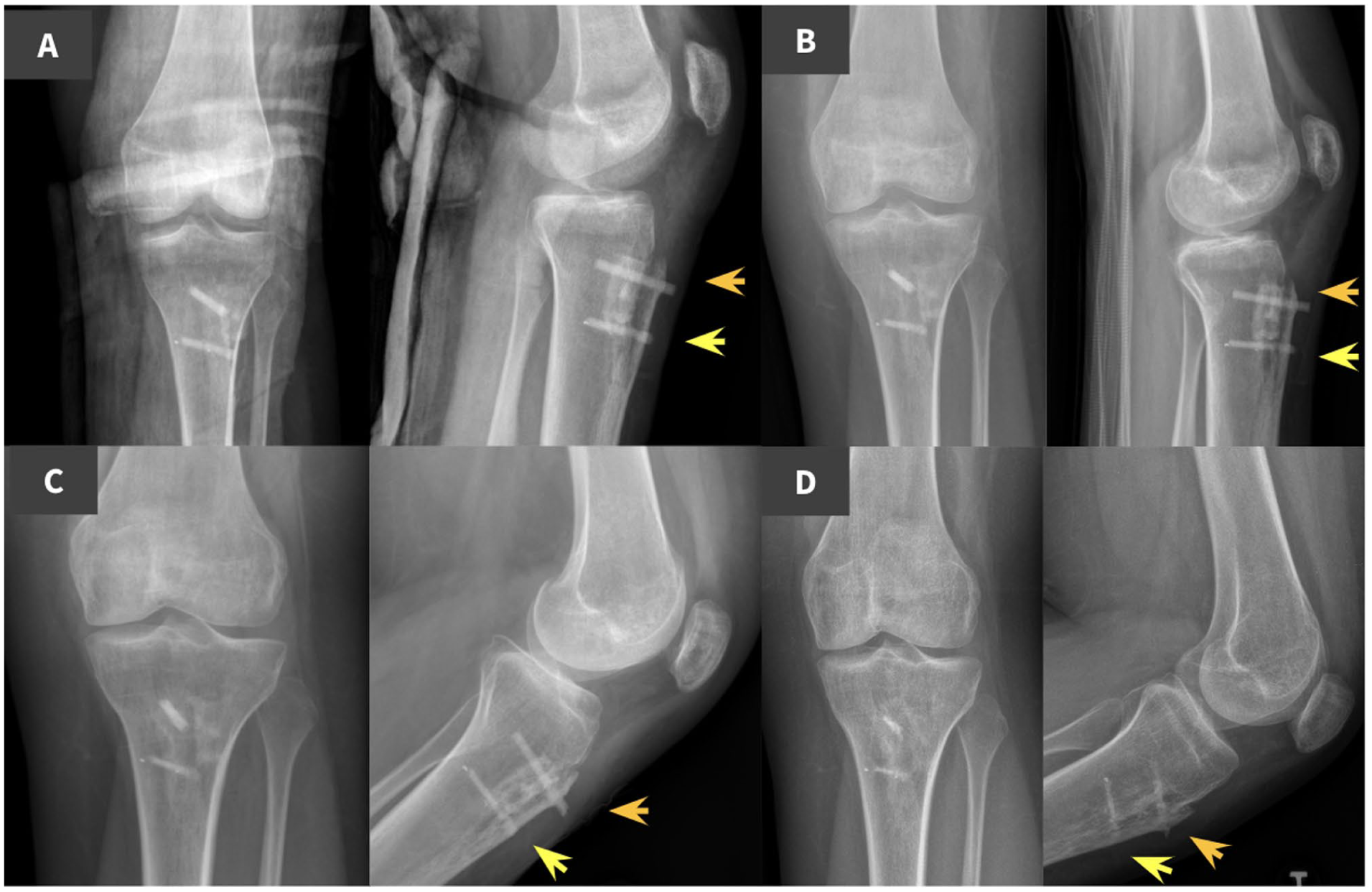
Emslie-Trillat procedure following recurrent knee dislocation in a 14-year-old female.
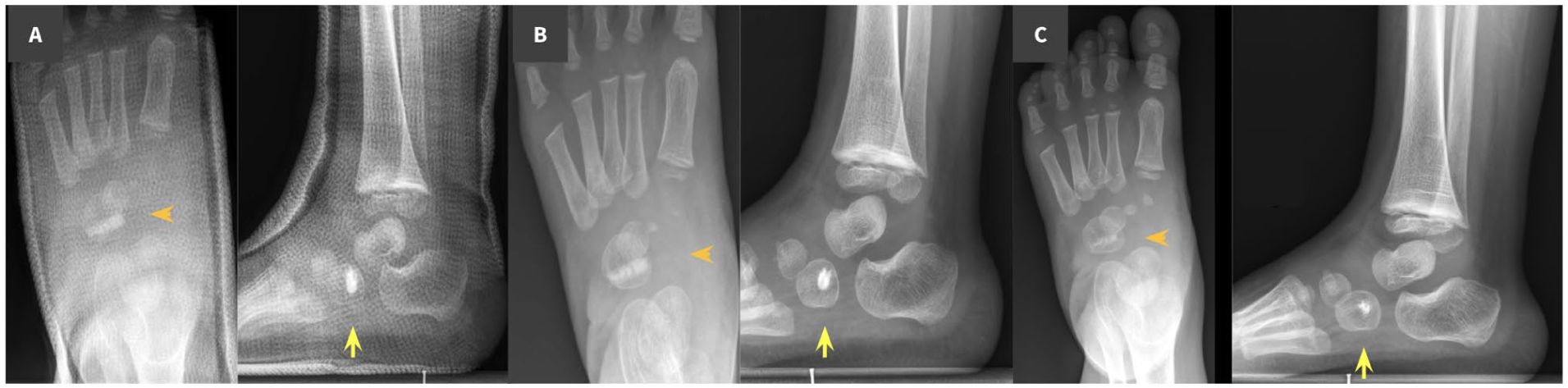
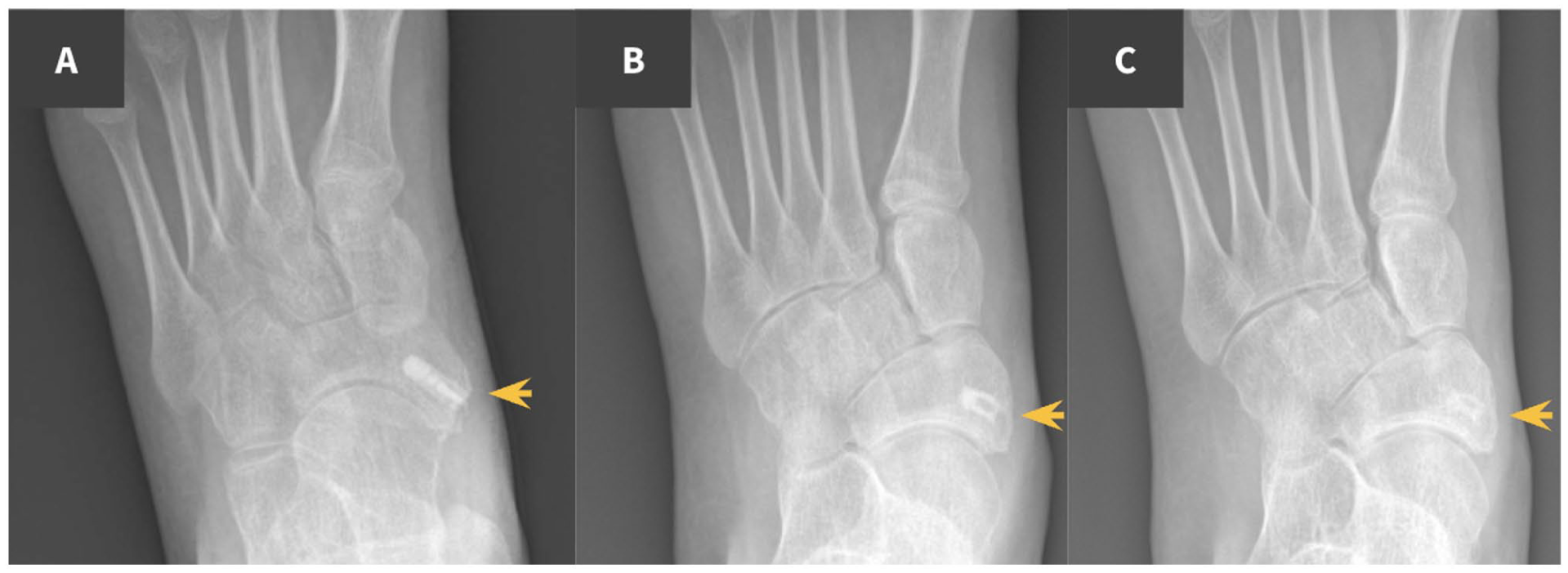
Anterior tibialis transposition to the cuboid bone in a 3-year-old male with clubfoot.
Demographic data of the case series.

M: male, F: female; MTP: metatarsophalangeal; IP: interphalangeal.
Regarding implant diameter, 27 screws of 3.5 Ø, 4 screws of 4.0 Ø, 3 screws of 4.5 Ø, and 16 screws of 5.0 Ø were used. In terms of length, 7 screws of 15 mm, 33 screws of 35 mm, and 10 screws of 45 mm were used.
After a mean follow-up of 44 months (range 24–66), no patients in the sample developed minor or major complications, implant rejection, delayed union, fever, eczema, or infection. Three patients reported local discomfort related to implant prominence. All cases occurred after interphalangeal (IP) arthrodesis (Figure 9), a superficial anatomical region with limited soft tissue coverage. In one patient, symptoms resolved spontaneously with progressive remodeling of the screw head. Two patients (7%) required a minor secondary procedure to trim the prominent portion of the implant.
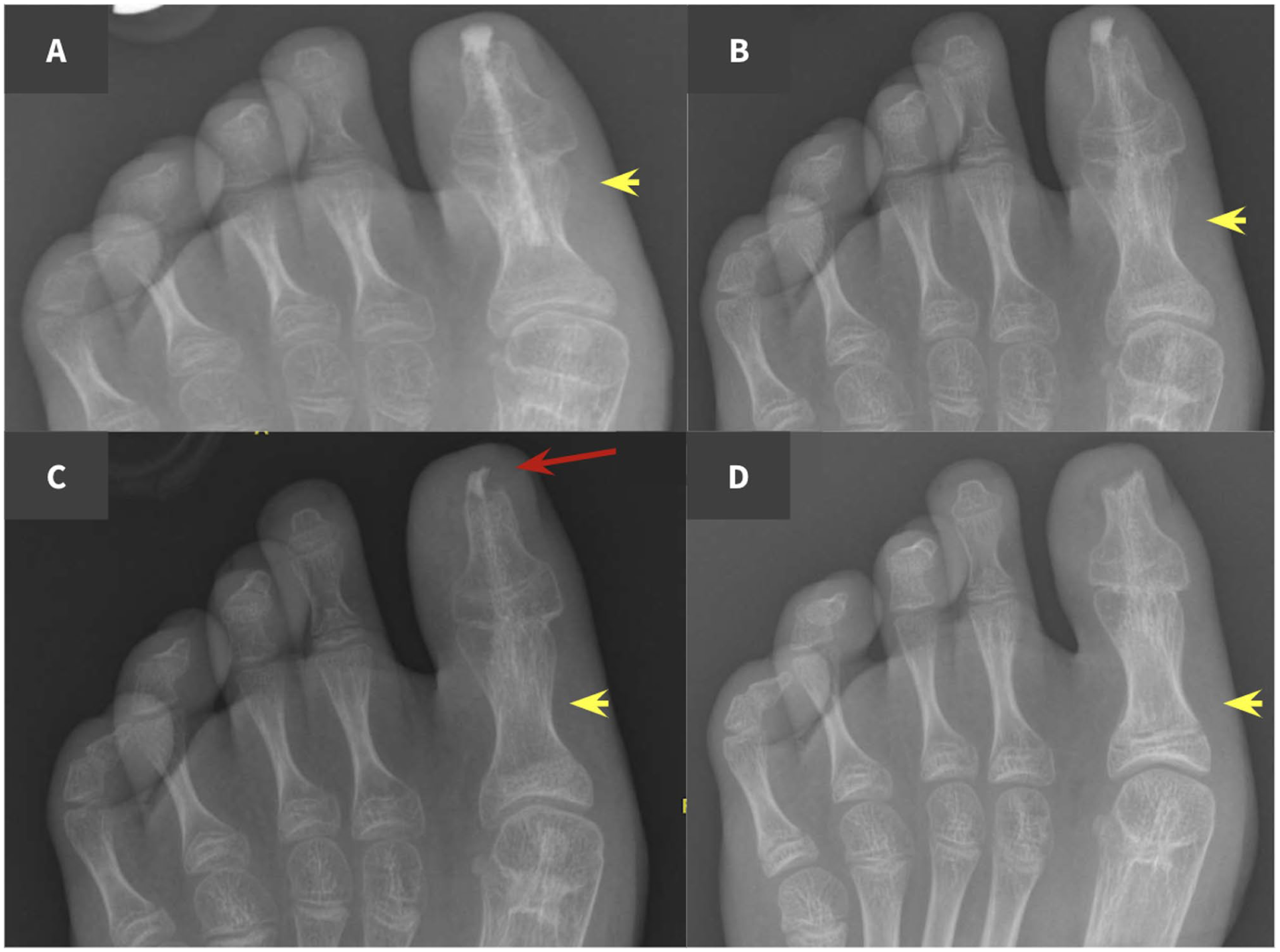
Interphalangeal arthrodesis for hallux extensus in an 11-year-old female.
Although no intraoperative screw breakage occurred in our series, this complication has been described and may arise if the drilling protocol specific to each screw type and diameter is not adequately followed. Proper surgical technique is therefore essential. In the two cases requiring a minor secondary procedure to trim the prominent portion of the implant, the screw was easily reshaped or shortened using standard instruments, such as a rongeur or oscillating saw, and demonstrated mechanical properties comparable to native adult cortical bone.
The mean absorption time was 9 months (range 5–15). It was observed that the absorption time was shorter in younger patients.
Regarding functionality, the mean preoperative AOFAS score was 42.00 ± 32.05, and the mean postoperative score was 80.28 ± 30.82. The mean AOFAS score improved by 38.28 points (95% CI, 25.7–50.9; p < 0.001), exceeding the reported minimal clinically important difference (MCID) for the AOFAS score (approximately 9–10 points).
The mean preoperative VAS score was 5.26 ± 4.30, and the mean postoperative score was 1.10 ± 1.85, with a mean reduction of 4.16 points (95% CI, 2.48–5.84; p < 0.05, Wilcoxon test). Postoperative patient satisfaction, assessed using a Likert scale (1–5), was high, with a mean score of 4.79 ± 0.42 (95% CI, 4.59–4.99).
Discussion
Our study demonstrates the efficacy, tolerance, and complication-free absorption of human cortical bone screws as a method of osteosynthesis in lower limb surgeries in Pediatric Orthopedics. After a mean follow-up of 44 months, all patients showed complete absorption and integration of the implant. None developed infections or any major complications. Regarding the AOFAS scale, measured to assess functionality, there was an average increase of 38.28 points. Importantly, the magnitude of improvement in the AOFAS score substantially exceeded the MCID, supporting the clinical relevance of the observed functional gains despite the limited sample size. Pain, as assessed by the VAS scale, decreased by 4.16 points. Although satisfaction is a subjective and debatable variable, the mean Likert scale score of 4.80 points reflects the general satisfaction of both patients and their legal guardians.
The youngest patients in the sample were 3 years old (patients No. 27 and No. 28, Table 1), both diagnosed with clubfoot; the first was treated with transposition and anterior tibial tendon reinsertion to the cuboid, while the second underwent stabilization through a cuboid osteotomy. These patients showed the fastest implant absorption time, less than 6 months. This finding was descriptive and did not reach statistical significance due to the limited number of cases in this age group. However, it is consistent with the known higher bone turnover and remodeling capacity of very young children. From a clinical perspective, faster graft incorporation may be advantageous, but it could also influence the duration of mechanical support provided by the implant. In very young patients, careful consideration of screw size, positioning, and fixation strategy may therefore be warranted to ensure adequate stability during the early healing phase. At present, no specific modifications to surgical technique can be recommended based on these findings alone, but they highlight the need for further studies focused on age-specific implant behavior.
In this series, no complete implant removals were required. Although two patients (7%) required a minor secondary procedure for screw head reshaping due to discomfort caused by its protrusion, all implants were otherwise retained, resulting in an implant removal rate of 0%. Screw prominence was observed exclusively in IP arthrodesis. This highlights the importance of avoiding implant prominence when using these screws, which could potentially be prevented by trimming the screw head during the initial surgery or by selecting the diver-type screw, which lacks a head and can be fully buried within the cortical bone. Similarly, a longer period could have been allowed for the biological process to facilitate the reabsorption of the screw head, as radiographic monitoring of these patients indicated partial reabsorption. Importantly, these procedures were performed after radiographic evidence of fusion had been achieved. At that stage, the primary compressive role of the screw was no longer required, and the implant functioned as an allogeneic bone scaffold undergoing progressive remodeling. Trimming the screw head, therefore, did not compromise fixation stability or clinical outcome.
After the second surgery, the patients no longer experienced discomfort or pain. This implant-related discomfort experienced by the patient is not specific to allograft screws and can also occur in patients with metallic screws. Similar rates of implant prominence and secondary procedures have been reported with conventional metallic screws in digital arthrodesis, where hardware irritation and removal are not uncommon. Although the reoperation rate in our series is notable, the secondary procedures were minor, foreseeable, and did not involve complete implant removal.
The Shark Screw® cortical bone screws (Surge Bright, Austria) are human-derived allografts that can be used as an alternative to plates, screws, or other metal implants for osteosynthesis.13–18 Once inserted into the bone, these screws are colonized by the body’s own cells, grow, and gradually transform into the patient’s own bone during the process of bone remodeling.
Cortical and trabecular bone allografts are used in various reconstructive bone procedures, such as fractures, tumor surgery, and bone discontinuity.19,20 Cortical allografts heal through osteoclastic resorption and new blood vessel formation, followed by osteoblastic activity and the production of new bone. 21
The risk of infection and immunological rejection associated with this type of implant exists but is lower than that associated with a transfusion of blood derivatives.22,23 One of the main advantages of cortical bone allograft screws lies in their dual function. Initially, they provide mechanical stabilization comparable to conventional fixation devices. Over time, however, they act as a structural bone graft, undergoing cellular colonization and remodeling into host bone. A relevant limitation of this product is its limited availability. Allografts exhibit osteoconductive properties and present a low risk of rejection. Nonetheless, their osteogenic and osteoinductive capacities are considered negligible due to the absence of living cells.24–27 The crucial factors for the incorporation of the allograft into the host bone are revascularization with new bone formation and stable osteosynthesis with good bone–graft union.
These implants also offer several added advantages: they provide mechanical stability, rapid resorption, and an absence of allergic reactions. Regarding the general contraindications for the use of bone allografts, the literature describes contaminated surgeries, open fractures, systemic immunodeficiency, and prior graft-versus-host reaction.22–23,28–30
From an economic and ethical perspective, the use of human cortical bone allograft screws presents several noteworthy advantages. The progressive biological integration of the implant eliminates the need for a second surgical procedure for hardware removal, thereby reducing hospital costs, anesthesia exposure, and overall patient morbidity. This single-stage approach is particularly advantageous in pediatric orthopedics, where minimizing surgical burden is ethically significant. Moreover, the use of certified allografts supplied by regulated tissue banks ensures strict compliance with donor screening, traceability, and sterilization standards, in accordance with current European directives and ISO quality frameworks. This guarantees both the ethical sourcing of human tissue and patient safety. The combination of reduced reintervention rates, lower long-term healthcare costs, and adherence to rigorous bioethical and regulatory principles supports the use of these implants as a cost-effective and ethically sound alternative to conventional metallic devices.
Although the initial cost of cortical bone allograft screws is higher than that of conventional metallic implants, a formal cost analysis was beyond the scope of this study. In pediatric orthopedic surgery, the economic impact of an implant should be considered in the context of potential secondary procedures, particularly implant removal. The avoidance of routine hardware removal and implant-related complications may partially offset the higher upfront cost, although dedicated cost-effectiveness studies are needed to address this issue.
When compared with other fixation materials commonly used in pediatric orthopedics, such as titanium alloy mini-screws, magnesium-based implants, or poly-L-lactic acid (PLA) devices, cortical bone allograft screws offer distinct biological and clinical advantages.
Titanium implants provide excellent mechanical stability but often require subsequent removal due to irritation, interference with growth plates, or hypersensitivity reactions.2–5 PLA and other polymer-based bioresorbable materials, while eliminating the need for removal, may induce local inflammatory responses during degradation and demonstrate slower load transfer to the host bone. Magnesium-based implants exhibit favorable mechanical strength and controlled resorption but carry the risk of gas formation and unpredictable degradation kinetics, particularly in younger patients with higher bone turnover. By contrast, human cortical bone allograft screws integrate into the host bone through natural remodeling, allowing progressive load sharing and complete substitution by viable bone tissue without generating foreign body reactions. This biological incorporation makes them particularly suitable for the pediatric population, where ongoing skeletal growth and metabolic activity demand materials that can adapt physiologically over time.
Hardware removal is a common issue in pediatric orthopedics and its frequency varies with indication and anatomical region. For example, a large pediatric series of physeal ankle fractures reported a hardware removal rate of 24.2%. 31 In broader pediatric series and systematic reviews, the reported incidence of implant removal spans widely, depending on the population and implant type. Importantly, hardware removal is not without risk: series in children report complication rates after removal on the order of 9%–10% (incomplete removal, infection, refracture, neurovascular injury). 32 A recent review outlined current knowledge on metal implants in children and emphasized that routine removal after fracture healing is not always medically justified. The indications for elective hardware removal remain poorly defined, and available evidence suggests that removal should be reserved for infection, mechanical failure, or implant-related symptoms rather than following tradition alone. 33
In our series, symptomatic prominence requiring surgical trimming occurred in two patients (7% of patients), all after interphalangeal arthrodesis. Although this reoperation rate is lower than the removal rates reported for some pediatric fracture cohorts, it is comparable to reported complication rates after hardware removal. These data suggest that, while cortical allograft screws may reduce the need for complete implant explantation in many cases, superficial locations with limited soft tissue coverage remain at risk for symptomatic prominence and may require specific intraoperative measures (head trimming, use of headless/diver-type screws) to minimize irritation.
Future research should aim to expand on the preliminary findings of this study through multicenter, prospective trials with larger pediatric cohorts and extended follow-up periods. Biomechanical investigations comparing cortical bone allograft screws with metallic and bioresorbable alternatives are essential to better quantify their load-bearing capacity, torsional resistance, and fatigue behavior under pediatric physiological conditions.17,18 Histological studies could provide valuable insights into the cellular mechanisms of graft remodeling and bone incorporation, elucidating the timeline and quality of biological integration. In addition, long-term follow-up until physeal closure would allow assessment of the potential influence of these implants on bone growth and alignment, ensuring their safety throughout skeletal development. Taken together, such studies would strengthen the evidence base for the use of allograft screws and refine their indications within pediatric orthopedic surgery.
The limitations of our study include those inherent to its retrospective and single-center design, the absence of a control group, sample size, diagnosis and procedure heterogeneity, growth differences by sex, and the challenges associated with studies in the pediatric population. The study did not include a control group treated with conventional metallic implants. Although such a comparison would be desirable, it was not feasible due to the retrospective nature of the study, the limited number of cases, and the heterogeneity of pathologies and surgical indications. Under these circumstances, an institutional comparison with metallic implants could introduce significant bias. Consequently, our results should be interpreted in light of published pediatric and adult series using metallic fixation rather than as a direct comparative analysis.
The risk of infection and immunological rejection associated with this type of implant appears to be low. Although no infections were observed in our cohort, infection management in the presence of cortical bone allograft implants warrants consideration. Adult series suggest that, in contrast to metallic hardware, allograft screws may not necessarily require removal in cases of superficial or early infection, as they lack permanent foreign material and undergo biological incorporation. However, in cases of deep or persistent infection, standard principles of infection control, including implant removal if required, should still be applied.
A larger sample size and longer follow-up could have helped clarify the absorption rate according to age, as well as further explore the potential contraindications of its use. Although the sample size is considerable for the clinical context, a larger sample could provide greater statistical power and allow for better analysis of subcategories based on age, sex, and pathology.
Future research should prioritize well-defined clinical indications and standardized surgical techniques to allow more robust evaluation of cortical bone allograft screws. Focused studies in specific anatomical regions or procedures, rather than broad heterogeneous applications, are necessary before wider adoption of this technology in pediatric orthopedic practice.
Conclusion
This is the first reported series assessing cortical bone allograft screws in the pediatric population. Our findings suggest that these implants may reduce the need for hardware removal and associated morbidity. It is essential to avoid implant prominences that may cause discomfort to the patient and necessitate reoperation.
Supplemental Material
sj-docx-2-cho-10.1177_18632521261420136 – Supplemental material for Osteosynthesis with allograft screws in pediatric orthopedics
Supplemental material, sj-docx-2-cho-10.1177_18632521261420136 for Osteosynthesis with allograft screws in pediatric orthopedics by Javier Gutierrez-Pereira, Flavio Paulos, Pedro Gutierrez-Carbonell and Antonio Garcia-Lopez in Journal of Children's Orthopaedics
Supplemental Material
sj-pdf-1-cho-10.1177_18632521261420136 – Supplemental material for Osteosynthesis with allograft screws in pediatric orthopedics
Supplemental material, sj-pdf-1-cho-10.1177_18632521261420136 for Osteosynthesis with allograft screws in pediatric orthopedics by Javier Gutierrez-Pereira, Flavio Paulos, Pedro Gutierrez-Carbonell and Antonio Garcia-Lopez in Journal of Children's Orthopaedics
Footnotes
Author contributions
JGP: study design, data collection, statistical analysis, data interpretation, manuscript preparation, and literature search. FP: study design, statistical analysis, and literature search. PGC: study design, data collection, and supervision. AGL: study design and supervision.
Declaration of conflicting interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Ethical statement
Full name of the ethical board that approved your study: COMITÉ ÉTICO DE INVESTIGACIÓN CON MEDICAMENTOS DEL DEPARTAMENTO HOSPITAL GENERAL UNIVERSITARIO DE ALICANTE. Approval number given by the ethical board: Code 2024-018. References: Ref. CEIm: 2024-018—Ref. ISABIAL: 2024-0009.
Informed consent
Confirmation that all your patients gave written informed consent: All participants gave informed consent.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
